Macular Dystrophy in Kjellin's Syndrome: A Case Report
Nathalie Massamba*, Claire Meyniel, Barham Bodaghi and Valerie Touitou
Department of Ophthalmology, University of Paris VI, Fra
Submission: October 02. 2017; Published: November 20, 2017
*Corresponding author: Nathalie Massamba, Departments of Ophthalmology, Pitie- Salpetriere Hospital, University of PARIS VI.DHU Handicaps and Vision, 125 rue de Stalingrad, 93000 Bobigny, France, Email: nathalie.massamba@gmail.com
How to cite this article: Nathalie M, Claire M, Barham B Valerie T. Macular Dystrophy in Kjellin's Syndrome: A Case Report. 2017; 5(4): 555667.DOI: 10.19080/JOJO.2018.06.555667
Abstract
Purpose: To report a case of macular dystrophy in patient with Kjellin syndrome and to analyze different multimodal imaging associated with neurologic manifestations
Methods: This is an observational and descriptive case report.
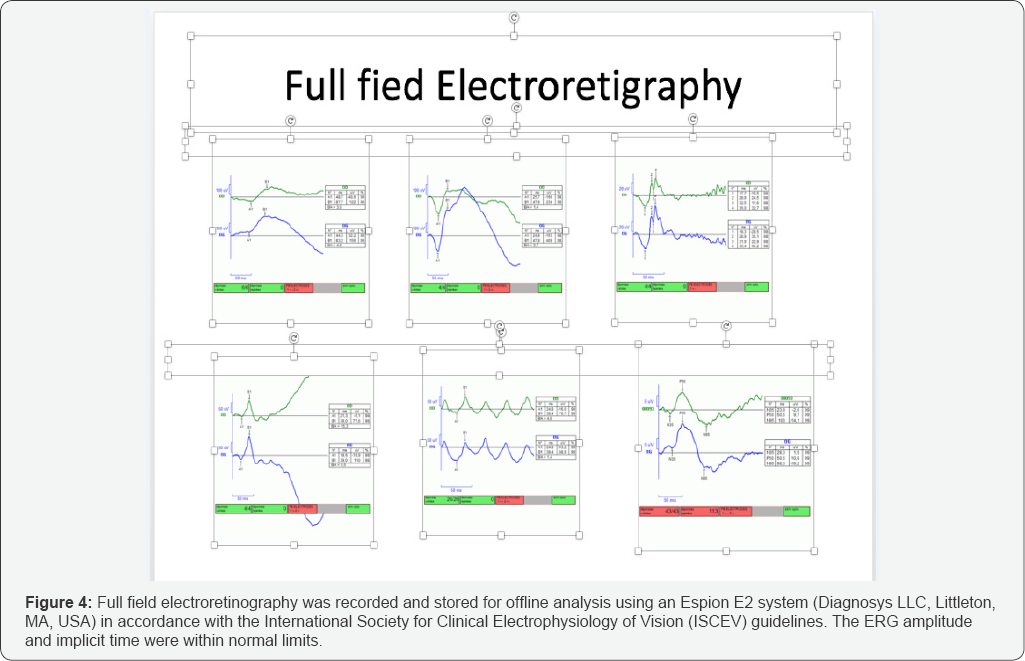
Results: A 48 -year-old female affected by spastic pa paresis, dysarthria, hearing loss associated with sphincter disorders was referred to our department because of neuro ophthalmologic examination. The BCVA was excellent 20/20 in both eyes, slip lamp examination was also normal. Color fundus photography, Fundus auto fluorescence, Fluorescein angiography, Green Indocyanine Angiography and spectral- domain optical coherence tomography imaging of both eyes were also performed using Heidelberg Engineering, Heidelberg, Germany. Full field electroretinography was recorded and stored for offline analysis using an Espion E2 system (Diagnosys LLC, Littleton, MA, USA) was also done.
Conclusion: We report a case of multimodal imaging in macular dystrophy associated with neurologic event, hearing loss as a possible Kjellin's syndrome with a complete SPG11 and SPG15 phenotype as abnormalities detected in multifocal retinal imaging.
Keywords: Kjellin's syndrome; Electro-oculography; Electroretinography; Spastic paraparesis; Cognitive impairment
Introduction
Kjellin's syndrome is an autosomal recessive disorder that is characterized by the progressive emergence in children of walking difficulty and leg stiffness, followed by spastic paraplegia accompanied by hyperreflexia and distal amyotrophy [1]. It is complicated with macular dystrophy. Individuals with this syndrome present with macular changes, most often described as fundus flavimaculatus or Stargardt disease-like, particularly on the basis of fluorescein angiography findings [1].
Case Presentation
A 48-year-old Caucasian female, was referred to our department, at Pitie-Salpetriere Hospital for neuro ophthalmic consultation. At his entrance in the examination box, we observed a difficulty walking, with spasm that made spastic and jerky walking. Since childhood, he had experienced learning difficulties and mild cognitive impairment combined with hypoacusis. Her attendant also reported associated vesico- sphincterian disorders. There was long-term history of visual acuity impairment; no worsening of vision over years Prior to admission, her family history, the attendant reported that two maternal aunts presented also a difficult working. non- consanguineous history of her parents
Neurologic examination released
A walking difficulty and leg stiffness, dysarthria, frontal release signs, preserved perception of touch and pain, spasticity of the lower limbs with a scissors gait associated with hearing loss.
Ophthalmological Evaluation
Best-corrected visual acuity (BCVA) was 20/20 within the right eye (RE) and 20/20 in the LE. The intraocular pressure was 15 mm/hg on the RE and 16 mm/hg on the LE. Slip lamp examination was normal. Color Fundus examination showed in both eyes, multiple round yellowish flecks at the level of the retinal pigment epithelium scattered at the posterior pole. Some of these flecks were elongated and showed partial confluence with neighboring flecks giving the appearance of fishing net (Figure 1). Retinal imaging including Fundus auto fluorescent (FAF) , Fluorescein Angiography ( AF) ,Green Indocyanine Angiography (ICG) and Spectral -Domain Optical Coherence Tomography was performed using Heidelberg Engineering, Heidelberg, Germany.
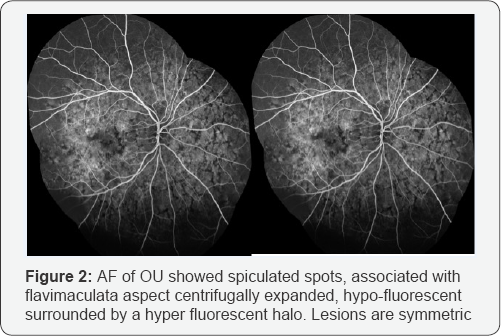
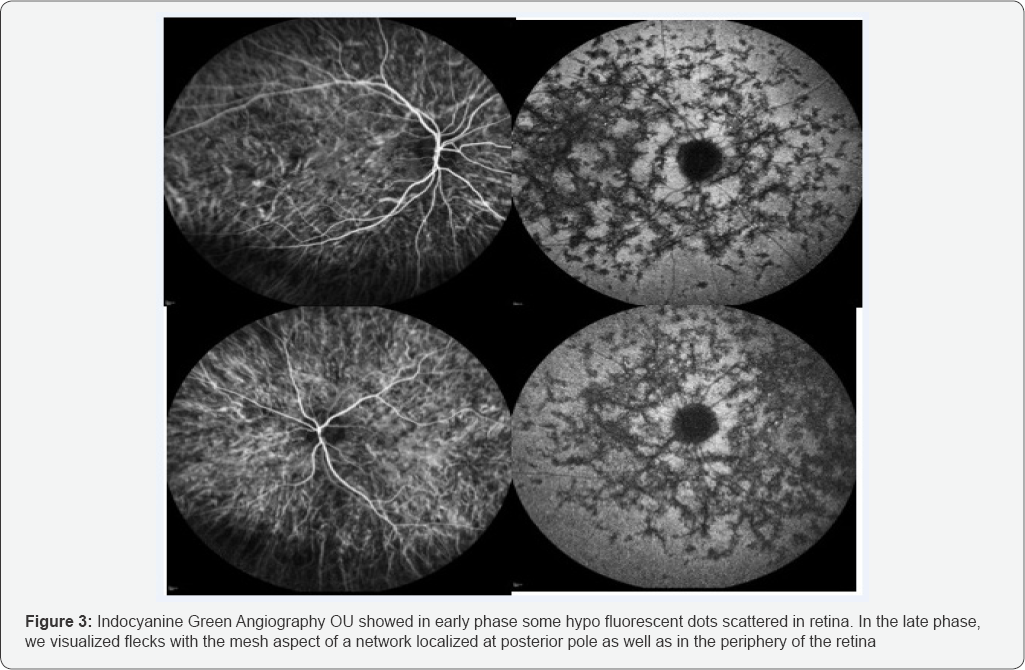
FAF showed flecks that were limited to the posterior pole, extended beyond the vascular arcades. The fovea zone still iso fluorescent and normal. The lesions appeared as hypo fluorescent periphery on the retina, consistent with local atrophy of the retinal (Figure 1) pigment epithelium. alternated with hyper fluorescent flecks due to the masking effect of the yellowish material. AF showed spiculated spots, associated with flavimaculata aspect centrifugally expanded, hypo-fluorescent surrounded by a hyper fluorescent halo (Figure 2). ICGA on the early phase showed some hypo fluorescent dots scattered in retina.

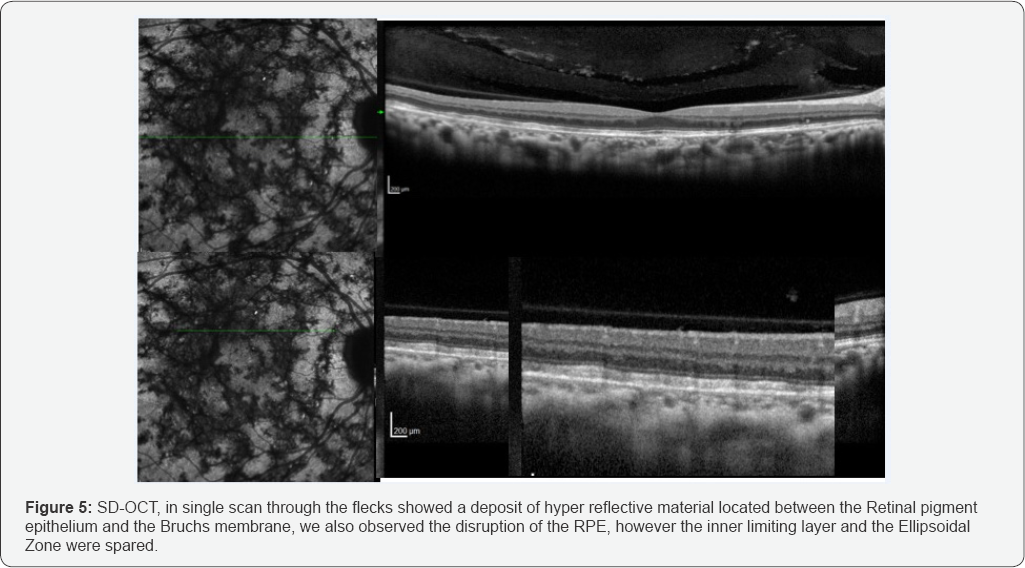
In the late phase, we visualized flecks with the mesh aspect of a network localized at posterior pole as well as in the periphery of the retina (Figure 3). Full field electroretinography was recorded and stored for offline analysis using an Espion E2 system (Diagnosys LLC, Littleton, MA, USA) in accordance with the International Society for Clinical Electrophysiology of Vision (ISCEV) guidelines. The ERG amplitude and implicit time were within normal limits; (Figure 4). SD-OCT, in single scan through the flecks showed a deposit of hyper reflective material located between the Retinal pigment epithelium and the Bruchs membrane, we also observed the disruption of the RPE, however the inner limiting layer and the Ellipsoidal Zone were spared (Figure 5).
Discussion
The earliest descriptions of Kjellin’s syndrome date from 1941, 1945, and 1959 [2,3]. More recently, Frisch et al. [4]. reported autofluorescence, electrophysiologic, and angiographic findings for Kjellin's syndrome. Kjellin's syndrome is an autosomal recessive disorder that is characterized by the progressive emergence in children of walking difficulty and leg stiffness, followed by spastic paraplegia accompanied by hyperreflexia and distal amyotrophy. It is complicated by dysarthria, urinary dysfunction, mental retardation, and progressive dementia. Ocular complications are secondary symptoms and consist of macular dystrophy. Cerebral magnetic resonance imaging (MRI) scans reveal characteristic anomalies such as cerebellar atrophy, with thin corpus callosum, and frontotemporal atrophy.
The patient showed typical phenotypic traits of Kjellin's syndrome characterized, also she presented Results of brain magnetic resonance imaging (MRI) scans for) show severe corpus callosum which have been done, 5 years before. From a neurologic point of view, the first symptoms of Kjellin’s syndrome appear well before the age of 10 years old. Patients have delayed walking and speak between the ages of 1 and 3 years. They later encounter walking and running impairment. Mental retardation becomes obvious, and memory deficits and early academic problems appear, sometimes at the end of the first decade [1]. During the second decade, unsteadiness and paraplegia become evident. They are rap- idly joined by severe cognitive problems and praxic or gnostic difficulties [2].
At presentation, the disease had been started for less than 5 years old with delayed walking. They reported that the family notice it immediately, but without wooied. it is only after about 7 years after the appearance of the difficulty of walking that they referred back to a neurologic physician. Whom continue the neurologic exam follow up. Thus, the patient had never complain of low visual acuity. This confirmed what previously study demonstrated that the natural history of macular dystrophy in Kjellin's syndrome is such that patients rarely complain of low visual acuity unless it is associated with cataract or optic neuropathy [2,3]. However, we did not performed yet the genotype analysis, but our findings, including :macula dystrophy, hearing loss, spastic para paresis associated with sphincter disorder and deformity of the corpus callosum are suggestive of HSP subtypes 11 and 15.
Bernard Puech et al. [1] found in 2009 that, ocular signs appear after 20 years old, when the neurologic problems are already advanced, they also added that the macular dystrophy in this entity are not changed much, but spread to periphery, this help them to have a good visual acuity. We agree with the authors findings because our patient had all neurologic manifestations associated with macular dystrophy extend to the periphery but the visual acuity still excellent.
Although this entity could have differential diagnosis especially with Stargardt disease, a well-performed clinical examination that excluded any neurological abnormality before retinal imaging is required. Our case represents Kjellin's syndrome with a complete SPG11 and SPG15 phenotype as abnormalities detected in multifocal retinal imaging. Vinicius Monteriro De Castro et al. [5] defined that the retinal changes in Kjellin’s syndrome might be the result of a metabolic defect, causing accumulation of products related to lipofuscin. We don't have more information about this affirmation. Future study with histologic, gentotype and phenotype analysis will help for the better understanding of the disease.
References
- Puech B, Lacour A, Stevanin G, Sautiere BG, Devos D, et al. (2011) Kjellin Syndrome Long-term Neuro-ophthalmologic Follow-up and Novel Mutations in the SPG11 Gene. Ophthalmology 118(3): 564-573.
- Kjellin K (1959) Familial spastic paraplegia with amyotrophy, oligophrenia, and central retinal degeneration. Arch Neurol 1: 133-140.
- Franceschetti A, Klein D (1941) Uber das Vorkommen von he- reditarer juveniler Maculadegeneration (Typ Stargardt) bei zwei Geschwisterpaaren der Friedreich Sippe 'Glaser'. Arch Julius Klaus Stift Vererbungsforch Sozialanthropol Rassen- hyg 16: 469-493.
- Arndt CF, Husson J, Derambure P, Hache JC, Arnaud B, et al. (2005) Retinal electrophys- iological results in patients receiving lamotrigine mono- therapy. Epilepsia 46: 1055-1060.
- Vinícius Monteiro de Castro, André Meirelles, Rafael Saran Arcieri, Katharina Messias, Andre Messias (2015) Macular dystrophy associated with Kjellin's syndrome: a case report. Arq Bras Oftalmol 78(2): 120-122.






























