Quality of Life among Post-Liver Transplant Recipients at a Private Tertiary Care Hospital, In Islamabad Pakistan
Shumaila Batool1*, Tazeen Saeed Ali2, Wardah Khalid3 and Faisal Saud Dar4
1Sr. Lecturer shifa College of Nursing Islamabad, Pakistan
2Assistant Dean, Research and Graduate Programs Aga Khan University Karachi, Pakistan
3PhD Scholar University of Edinburg, UK
4Chairman Hepatobiliary & Liver Transplant Surgery Pakistan Kidney and Liver Institute and Research Center Lahore, Pakistan
Submission: June 26, 2022; Published: July 07, 2022
*Corresponding author: Shumaila Batool, Sr. Lecturer shifa College of Nursing Islamabad, Pakistan
How to cite this article: Shumaila B, Tazeen Saeed A, Wardah K, Faisal S D. Quality of Life among Post-Liver Transplant Recipients at a Private Tertiary Care Hospital, In Islamabad Pakistan. JOJ Nurse Health Care. 2022; 12(3): 555840. DOI:10.19080/JOJNHC.2022.12.555840
Abstract
Background: Liver transplantation is the choice of treatment for patients with the End Stage Liver Disease, as it can save and extend their lives. The aim of the transplant procedure is not only to ensure the survival of the patients, but also to help individuals regain the same health which they had before the occurrence of the disease.
Material and Methods: An analytical cross-sectional study was conducted in a tertiary care hospital. Multiple linear regression was done for analysis, as the outcome variable, Quality of Life (QoL), was on a continuous scale. β coefficients with 95% C.I were accounted for critical affiliations.
Results: A total of 182 post liver transplant recipients, with a median age 47 (IQR 14) participated in the current study. The median QoL score of the post liver transplant participants was 200.5 (IQR 35). The results showed that co-morbid condition diabetes, more than one transplant indication, and older age negatively impacts the QoL of the post liver transplant recipients.
Conclusion: The current study identified some of the important factors that were associated with the QoL of the post liver transplant recipients. The findings of the current study suggest the need for the development of some new and more innovative strategies, so that the patients, after the liver transplant procedure, can resume their normal lives and have the health which they possessed before the occurrence of the disease.
Introduction
Liver transplant is the only beneficial and life-saving curative intervention for the End-Stage Liver Disease (ESLD). Pakistan already carries one of the highest burdens of ESLD, as it can be roughly estimated that about 10 million people are suffering from the infection of Hepatitis C Virus (HCV), which is a major cause of ESLD [1]. According to Dar et al. [2], ESLD due to Hepatitis C virus was the most common indication for liver transplantation in their retrospective study. They also indicated that out of 100 liver transplant recipients, 66% were Child-Pugh Class C. Thus, the combined burden of both hepatitis B and C viruses is about 7% in general, and 13% in high-risk population [3]. Therefore, because of its high prevalence, it can be estimated that half of a million people will develop liver disease and will be on the waiting list of liver transplant. Liver transplant is an extensive surgical procedure in which an unhealthy liver is removed and then replaced by a healthy liver from the donor [4]. Resultantly, after a risky procedure, one’s life can be saved, and the health-related quality of life (HRQoL) can be improved. In the past, the outcome of the liver transplant was only evaluated in terms of patients’ survival rate.
Statistical facts show that the liver transplant survival rates have appreciably improved in the past years, due to advancement in the medical treatment and by quality nursing care, in the first year of transplant by 90% and in the fifth year by 70% [5,6]. Improvement in the survival rate and graft functioning are not now the only focus, as the attention has moved now towards the other indicators to assess liver transplantation outcomes i.e., recurrence of an underlying cause, transplant-related complications, and the HRQoL of transplant recipients [7]. Therefore, the most expected outcome of a liver transplant intervention is the improvement in patients’ HRQoL. During the last 25 years, the concept of measuring HRQoL was only considered in terms of mortality and morbidity, which was not sufficient. HRQoL has multidimensional aspects, and it is difficult to define and quantify. But evaluating patients’ HRQoL has an important role in diseases’ management and need to be considered. Liver transplant is a new area in terms of recent advanced surgical procedure in the health care settings of Pakistan and there is a dire need to identify the physical, social, and psychological needs of the liver transplant recipients in their own context to adopt holistic care approach and to provide optimum quality care. Also, with the advancement in medical sciences, there is a great need for nurses to enhance their knowledge to meet the current demands. This study may help nurses to assess patients’ survival rate and HRQoL not only from the perspective of a successful liver transplant procedure, but also from the multidimensional aspect. Therefore, this study aims to answer the following questions.
i. What is the level of HRQoL in Post-Liver Transplant patients?
ii. What are the important associated factors of HRQoL in Post-Liver Transplant patients?
Material and Methods
Study Design
An analytical cross- sectional study design was adopted to answer the research questions. This is an effective study design where a researcher is inclined to demonstrate a relationship among the contributing factors and the phenomenon of interest, at a predetermined time [8].
Study Setting and Study Period
The study was conducted from March to September 2017 at the Gastroenterology and Hepatobiliary outpatient departments of the private tertiary care hospital in Islamabad, Pakistan. The hospital is a pioneer in conducting the first successful liver transplant surgical treatment in Pakistan and performing around 10-12 liver transplants for each month [9].
Study Population
The study population comprised men and women, more than 18 years of age, who had undergone liver transplant at the Private Tertiary Care Hospital, and who fulfilled the eligibility criteria, and were willing to give informed consent.
Eligibility Criteria
Inclusion Criteria
a. Adult (age ≥ 18 years of age) with single organ transplant.
b. Living donor liver transplantation cases.
c. Had undergone liver transplant more than ≥ 6 months ago.
d. Clinically stable, with normal vital signs, such as Heart Rate, Blood Pressure, Temperature, Respiratory Rate and Oxygen Saturation.
e. Cognitively able to understand and respond to the questionnaire.
Exclusion Criteria
i. Those who had received any other transplant previously.
ii. Who had a disease like, last stage cancer or were on palliative therapy and end-stage renal disease?
Data Collection Instrument
The disease specific post liver transplant QoL questionnaire (pLTQ) was used to assess the HRQoL of liver transplant recipients [10]. This was the first disease targeted questionnaire developed in the United States and was further validated on the Brazilian population, and it is recommended as a useful tool for measuring the HRQoL of post liver transplant recipients [11]. The 32-items questionnaire was developed after a methodological rigor, for patients who had undergone liver transplant [12]. It has eight domains: measure of physical function, emotional function, worry, medications, graft rejection concerns, pain, health care, and finances.
Reliability and Validity of the Instrument
Questionnaire was originally in the English language and was not validated in Pakistani context so, to ensure the validity of tool in Pakistani context Content Validity Context (CVI) was computed. Panel of experts included gastroenterologists, transplant surgeons, epidemiologists, biostatistician, and clinical expert nurses. They reviewed and rated the scale for its relevancy as well as for the clarity. Moreover, based on the experts’ scores, CVI was computed by dividing the number of scores judged by the expert panels by aggregated items in the scale [13,14]. CVI result for relevancy was 0.99 and for clarity 0.98 and which indicates harmony among the experts’ opinion [15]. Moreover, post analysis, the reliability of the scale was considered and cronbach`s-alpha was calculated and it came to be 0.94 and according to literature 0.70 or greater value of Cronbach`s-alpha is considered as good [16].
Translation and Pilot Testing of the Questionnaire
The contextual aspect was considered, and the questionnaire was translated and converted into the Urdu language through a language specialist and then back translated into English, considering the steps suggested by Polit and Beck [17]. No loss of meaning was detected in the back translation. Pretesting was performed on 10% of the total sample size in the Gastroenterology and Hepatobiliary outpatient departments of the Shifa International Hospital Islamabad, and no ambiguity was found in the questions and no question was eliminated from the questionnaire. Those participants who took part in the pilot testing were not included in the study sample size and in the analysis.
Sample Size
The calculated range of sample size was from 165 (minimum) to 387 (maximum). The sample size calculation was based on the prevalence of associated factors related to post liver transplant quality of life [18] with 95% CI, power 80%, and an assumed percentage of exposure among the controls of 40%. Moreover, with a hypothetical proportion of cases with exposures in the range of 60, an odds ratio of 2.25, and10% adjusted for the nonresponse rate and the sample size came to be 218. So, 218 was the final sample size for the current study. The sample size calculation was carried on Open Epi version 3.04. Thus, a sample of 182 participants was achieved from the calculated range within the specified period of the study.
Sampling Technique
The purposive sampling technique was applied to recruit participants. Patients were first identified through their medical record numbers. The list with contact numbers was collected from the patients’ record system, after approval from the Institutional Review Board (IRB Reference # 702-150-2016) of the hospital and the Ethical Review Committee (ERC Reference # 4626-SONERC- 17) of the university. Post-transplant recipients were approached through telephone and were ask for participation in the study. Those who fulfilled the study’s eligibility criteria and agreed for consent were included and invited for the interview. Their interviews were conducted in the Hepatobiliary outpatient departments of the hospital, at a time that was convenient and feasible for them. A room in the Gastroenterology and Hepatobiliary Outpatient Departments was designated specifically for the data collection, where a calm and relaxed environment was provided and throughout privacy and confidentiality of the participants were maintained. Questions were asked through the interview and questionnaire was filled by the primary data collector. Moreover, the transportation expenses of the participants were reimbursed from the fund given by the Aga Khan University Karachi, Pakistan.
Data Entry, Cleaning, and Data Management
Post coding of the data was performed by the principal investigator and then it was entered in the STATA VERSION 12. To check the error rate, 50 questionnaires were selected randomly picked and were counterchecked by the principal investigator on daily basis during the data entry period
Study Variables
Independent Variable
The data was collected on the following: demographics, socio-economic status, co-morbid conditions, transplant duration, transplant indication, immunosuppressant agents, and immunosuppressants’ frequency.
Outcome Variable
HRQoL was the outcome variable and was measured through post liver transplant disease specific HRQoL questionnaire.
Statistical Analysis
The descriptive analysis of quantitative and qualitative variables was done. Histograms were plotted for all the continuous variables to check their distribution. Moreover, Shapiro-Wilk test was also done for the assessment of the normality. For the normally distributed variables mean and standard deviations were reported. Furthermore, for the variables that were not following normal distribution median and interquartile ranges were calculated. For categorical data, frequencies and proportions were measured.
Multiple linear regression was done, as the outcome variable, HRQoL, was on a continuous scale. The scale examination of independent continuous variables was done by making scatter plots against the HRQoL scores to assess the linearity. Moreover, a histogram and a normal probability plot were also plotted to assess the normality of outcome variable.
Simple linear regression was carried out to calculate the unadjusted β-coefficient with 95% C.I. Each independent variable was regressed with the overall HRQoL scores, and p-value ≤ 0.25 was significant at the univariate level. To identify the predictors of HRQoL of post liver transplant recipients, a stepwise approach for model building was adopted to develop a multiple linear regression model and for the calculation of adjusted β-coefficients by keeping the CI 95%. All the clinically important co-variants, along with the variables which were significant (α=0.25) at the univariate level and were considered for the model building. All biologically plausible interactions were also checked, and none was found to be significant. After that, Multicollinearity was evaluated by using the scatter matrix, correlation coefficient, variance inflation factor (VIF), and tolerance (1/VIF), and no variable was found to be multicollinear. Before finalizing the model, outliers and influential observations were evaluated through the computation of Standardized Residuals, Cook’s Residuals, Leverages, and DFBETA.
Outliers were reported as around 6.04%. To deal with the outliers, robust regression was run. Since there was no major improvement in the model adjusted R2, after robust regression, it was decided to keep the original model as the final. Model adequacy was evaluated through the model adequacy plots, residuals were plotted against the fitted values and against the independent variables as well. The plots were distributed equally, and no heteroscedasticity was found. Normal probability plot was plotted, and it showed not following normal deviation. Hence, as the normality assumptions were not fulfilled, cubic transformation on the outcome variable was performed and model building was done again. Then model adequacy checks were performed to evaluate the assumptions. As no major improvement was identified in the NPP, so finally came to a decision to keep the outcome variable as in its original form
Ethical Considerations
The study proposal and the informed consent document were reviewed and then endorsed by the Ethical Review Committee (ERC) of the Aga Khan University, with ERC reference # 4626-SONERC- 17 and the Institutional Review Board (IRB) of the Shifa International Hospital, with IRB reference # 702-150-2016. Moreover, permission of the study tool was taken by the authorized departments. All the study participants were informed in detail about the aim and purpose of the study. The informed consent form was provided to the participants before the interview, and consents were taken. Participation of the participants was truly voluntary, and they had a right to withdraw from the study at any time or at any point of data collection. All the personal information and questionnaires were stored separately and secured in locked cupboards. The participants were guaranteed that the information from individuals will be published in an aggregated form.
Results
Socio-demographic characteristics
The demographic and socio-economic features of the post liver transplant recipients are presented in Table 1. A total of 201 study participants from the Gastroenterology and Hepatobiliary outpatient clinics of the Shifa International Hospital in Islamabad, Pakistan, were approached. Among them, 19 participants, i.e., 11 men and 8 women wished to withdraw from data collection. A total of 182 participants were, therefore, interviewed between March till June 2017. The median age of the post liver transplant recipients in years was 47 (IQR 14). Most of the participants (80.77%) were male, undergraduate (41.21%), living in a nuclear family (53.85%) having one to five adults (64.84%), one to two children (60.99%), and two caregivers (92.31%). Most of the participants were permanently employed (34.62%) and belongs to the middle socio-economic group (40.11%) (Table 1).
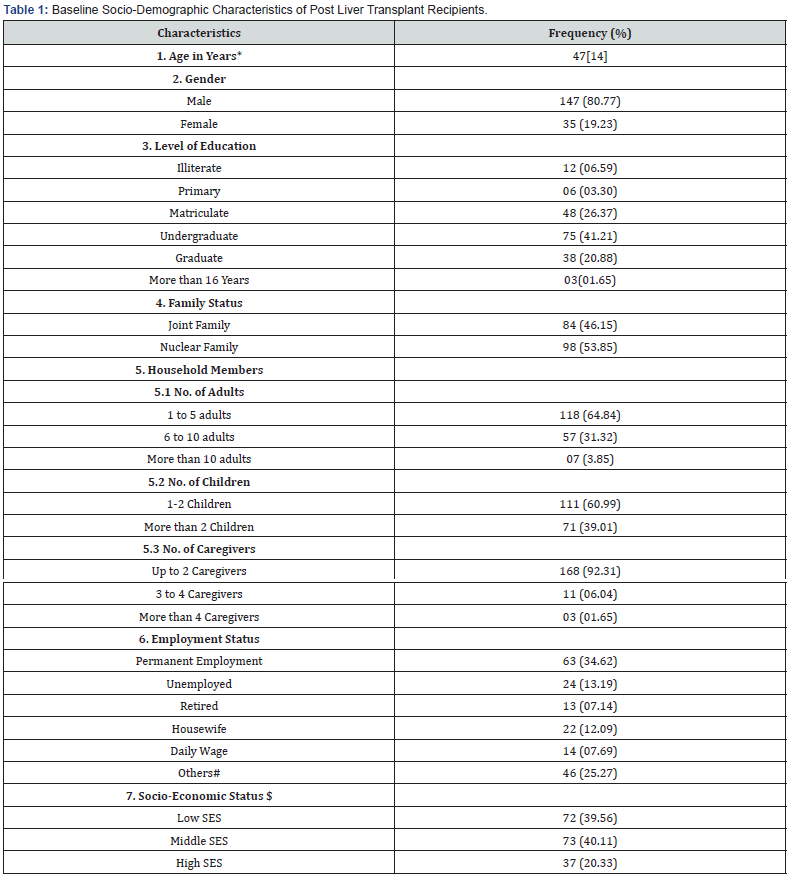
*Median [IQR].
#Others category in the employment status includes self-employed, private job, student, housewife, contractor, intern, the volunteer, peripatetic worker, part time worker, and own business
$ The Factor component analysis was performed to assess the socio-economic status.
Clinical Characteristics of the Participants
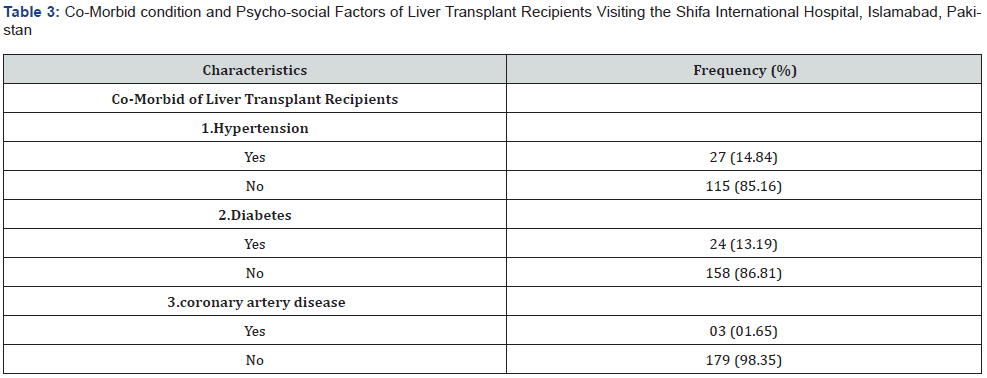
Clinical variables which were important and might influence the QoL of post liver transplant recipients, such as transplant indication, transplant duration, immunosuppressant agents, and frequency of immunosuppressant agents, were also assessed, as shown in Table 2. The Hepatitis C virus infection (62.09%) was the most common indication for liver transplantation. The mean time since transplantation, in months, was 24.34±13.60. Most of the participants (81.87%) were taking Tacrolimus (43.41 %,) as an immunosuppressant agent. Around 14.84% were hypertensive, 13.19% were diabetic, and 13.19% were suffering from coronary artery disease (Tables 2 & 3).
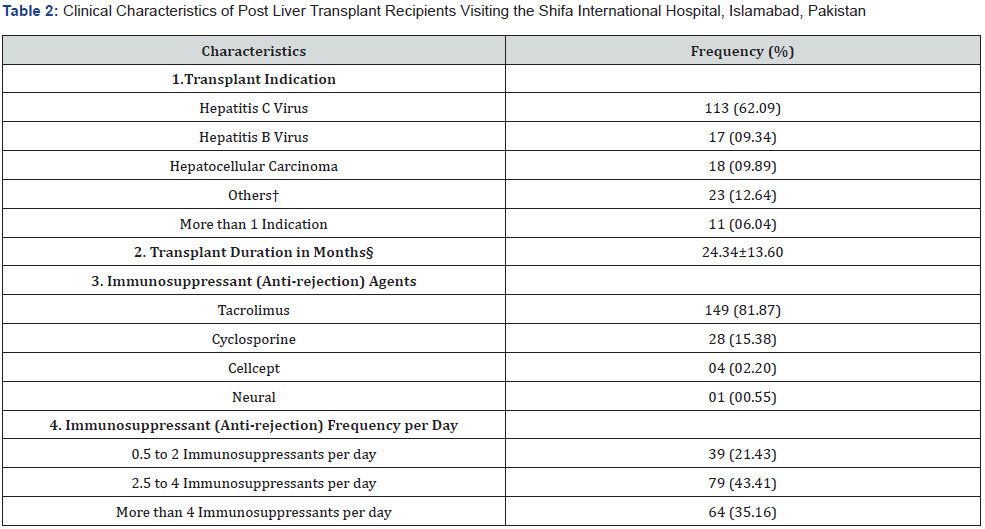
§Mean (SD)
†The others category in Transplant Indication includes Alcoholic Cirrhosis, Cryptogenic Cirrhosis, Non-Alcoholic Fatty Liver Disease, Primary Sclerosing Cholangitis or Primary Biliary Sclerosis, and Fulminant Hepatic Failure
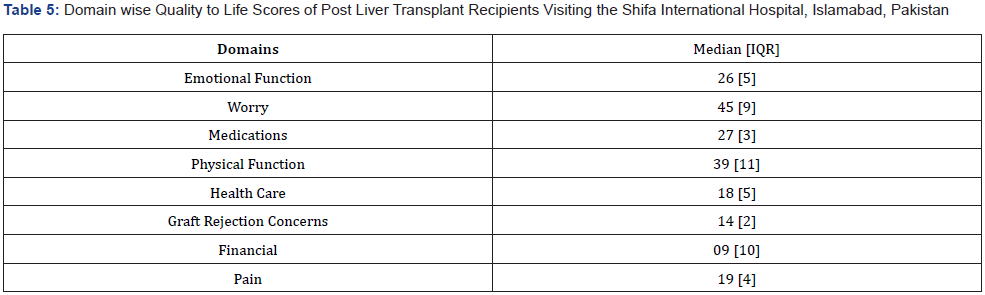
Health Related Quality of Life Score
The median QoL score of the post liver transplant participants was 200.5 (IQR 35) shown in Table 4. Table 5 presents the results of the HRQoL of post liver transplant recipients from the perspective of different domains. 45 [9] was the highest median score in the domain of worry and 9 [10] was the lowest median score which was reported in the domain of concerns about graft rejection (Table 5).

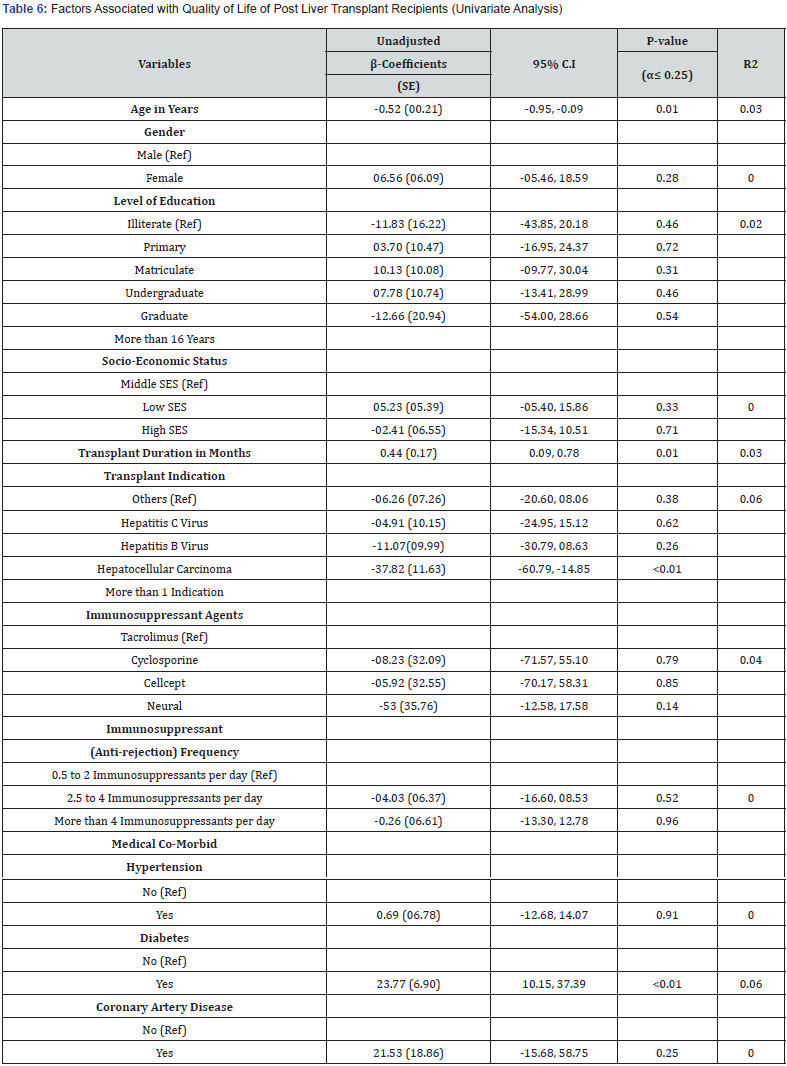
Simple Linear Regression
Table 6 presents the simple linear regression results of all the independent variables. The results of the simple linear regression show that increasing age, more than 16 years of education level, and high socio-economic group were inversely related with the HRQoL of the post liver transplant recipients. Whereas it was observed that female gender and low socio-economic group positively associated with the HRQoL. Moreover, transplant indications, immunosuppressant agents, and frequency of immunosuppressant agents per day have a negative impact on HRQoL. More time since transplantation, diabetes, and cardiovascular diseases as co-morbid conditions were positively associated with HRQoL (Table 6).
Multiple Linear Regression
Multivariable analysis is shown in Table 7. All the clinically important co-variants, along with the variables which were significant at the univariate level, were added in the model. The findings of the current study suggest young age recipients had better HRQoL by -0.38(95% C.I; -0.77, 0.02). Moreover, model suggest that those patients who had more than one transplant indication had a significantly poorer HRQoL by -35(95% C.I; -109.89, -33.09). It was identified that Diabetes as a co-morbid condition had a negative impact on the HRQoL by -14.34 (95% C. I; -26.80, -01.88). The final model signified that 32% (Adj. R2= 0.3284) of the variability in the HRQoL of post liver transplant recipients explained by age, transplant duration, transplant indication, and co-morbid (Diabetes) (Table 7).

The model was adjusted for cardiovascular diseases
Overall P-value of model is <0.01
Adjusted R2=0.32
Discussion
HRQoL is a multifaceted concept; however, the discussion focused on the main area of HRQoL and focuses on the different factors which affect the HRQoL of post liver transplant recipients. The current study identified different factors which were associated with HRQoL. The data showed that the age, gender, more than one transplant indication and among co-morbid condition diabetes, were the significant factors associated with the post liver transplant HRQoL. Current study suggested that the young age patients had better HRQoL after liver transplantation. The effect of a recipient’s age on the outcomes of liver transplantation has been examined in the past years and different studies come up with different conflicting results. A study of a recent era on the outcomes of liver transplantation suggests that the recipient’s age significantly affects the graft survival; the findings indicate that the long-term effects of the graft could be more beneficial in the younger age as compared to the older age [19].
Similar findings were reported in another study, in which researchers indicated the association of reduced HRQoL with the older age [20]. The findings of the current study also highlighted that the there is no gender wise differences in the HRQoL scores and this finding conflicts with the other studies. Alkatheri et al. [21] reported that male liver transplant recipients reliably revealed higher scores in all HRQoL domains as compared to the female recipients. The same results have also been reported in another study, in which HRQoL was measured by the Medical Outcomes Study Short Form-36, and in which women scored lower in all domains than the men, which indicates that gender is a potential factor which significantly affects HRQoL after liver transplantation [22]. Moreover, the current study reported 69.04% hepatitis C as an etiology of the liver transplantation. Similar findings were obtained from another study, which evaluated the sustainability of HRQoL by monitoring recipients according to their diagnosis. They found that the patients with the hepatitis C along with any other cause had a consistent decline in their health related HRQoL, with significant worsening in their physical and mental functioning.
Furthermore, the current study findings are consistent with the results of Saab et al. [23] in which the same disease specific HRQoL tool was used as the one adopted for this study. Their result showed that the patients with the indication of hepatitis C had lower HRQoL as compared to the patients who underwent transplant with some other cause. Co-morbid conditions are an important determinant of health outcomes in any medical or surgical condition. The current study also revealed type II Diabetes as a co-morbid condition that negatively impacted HRQoL. Similarly, a study from the liver transplant centers in England identified that the most common combinations of co-morbidities among transplant patients were identified with diabetes, i.e., diabetes with cardiovascular disease, diabetes with renal disease, and diabetes with pulmonary disease. Moreover, they established the fact that diabetes was the most common co- morbid among the liver transplant recipients, with a significant impact on posttransplant mortality [24].
Additionally, these findings were like the findings of another study; also highlighted that, post operatively, liver transplant patients suffered from different metabolic disorders, and amongst them diabetes was the most common with its negative impacts on the HRQoL. Along with this, they also pointed out that the post liver transplant patients with diabetes frequently experienced more fungal, viral, and bacterial infections. Similarly, steroid resistance and episodes of rejection were the most significant risk factors among liver recipients with diabetes [25].
Strengths of the Study
The current study has several strengths, which are as follows.
To the best of the researcher’s knowledge, this is the first study in Pakistan on the HRQoL and its associated factors, among liver transplant recipients. This study projected to seek a relationship between HRQoL and its associated factors, so the cross-sectional study design was found to be appropriate in this regard. The study site being first liver transplant center catering majority of the patients, so it can be assumed that the population covered in this study was from almost all regions of Pakistan. Moreover, robust measures of internal validity were ensured throughout the study.
Limitations of the Study
The study has some limitations which should be acknowledged to guide the interpretations of its results. The change in HRQoL could not be assessed from the longitudinal perspective due to the time limitations of the academic research. Time limitation also affected the sample size; the highest estimated sample size of the study could not be achieved which might have affected the power and internal validity of the study. The study was limited to only one setting, so the generalizability of findings is restricted.
Recommendations of the Study
HRQoL has become an important and frequent expression in liver transplant outcomes because of the patients’ long-term dependency on clinics for treatment and care. So, the evaluation of HRQoL is crucial due to its long-lasting treatment procedures. Thus, to rehabilitate the patients transplant nurses and physicians need to have a holistic approach towards the restoration of HRQoL in patients. Thus, nursing care plans and post-transplant discharge teachings should be more specific and targeted. Based on the findings of current study it is recommended that, elderly transplant recipients, and patients with more than one transplant indications and those who are known case of diabetes require more attention from nurses and doctors. Moreover, establishment of Tele-Health services in transplant hospitals can be helpful for providing guidance and assisting recovery.
Future longitudinal studies can be conducted to evaluate the effects of different factors on the HRQoL in the Pakistani context. Moreover, there is also a need for studies on the role of informal caregivers, because before, during, and after liver transplantation, patients remain in the care of their family members and the role of family members suddenly shifts and they become caregivers. Also, prospective studies are needed to investigate customized interventions to help liver transplant recipients to overcome their physical and psychological discomforts, and additionally social inconveniences. Additionally, more studies may be conducted to further validate the study tools employed in this study.
Conclusion
In conclusion, the current study has identified that liver transplant recipients of Pakistan are maintaining their HRQoL near to the maximum level of scores. Moreover, current study also identified some factors which may impact on HRQoL such transplantation in old age, having more than one transplant indication and known case of diabetes. Moreover, some recommendations are proposed to address these factors.
References
- Umer M, Iqbal M. (2016) Hepatitis C virus prevalence and genotype distribution in Pakistan: Comprehensive review of recent data. World J Gastroenterol 22(4): 1684–1700.
- Dar FS, Bhatti AB, Dogar AW, Zia H, Amin S, et al. (2015) The travails of setting up a living donor liver transplant program: experience from Pakistan and lessons learned. Liver Transpl 21(7): 982-990.
- Dar FS, Bhatti AB, Dogar AW, Zia H, Amin S, et al. (2015) The travails of setting up a living donor liver transplant program: experience from Pakistan and lessons learned. Liver Transpl 21(7): 982-990.
- Starzl TE, Fung JJ. (2010) Themes of liver transplantation. Hepatology Jun 51(6): 1869-1884.
- Tome S, Wells JT, Said A, Lucey MR. (2008) Quality of life after liver transplantation. A systematic review. J Hepatol 48(4): 567-577.
- Braun F, Teren K, Wilms P, Günther R, Allmann J,et al (2009)Quality of life after liver transplantation. InTransplantation proceedings 41 6, pp. 2564-2566.
- Desai R, Jamieson NV, Gimson AE, Watson CJ, Gibbs P, et al. (2008) Quality of life up to 30 years following liver transplantation. Liver Transpl 14(10): 1473-1479.
- Polit DF, Beck CT (2008) Nursing research: Generating and assessing evidence for nursing practice. Lippincott Williams & Wilkins.
- Bhatti AB, Zia H, Dar FS, Zia MQ, Nasir A, et al. (2015) Quality of life after living donor hepatectomy for liver transplantation. World journal of surgery 39(9): 2300-2305.
- Saab S, Ng V, Landaverde C, Lee SJ, Comulada WS (2011) Development of a disease‐specific questionnaire to measure health‐related quality of life in liver transplant recipients. Liver Transpl 17(5): 567-579.
- Molski C, Mattiello R, Sarria EE, Saab S, Medeiros R, et al. (2016) Cultural validation of the post-Liver transplant quality of life (pLTQ) questionnaire for the Brazilian population. Annals of hepatology 15(3): 377-385.
- Saab S, Ng V, Landaverde C, Lee SJ, Comulada WS (2011) Development of a disease‐specific questionnaire to measure health‐related quality of life in liver transplant recipients. Liver Transpl 17(5): 567-579.
- Polit DF, Beck CT (2006) The content validity index: are you sure you know what's being reported? Critique and recommendations. Research in nursing & health 29(5): 489-497.
- Khalid W, Rozi S, Ali TS, Azam I, Mullen MT, et al. (2016) Quality of life of stroke survivors and their primary caregivers in Pakistan 11(3): 34-35.
- Bolarinwa OA. (2015) Principles and methods of validity and reliability testing of questionnaires used in social and health science researches. Niger Postgrad Med J 22(4): 195.
- Khalid W, Rozi S, Ali TS, Azam I, Mullen MT, et al. (2016) Quality of life of stroke survivors and their primary caregivers in Pakistan 11(3): 34-35.
- Polit DF, Beck CT (2008) Nursing research: Generating and assessing evidence for nursing practice. Lippincott Williams & Wilkins.
- Chen PX, Yan LN, Wang WT (2012) Health-related quality of life of 256 recipients after liver transplantation. World J Gastroenterol 18(36): 5114.
- Selzner N, Renner EL, Selzner M, Adeyi O, Kashfi A, et al. (2009) Antiviral treatment of recurrent hepatitis C after liver transplantation: predictors of response and long-term outcome. Transplantation 88(10): 1214-1221.
- Alkatheri A, Al Bekairy A, Aburuz S, Qandil A, Khalidi N, et al. (2015) Exploring quality of life among renal and liver transplant recipients. Annals of Saudi medicine 35(5): 368-376.
- Alkatheri A, Al Bekairy A, Aburuz S, Qandil A, Khalidi N, et al (2015) Exploring quality of life among renal and liver transplant recipients. Annals of Saudi medicine 35(5): 368-376.
- Jin S, Yan L, Li B, Wen T, Zhao J (2010) Quality of life and psychologic distress of recipients after adult living-donor liver transplantation (LDLT)—a study from mainland China. In Transplant proc 42(7): 2611-2616.
- Saab S, Ng V, Landaverde C, Lee SJ, Comulada WS (2011) Development of a disease‐specific questionnaire to measure health‐related quality of life in liver transplant recipients. Liver Transpl 17(5): 567-579.
- Tovikkai C, Charman SC, Praseedom RK, Gimson AE, van der Meulen J (2015) Time-varying impact of comorbidities on mortality after liver transplantation: a national cohort study using linked clinical and administrative data. BMJ open 1: 5(5).
- Stilley CS, Bender CM, Dunbar-Jacob J, Sereika S, Ryan CM (2010) The impact of cognitive function on medication management: three studies. Health Psychol 29(1): 50.






























