Influence of a Kefir-Derived Antimicrobial Fraction on Zika Virus Cytopathic Effects and Lymphocyte Proliferation
Marta de Oliveira Domingos*1, Irys Viana Neves1, Monamaris Martins Borges1, Thiago Martinhago Broring1, Gabrielle Ribeiro de Andrade1, Cinthia Soarez Galhardo1, Marina Tavares dos Anjos1 and Renato Mancini AstrayAuthors2
Laboratory of Bacteriology, Instituto Butantan, Brazil
Laboratory of Immunovirology, Instituto Butantan, Brazil
Submission: June 16, 2017; Published: July 07, 2017
*Corresponding author: Marta de Oliveira Domingos, Laboratory of Bacteriology, Instituto Butantan, Avenida Vita Brasil, 1500, CEP 05503000, Sao Paulo, Brazil, Tel: 55 11 26279708; Email: marta.domingos@butantan.gov.br
How to cite this article: Marta d O D, Irys V N, Monamaris M B, Thiago M B, Gabrielle R d A, et al. Influence of a Kefir-Derived Antimicrobial Fraction on Zika Virus Cytopathic Effects and Lymphocyte Proliferation. J Virol Curr Res. 2017; 2(2):555584. 10.19080/004 JOJIV.2017.02.555584.
Abstract
Several studies have already demonstrated the ability of probiotics to fight virus infection either by enhancing the host immune response or by inhibiting viral adhesion to the host cells. Accordingly, it has been decided to determine the ability of an antimicrobial fraction derived from kefir to inhibit the cytopathic effect of Zika virus on epithelial cells. In addition, the influence of this fraction on lymphocytes was also investigated. The results demonstrated that after 72h of infection the antimicrobial fraction, at a concentration of 37.5µg/mL, was able to inhibit 44% of Zika virus cytopathic effect on Vero cells. The results have also shown that the antimicrobial fraction did not influence the number of T and B lymphocytes in the spleens of mice immunized subcutaneously with BSA. However, the fraction was able to inhibit fully the proliferative effect of Concanavalin A on T cells. The results indicate that the antimicrobial fraction derived from kefir has an anti-virus effect that can be improved either by exploiting its ability to reduce viral cytopathic effect or by antagonizing the influence that some types of virus have on T cell proliferation.
Keywords: Zika virus; Kefir antimicrobial fraction; T cells; ConA
Introduction
A huge increase in the number of new emerging viral diseases such as dengue, Chikungunya, Ebola and lately Zika, a flavivirus transmitted by Aedes mosquitos, has become a huge problem of public health worldwide [1], especially taking into consideration the fact that there are not enough drugs in the pharmaceutical pipeline to control the evolution of viral drug- resistance. For these reasons, there is a continual search for new antiviral agents to replace the current ones used in the medical practice when they become ineffective. In the search for anti-viral drugs with few side effects, several studies have investigated whether probiotic products could be utilized as remedies against viral diseases. The results obtained by these studies have demonstrated that probiotic products have the potential be used as medicines against viral infection [2-4]. In this regard, a very interesting study was published by Morsy and colleagues [5], who demonstrated that patients with chronic hepatitis C virus infection could benefit by drinking kefir. Our results also support these data by showing that a molecular fraction with mass lower than 700kDa derived from milk kefir was able to inhibit the cytopathic effect of Zika virus on Vero cells in VP SFM-Thermo, (a serum-free, ultra-low protein medium containing no proteins, peptides, or other components of animal or human origin, designed for the growth of Vero cells for virus production) [6]. In this experiment, the cytopathic effect of Zika virus was observed after 72 hours incubation of cells in 96 well cell culture plates containing 5 x 104 cells/ml (100µl/well) of fresh VP SFM medium diluted serially with kefir antimicrobial fraction at the following concentrations: 150Ml/ml to 0.29µl/ ml. After 2 hours incubation, a MOI of 0.1 PFU/cell of Zika virus culture was added to the wells at a final volume of 100µL per/ well. After 72h of incubation, the plates were stained and read, revealing a 44% CPE (cytopathic effect) inhibition when the antimicrobial fraction of Kefir was used at 37.5µgI/mL (Figure 1).
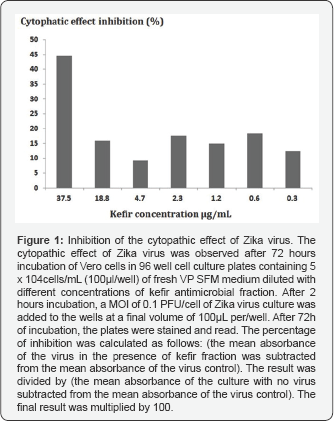
Other studies have also demonstrated that kefir metabolites are able to arrest proliferation and induce apoptosis of HTLV- 1 negative malignant lymphocytes [7]. Studies using probiotic bacteria instead of metabolites have also demonstrated that the probiotic bacteria themselves can also help the immune system to fight viral infection by enhancing the host's immune response. A good example of the latter is the work of Sugimura and colleagues who demonstrated that Lactococcus lactis JCM5808 can directly activate plasmocytoid dendritic cells in-vivo and increase their ability to produce IFNs [8]. Yang and coworkers [9] have also demonstrated that oral immunization of chickens with a recombinant probiotic L. plantarum expressing NP-M1- DCpep from H9N2 avian influenza virus significantly increased T-cell-mediated immune responses, mucosal sIgA and IgG levels and provided protection against H9N2 AIV challenge.
Accordingly, it was decided in the present work to determine whether the humoral immune response induced by CTB (the B subunit of Cholera Toxin), a very potent immune adjuvant would be influenced by co-administration with the antimicrobial fraction of kefir. The results demonstrated that the humoral immune response against CTB of mice immunized subcutaneously with 1µg of CTB (100µl/animal) diluted in 100µl of the antimicrobial fraction was equivalent to the response generated by the group immunized with 1µg of CTB in PBS, showing that in this case the kefir fraction did not enhance the immune response generated by CTB. In order to determine whether the fraction would interfere with the humoral response generated in the presence of another adjuvant such as alum, Balb/c mice were immunized subcutaneously twice with BSA adsorbed on alum in the presence or absence of the antimicrobial fraction. Seven days after the last immunization blood was collected and the IgG response against BSA was measured. Contrary to what was observed previously with CTB, the IgG response against BSA generated by the group of the animals immunized with BSA absorbed on alum in the presence of the fraction was much lower than the response generated by the group control immunized with BSA adsorbed on alum alone. These data indicate that the antimicrobial fraction can influence the immune response in different ways in the presence of molecules that act upon different cell signaling pathways.
Therefore, in this study, the influence of the antimicrobial fraction of kefir on the proliferation of T and B cells in-vivo was determined. For this experiment, seven days after the last immunization, the splenic cells of the animals immunized subcutaneously with BSA adsorbed onto alum in the presence or absence of the antimicrobial fraction were incubated for 30 minutes with the following antibodies: rat IgG anti-mouse CD3- Tricolor, rat IgG anti-mouse CD4 labeled with FITC, rat IgG anti mouse CD8 labeled with (PE) and rat IgG anti mouse CD 45R labeled with PE. After 30 minutes of incubation with the aforementioned antibodies, the lymphocyte population profiles were determined by FACS according to the manufacturer's instructions. The results obtained in these experiments showed that immunization of BSA adsorbed onto alum in the presence of the antimicrobial fraction does alter the number of CD8 and CD4 T cells in the spleen (Figure 2). In addition the number of B cells was also equivalent to the number encountered in the group of mice immunized with BSA adsorbed onto alum (Figure 3).
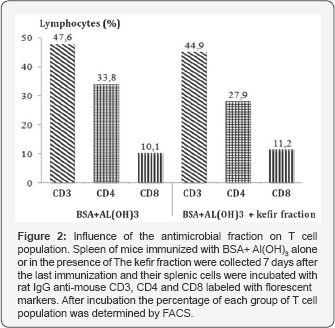
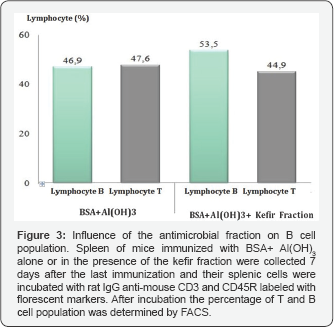
Despite the fact that no alteration in the number of T and B cell populations was noted, the humoral immune response of the animals was altered by the fraction, indicating that the fraction has a huge influence on the immune response. Apart from probiotic metabolites, other studies have indicated that concanavalin A (ConA) may also have antimicrobial properties since it has been shown that ConA is able to inhibit dengue virus adhesion to Vero cells [10]. Therefore, it was decided to determine the influence of the kefir fraction on T cell proliferation induced by ConA. For this experiment, seven days after the last immunization, the splenic cells of the animals immunized subcutaneously with BSA adsorbed onto alum in the presence or absence of the antimicrobial fraction were incubated with ConA in RPMI-1640 medium either alone or in the presence of the antimicrobial fraction diluted two-fold in RPMI-1640 medium (Sigma). The incubation was performed for 72h at 37 °C in a 5% carbon dioxide atmosphere. After incubation, the ability of ConA to induce T cell proliferation was determined by the MTT methodology [11]. The results demonstrated that the kefir fraction was able to abolish the proliferation effect of ConA on T cells. This results support the studies that have demonstrated that kefir metabolites can inhibit the proliferation of malignant lymphocytes [7,12].
Conclusion
The results obtained in the present work suggest that the antimicrobial fraction isolated from kefir has the potential to be used as an anti-viral medicine. The fraction can act directly on the virus or by influencing the host immune response to fight the infection. However, in order to be used as a product, the fraction needs to be better characterized and its mechanism of action has to be elucidated further.
Acknowledgement
The authors would like to thank Dr. Roger R C New for correcting the English of the manuscript.
References
- Saiz JC, Vazquez-Calvo A, Blázquez AB, Merino-Ramos T, Escribano- Romero E, et al. (2016) Zika Virus: the Latest Newcomer. Front Microbiol 7: 496.
- Olaya Galán NN, Ulloa Rubiano JC, Velez Reyes FA, Fernandez Duarte KP, Salas Cárdenas SP, et al. (2016) In-vitro antiviral activity of Lactobacillus casei and Bifidobacterium adolescentis against rotavirus infection monitored by NSP4 protein production. J Appl Microbiol 120(4): 1041-1051.
- Fernandez-Duarte KP, Olaya-Galán NN, Salas-Cárdenas SP, Lopez- Rozo J, Gutierrez-Fernandez MF (2017) Bifidobacterium adolescentis (DSM 20083) and Lactobacilus casei (Lafti L26-DSL) Probiotics able to Block the in vitro adherence of Rotavirus in MA104 cells. Probiotics Antimicrob Proteins.
- Starosila D, Rybalko S, Varbanetz L, Ivanskaya N, Sorokulova I (2017) Anti-influenza activity of Bacillus subtilis probiotic strain. Antimicrob Agents Chemother 61(7): e00539-17.
- Morsy RM Mahmoud AM, Zanaty MI, Abdel-Moneim A, Abo-Self MA (2014) Beneficial Effects of Milk Kefir in Patients with Chronic Hepatitis C Virus-Infection. International Hournal of Bioassays 3(6): 3086-3091.
- Frazzati-Gallina NM, Paoli RL, Mourao-Fuches RM, Jorge SA, Pereira CA (2001) Higher production of rabies virus in serum-free medium cell cultures on microcarriers. J Biotechnol 92(1):67-72.
- Maalouf K, Baydon E, Rizk S (2011) Kefir induces cell-cycle arrest and apoptosis in HTLV-I-negative malignant T-lymphocytes. Cancer Manag Res 14(3): 39-47.
- Sugimura T, Jounai K, Ohshio K, Tanaka T, Suwia M, et al. (2013) Immunomodulatory effect of Lactococcus lactis JCM5805 on human plasmacytoid dendritic cells. Clin Immunol 149(3): 509-518.
- Yang WT, Yang GL, Shi SH, Liu YY, Huang HB, et al. (2017) Protection of chickens against H9N2 avian influenza virus challenge with recombinant Lactobacillus planetarium expressing conserved antigens. Applied Microbiol. Appl Microbiol Biotechnol 101(11): 45934603.
- Martinez-Barragan JJ, Angel RM (2001) Identification of a putative Coreceptor on Vero Cells that Participates in Dengue 4 Virus Infection. J Virol 75(17): 7818-7827.
- Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1-2): 55-63.
- Pelopose JJM, Jean KT (2006) Role for Akt/protein kinase B and activator protein-1 in cellular proliferation induced by the human T-cell leukemia virus type 1 tax oncoprotein. J Biol Chem 281: 8927






























