Indices of Oxidative Stress in Egyptian Children Infected by Helicobacter Pylori
Hasan Tarek Ebrahim1* and Eman Gamal El-Behery2
1Department of Pediatrics, Benha Teaching Hospital, Qalubia Governate, Egypt
2Department of Clinical Pathology, Benha University, Egypt
Submission: September 16, 2017; Published: September 25, 2017
*Corresponding author: Hasan Tarek Ebrahim,Department of Pediatrics,Benha Teaching Hospital,Qalubia Governate, Egypt, Email: mgamsy@gmail.com
How to cite this article: Ebrahim HT, El-Behery EG. Indices of Oxidative Stress in Egyptian Children Infected by Helicobacter Pylori. JOJ Nurse Health Care. 2017; 4(4): 555643. DOI: 10.19080/JOJNHC.2017.04.555643
Abstract
Background: The mucosal oxidative stress could be considered as an important mechanism in the pathology of Helicobacter pylori (H.pylori) infection in the stomach.
Aim of the work: Aim of the work is to assess the levels of serum and gastric mucosal oxidative stress markers of H. pylori-infected children.
Materials and methods: This study was conducted on 45 children infected with H. pylori from Pediatric Department of Benha Teaching Hospital. 45 children, age and sex-matched were chosen as controls. All children were subjected to history taking, anthropometric measurements. Laboratory investigations (complete urine and stool analysis, Complete blood count (CBC), routine liver and renal function tests), Upper Gastrointestinal Endoscopy with antral gastric mucosal biopsies which were submitted to rapid unease test, measurement of serum and gastric mucosal oxidative stress markers including tissue Malondialdehyde(MDA), superoxide dismutase (SOD), reduced glutathione (GSH), nitric oxide (NO), and catalyse.
Results: The serum and gastric mucosal levels of SOD and catalane are significantly higher in patients than in controls, while serum and gastric mucosal levels of NO and GSH are significantly lower in patients than controls. There was significantly higher gastric mucosal level of superoxide dismutase in the studied females as compared with males. There was a significant positive correlation between duration of abdominal pain and both gastric mucosal catalyses and reduced glutathione tissue levels in our studied patient group. There was a significant inverse correlation between serum reduced glutathione and serum catalyses levels in our studied patients.
Conclusion: The oxidative stress response to H. pylori infection is a mechanism involved in H. pylori infection in children
Keywords: Oxidative stress; Indices; Helicobacter pylori; Egyptian children
Introduction
Background
The global human population gets infected by H.pylori as early as in childhood and adolescence [1]. It may causes chronic gastritis, peptic ulcer disease, an increased risk of or gastric mucosa-associated lymphoid tissue (MALT) lymphoma or gastric adenocarcinoma [2]. Recently, reactive species (oxygen or nitrogen) are considered factors that influence gastro duodenal mucosal changes in H.pylori-infected patients [3]. Create a conducive environment to DNA damage leading to genetic instability and eventually neoplastic transformation [4]. Variable levels of mean serum NO, SOD, and GSH were reported in H.pylori-infected adult patients [5]. However, there is paucity of studies regarding this issue in the paediatric population.
The aim of the work
The aim of the work is to assess the levels of oxidative stress markers in the gastric mucosa of H. pylori-infected children.
Material and Methods
Design of the study and setting
This study was carried out upon 45 children infected with Helicobacter pylori with their ages ranged from 7-15 years with a mean value of 11±2.65 years who were attending the Pediatric Department of Benha Teaching Hospital in the period from July 2016 to July 2017. Forty five children, age and sex-matched with no evidence of H pylori infection were chosen and served as controls. The study was conducted after approval from the ethical committee of the Faculty of Medicine, University and informed written parental consents.
Inclusion criteria: Children with recurrent abdominal pain, vomiting, or dyspepsia.
Exclusion criteria: Children with conditions associated with systemic oxidative stress e.g. bronchial asthma, liver disease, renal disease, inflammatory bowel disease, or rheumatoid arthritis. Children receiving antibiotics, antacids, proton pump inhibitors, non-steroidal anti-inflammatory (NSAI) drugs, or corticosteroids within two weeks of the start of the study. Active gastrointestinal bleeding.
All children were subjected to: Full history taking, thorough clinical examination especially anthropometric measurements (weight and height).
a) Laboratory investigations: Including complete urine and stool analysis, Complete blood count including hemoglobin level, total leucocyte and platelet counts, Liver function tests including serum bilirubin, alanine aminotransferase, aspartate aminotransferase and albumin and renal function tests include blood urea and creatinine.
b) Upper Gastrointestinal Endoscopy: This was done for all patients with taking multiple gastric mucosal biopsies from the antrum of stomach. Biopsy specimens were submitted to rapid urease test for diagnosis of H pylori infection [6,7].
c) Measurement of oxidative stress markers(in serum and in gastric mucosal biopsy) which include:
A. Estimation of serum and gastric mucosal tissue Malondialdehyde (MDA) [8]
B. Estimation of serum and gastric mucosal tissue superoxide dismutase (SOD) [9]
C. Estimation of serum and gastric mucosal tissue reduced glutathione (GSH) [10]
D. Estimation of serum and gastric mucosal tissue Nitric Oxide (NO) [11]
E. Estimation of serum and gastric mucosal tissue catalyse [12]
Statistical analysis
The collected data were tabulated and statistically analyzed using Statistical Package for the Social Sciences (SPSS) software, version 17. For quantitative data, the range, mean and standard deviation were calculated. For qualitative data, comparison between two groups and more was done using Chi-square test ( 2). For comparison between means of two groups of parametric data of independent samples, Student t-test was used. Univariate parametric analysis of variance was used for comparison between more than two means of more than two different groups (F) value of analysis of variance (ANOVA). Significance was adopted at p<0.05 for interpretation of results of tests of significance [13].
2). For comparison between means of two groups of parametric data of independent samples, Student t-test was used. Univariate parametric analysis of variance was used for comparison between more than two means of more than two different groups (F) value of analysis of variance (ANOVA). Significance was adopted at p<0.05 for interpretation of results of tests of significance [13].
Results
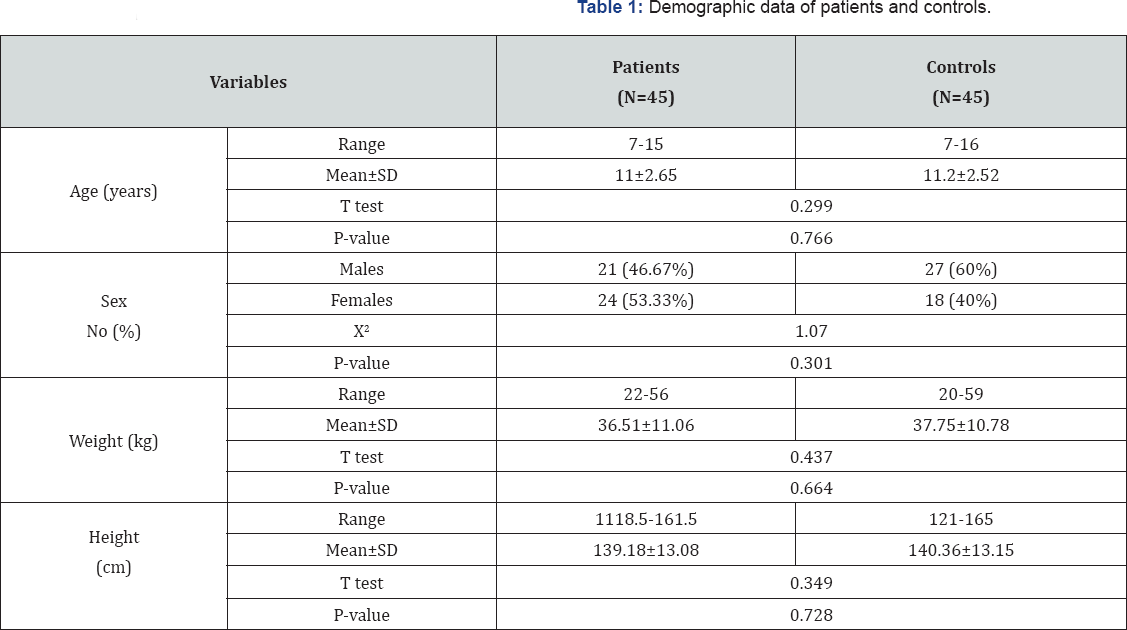
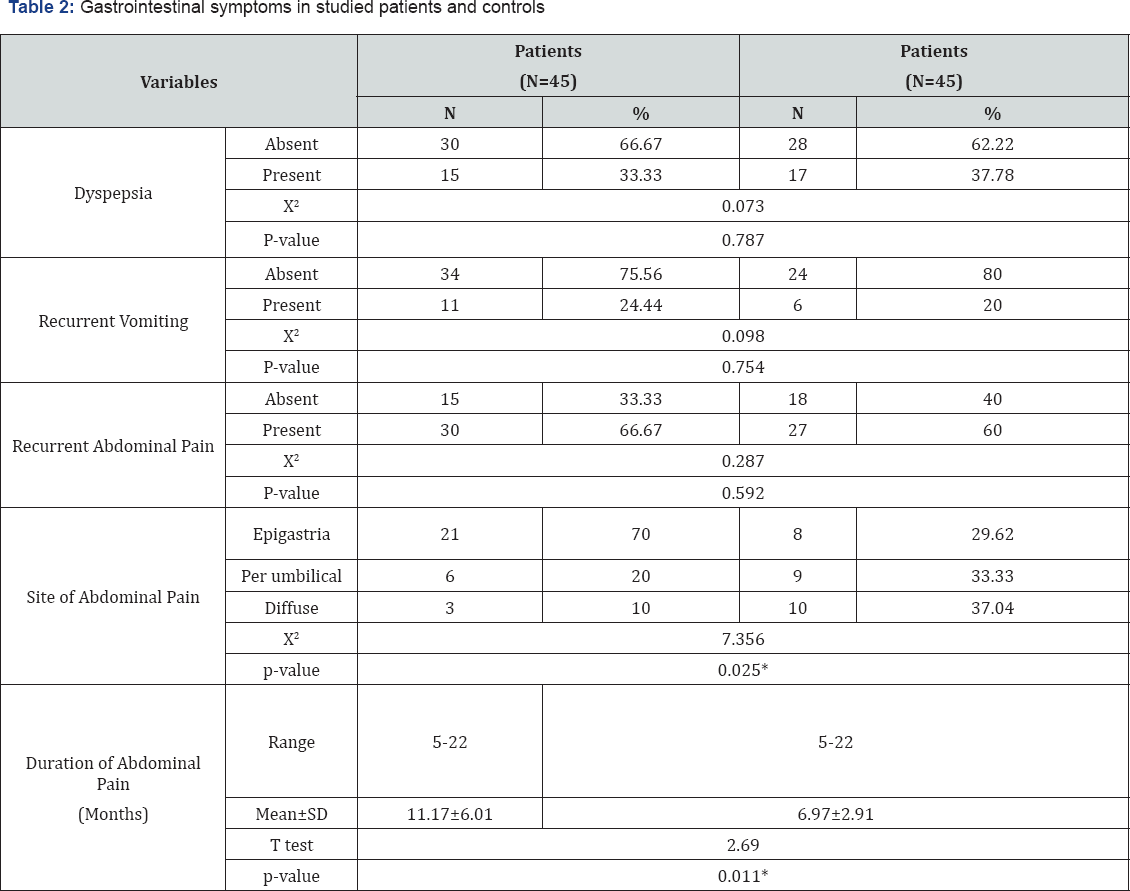
Table 1 summarize demographic data of studied patients and controls, there was no significant difference between patients and controls as regard age, sex, weight and height. Table 2 summarize gastrointestinal manifestations of studied patients and controls there was no significant difference between patients and controls as regard dyspepsia, recurrent vomiting, or recurrent abdominal pain but there was a significant increase in duration of abdominal pain in patients when compared to controls (p>0.05). The epigastriums were the commonest site of abdominal pain in the studied patients (70%), followed by per umbilical (20%) and diffuse (10%) abdominal pain.
*significant
ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase.
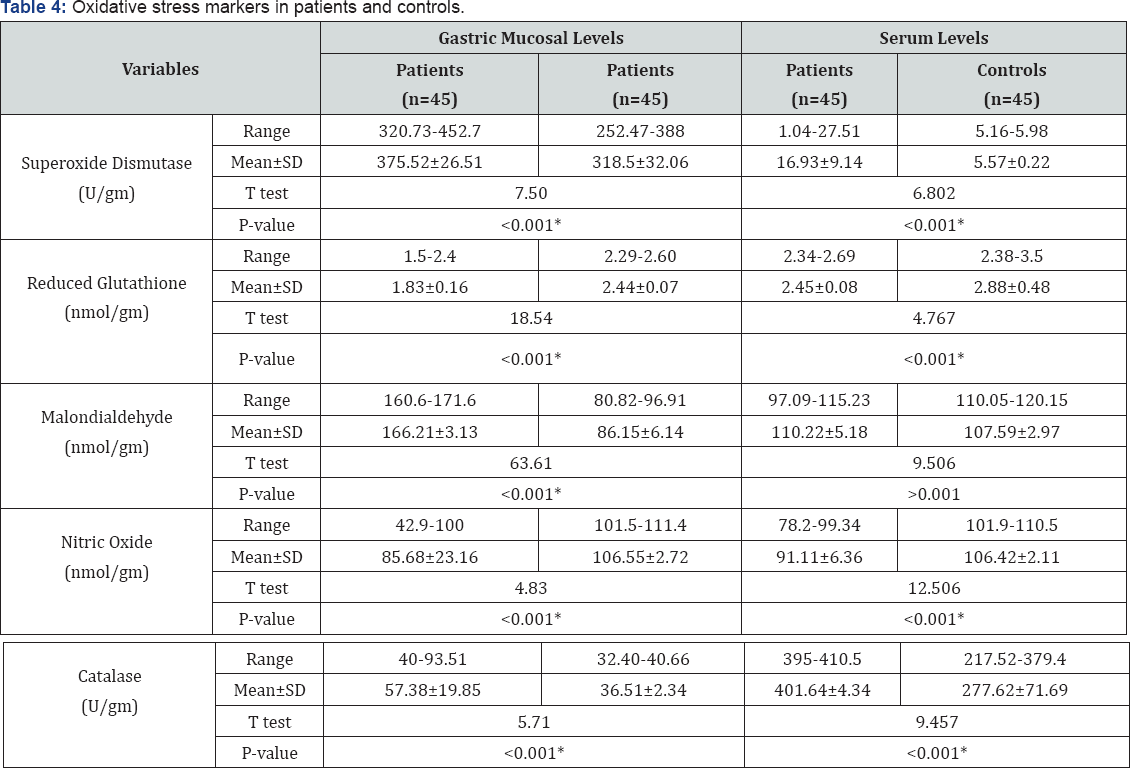
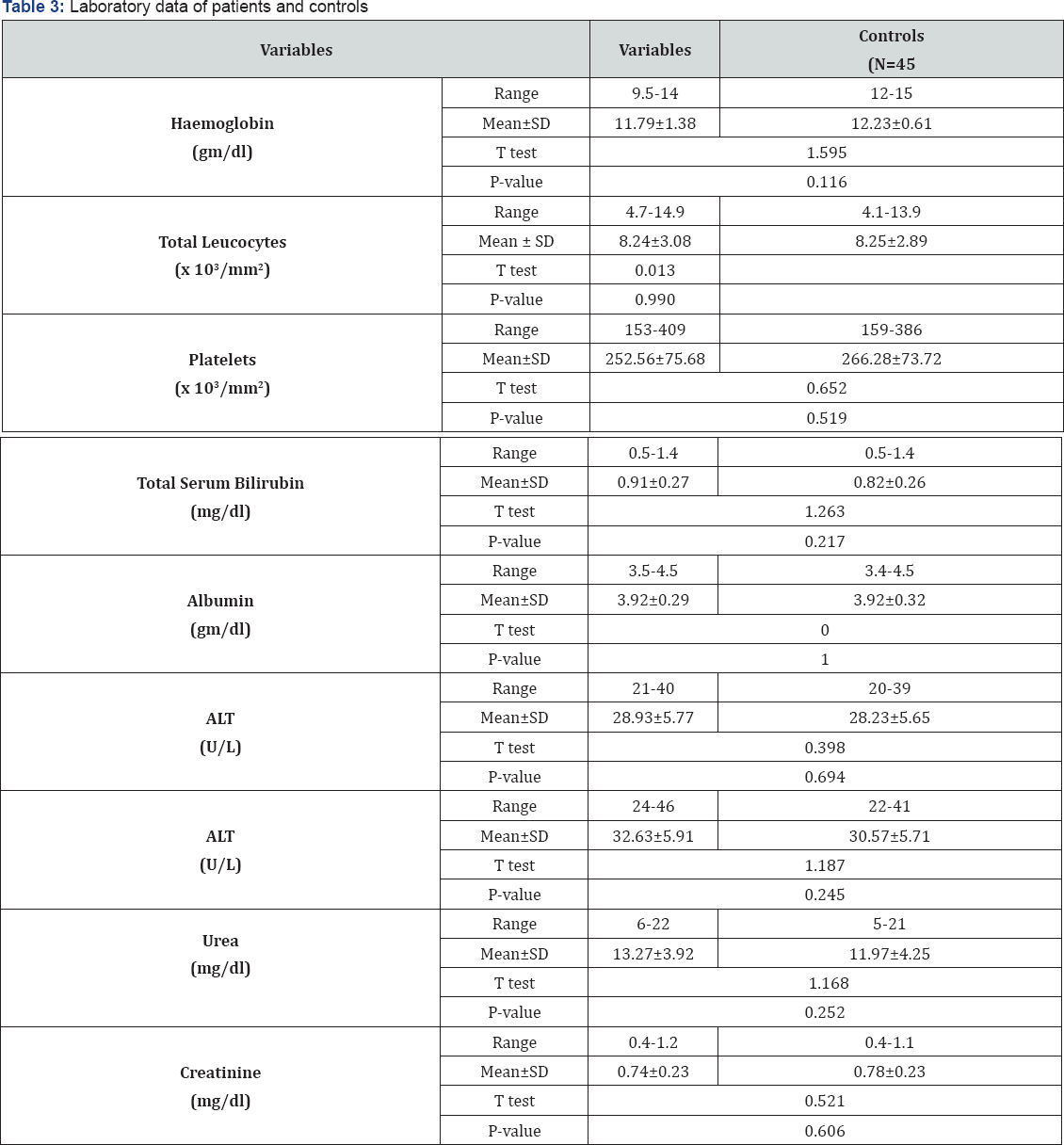
Table 3 summarize laboratory data of studied patients and controls there was no significant difference between patients and controls as regard haemoglobin %, total leucocytic count, platelets count, serum creatinine, urea levels, total serum bilirubin, alanine aminotransferase, aspartate aminotransferase, serum albumin levels. Table 4 shows comparison between patients and controls as regard serum and gastric mucosal oxidative stress markers, the serum and gastric mucosal levels of SOD, and catalyse are significantly higher in patients than in controls, the gastric mucosal level of MDA are significantly higher in patients than in controls. The present study demonstrates comparable serum Malondialdehyde concentrations in H. pylori -infected patients and control subjects. While serum and gastric mucosal levels of NO and GSH are significantly lower in patients than controls (p<0.05). There was significantly higher gastric mucosal level of SOD in the studied females as compared with males (Table 5).
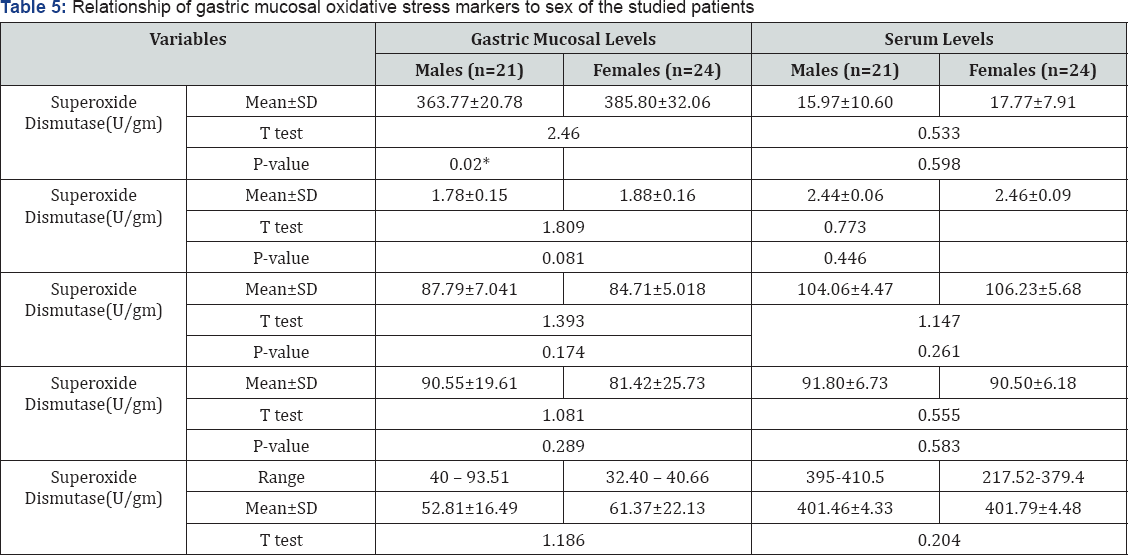
*significant
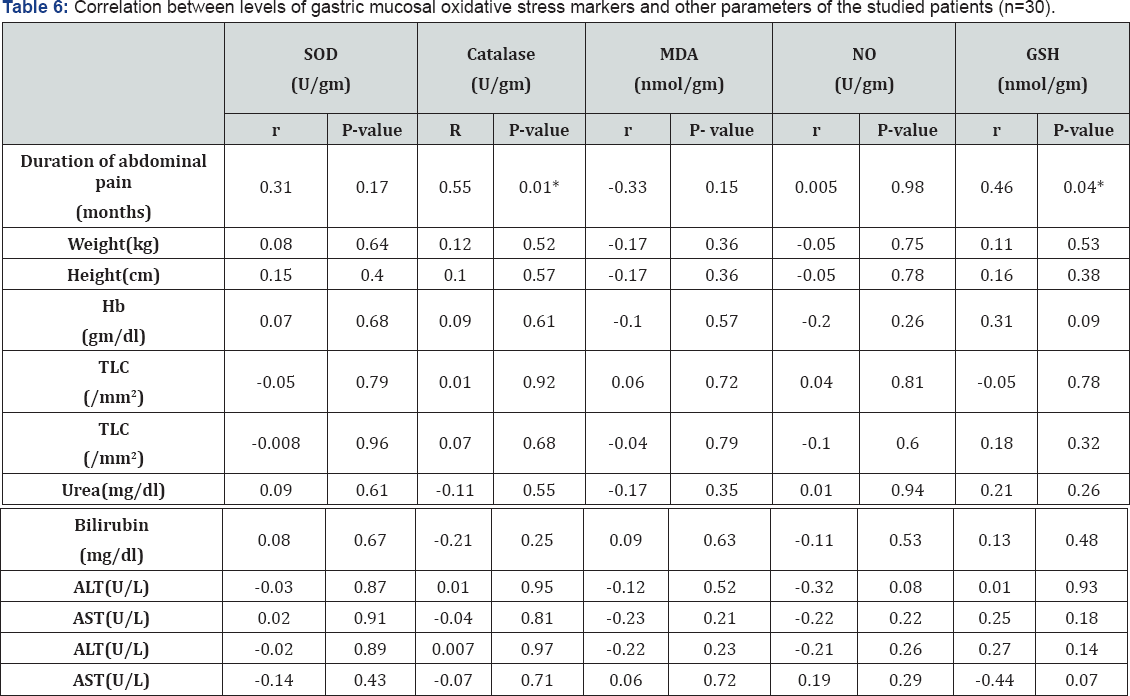
P-value: Patients Vs Control; SOD: Superoxide Dismutase; MDA: Malondialdehyde; NO: Nitric Oxide; GSH: Reduced Glutathione; Hb: Hemoglobin; TLC: Total Leucocytic Count; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase.
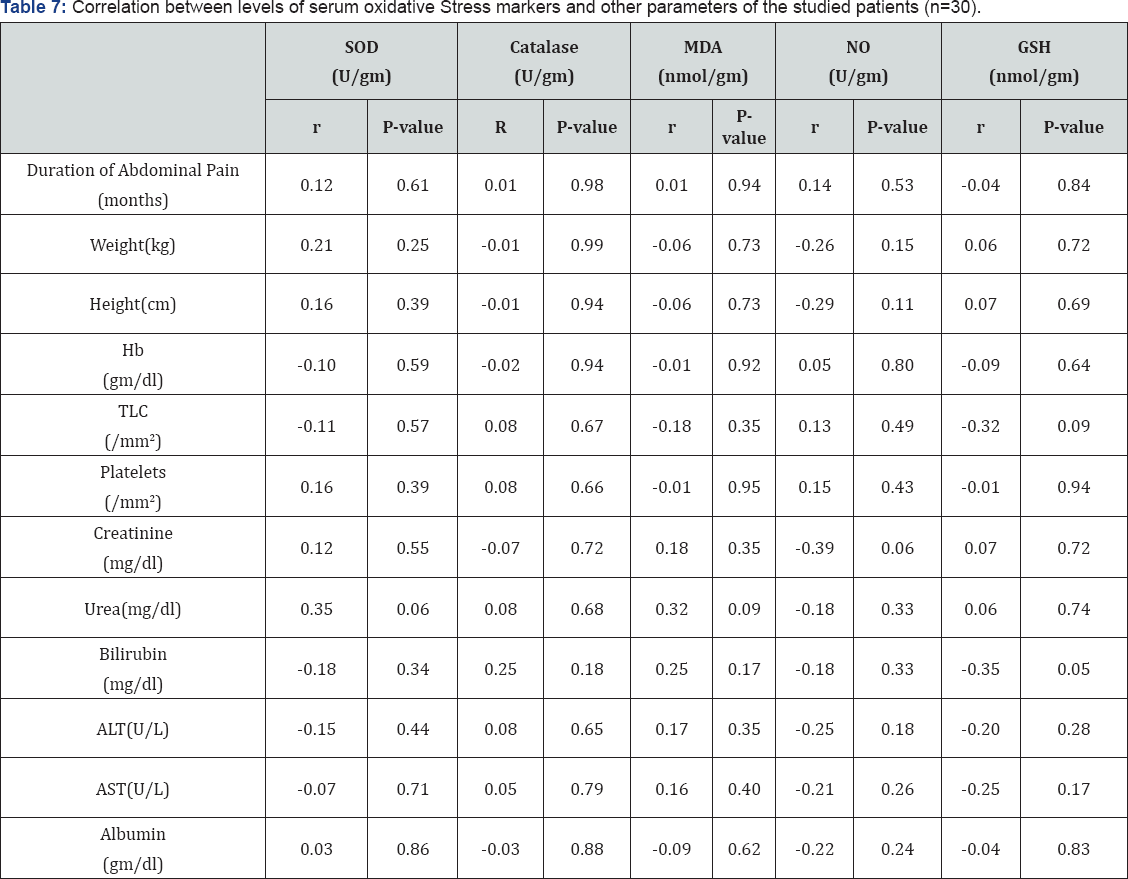
*SOD: Superoxide Dismutase; MDA: Malondialdehyde; NO: Nitric Oxide; GSH: Reduced Glutathione; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase.
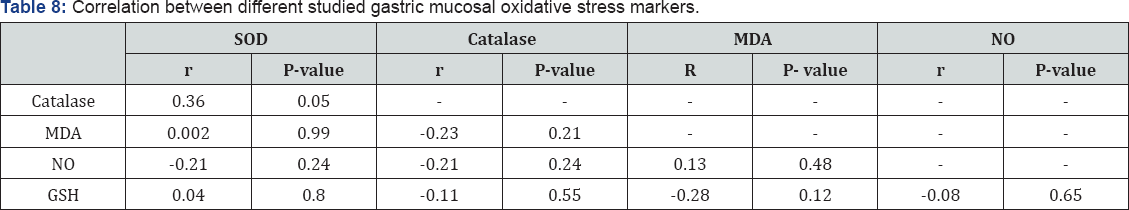
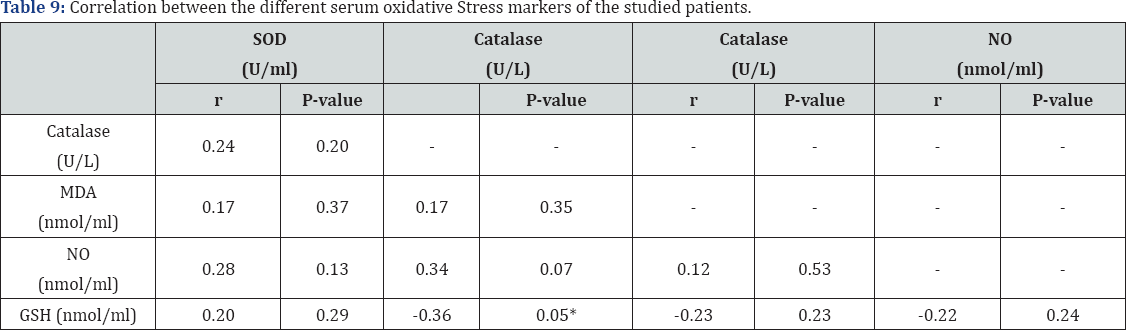
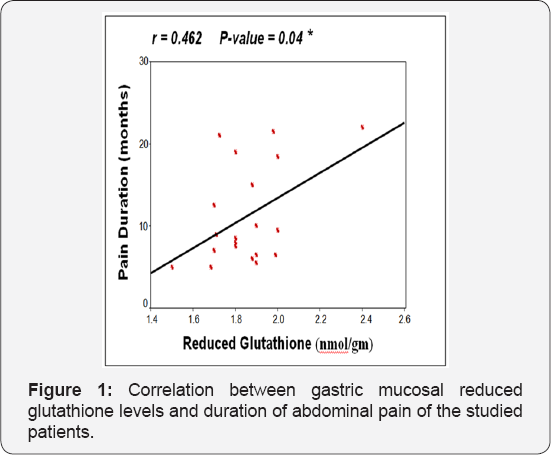
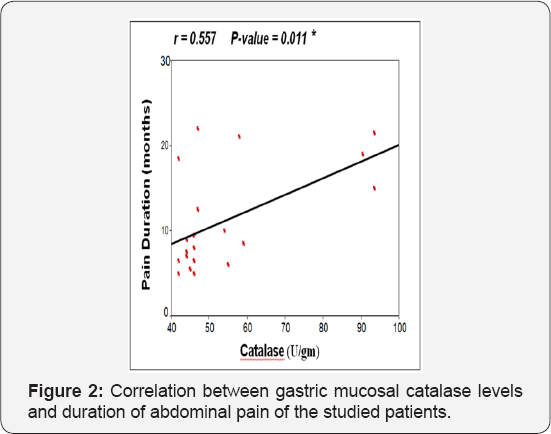
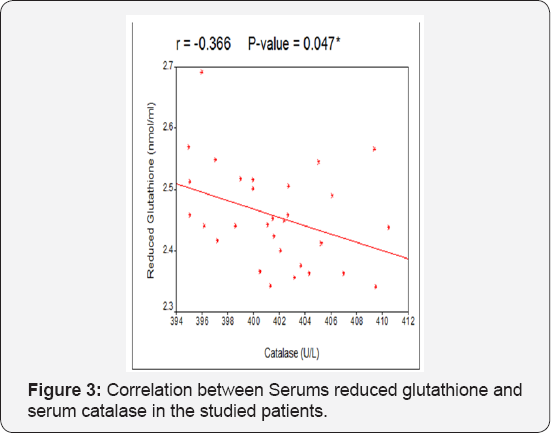
Table 6-9 summarized correlations between the studied serum and gastric mucosal oxidative stress markers and different studied parameters and correlations between each others, there was a significant positive correlation between duration of abdominal pain and both gastric mucosal catalyse and GSH tissue levels (Figure 1 & 2) and there was a significant negative correlation between serum catalyse and serum reduced glutathione the studied patients (Figure 3).
Discussion
Enhanced reactive oxygen species (ROS) levels due to neutrophil infiltration in H.pylori infected patients as well as increased oxidative DNA damage in H.pylori infected mucosa has been reported [14]. Superoxide radical anion [O2--] produced by infiltrating neutrophil reacts with cellular membrane lipids leading to the formation of lipid peroxides that are metabolized to Malondialdehyde (MDA) [15]. Further, the intimate interaction between the highly virulent H. pylori with host epithelium could be another mitigating factor for increased flares and higher levels of MDA and nitric oxide (NO) [16].
In our study, there was no significant difference between H.pylori infected children and controls as regard to age. Mahmud et al. [17] who showed that although two thirds of the children infected with H.pylori belong to the age group of 5-10 years, there was no significant relationship between the age groups and H. pylori infection. Malaty et al. [18] found that the majority of children get infected with H. pylori before age of 10 years. An Egyptian study concluded that H pylori infection is alarmingly high among school children aged 6-15 years, with an overall prevalence of 72 % [19]. While Mohran et al. [20] demonstrated prevalence of 46% in their study using endoscopy on 2-17 years old Egyptian children.
In our study, there was no significant difference between H.pylori infected children and controls as regard to sex. Several previous studies [21,22] have not been identified sex as a relevant influencing characteristic for H. pylori infection in their studies. On other side, Bin Mohanna et al. [23] revealed that females were more affected than males by H.pylori infection in their study. Naji et al. [24] found that higher level of H.pylori infection was observed among female patients in both blood antibody and stool antigen test methods (76.4% and 50%) rather than males (68.7% and 43.7%) respectively.
Females were also reported with higher prevalence of H. pylori infection than males in a study conducted about seroprevalence of H.pylori infection in large series of patients in an urban area of Saudi Arabia [25]. Hestvik et al. [26] found a significant increase in the prevalence of H. pylori colonization in boys more often than girls. These differences may be attributed to difference in sample size in each study. As regards anthropometric measurements (weight and height) of children in the present work, there was no significant difference between H.pylori-infected and non-infected children. Gokce et al. [27] results were in consistence to our results. In contrast to the present work, the study done by Pehlivanoglu et al. [28] suggested that H.pylori was associated with growth failure and short stature.
The present study showed that two thirds of H.pylori infected patients presented with recurrent abdominal pain. This finding is in agreement with study of Das B et al. [29], Liberato et al. [30] and Hasosah et al. [31] In contrast, Bode et al. [32] identified only 22% of their studied children having recurrent abdominal pain.
The epigastria area was the commonest site of abdominal pain in our studied H.pylori infected children. They reported epigastria pain is an exclusive characteristic of H.pylori infection in children [32]. This could be explained on the basis of previous studies which reported that H.pylori is the major etiologic factor in the development of chronic gastritis and peptic ulcer disease in children [33]. In this work, there were significantly higher gastric mucosal Malondialdehyde (MDA) levels in patients compared with controls. Similar results were obtained by Duran A et al. [34].
Drake et al. [35] investigated reactive oxygen species and lipid per oxidation in H.pylori associated gastritis and the effect of H.pylori eradication and found that MDA concentrations fell significantly in the mucosa of patients in whom H.pylori was successfully eradicated, but not in those in whom eradication failed. Farinati et al. [36] reported that H.pylori positive patients had significantly higher gastric tissue levels of MDA, which correlated with the level of free radical products. Ebru et al. [37] observed that there is a significant increase in MDA levels as the severity of inflammation caused by H.pylori increased.
The present study demonstrates comparable serum Malondialdehyde concentrations in H. pylori -infected patients and control subjects. However, some authors [34-38] reported that serum Malondialdehyde levels increased significantly in H. pylori- positive patients. Possible explanation to our finding might be that early clinical presentation of the studied cases reflects the existing mild degree of lipid per oxidation. It has been shown higher accumulation of lipid peroxides was found more and concentration of serum MDA increases as the disease progresses [39].
In the current work, the mean gastric mucosal level of reduced glutathione (GSH) was decreased significantly in patients infected with H. pylori when compared with controls. This agrees with Haim et al. [40] who found that gastric mucosal glutathione concentrations are depleted during chronic colonization by H.pylori. Our results is also in agreement with Obst et al. [41] who showed that addition of a cytosolic extract of H.pylori to gastric epithelial cells directly stimulated reactive oxygen formation and caused a modest decline in glutathione concentrations after 24 hours. Our results is also in agreement with Verhulst et al. [42] reported that antral mucosal glutathione concentrations were approximately 40% lower in patients infected with H.pylori compared with non-infected controls.
In the current work, the mean serum level of reduced glutathione (GSH) was decreased significantly in patients infected with H. pylori when compared with controls. Similarly, Santra [39], Ansari et al. [43] showed that levels of reduced glutathione were significantly lower in H.pylori positive patients. Depletion of glutathione in H. pylori-infected patients may be due to failure of the glutathione-dependent antioxidant defence system which results in accumulation of free radicals thus initiating membrane damage by lipid per oxidation [43]. The study of Beil et al. [44], Shirin et al. [45] found a decrease in serum reduced glutathione in H.pylori infected patients. They suggested that diminished levels of reduced glutathione may be due to direct effect of the bacterium as well as through the associated inflammatory response. Beil et al. [44] found that the level of glutathione correlates with parameters of acute and chronic inflammation, but does not correlate significantly with bacterial density, supporting the hypothesis that the depletion in glutathione is secondary to the inflammatory response to H.pylori rather than to direct interaction with the bacteria. They found that addition of H.pylori does directly affect glutathione levels, causing a very early increase in glutathione concentration followed by complete depletion of glutathione within 24 hours. Thus, a direct effect of H.pylori on reduced GSH concentrations cannot be excluded.
In this work, there were significantly higher gastric mucosal superoxide dismutase (SOD) levels in the studied patients compared with controls. These results were consistent with those of Nagata et al. [46] who supports the role of H.pylori in generation of superoxide radicals, Brodie et al. [47] who found that SOD level was higher in the antral mucosa of H.pylori positive children than in controls and Gotz et al. [48] who reported that amount and activity of magnesium-SOD in H.pylori infected antrum and corpus were found to be significantly higher than in controls.
Hazell et al. [49] found that the bacterial survival in the presence of ROS is largely due to its antioxidant defence mechanisms, including SOD, alkyl hydro peroxide reductive, thiol peroxidise, and catalase. Ansari et al. [43] reported that formation of high levels of superoxide radical by H.pylori will increase the activity of SOD in gastric mucosa. Kyoko et al. [50] demonstrated that H.pylori infection was associated with a significant increase of the expression of Magnesium-SOD in the antrum and also reported that successful eradication of the organism resulted in a decrease of the expression of Magnesium SOD.
Farkas et al. [51] declared that SOD activity was significantly higher in H.pylori-positive patients than in H.pylori-negative ones, and SOD level was significantly higher with increased H.pylori density and H.pylori positivity. Contrary to results of the present work, Akcam et al. [52] who found that myeloperoxidase, xanthine oxidase, and SOD were not different in the gastric mucosa of children who were infected and non-infected with H.pylori. The serum level of SOD was increased significantly in H.pylori -infected patients as compared to controls in the current study.
Our findings confirms earlier work of Khanzode et al. [53] who showed that serum SOD level increased significantly in H. pylori patients, diagnosed by upper gastrointestinal endoscopy, compared to controls. The rise in SOD concentrations correlated well with the rise in the degree of gastric pathology. In contrast, Blattacharjee et al. [54] found that the total level of SOD decreases significantly in Helicobacter Pylori leading to accumulation of reactive oxygen species. Gastric ulcer is associated with oxidative damage as evidenced by a significant increase in lipid per oxidation, depletion of superoxide dismutase leading to accumulation of free radicals.
In the present study serum and gastric mucosal levels of catalase increased significantly in H. pylori- positive patients as compared to controls. Moreover, our work also showed a significant inverse correlation between catalase and reduced glutathione serum levels of the studied H.pylori infected patients. H.pylori catalase proves to be important in bacterial protection from exogenous H202. This enzyme may represent another primary virulence factor in that it allows the bacterium to survive on the surface of inflamed gastric mucosa [55]. However, Bulbuloglu et al. [56] studied the association of adenosine deaminase, SOD, and catalase activities with H.pylori and found no catalase activity in some antral tissue specimens in H pylori infected patients.
In this work, there were significantly lower serum and gastric mucosal NO levels in patients compared with controls. This may be explained by the study of alain et al. [57] who found that extra cellular H. pylori can be killed by nitric oxide (NO) released from activated macrophages. H. pylori arginase competes with host cell inducible NO synthase (iNOS) for the common substrate L-arginine. Thus, H. pylori prevent NO production by host cells [58].
Similar to us, Shiotani et al. [59] found that H.pylori- positive patients had a significantly lower concentration of NO in the gastric lumen. After completing eradication therapy, the intraluminal concentrations of NO returned to the same levels as those of H.pylori-negative subjects. In contrast to this study, Lim et al. [60] found that NO which is produced by iNOS induced by H.pylori in gastric mucosal epithelial cells, can contributes to apoptotic cell death. Dominique et al. [61] studied cytotoxicity associated with induction of iNOS and reported that release of H. pylori lip polysaccharides in vivo leads to the local production of elevated concentrations of NO from duodenal epithelium and possibly other mucosal cells.
Conclusion
There is a clear evidence of significant oxidative alteration of the gastric mucosal cells in children infected with H. pylori. It appears that H.pylori have developed various mechanisms to escape the effect of the immune system and to defense against oxidative stress, such as constitutively expressing superoxide dismutase and catalase, which detoxify superoxide anion and hydrogen peroxide, respectively. Also, H.pylori arginase prevents host NO production. This allows a successful adaptation of H. pylori to its hostile environment of the gastric mucosa. This was proved by the lower level of reduced glutathione in the infected patients than controls mostly due to its utilization to provide protection against mucosal damage by the free radicals. Also, there were high Malondialdehyde, superoxide dismutase, and catalase levels as a response to the oxidative.
References
- Feng H, Zhou X, Zhang G (2014) Association between cirrhosis and Helicobacter pylori infection: a meta-analysis. Eur J Gastroenterol Hepatol 26(12): 1309-1319.
- Fu HW (2014) Helicobacter pylori neutrophil-activating protein: from molecular pathogenesis to clinical applications. World J Gastroenterol 20(18): 5294-5301.
- Handa O, Naito Y, Yoshikawa T (2014) Redox biology and gastric carcinogenesis: the role of Helicobacter pylori. Redox Rep 16(1): 1-7.
- Hardbower D, Peek R, Wilson K (2014) At the Bench: Helicobacter pylori, dysregulated host responses, DNA damage, and gastric cancer. J Leukoc Biol 96(2): 201-212.
- Tiwari SK, Sharma V, Bardia A, Sivaram G, Saikant R, et al. (2010) Relevance of Helicobacter pylori genotypes in gastric pathology and its association with plasma malondialdehyde and nitric oxide levels. Inflammopharmacology 18(2): 59-64.
- Takahiro U, David Y (2015) Diagnosis of Helicobacter pylori using the rapid urease test. Ann Transl Med 3(1): 9.
- Buharideen S, Wijetunge S, Kotakadeniya R, Galketiya KB, Samarasinghe B, et al. (2015) A low cost rapid urease test to detect Helicobacter Pylori infection in resourse limited settings. Celyon Med J 60(1): 21-23.
- Esterbauer H (1996) Estimation of peroxidative damage; a critical review. Pathol Biol 44(1): 25.
- Zhang J, Masciocchi M, Lewis D, Sun W, Liu A (2008) Placental antioxidant gene polymorphisms, enzyme activity, and oxidative stress in preeclampsia. Placenta 29(5): 439-443.
- Shaik I, Mehvar R (2006) Rapid determination of reduced and oxidized glutathione levels using a new thiol-masking reagent and the enzymatic recycling method: application to the rat liver and bile samples. Anal Bioanal Chem 385(1): 105-113.
- Guevara I, Iwanejko J, Dembinska A (1998) Determination of nitrite/ nitrate in human biological material by the simple Greiss reaction. Clin Chim Acta 274(2): 177-188.
- Stuart H, Doyle E, David G (1991) Helicobacter pylori catalase,J Gen Microbiol 137(1): 57-61.
- Armitage P, Berry G, Matthews J (2002) Statistical methods in medical research. (4th edn), Oxford. Blackwell, UK, 4: 125.
- Morishita K, Takeuchi H, Morimoto N, Shimamura T, Kadota Y, et al. (2012) Superoxide dismutase activity of Helicobacter pylori per se from 158 clinical isolates and the characteristics. Microbiol Immunol 56(4): 262-272.
- Kwiecien S, Brzozowski T, Konturek PC, Pawlik MW, Pawlik WW, et al. (2004) Gastroprotection by pentoxyfilline against stress induced gastric damage. Role of lipid peroxidation, antioxidizing enzymes and proinflammatory cytokines. J Physiol Pharmacol 55(2): 337-355.
- Karita M, Matsumoto S, Kamei T (2003) The size of cag A based on repeat sequence has the responsibility of the location of Helicobacter pylori in the gastric mucus and the degree of gastric mucosal inflammation. Microbiol Immunol 47(9): 619-630.
- Mahmud S, Shah SA, Ali S (2015) Frequency of helicobacter pylori (hp) infection in children with recurrent abdominal pain (RAP). Pak Armed Forces Med J 65(3): 358-362.
- Malaty H, El Kasabany A (2002) Age at acquisition of Helicobacter pylori infection: a follow up study from infancy to adulthood. Lancet 359(9310): 931-935.
- Mahmoud MA, Hussein L, Coward A, Jackson SJ (2008) Prevalence of Helicobacter pylori infection among Egyptian children: impact of social background and effect on growth. Public Health Nutr 11(3): 230-236.
- Mohran Z, Sherif M, Fathy H (2008) Universal high-level primary metronidazole resistance in Helicobacter Pylori isolated from children. Public Health Nutrition 8: 250.
- Tsai HF, Hsu PN (2016) Modulation of tumor necrosis factor- related apoptosis-inducing ligand (TRAIL)-mediated apoptosis by Helicobacter pylori in immune pathogenesis of gastric mucosal damage. J Microbiol Immunol Infect 50(1): 4-9.
- Goodman KJ, Correa P, Mera R, Yepez MC, Cerón C, et al. (2011) Effect of Helicobacter pylori infection on growth velocity of school age Andean children. Epidemiology 22(1): 118-126.
- Al Mendalawi MD (2015) Prevalence of Helicobacter pylori and parasites in symptomatic children examined for Helicobacter pylori antibodies, antigens, and parasites in Yemen. Saudi Med J 36(1): 126.
- Naji AS, Ameri GAA, Alkadasi MN, Hanash S, Ali WAM, et al. (2014) Comparison of stool antigen and blood antibody test methods for detection of Helicobacter pylori infection and the risk factors. Int J Curr Microbiol App Sci 3(12): 118-127.
- Marie MA (2008) Seroprevalence of Helicobacter pylori infection in large series of patients in an urban area of Saudi Arabia. Korean J Gastroenterol 52(4): 226-229.
- Hestvik E, Tylleskar, Kaddu Mulindwa DH (2010) Helicobacter pylori in apparently healthy children aged 6-10 years in urban Kampala, Uganda: a community- based cross sectional survey. BMC Gastroenterol 10: 62.
- Süoglu OD, Gökfe S, Saglam AT, Sökücü S, Saner G (2007) Association of Helicobacter pylori infection with gastroduodenal disease: epidemiological factors and iron-deficiency anemia in Turkish children undergoing endoscopy, and impact on growth. Pediatr Int 49(6): 858-863.
- Pehlivanoglu E, Ertem D (2002) Helicobacter pylori may influence height in children independent of socio-economic factors. J Pediatr Gastroenterol Nutr 35(2): 232-233.
- Das BK, Kakkar S, Dixit VK, Kumar M, Nath G, et al. (2003) Helicobacter pylori infection and recurrent abdominal pain in children. J Trop Pediatr 49(4): 250-252.
- Liberato SV, Galindo M, Alvarez L, Sánchez Miramón F, Leandro Ciriza SE (2005) Helicobacter pylori infection in the child population in Spain: prevalence, related factors and influence on growth. An Pediatr (Barc) 63(6): 489-494.
- Hasosah M, Satti M, Shehzad A, Alsahafi A, Sukkar G, et al. (2015) Prevalence and risk factors of Helicobacter pylori infection in Saudi children: a three-year prospective controlled study. Helicobacter 20(1): 56-63.
- Bode G, Brenner H, Adler G, Rothenbacher D (2003) Recurrent abdominal pain in children: evidence from a population-based study that social and familial factors play a major role but not Helicobacter pylori infection. J Psychosom Res 54(5): 417-421.
- Hardikar W, Davidson P, Cameron D (2008) Helicobacter Pylori infection in children. J Gastroenterol Hepatol 6: 450.
- Arslan D, Kose K, Patiroglu TE (2007) Is there oxidative stress in children with Helicobacter pylori? Saudi Med J 28(8): 1222-1226.
- Drake IM, Mapstone NP, Schorah CJ, White KL, Chalmers DM, et al. (1998) Reactive oxygen species and lipid peroxidation in Helicobacter pylori associated gastritis relation to gastric mucosal ascorbic acid concentrations and effect of Helicobacter pylori eradication. Gut 42(6): 768-771.
- Farinati F, Della G, Cardin R, Molari A, Plebani M, et al. (1996) Gastric antioxidants, nitrites, and mucosal lipoperoxidation in chronic gastritis and Helicobacter pylori infection. J Clin Gastroenterol 22(4): 275-281.
- Ebru T, Ihsan U, gursel A, Nevin T, Ahmet K, et al. (2009) Does Helicobacter pylori-induced inflammation of gastric mucosa determine the severity of symptoms in functional dyspepsia? J Gastroenterol 44(1): 66-70.
- Vijayan G, Sundaram RC, Boby Z, Hamide A, Selvaraj N, et al. (2007) Increased plasma malondialdehyde and fructosaminein anemic H. pylori infected patients: Effect of treatment. World J Gastroenterol 13(5): 796-800.
- Santra A (2000) Oxidative stress in gastric mucosa in Helicobacter pylori infection. Indian J Gastroenterol 19: 21-23.
- Shirin H, Pinto JT, Liu LU, Merzianu M, Sordillo EM, et al. (2001) Helicobacter pylori decreases mucosal glutathione. Cancer Letters 164(2): 127-133.
- Obst B, Wagner S, Sewing KF, Beil W (2000) Helicobacter pylori causes DNA damage in gastric epithelial cells. Carcinogenesis 21(6): 11111115.
- Verhulst ML, van Oijen AH, Roelofs HM, Peters WH, Jansen JB (2000) Antral glutathione concentration and glutathione S-transferase activity in patients with and without Helicobacter pylori. Dig Dis Sci 45(3): 629-632.
- Ansari M, Rahbani M, Dolatkhah H, Fattahi E, Aghazade AM, et al. (2006) Comparison of levels of nitric oxide, superoxide dismutase, and glutathione peroxidase of gastric juice in infected and noninfected patients with helicobacter pylori. Acta Medica Iranica 44: 159.
- Beil W, Obst B, Sewing KF, Wagner S (2000) Helicobacter pylori reduces intracellular glutathione in gastric epithelial cells. Dig. Dis Sci 45(9): 1769-1773.
- Shirin H, Pinto JT, Lui LU, Merzianu M, Sordillo EM, et al. (2001) Helicobacter pylori reduces gastric mucosal glutathione. Cancer Lett 164(2): 127-133.
- Nagata K, Nishikawa M, Kashiba M, Nakamura A, Sato EF, et al. (1998) Helicobacter pylori generates superoxide radicals and modulates nitric oxide metabolism. J Biol Chem 273(23): 14071-14073.
- Bordie E, Klinowski E, Varsano R, Eshchar J, Herbert M, et al. (1996) Superoxide dismutase activity in Helicobacter pylori-positive antral gastritis in children. J Pediatr Gastroenterol Nutr 23(5): 609-613.
- Götz JM, van Kan CI, Verspaget HW, Biemond I, Lamers CB, et al. (1996) Gastric mucosal superoxide dismutases in helicobacter pylori infection. Gut 38(4): 502-506.
- Hazell S, Harris A, Trend M (2001) Helicobacter pylori: physiology and genetics. Amer Soc Microbiol 15: 167.
- Ozawa K, Kato S, Sekine H, Koike T, Minoura T, et al. (2005) Gastric epithelial cell turnover and mucosal protection in Japanese children with Helicobacter Pylori infection. J Gastroenterol 40(3): 236-246.
- Farkas R, Selmeci L, Tulassay Z, Pronai L (2003) Superoxide dismutase activity of the gastric mucosa in patients with Helicobacter pylori infection. Anticancer Res 23(5b): 4309-4312.
- Akcam M, Elmas O, Yilmaz A, Caglar S, Artan R, et al. (2006) Myleoperoxidase, xanthine oxidase and superoxide dismutase in the gastric mucosa of Helicobacter pylori positive and negative pediatric patients. Mol Cell Biochem 290(1-2): 125-130.
- Khanzode SS, Khanzode SD, Dakhale GN (2003) Serum and plasma concentration of oxidant and antioxidants in patients of Helicobacter pylori gastritis and its correlation with gastric cancer. Cancer Let 195(1): 27-31.
- Bhattacharjee M, Bhattacharjee S, Gupta A, Banerjee RK (2002) Critical role of an endogenous gastric peroxidase in controlling oxidative damage in Helicobacter pylori mediated and non-mediated gastric ulcer. Free Radic Biol 32(8): 731-743.
- Harris AG, Wilson JE, Danon SJ, Dixon MF, Donegan K, et al. (2003) Catalase (KatA) and KatA-associated protein (KapA) are essential to persistent colonization in the Helicobacter pylori SS1 mouse model. Microbiology 149(Pt 3): 665-672.
- Bulbuloglu E, Inanc F, Bakaris S, Kantarceken B, Cetinkaya A, et al. (2005) Association of adenosine deaminase, superoxide dismutase, and catalase activities with Helicobacter pylori. Dig Dis Sci 50(12): 2296-2299.
- Gobert AP, McGee DJ, Akhtar M, Mendz GL, Newton JC, et al. (2001) Helicobacter pylori arginase inhibits nitric oxide production by eukaryotic cells: A strategy for bacterial survival. Proc Natl Acad Sci USA 98(24): 13844-13849.
- Kavermann H, Burns BP, Angermuller K, Odenbreit S, Fischer W, et al. (2003) Identification and characterization of Helicobacter pylori genes essential for gastric colonization. J Exp Med 197(7): 813-822.
- Shiotani A, Yahaoka K, Iguchi M, Saika A, Itoh H, et al. (1999) Helicobacter pylori infection induces intraluminal nitric oxide in humans. J Gastroenterol 34(6): 668-674.
- Lim JW, Kim H, Kim KH, NF Kappa B (2001) Inducible nitric oxide synthase and apoptosis by Helicobacter Pylori infection. Free Radic Biol Med 31(3): 355-366.
- Lamarque D, Moran AP, Szepes Z, Delchier JC, Whittle BJ (2000) Cytotoxicity associated with induction of nitric oxide synthase in rat duodenal epithelial cells in vivo by lipopolysaccharide of Helicobacter Pylori: inhibition by superoxide dismutase. Br J Pharmacol 130(7): 1531-1538.






























