Long Term Ophthalmic Follow Up in LCHAD Deficiency
Levy N*, Paz T, Leiba H, Hadas B and Parness R
Ophthalmology Department, Kaplan Medical Center, Israel
Submission: November 11, 2017; Published: December 11, 2017
*Corresponding author: Levy Niv, Ophthalmology Department, Kaplan Medical Center, 1 Pasternak Road, Rehovot 76100, Israel, Tel: +972-54-5967283; Email: Niv.levi@mail.huji.ac.il
How to cite this article: Levy N, Paz T, Leiba H, Hadas B, Parness R.Long Term Ophthalmic Follow Up in LCHAD Deficiency. JOJ Ophthal. 2017; 5(5): 555673. DOI: 10.19080/JOJO.2017.05.555673.
Abstract
Long-chain 3-hydroxyacyl-CoA dehydrogenase (LCHAD) deficiency is an autosomal recessive disorder of mitochondrial fatty acid beta oxidation, associated with hypoketotic hypoglycemia, hepatic steatosis, rhabdomyolysis, cardiomyopathy, polyneuropathy and retinal changes. We present the course of retinal findings in a case of a 6-year-old girl with LCHAD deficiency diagnosed at birth, and hence early treated and followed. Our patient had annual eye exams from the age of 1 year. Clinical examinations, ocular coherence tomography (OCT) and electroretinogram (ERG) findings during follow up are presented. At the age of 3 years, after systemic deteriorations, nyctalopia appeared with pigmentary retinopathy changes in both eyes. ERG was subnormal while Infra-red reflectance imaging with OCT displayed more advanced stage of the disease. Progressive chorioretinopathy with visual impairment was observed along the follow up on clinical exams, as well as on repeated OCTs and ERGs.
Conclusion: Retinal dystrophy in children can be related to metabolic disorders, including LCHAD deficiency. The retinal findings may rapidly progress despite dietary treatment, in relation to frequent metabolic decompensations. On early phase of the disease, Infra-red reflectance imaging with OCT may better identify the severity of the chorioretinopathy. Regular follow-up including OCT is recommended in LCHAD deficiency patients to monitor the ocular status.
Keywords: Long-chain 3-hydroxyacyl-CoA dehydrogenase (LCHAD); Retinal dystrophy; Ocular coherence tomography (OCT)
Introduction
Mitochondrial beta oxidation is the principal pathway by which energy is obtained from fatty acids [1]. Defect in this pathway are associated with hypoketotic hypoglycemia, hepatic steatosis, rhabdomyolysis, cardiomyopathy, polyneuropathy and abnormal urinary metabolites [1-4]. Long-chain 3-hydroxy-acyl- CoA dehydrogenase (LCHAD) deficiency disrupts this pathway and was first described in 1989 [1]. LCHAD is an autosomal recessive disorder [2] with unknown occurrence frequency in Israel or the United States, but in the Swedish population the incidence is 1:100,000 [3]. Analysis of the frequency of the most common mutation (G1528C), comprising more than 90% of mutated alleles in Swedish population [3], revealed a carrier frequency of 1:240 in Finland. The ocular findings associated with this rare metabolic disorder may include pigmentary retinopathy, developmental cataract, progressive myopia, visual fields defects and color vision abnormalities [1]. Here, we present the course of retinal findings in a case with LCHAD deficiency. Clinical examination, ocular coherence tomography (OCT) and electroretinogram (ERG) findings during follow up are presented.
Case Report
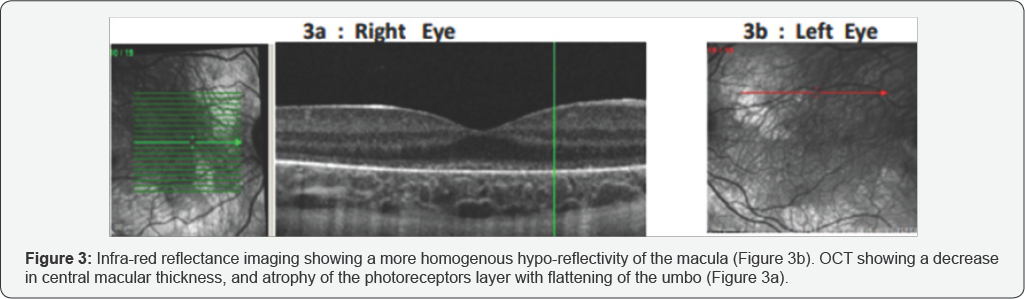
We present the history of a 6-year-old girl who was diagnosed at birth with LCHAD deficiency. She was started on a diet with low-fat high-carbohydrate content and docozahexanoic acid (DHA) supplements, and she had annual eye exams. At the age of 1 year fundoscopy was normal. At the age of 2 years the fovea had a very dark appearance, as a result of perifoveal atrophy. After systemic deterioration and repeated hospitalizations due to hypoglycemia, vomiting and rhabdomyolisis, nyctalopia appeared. Examination at the age of 3 years revealed visual acuity of 6/7.5 in both eyes, and ophthalmoscopy demonstrated a dark macula and a "salt & pepper" retinopathy extending to the arcades. Infra-red reflectance imaging showed dark center bordered by well-defined hypo-reflective spots, and surrounded by hypo-reflective zone (Figure 1b). On OCT there were extra- foveal irregularities in the ellipsoid zone of the photoreceptor layer (Figure 1a). On ERG there were slightly reduced responses. Examination at the age of 6 years revealed deterioration of the visual acuity to 6/30. She had myopia in both eye (-1.5), and her color vision was 1/13. Fundoscopy revealed extensive atrophy of the retina and retinal pigment epithelium (RPE) with hypopigmentation and hyperpigmentation in the posterior pole, and pigment clumping in the periphery (Figure 2a+b). Infrared reflectance imaging showed a more homogenous hypo- reflectivity of the macula (Figure 3b). OCT showed a decrease in central macular thickness, and atrophy of the photoreceptors layer with flattening of the umbo (Figure 3a). ERG showed severely reduced photopic and scotopic responses.


Discussion
Retinal dystrophy is one of the manifestations of LCHAD deficiency [1-4]. Treatment is aimed at ensuring sufficient caloric intake during periods of metabolic stress and fasting, and DHA supplementation is believed to slow retinal dystrophy [3]. LCHAD activity has been found in human RPE cells, and the metabolic products of the p-oxidation pathway in this enzyme deficiency might contribute to the retinal damage seen in this rare metabolic disorder [5].
The chorioretinopathy in LCHAD deficiency can be classified into 4 stages [6]:
At stage 1, the fundus appears normal to diffusely hypo- pigmented.
At stage 2, pigment clumping in the RPE gives rise to pigmentary changes.
At stage 3, the granular fundus pigmentation disappears centrifugally with concomitant atrophy of the choroid and the posterior pole, leaving bare sclera.
At stage 4, the central island of the remaining choroids and photoreceptors is lost, and the posterior pole becomes atrophic and devoid of RPE.
At the periphery choroidal vessels and RPE remain relatively normal. This case of a 6-year-old girl with LCHAD deficiency diagnosed at birth, presented the clinical examination, OCT and ERG findings during follow up, and demonstrated that early diagnosis and adequate therapy may delay but not prevent the progression of retinal dystrophy. In the early phase of the disease, ERG findings did not reflect the clinical findings. A rather good ERG can sometimes occur in secondary retinal degenerations, compared with fundus changes and visual field abnormalities [2]. On the other hand, OCT and infra-red reflectance imaging better demonstrated the severity of the chorioretinopathy. In the early phase of the disease the Infra-red reflectance imaging and OCT showed a dark center, bordered by well-defined hypo- reflective spots and surrounded by hypo-reflective zone, as a result of the perifoveal atrophy developing early in the disease course. Thus, OCT and Infra-red reflectance imaging are more sensitive and easier to implement in the early phases of LCHAD deficiency.
Conclusion
Retinal dystrophy in children can be related to metabolic disorders, including LCHAD deficiency. The retinal findings may rapidly progress despite dietary treatment, in relation to frequent metabolic decompensations. On early phases of the disease, Infra-red reflectance imaging and OCT may better identify the severity of the chorioretinopathy. Regular follow-up including OCT is recommended in LCHAD deficiency patients to monitor the ocular status.
Conflict of Interest
The authors have no conflict of interest relevant to this article.
Patient Consent
This report does not contain any personal information that could lead to the identification of the patient.
References
- McGimpsey SJ, Williams M, Mulholland DA (2006) Ten year follow up of pigmentary retinopathy associated with LCHAD deficiency. Eye (20): 1074-1075.
- Schrijver-weilingl, van Rens GH, Wittebol-Post D, Smeitink JA, de Jaker JP, et al. (1998) Retinal dystrophy in LCHAD deficiency. Br J Ophthalmol 81(4): 291-294.
- Fahnehjelm KT, Holmstrom G, Ying L, Haglind CB, Nordenstrom A, et al. (2008) Ocular characteristic in 10 children with LCHAD deficiency: a cross-sectional study with long-term follow up. Acta Ophthalmol 86(3): 329-337.
- Turaka K, Bryan JS, Gordon AJ, Kwong HM Jr, Reddy R, et al. (2012) Clinical and image-guided chorioretinal findings in LCHAD deficiency. J Pediarc Endocrinol Metabol 25(5-6): 565-567.
- Tyni T, Johnson M, Eaton S, Pourfarzam M, Andrews R, et al. (2002) Mitochondrial fatty acid ß-oxidation in the retinal pigment epithelium. Pediatr Res 52(4): 595-600.
- Tyni T, Kivela T, Lappi M, Summanen P, Nikoskelainen E, et al. (1998) Ophthalmological findings in long-chain 3-hydroxyacyl CoA dehydrogenase deficiency caused by the G1528C mutation, A new type of chorioretinopathy. Opthalmology 105(5): 810-824.






























