An Improved Method for the Detection of Viral Infections Using Multiplex Molecular Diagnostic Technology
Mohammad M Hossain* and Raymond R Rowland
Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, Kansas State University, USA
Submission: April 08, 2017; Published: May 05, 2017
*Corresponding author: Mohammad M Hossain, Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, Kansas State University, USA, Tel: 785-532-4855; Fax: 785-532-4481; Email; mofazzal@vet.k-state.edu
How to cite this article: Mohammad M H, Raymond R R. An Improved Method for the Detection of Viral Infections Using Multiplex Molecular Diagnostic Technology. J Virol 004 irol Curr Res. 2017;2(1):555577.DOI: 10.19080/JOJIV.2017.02.555577
Abstract
The purpose of this study was the development of multiplex fluorescent microsphere immunoassay (FMIA) for the detection of antibody response against several different viral antigens by incorporation of non-species-specific conjugates Protein A, G and A/G in place of the secondary antibody. To evaluate multiplex detection of antibody responses against structural and non-structural recombinant antigens from seven different animal viruses: porcine reproductive and respiratory syndrome virus (PRRSV), porcine circovirus type 2 (PCV2), swine influenza virus (SIV), African swine fever virus (ASFV), classical swine fever virus (CSFV), bovine viral diarrhea virus (BVDV), and Rift Valley fever virus (RVFV) conjugated to microsphere beads were tested. Seven target antigens were assembled into a single multiplex and tested in sera infected with PRRSV, PCV2, SIV, ASFV, CSFV in swine, BVDV in cattle, and RVFV in sheep. All conjugates were capable of detecting antibody and the detection of IgG responses against PRRSV, PCV2, SIV, ASFV, CSFV antigen targets were varied with A>A/G>G. IgG response to BVDV and RVFV were different than swine as, G>A/G>A. In this study, PCV2 antigen-specific IgG was detected in oral fluid using Protein A. The results suggested that Protein A, G, and A/G can be an alternative to species specific reagents IgG.
Keywords: Fluorescent microsphere immunoassay; Luminex; Protein A, G, A/G; Nucleocapsid protein; Capsid protein; Multiplex assay; Animal viruses
Abbreviations: FMIA: Fluorescent Microsphere Immunoassay; EDC: 1-Ethyl-3-(3-Dimethylaminopropyl) Carbodiimide Hydrochloride; NHS: N-Hydroxy Sulfosuccinimide; PRRSV: Porcine Reproductive and Respiratory Syndrome Virus; PCV2: Porcine Circovirus Type 2; SIV: Swine Influenza Virus; ASFV: African Swine Fever Virus; CSFV: Classical Swine Fever Virus; BVDV: Bovine Viral Diarrhea Virus; RVFV: Rift Valley Fever Virus
Introduction
Emerging viral diseases are a major problem to human and veterinary public health and continue to be an eminent threat to the world economy. Fluorescent microsphere immunoassay (FMIA) can be used for the detection of emerging infectious diseases in humans and animals. FMIA is a novel molecular diagnostic technology for the detection of antibodies to at least 100 target antigens in a small sample volume. FMIA incorporates the incubation of serum or oral fluid with antigen- coated microsphere beads, each internally labeled with a unique spectral address.
Antigen-specific total immunoglobulin (Ig) is detected by a secondary antibody conjugated with biotin. Biotinylated secondary antibody captured analyte-specific Ig can be detected using streptavidin-labelled phycoerythrin (SA-PE) reporter molecule. Two light emitting diodes (LEDs) and a CCD imager are used for bead and analyte identification. Excitation with one LED identifies the unique fluorescent signature of each antigen- coated microsphere. Excitation with the second LED detects the phycoerythrin reporter molecule. The system can detect up to 50 antigen targets in a single sample.
Recently, detection of IgA, IgG, and IgM response against several viral antigens including classical swine fever virus (CSFV), Rift Valley fever virus (RVFV) have been studied [1,2]. In the FMIA, traditionally host specific IgG conjugates is being used. Detection of IgG responses to recombinant antigens from CSFV, RVFV, BVDV using FMIA have been summarized in a recent publication [1-3]. In the previous report, PA, PG, and PA/PG have been used into ELISA-based studies [4,5].
Protein A and G were isolated from bacterial cell wall of Staphylococcus aureus and Streptococcus species, respectively. Protein A/G is a recombinant form of Protein A and Protein G [6]. All of the reagents are reactive to mammalian immunoglobulin G (IgG). Recently, IgG responses to PRRSV and PCV2 antigens in swine using non-species-specific conjugates such as Protein A (PA), G (PG), and (PA/G), all of which have been tested [7]. In addition to PRRSV and PCV2 in swine, BVDV in cattle and RVFV in sheep sera in the presence of alternative conjugate have been evaluated in this study.
Materials and Methods
Serum and oral fluid samples
Whole blood from virus infected animals were collected in tubes containing EDTA and processed for serum isolation. Serum was separated by centrifugation and stored at -80 °C until use. Oral fluids were collected from group of pig (pens) infected with PCV2 and used in this study.
Recombinant proteins
Recombinant structural and non-structural proteins from PRRSV, PCV2, SIV, ASFV, CSFV, BVDV, RVFV were cloned into the pHUE expression vector [8] and expressed in Escherichia coli. The 6xHis-tagged protein was affinity purified using a PrepEase His-Tagged Protein Purification high yield Kit (Affymetrix, USB, Santa Clara, CA). The concentration of protein was measured using BIO-RAD protein assay kit (BioRad, Hercules, CA) on a precision microplate reader (Molecular Devices, Sunnyvale, CA).
Conjugation of recombinant proteins to microsphere beads
Recombinant proteins were covalently coupled to Luminex MagPlex® polystyrene, carboxylated magnetic microsphere beads (Luminex Corporation, Austin, TX) using the method as previously described [9]. Briefly, 500|il of beads at a concentration of 1.25x107 beads/ml were conjugated to 25|ig of recombinant protein. At the final step, the coupled beads were re-suspended in 1 ml of PBST with 1% fish gelatin (Sigma- Aldrich, St. Louis, MO) and stored at 4 °C in the dark until use.
Fluorescent microsphere immunoassay procedure (FMIA)
For the multiplex FMIA, the seven recombinant antigen bead sets were combined with an unconjugated bead set as a background control. A penta-His monoclonal antibody was used to estimate the relative amount of recombinant His-tagged protein attached to each bead set as described previously [1]. Approximately 1,250 beads in 50μl of PBS with 4% goat serum (PBS-GS) and 50μl serum samples or serial 2-fold diluted penta- HisTM mouse monoclonal IgG (Qiagen GmbH, Valencia, CA) were placed in each well of a CorningTM CostarTM polystyrene white round-bottom 96-well plate (Corning Incorporated, Kennebunk, ME ). Biotin-conjugated protein (4μg/ml): PA or PG (Thermo Scientific, Waltham, MA) or PA/G (BioVision, Inc., Milpitas, CA); biotin-SP-conjugated goat anti-swine IgG (2μg/ ml) (Jackson Immuno Research Laboratories, Inc., West Grove, PA), and biotinylated anti-mouse IgG (H+L) (2μg/ml) (Vector Laboratories, Inc., Burlingame, CA) were diluted in PBS-GS. Data were analyzed using xPONENT 4.2 software, and the results reported as mean fluorescent intensity (MFI) obtained from the median value for at least 100 beads. Results were reported as MFI of the test sample minus MFI of the background bead set [2].
Results
Detection of microsphere bead-bound protein using alternative conjugate PA
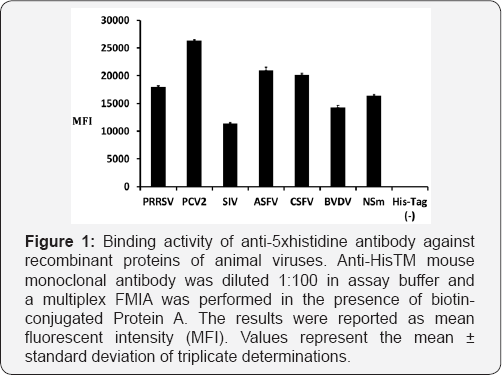
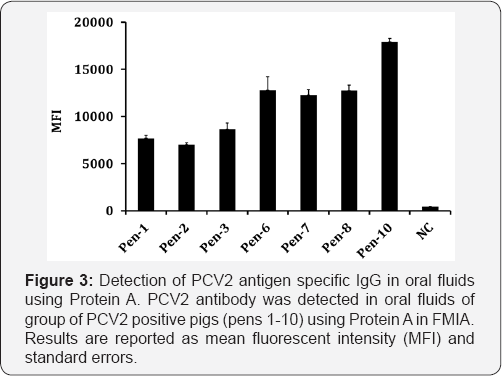
As shown in Table 1, recombinant proteins were purified from animal viruses (PRRSV, PCV2, SIV, ASFV, CSFV, BVDV, RVFV) and all of the proteins possessed His-tag for successful purification. In addition to protein purity, penta-His mAb was used to evaluate the relative amount of protein attached to each bead set. As shown in Figure 1, all of the bead sets bound to recombinant His-tag proteins was positive. All of the seven recombinant proteins with His-tag showed binding activities whereas recombinant proteins without penta-His mAb did not show binding activity. PCV2 IgG antibody was highly reactive to PCV2 antigen detected in oral fluids of group of PCV2 positive pigs (pens 1-10) using Protein A (Figure 2). Overall, the results show that the use of Protein A provides an alternative method for assessing antigen binding to the beads.
PRRSV: Porcine Reproductive and Respiratory Syndrome
Virus; PCV2: Porcine Circovirus Type 2; SIV: Swine Influenza
Virus; ASFV: African Swine Virus; CSFV: Classical Swine Fever
Virus; BVDV: Bovine Viral Diarrhea Virus; RVFV: Rift Valley Fever Virus
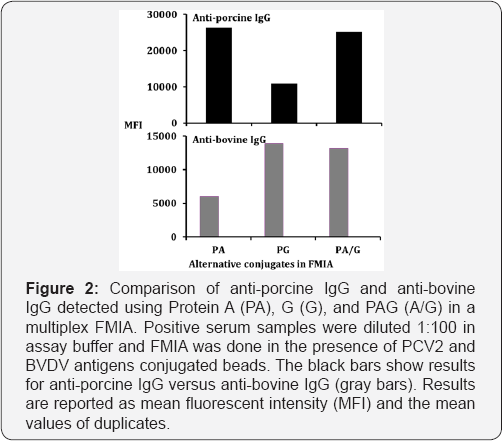
Comparison of PA, PG and PA/G for the detection of anti-porcine and anti-bovine IgG
The binding activities of PA, PG and PA/G with antibodies in different mammalian species are illustrated in Figure 3. For the species-specific IgG binding activity, PCV2 and BVDV infected animal sera samples were used for the detection of IgG in FMIA using biotinylated PA, PG or PA/G. The MFI values were higher for PA and PA/G, followed by PG in swine. The MFI values for PG were reduced by about 50% compared to PA and PA/G. The MFI values were higher for PG and A/G in cattle (Figure 3). IgG in swine are highly reactive to Protein A, whereas IgG in cattle and sheep are highly reactive to Protein G (Table 2).
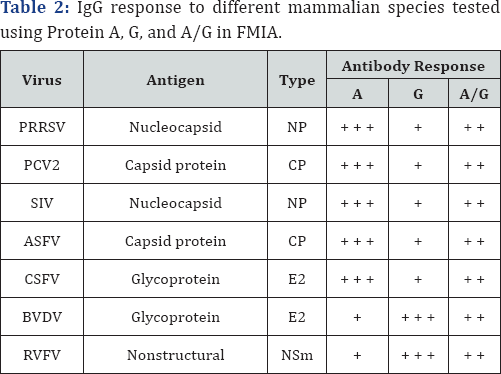
PRRSV: Porcine Reproductive and Respiratory Syndrome Virus;
PCV2: Porcine Circovirus Type 2; SIV: Swine
Influenza Virus; ASFV: African Swine FeverVirus; CSFV: Classical
Swine Fever Virus; BVDV: Bovine Viral
DiarrheaVirus; RVFV: Rift Valley Fever Virus; MFI = Mean
fluorescence intensity
+++ = highly responsive
++ = moderatelyresponsive
+ = weakly responsive
Discussion
In this study, multiplex serological assay was developed using Protein A, G and A/G which was derived from bacterial wall. These proteins were used as conjugates in place of the secondary antibody. All of the conjugates bind to mammalian IgG and the binding activity are species-specific. In representative results for a multiplex FMIA incorporating the seven viral antigens, all of the antigens bound to beads were detected using Protein A. Of the three non-species specific bacterial cell wall derived proteins used in this study as an alternative conjugates, the highest MFI values were recorded for PA in swine and PG in cattle suggested that the binding activity of PA and PG are species-specific. In our previous studies, antibody responses to PRRSV, and PCV2 were evaluated in multiplex FMIA using alternative conjugates Protein A, G, and A/G [7]. The data in the present study support the previously reported findings.
The results in this study suggested that Protein A is a superior alternative conjugate for the detection of Ig in swine and PG is the best alternative conjugate in cattle (Figure 3). Protein A/G is the second best non-species specific alternative conjugate in both swine and cattle suggested that Protein A/G has a common binding activity in all mammalian species (Table 2). In the previous report, Protein A/G was used for the ELISA- based diagnosis of infection in wide range of mammalian species [10,11], suggested that the data in the current studies are supported by previous finding in Protein A/G based ELISA. PCV2 antigen specific IgG was detected in oral fluids of PCV2 positive pigs (Figure 3), suggested that Protein A is a suitable conjugate in FMIA for the detection of antibody response in oral fluids.
Further, it has been reported that native forms of bacterial cell wall derived PA and combined form of chimeric PA/G can bind to the immunoglobulins of many mammalian species [12,13]. Therefore, non-species-specific secondary conjugates (PA, PG, and PA/G) can be used for the detection of IgG in wide range of mammalian species. The geographical regions where species-specific antibodies are not available and production is exceedingly time consuming PA, PG and PA/G are the best choice in immunological assays for the control of emerging of new pathogens. The use of PA, PG, and PA/G in the place of secondary antibody in FMIA is an efficient and cost-effective approach to animal virus surveillance.
Conclusion
The use of alternative conjugates Protein A, G, A/G for the detection of IgG is a new development in FMIA. Monitoring immune responses in a wide variety of mammalian species using an alternative conjugate based FMIA is reliable, cheap, and fast. The present study suggests that alternative conjugate-based FMIA have a wide variety of applications in veterinary diagnostic laboratory because of the multiple mammalian species that are dealt with in veterinary professions.
References
- Hossain MM, Rowland RRR (2017) Evaluation of classical swine fever virus (CSFV)-specific IgA, IgG, and IgM antibody response in swine vaccinated with alphavirus replicon particles-expressed antigens. Adv Tech Biol Med 5(1): 213.
- Hossain MM, Wilson WC, Faburay B, Richt J, McVey DS, et al. (2016) Multiplex detection of IgG and IgM to Rift Valley fever virus nucleoprotein, nonstructural proteins, and glycoprotein in ovine and bovine. Vector Borne Zoonotic Dis 16(8): 550-557.
- Hossain MM, Rowland RR (2016) A new development in multiplex platform for the diagnosis of bovine viral diarrhea virus (BVDV) infection in cattle. 7th BVDV Symposium, Conference of Research Workers in Animal Diseases (CRWAD), Illinois, USA.
- Madore HP, Baumgarten A (1979) Enzyme-linked protein A: an enzyme-linked immunosorbent assay reagent for detection of human immunoglobulin G and virus-specific antibody. J Clin Microbiol 10(4): 529-532.
- Inoshima Y, Shimizu S, Minamoto N, Hirai K, Sentsui H (1999) Use of protein AG in an enzyme-linked immunosorbent assay for screening for antibodies against parapoxvirus in wild animals in Japan. Clin Diagn Lab Immunol 6(3): 388-391.
- Eliasson M, Andersson R, Olsson A, Wigzell H, Uhlen M (1989) Differential IgG-binding characteristics of staphylococcal protein A, streptococcal protein G, and a chimeric protein AG. J Immunol 142:575-581.
- Hossain MM, Rowland RR (2017) Fluorescence microsphere immunoassay for detection of antibodies to porcine reproductive and respiratory syndrome virus and porcine circovirus type 2 using protein A, protein G, and protein A/G. J Immunol Tech Infect Dis 6(1):1-6.
- Catanzariti AM, Soboleva TA, Jans DA, Board PG, Baker RT (2004) An efficient system for high-level expression and easy purification of authentic recombinant proteins. Protein Sci 13(5): 1331-1339.
- Hossain MM, Wilson WC, Rowland RR (2017) Incorporation of antigens from whole cell lysates and purified virions from MP12 into fluorescence microsphere immunoassays for the detection of antibodies against Rift Valley fever virus. Virology Research Journal 1(1):24-31.
- Richman DD, Cleveland PH, Oxman MN, Johnson KM (1982) The binding of staphylococcal protein A by the sera of different animal species. J Immunol 128(8): 2300-2305.
- Bjorck L, Kronvall G (1984) Purification and some properties of streptococcal protein G, a novel IgG-binding reagent. J Immunol 133(2): 969-974.
- Kelly PJ, Tagwira M, Matthewman L, Mason PR, Wright EP (1993) Reactions of sera from laboratory, domestic and wild animals in Africa with protein A and a recombinant chimeric protein AG. Comp Immunol Microbiol Infect Dis 16(4): 299-305.
- Feir D, Lau C, Junge R (1993) Protein A and Protein G in the diagnosis of diseases in zoo animals. Trans Mo Acad Sci 27:9-14.






























