Mineral Fertilizer Effects on Anthracnose Disease Caused by Colletotrichum gloeosporioides Penz, Tuber Rot by Botryodiplodia theobromae Pat and yield in Dioscorea rotundata Variety Oniyere
Otusanya MO*
Department of Crop protection, Federal University of Agriculture Abeokuta, Nigeria
Submission: June 13, 2018; Published: August 24, 2018
*Corresponding author: Otusanya MO, Department of Crop protection, College of Plant Science and Crop Production (COLPLANT), Federal University of Agriculture Abeokuta (FUNAAB), Nigeria; Email: otusanyaoluleke@gmail.com
How to cite this article: Otusanya MO. Mineral Fertilizer Effects on Anthracnose Disease Caused by Colletotrichum gloeosporioides Penz, Tuber Rot by Botryodiplodia theobromae Pat and yield in Dioscorea rotundata Variety Oniyere. JOJ Hortic Arboric. 2018; 2(1): 555580.
Abstract
Mineral nutrition effect on anthracnose caused by Colletotrichum gloeosporioides, tuber rot by Botryodiplodia theobromae and yield in Dioscorea rotundata var. Iseosi was investigated in this study. The field plot used had been monoropped to Dioscorea species in the two previous consecutive years with only calcium nitrate fertilizer. But nitrogen and calcium in the plot soil were still below the critical (2% and 0.6 cmol kg-1 respectively) determined for optimum yam production (South West Nigeria). The field design was RCBD with 3 treatments of 0, 0.6, 1.2ml/l-1 Bounty fertilizer applied at 3 MAP, and three replicates. Bounty fertilizer favoured higher concentration of the phytoanticipins (with antimicrobial activity) flavonoid, lignin and tannin over the control. Leaf N and Mg had significant correlation with leaf flavonoid content (r = 0.7634, 0.6786). Leaf flavonoid had significant correlation with leaf lignin content, r = 0.7189. Leaf lignin in turn had significant correlation with leaf tannin content, r = 0.7556. Leaf Sulphur correlated significantly with leaf flavonoid and leaf phosphorus, r = 0.7218 and 0.8467 respectively. Bounty fertilizer also favoured higher leaf magnesium 234.73 and 230.27mg/100gdm (0.60ml/l-1 and 1.20ml/l-1) than the control, 182.40mg/100gdm. Leaf nitrogen, phosphorus and sulphur increased with bounty fertilizer treatment, then the control. Only leaf potassium was higher in the control (116.72mg/100gdm) than in the Bounty-fertilized plants with 87.31 and 87.53mg/100gdm in the 0.60mll-1 and 1.20mll-1 treatments.
The following correlations were also significant respectively: Leaf N and Leaf P, S, r = 0.8484 and 0.9836 respectively.
Leaf Ca and leaf Mg, r = 0.9192
Anthraconse incidence did not increase after the 3 MAP bounty fertilizer soil amendment. It was 11%, 6% and 0% in the control, 0.60ml/l-1 and 1.20ml/l-1 treatments respectively at 5 MAPS. It reduced to 5.6% (control and 0.60ml/l-1 treatment) and remained 0% in the 1.20ml/l-1 treatment at 6MAP. Severity of anthracnose was 1, category of resistant, both at 5 MAP and 6 MAPS across all the three treatments. The interplay/enhancement of leaf minerals with the three phytochemicals/anticipants, as well as the latter with one another should be responsible for the low disease incidence and resistant severity status of variety Oniyere. Mean tuber weight loss after inoculation and incubation with B. theobromae for 2 weeks was 0% and infection range over the three treatments was low, that is 1.21 to 2.95% with no significant differences. Mean tuber weight per plant was in the range 0.88kg to 1.35kg with no significant differences between treatments. The 1.20ml/l-1 bounty fertilizer treatment gave equivalent yield of 13.46 tons ha-1. Bounty fertilizer at 0.60ml/l-1 or 1.20ml/l-1 is recommended for local varieties of white guinea yam in South West Nigeria especially where monocropping is desired.
Introduction
White guinea yam, Dioscorea rotundata Poir is one of the three more popular species of yams in Nigeria, the largest world producer [1]. Production of this food security and income generating crop is constrained especially by necessary fallow periods as it drains heavily on soil nutrients [2,3]. High export and commercialization are also hampered by storage losses of the tune of 7million metric tons annually [4]. Anthracnose disease now prevalent on all cultivated Dioscorea species also reduces yield as it affects the leaves responsible for photosynthesis and subsequent tuber bulking. Yield losses are reported to be high, 80 to 85% in susceptible varieties in Nigeria [5]. Resistance breeding, regular fungicide usage where affordable and good cultural practices such as improved storage are management practices to combat yield losses in the field and in storage. Mineral nutrition for disease reduction in agricultural crops is regarded as cost effective management of crop loss compared to pesticide usage with environmental /health hazards [6]. Biofortification of crops through the application of mineral fertilizers combined with breeding varieties with an increased ability to acquire mineral elements is also advocated as an immediate strategy not only to increase mineral concentrations in edible crops but also to improve yields in infertile soils [7,8].
This project aimed at investigating the effect of the fertilizer Bounty, which is composed of 8 mineral elements (N, Ca, Mg, Mn, Zn, Cu, Fe and B) on anthracnose disease and tuber rot by Botrydiplodia theobromae in Discorea rotundata var. Oniyere, a popular white guinea yam variety in South West Nigeria.
Materials and Methods
Field layout, soil sampling and planting
The plot used had been monocropped to Dioscorea species for two previous consecutive years (2015 and 2016) with only calcium nitrate fertilizer. It was sited at the Teaching and Research Farms of the Directorate of University Farms (DUFARMS), Federal University of Agriculture (FUNAAB), Abeokuta, Ogun State, Nigeria. The soil site was sandy-loamy soil. The plot was 14m by 9.5m with 1m field border on all four sides. Soil samples were collected using standard methods from each of 3 replicates in the plot [9]. Twenty core samples were lifted at 20cm depth with a slanting soil auger, into labeled polythene bags. They were transferred to the laboratory for air-drying and sieving (2mm sieve) before bulking per replicate for analysis. Soil analysis of exchangeable cations was by the method of [10].
After soil sampling, mounds of 1m by 1m size and 80cm high were made, 18 per replicate. Intra-row inter -row distances were 0.5m and 1m respectively. 2m length robust bamboo stakes were established per mound. Planting of whole seed yams of 0.4kg to 0.5kg average size was at the onset of heavy rains (April 5, 2017).
Fertilization with bounty fertilizer
At 3MAP, three treatments of Bounty fertilizer per replicate, of 1.20ml/l-1, 0.60ml/l-1 and 0ml/l-1 (control) was applied in a 15cm deep groove, at 25cm radius from the plant roots in the mound [3]. Fertilizer was mixed in deionised water and poured evenly into the groove, which was thereafter covered well with soil
Disease incidence and severity assessment
Incidence of Anthracnose was assessed at 5MAP and 6MAPS. Percent incidence was determined using the formula:
%Incidence in a replicate plot( Number of plants with Anthrocnose symptoms/ Total number of plants in the replicate plot)100
Anthracnose severity was determined on a scale of 1 (resistant) to 5 (very susceptible) according to the method [11] shown in (Figures 1-3).
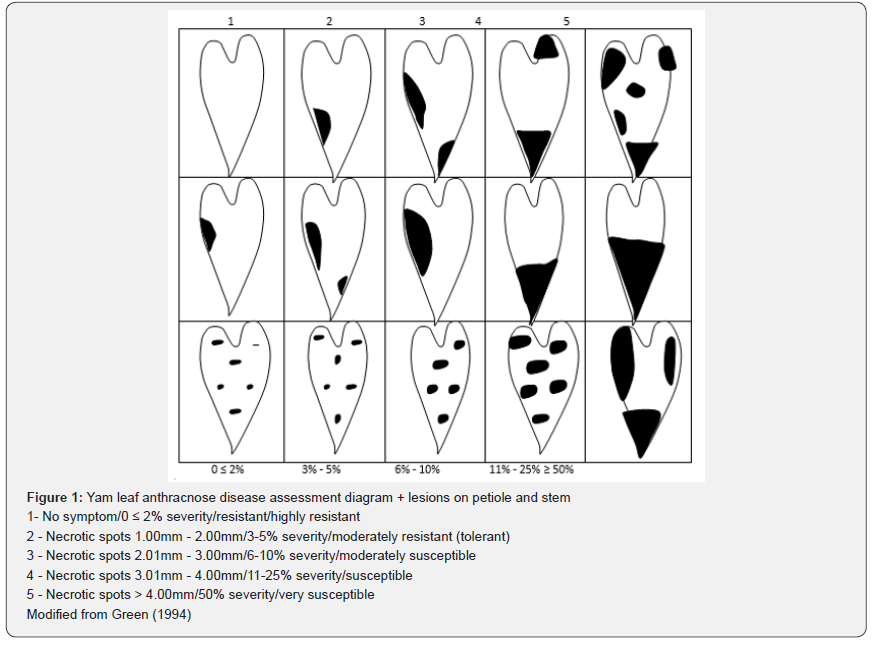
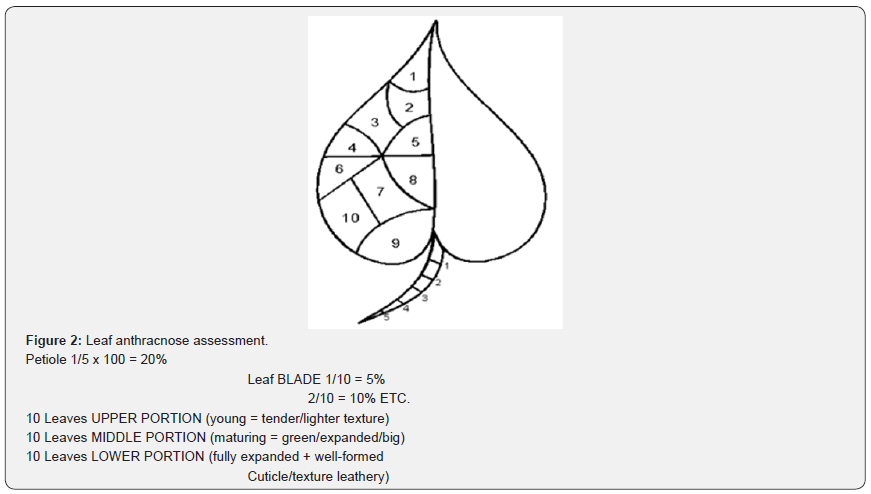
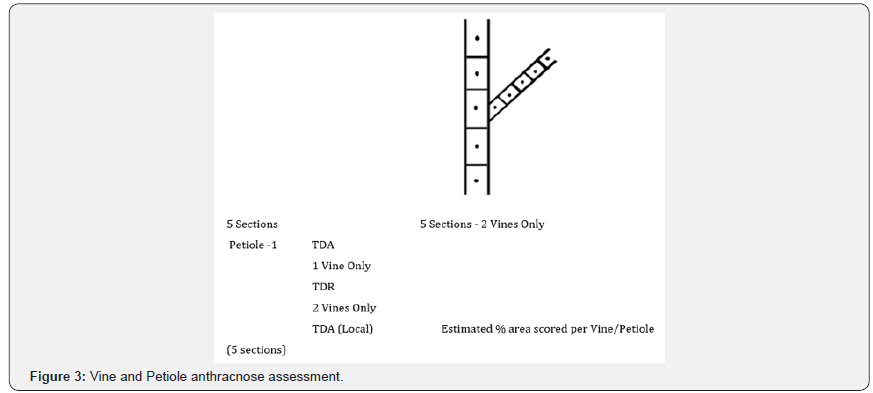
Leaf sampling and analysis
Leaf sampling was done at 6MAP, along a diagonal transect in the field plot. Sampling was done according to treatments, and 12 leaves (from top middle and lower portion) were plucked with the petiole attached from each sampled plant, into new paper envelopes, in the early morning (7:00am). They were transferred to the Crop Protection Laboratory, COLPLANT (College of Plant Science and Crop Production), FUNAAB. They were washed in three changes of distilled deionized water and left to dry on new food paper packs under a low speed fan, for an hour. They were transferred for subsequent drying on raised wooden structures in the COLPLANT Screen house under shade for 2 days. They were then milled with a waring 2 speed Saisho blender after drying and repacked into new paper envelopes for analysis. Analysis of the three phytochemicals flavonoid lignin and tannin was carried out in the Biological Sciences Tet fund Laboratories, FUNAAB, using standard methods of the [12].
Seven minerals were analyzed in the leaves namely Calcium (Ca), Magnesium (Mg), Potassium (K), Nitrogen (N), Phosphorus (P), Iron (Fe) and Sulphur (S) using standard methods [12].
Yield at 6MAP
Careful harvest with smooth wooden spoons was undertaken to avoid bruising /wounding of tubers. Tuber number per plant was assessed after freeing of all tubers in a mound from soil. Thereafter they were left to dry for about 3 hours, when total tuber weight per mound was recorded with a top loading field mettler balance.
Infection and weight loss experiment
Triplicate tubers from treatment plots were also used in the infection and weight loss experiment. Inoculation of tubers was carried out in an Inoculation Hood Structure in the Crop Protection Laboratory, COLPLANT Building, FUNAAB. Three tubers per treatment were surface-sterilized by swabbing a point on the yam surface (inoculation site) with cotton wool dipped in 80% ethanol. Two cork borers (6mm and 4mm), scalpel and pair of forceps were surface-sterilized by dipping into 80% ethanol and flaming until red-hot over a lighted spirit flame bottle. They were thereafter cooled, slanting against another cork borer. The surface-sterilized 6mm borer was then used to bore a 10 to 12mm (depending on the size of the tuber) hole through the inoculation site in each tuber. The incised portion was lifted out with the sterilized scalpel and forceps. A 4mm disc from a 7-day old pure culture (growing on potato dextrose agar) of Botryodiplopodia theobromae in a 9cm petri-dish was removed with the sterile 4mm cork borer and placed inside the hole created in the tuber. The incised tissue was replaced and the incision on the inoculation site sealed with vaseline (petroleum jelly). Each tuber weight was recorded before inoculation. Inoculated tubers were transferred into the raised wooden netted and ventilated yam storage structures in the COLPLANT Screenhouse for a period of 2 weeks. After the 2week incubation period each tuber weight was again measured with a field scale. Percent weight loss in the tuber was determined with the formula:
%weightloss(Y)=(A-B)100/A
where A and B are the weight of the tuber before inoculation and at the end of incubation respectively. Percent infection was determined with the formula:
%Infection=(C/A)X100
where C and A are, corrected weight of infected tissue and weight of the tuber at the beginning of the experiment (before inoculation) respectively. Also “C” is determined with the
formula C= 100X/100-Y
where X and Y are, weight of infected tissue and percent weight loss respectively [13].
Data analysis
Percentage data obtained were transformed appropriately (arcsine, square root or logarithm) before analysis of variance. Means were separated with Turkey’s Studentized (HSD) test.
Results
Soil analysis
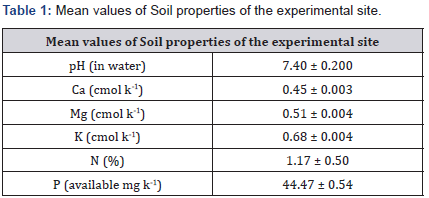
Nitrogen and Calcium in the field plot soil were 0.14% and 0.48cmol/kg-1 Table 1, below the critical required for optimum yam production in South West Nigeria [14]. Phosphorus, potassium and magnesium were 42.51mg/kg-1, 0.67cmol/kg-1 and 0.54cmol/kg-1 respectively Table 1.
Leaf tissue analysis of phytochemicals and minerals at 6MAP
Flavonoid content, 920mg/100gdm in the 1.20ml l-1 soil amendment treatment was higher than the control, 876.67mg/100gdm, and not significantly different from that of the 0.60ml/l-1 soil amendment (916.67 mg/100gdm) as shown in Table 2.
Leaf lignin content was also significantly higher 1943.33 mg/100gdm in the 0.60 ml/l-1 treatment than the control, which was 1763.33mg/100gdm Table 2. The 1.20 ml/l-1 treatment leaf lignin (1876.67 mg/100gdm) was not significantly different from the 0.60ml-1 treatment. Leaf tannin content in the 0.60 ml-1 soil amendment was higher (4.16mg/100gdm) than the control (3.39mg/100gdm). The control and the 1.20ml l-1 soil amendment treatments were not significantly different in leaf lignin. Leaf calcium and iron were not significantly different in the three treatments Table 2. The 0.60 and 1.20ml/l-1 soil amended plants were higher in leaf magnesium (234.73 and 230.27mg/100gdm) than the control which is 182.40mg/100gdm Table 2. Leaf nitrogen phosphorus and Sulphur increased with bounty fertilizer treatment, then the control Table 2 in the following manner:
a) Leaf nitrogen: 707.67mg/100gdm (control)<748.33mg/100gdm (0.60mll-1) <777.33mg/100gdm (1.20ml/l-1).
b) Leaf Phosphorus: 76.81mg/100gdm (control)< 76.95mg/100gdm (0.60mll-1) < 78.52mg/100gdm (1.20ml/l- 1).
c) Leaf sulphur: 809mg/100gdm (control)<834.33mg/100gdm (0.60mll-1) <854.33mg/100gdm (1.20ml/l-1).
Only leaf potassium content was higher in the control (116.72mg/100gdm) than in the bounty-fertilized plants which had 87.31mg/100gdm and 87.53mg/100gdm respectively in the 0.60ml/l-1 and 11.20ml/l-1 treatments Table 2.
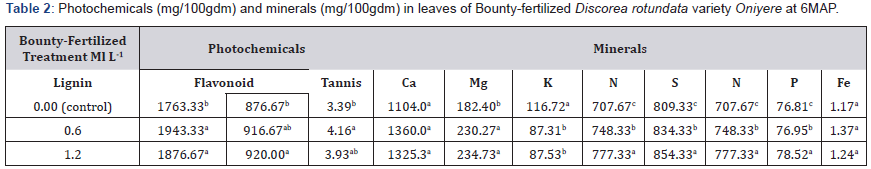
Means in a column followed by the same letter are not significantly different at P ≤ 0.05 (Tukey’s HSD test)
Disease incidence and severity
Incidence of Anthracnose was not high before the 3MAP soil amendment with Bounty fertilizer. After this, the plants were observed closely so as to administer foliar-spray of Ca(NO3)2 if necessary. However, anthracnose symptoms did not increase and at 5MAP was 11.11% in the control 5.56% in the 0.60mll-1 treated plants and 0% in the 1.20ml l-1 treated plants as shown in Table 3. It reduced further at 6MAP to 5.56%/ 5.57% in the control and 0.60ml l-1 treated plants and was still 0% in the higher treatment of 1.20 ml/l-1 Table 3. Anthracnose severity was low both at 5MAP and 6MAP, when all three treatments including the control had a score of I in the category of resistant Table 3.
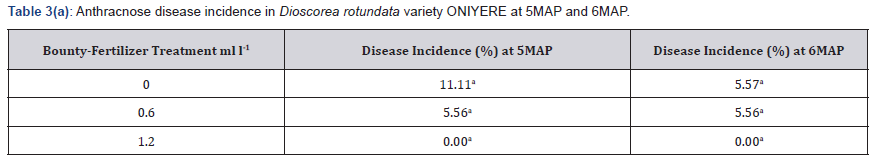
Means in a column followed by the same letter are not significantly different at P ≤ 0.05 (Tukey’s HSD test).
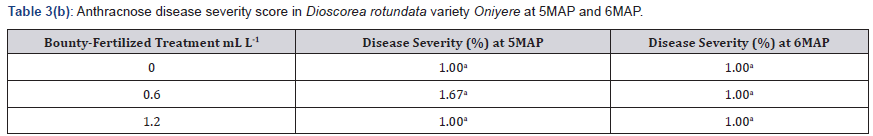
Means in a column followed by the same letter are not significantly different at P ≤ 0.05 (Tukey’s HSD test)
MAP = Months After Planting.
Correlations of anthracnose incidence/severity, leaf phytoanticipins, leaf minerals and yield
Only significant correlations in all parameters assessed are presented in Table 4. Leaf nitrogen correlated positively with leaf phosphorus and leaf Sulphur, r = 0.8484 and 0.9836. Leaf Nitrogen also correlated positively with leaf flavonoid content r = 0.7634. Leaf Calcium correlated positively with leaf magnesium, rr = 0.9192 while leaf magnesium correlated positively with leaf flavonoid content, r = 6786. Leaf flavonoid content correlated positively with leaf lignin content, r = 0.7189. Leaf lignin content correlated positively with leaf tannin content, r = 0.7556. Leaf Sulphur correlated both with flavonoid and phosphorus, r = 0.7218 and r = 0.8467. Leaf tannin content correlated negatively with disease incidence at 5 MAPS Table 4.
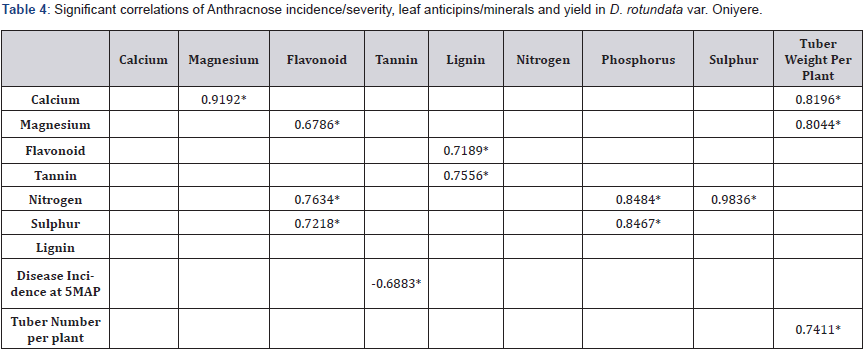
Significant Correlations at P = 0.001 or 0.05.
Infection and Weight loss experiment
Tubers inoculated and incubated with Botryodiplodia theobromae for 2 weeks had mean infection range of 1.213% to 2.95% across the three treatments. There were no significant differences in infection. Weight loss was 0% across all three treatments Table 5.
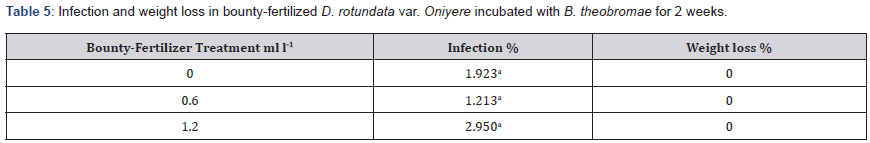
Means in a column followed by the same letter are not significantly different at P ≤ 0.05 (Tukey’s HSD test).
Yield at 6MAP
Tuber number per plant was in the range 1.443 to 2.333 Table 6. There were no significant differences across the three treatments. Tuber weight significantly correlated positively with tuber number per plant, r = 0.7411 Table 4. Tuber weight per plant range was 0.877kg to 1.347kg. There were no significant differences between treatments. Tuber weight in tons per hectare is 8.77 to 13.47tons ha-1 Table 4. Leaf calcium and leaf magnesium had significant positive correlation with tuber weight per plant, r = 8196 and r = 0.8044 respectively Table 6.
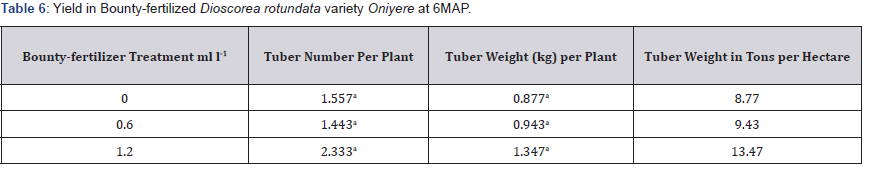
Means in a column followed by the same letter are not significantly different at P ≤ 0.05 (Tukey’s HSD test).
Discussion
The field plot used in this study had been monocropped to yams, Dioscorea species with calcium nitrate fertilizer, for two previous consecutive years (2015 and 2016). However, soil Nitrogen and Calcium were still below 2% and 0.65cmol/kg-1, critical levels determined for optimum yam production in South West Nigeria [14]. Lands fallowed for 2 -3 years in Nigeria may still require soil testing for mineral replenishment for optimum yam production. Phosphorus and potassium were however above the critical. Yam, Dioscorea species drain heavily on soil nutrients [3]. This fact informed the choice of fertilizer containing 8 minerals, coupled with the aim of the study which was to investigate the effect of mineral nutrition on yam anthracnose and tuber rot, especially on a field plot monocropped to Dioscorea species with only Ca(NO3)2 fertilizer for two previous consecutive years. Performed secondary metabolites or phytoanticipins such as flavonoid, lignin and tannins which are formed on the surface cell layers or within leaf cells have antimicrobial properties as they provide a chemical barrier that resists pathogen enzymatic degradation, or release toxic chemicals within the cells, conferring chemical protection against pathogen [15]. The three phytoanticipins (flavonoid, lignin, tannins) were higher in the leaves of the Bounty-fertilized plants in this study than the control. Furthermore, nitrogen and magnesium supplied from the fertilizer increased flavonoid content in the leaves (positive significant correlation). Nitrogen also promoted increase in phosphorus and Sulphur (both of which were not supplied from the fertilizer) in the leaves, while Sulphur in turn promoted leaf flavonoid content. Calcium supplied from the fertilizer promoted leaf magnesium content (significant positive correlation). Leaf flavonoid correlated significantly positively with lignin and lignin with tannin. The interplay/enhancement of the aforementioned minerals with phyto anticipins (three of them) and the latter with one another should be responsible for the low incidence of anthracnose at 5MAP and a lower incidence at 6MAP, and the resistance of the variety (severity score of 1 = resistant, at 5MAP and 6MAP). Anthracnose severity score, of 3, moderately susceptible was reported for an improved white yam variety D. rotundata TDr 131 at 6MAP, in the only report of use of Ca(NO3)2 foliar spray to control anthracnose in Dioscorea species in literature [11].
Mean infection in D. rotundata var. Oniyere tubers harvested at 6MAP, inoculated and incubated with B. theobromae for 2 weeks was 2.03%. This infection is 43% lower than that obtained in an improved variety of white guinea yam D. rotundata TDr 131 inoculated with B. theobromae after fertilization with CaCO3 and NPK [16]. Weight loss in Variety Oniyere in this study was 0% whereas D. rotundata TDr 131 had 3.4% weight loss in the cited report [16]. Bounty fertilizer with eight minerals may be recommended for management of both anthracnose disease and tuber rot by B. theobromae in D. rotundata var. Oniyere. At the 6 MAP harvest, tuber weight was 0.877kg, 0.94kg and 1.347kg per plant in the control, 0.60 ml/l-1 and 1.20ml/l-1 Bounty-fertilized plants respectively. The higher treatment of 1.2ml/l-1 Boutny fertilizer gave the yield equivalent of 13.47tons ha-1 which is higher than the 9-12 metric tons/ha in smallholder yam cultivation in shifting agricultural systems without external input in West Africa [17], and it is within the expected range of 10-15t/ha for white guinea yam under good management of NPK/NPK+Urea [18]. Bounty fertilizer at 1.20ml/l-1 is recommended for the production of local white guinea yam varieties such as variety Oniyere by smallholder farmers in Nigeria, after soil testing, especially where monocoping is desired.
References
- FAO (2014) FAOSTAT. Crop Production data.
- Carsky RJ, Wolo N, Manyong VM, Tian G (2001) Proceedings of the International Society for Tropical Root Crops-Africa Branch, Cotonou, Benin.
- Obigbesan GO, Agboola AA (1978) Uptake and distribution of nutrients by yams (Dioscorea spp.) in Western Nigeria. Experimental Agriculture 14(4): 349-355.
- Taiga A (2011) Comparative studies of the efficiency of some selected fungicidal aqueous plant extracts on yam tuber dry rot disease. Annals of Biological Research 2(2): 332-336.
- Green KR (1994) Studies on the epidemiology and control of Yam Anthracnose Ph.D. Thesis, University of Reading, UK, pp. 167.
- Nwankiti AO, Ene LSO (1984) Advancers in the study of anthracnose/ blotch disease of Dioscorea alata in Nigeria. In: Shilder FS, & Rincon H (Eds.), Proceedings of the 6th Symposium of the international society of Tropical Root Crops, Lima, Peru: 633-640.
- Dordas C (2008) Role of Nutrients in controlling plant disease in sustainable Agriculture: A review. In: Lichtfouse E (Ed), Sustainable Agriculture, springer Science -Business, Media BV, EDP Sciences 2009.
- Graham RD, Welch RM, Bouis HE (2001) Addressing micronutrients malnutrition through enhancing the nutritional quality of staple foods: Principles perspectives and knowledge gaps. Advances in Agronomy 70: 77-142.
- White P J, Broadley MR (2003) Calcium in plants. Annals of Botany 92(4): 487-511.
- Page AL, Miller R, Keeney D (1982) Methods of soil analysis. Madison, ASA-SSSA: 199-224.
- Otusanya MO, Enikuomehin O, Popoola A, Adetunji M, Kehinde O, et al. (2017) Effects of foliar application of calcium nitrate on the susceptibility of Dioscorea species to yam anthracnose caused by colletotrichum gloeosporiodes Penz (Penz and Sacc.). Journal of Plant Physiology and pathology.
- AOAC (2001) Association of Official Analytical Chemist, Official Methods of Analysis(edn.). II Washington DC, USA.
- Otusanya M O, Jeger M J (1994) Infection of Yam Tubers by Aspergillus niger in relation to Isolate, Yam Species and Temperatures. International Biodeterioration and Biodegradation 33(4): 319-331
- Ibedu MA, Unambra KRP, Udealor A (1988) Soil management strategies in relation to farming system development in Southwestern agricultural zone of Nigeria. Papers presented at the National Farming System Research Workshop, Jos, Plateau State, Nigeria: 22-29.
- Dube H C (2014) Modern Plant Pathology, (2nd edn.), Saraswati Purohit, Jodhpur, India, pp. 576.
- Otusanya MO, Enikuomehin O, Popoola A, Adetunji M, Kehinde O, et al. (2016) Effects of calcium fertilization on the susceptibility of Dioscorea species to the yam storage pathogens Aspergillus niger van Tiegh and Botryodiplodia theobromae pat. Journal of Plant Physiology and pathology 4(3).
- Gondo E, Diby NL, Nindjin C, Soro D, Dao D, et al. (2003) Selection participative de nouvelles varieties d’igname (Dioscorea sp.) dans la region de la vallae du Bandaman. Agronomie Africaine Numero special (4): 9-14.
- ICS-Nigeria (2017) Information and Communication Support for Agricultural Growth in Nigeria.






























