Adipose Derived-Stem Cells and their Cell-Free Supernatant Combined with a Nanofiber Scaffold for Skin Rejuvenation: The Avancell Stem Cell SecretomeTM - A Technical Note
Patrick Weninger*, Murat Özcelik and Angelika Reich
Avancell Medical Group, Liechtensteinstrasse 96, 1090 Vienna, Austria
Submission: December 30, 2024;Published: January 09, 2024
*Corresponding author: Patrick Weninger, Avancell Medical, Liechtensteinstrasse 96 1090 Vienna, Austria
How to cite this article: Patrick W, Murat Ö, Angelika R. Adipose Derived-Stem Cells and their Cell-Free Supernatant Combined with a Nanofiber Scaffold for Skin Rejuvenation: The Avancell Stem Cell SecretomeTM - A Technical Note. JOJ Dermatol & Cosmet. 2024; 5(5): 555672. DOI: 10.19080/JOJDC.2024.05.555672
Keywords: Patrick Weninger, Avancell Medical, Liechtensteinstrasse 96 1090 Vienna, Austria
Introduction
The skin is the largest body organ and acts as the first defense line against various external agents and stimuli such as toxins, ultraviolet radiation or environmental agents [1,2]. With the process of ageing, skin loses collagen and elasticity and is less hydrated and less plump [3,4]. These effects cause wrinkles and sagging of the skin which is not well tolerated in our society [5]. Therefore, several strategies are available to enhance aesthetic appearance of the ageing skin. Beauty industry was worth 511 billion US-Dollars and the annual total revenue is expected to rise to 716 billion by 2025. Skincare alone accounted for 42 percent of the global market. Monthly, Americans spend about 300 US-Dollars on cosmetic products. The Asian-Pacific and the North-American regions account for more than 60 percent of the global cosmetic market. L’Oreal, the largest beauty company in the industry, made 34.4 billion US-Dollar in 2019. 2021 marked the best year ever for cosmetics and the beauty industry revenue is expected to surpass 120 billion US-Dollars by 2025 [6]. During the past years, Ortho biological treatment strategies started to emerge continuously in the aesthetic field [7-9]. Adipose tissue has recently been identified as a promising source of multipotent cells for use in regenerative medicine [10,11]. Adipose-derived stem cells (ASCs) are cells of mesenchymal origin with a capacity to differentiate through adipogenicity, osteogenic, and chondrogenic lineages, among others [12]. In contrast to bone marrow-derived mesenchymal stem cells (BM-MSCs), ASC derived from adipose tissue are abundant, relatively easily obtainable and is not limited by the donor’s age [13,14]. Due to their high yield in adipose tissue, ASCs additionally have the potential to be used in clinical therapy without the need for expansion in culture [15]. The potential utility of ASCs in tissue engineering and cell-based regenerative therapies has been confirmed in a variety of pre-clinical and clinical applications. For example, pullulan-collagen hydrogel scaffolds seeded with ASCs have been demonstrated to increase vascularity and improve wound healing [16]. About skeletal regenerative potential, ASCs have the potential to heal critical size calvarial defects in murines [17,18]. Finally, the adipogenic and angiogenic capabilities of ASCs have been utilized in the technique of cell-assisted lipotransfer, in which fat grafts are enriched with their native ASCs to improve retention and variability [19]. Also, for wound healing, ASCs-based therapies were reported in the past [20-22]. Skin wound healing and skin regeneration is a complex and well-orchestrated regenerative event by multiple cells in the epidermal and dermal layers to re-establish the barrier. Upon injury, keratinocytes migrate from the basal layer to the wound area and proliferate for the restoration of the epidermal layer. Fibroblasts also move to the wound site in a spatio-temporal manner and release soluble factors such as growth factors and cytokines and produce the extracellular matrix to reconstitute the dermal layer [23,24].
Skin Rejuvenation, ASCs and ASCs-derived secretome
Aged skin is highly associated with an unpleasing esthetic, which occurs due to loss of function and structural degeneration of the skin. This can result in more serious complications, including more susceptibility to diseases such as eczema, dermatitis, autoimmune disorders, and melanoma [25,26]. With ageing, the skin naturally loses its collagen content and elastic fibers become deranged. Additionally, aged skin demonstrates an increase in oxidant activity, and increase in the production of matrix metalloproteases (MMP) which are typically involved in matrix degeneration [27]. Additionally, exposure to UV lights is known to promote premature aging of the skin, namely photoaging [28]. Thus, rejuvenation therapies, which focus on the prevention and reversal of skin aging are in high demand in our society, which increasingly aims to maintain a youthful appearance and improve their health. ASCs have been gaining attention in skin antiaging therapy because of their efficient re-epithelialization and secretion of several growth factors necessary for skin regeneration [29-31]. Charles-de-Sa et al [32] observed the histological and structural modifications in aged facial skin after the injection of expanded ASCs, collected from fat removed by liposuction. Treatment with ASCs caused an increase in elastic fibers in the superficial layer of the dermis and modified the collagen and reticular fiber networks, which became more arranged. Subsequently, ASCs were observed to induce complete regeneration of solar elastosis of photoaged skin. Another way to use stem cells in skin disease therapy and also in skin antiaging therapy, in a “cell-free” method of treatment, is by using its supernatants (secretome) such as extra-cellular vehicles (EVs, exosomes), endosomes, proteins or mRNA [33-35].
EV or exosomes are natural nanoparticles enclosed in a lipid bilayer that are released in response to the microenvironment. EVs deliver their membrane signals or internal contents of parental cells, including nucleic acids, lipids, mitochondria, and proteins, into the target cell, resulting in the physiological changes in the target [36]. Among EV subpopulation, exosome is gaining increasing interests in immune modulation and regenerative therapy [37-39]. ASCs EV have anti-photoaging potential and were analyzed as subcutaneous injections in photoaged mice models. The treatment resulted in a decrease in skin wrinkles and promotion of epidermal cell proliferation. Additionally, macrophage infiltration and reactive oxygen species production were reduced, which inhibited MMP activation and collagen degradation. Moreover, in vitro analysis showed increased fibroblast activity and protection from UVB-induced senescence [40]. Recently, it has been shown that some rare skin disorders, such as vitiligo or alopecia might be treatable by topic use of ASCs and secretomes derived from ASCs [41,42]. Accordingly, stem cell exosomes and the secretome are stem cell-derived regenerative mediators for skin rejuvenation. These secretomes have several advantages over stem cells and their safety issues. In contrast to stem cells, the stem cell-derived secretome cannot self-replicate [36-38]. Secretomes are also stable enough for long-term frozen storage and storage at room temperature after lyophilization. The smaller size further allows sterilization by filtration [37]. In addition, exosomes can be administered by several routes; for example, nebulized or lyophilized lung stem cell-derived exosomes can be administered by inhalation to treat lung diseases [43]. Moreover, their hydrophilic lumen and phospholipid bilayer containing membrane proteins can be engineered and modified to display molecules or for drug loading [44].
Nanofiber-Scaffolds and ASCs
Nanofibers are polymeric biomaterials that are created by spinning. Currently, different spinning methods are used for nanofiber fabrication [45]. Nanofibers have been observed to be an optimal scaffold for stem cells in pre-clinical and clinical studies [46]. Be it wound healing, myocardial regeneration, bone healing or nerve regeneration, nanofiber scaffolds enhance cell performance, quality of extracellular matrix speed up tissue regeneration and avoid tissue degeneration [47,48]. Recently, it was reported that epidermal stem cells maintain stemness via a biomimetic micro/nanofiber scaffold that promotes wound healing by activating the Notch signaling pathway [49]. Especially for wound healing, the use of nanofiber-acellular dermal matrix as a bilayer scaffolding containing mesenchymal stem cells for healing of full-thickness skin wounds was described [50]. Recently, nanofibers have been described to be optimal as drug delivery systems [51]. Also, it has been shown that collagen-based nanofibers can be used for skin regeneration and promote wound healing [52]. Furthermore, combining ASCs and their exosomes or secretomes with nanofiber technology has also been shown to have promising results for skin rejuvenation and /or wound healing [53]. In this publication, we describe a technique for in-office use of ASCs and ASCs-derived secretomes which are applied to the skin layers by using a nanofiber scaffold for skin rejuvenation. Furthermore, we describe a technique to create the Avancell Stem Cell Secretome which contains the supernatants of ASCs and other ingredients for skin-rejuvenation to allow at-home application combined with the nanofiber scaffold (“skin patching”).
Technique
Harvesting ASCs and in-office application
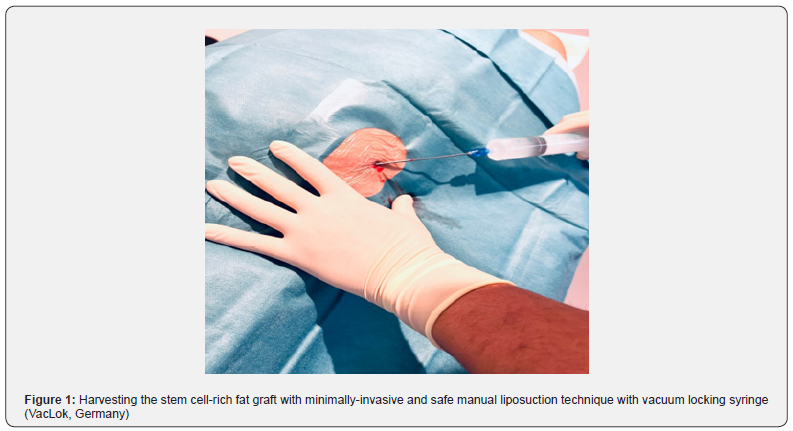
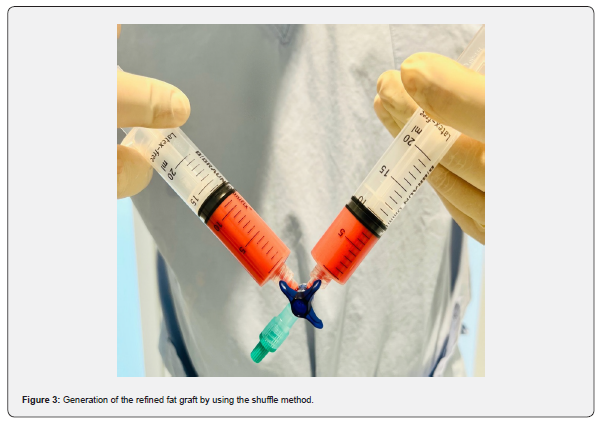
The technique has recently been described by the first author for cartilage regeneration [54-56]. First, the patient is placed in supine position for harvesting the autologous abdominal fat. Peri-operative intra-venous singleshot antibiotic (Cephazoline) is administered 15 minutes preoperatively. The abdominal area is then draped with square drapes and sterile washing is performed. Lipoaspirate is harvested in regional anesthesia using 1% lidocaine and epinephrine 1:10,000 from the lower abdominal region or from the flank. After infiltrating 100 milliliters of tumescent solution (1000 mL of saline solution, 1 mL of 1:200,000 adrenalin, and 600 mg of lidocaine and natrium bicarbonate), The fat graft is harvested. For harvesting, a 20mL syringe is connected to a blunt steel liposuction cannula of 3mm diameter (Tulip Medical, USA). The cannula is inserted into the subcutaneous fat and by aspirating the syringe a vacuum is obtained. Liposuction then is performed by back-and-forth motions of the cannula, oriented parallel to the underlying fascia. Meanwhile, the non-dominant hand applies pressure to the abdominal wall ensuring depth and direction of the cannula (Figure 1). The fat tissue is separated from fluids and oils using the decanting method without the need for centrifuged (Figure 2). Therefore, the syringes are placed upright and after a few minutes the lipoaspirate separates. The fluid layer then is decanted by disposing it out of the syringe. Next, the fat is mechanically homogenized with two syringes (shuffling method): One fat-containing syringe is connected to a second, empty syringe by a three-way valve. The fat is shuffled by transferring the content of one syringe to the other one 10 times, thus creating a refined fat graft (Figure 3). It is generally accepted that these refined fat grafts contain high numbers of ASCs and that there is no need to expand such in cultures. A single-use 20-gauge needle is used to inject 10 mL of the ASC graft onto the nanofiber patch. In case of in-office use, the ASC graft will be applied topically to the skin as the stem cell-rich fat graft will be processed immediately and no storage is needed. Consequently, no legislative rules for tissue banking are required or need to be obeyed.

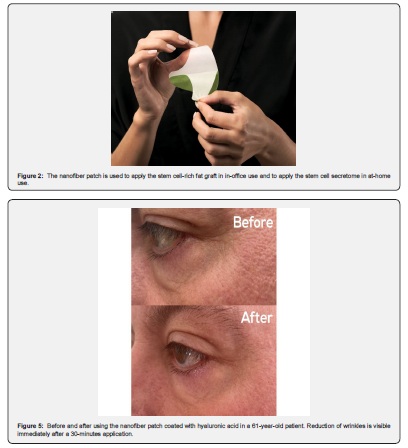
ASCs-derived secretome and at-home application
We use an easy and reproducible technique to further process the ASC graft to elude the cell-free ASC supernatant. Therefore, we add PBS buffer and collagenase to the graft and then centrifuge it three times in a standard centrifuge (Hettich Rotofix 32A, Germany). The supernatant of the graft is isolated by using this technique. In a third step, a VialTweeter (Hielscher Diagnostics, Germany) is used to disrupt the stem cells and to ensure the production of a cell free ASC-derived secretome. Then, after lyophilization, the serum can be stored even at room temperature. In total, 8 mL of ASC-derived secretome are isolated. Of this, 1 mL is used to create the Avancell Stem Cell SecretomeTM: 1 mL of ASC-derived secretome is mixed with 10 mL glycerine, 12 mL of hyaluronic acid and 2 mL of stabilizers to process a 25 mL product available for topic application. On two days per month (every two weeks), we recommend applying 1 mL of the concentrate for dermal application at anybody location (predominantly skin of the face or decolletee). 7 mL of the ASC supernatant will be stored and frozen at our laboratories for future use (for the next seven years). The 1 mL ASCs-derived supernatant will be thawed and immediately used to prepare the serum. The serum can be picked up at one of our Avancell Skin Rejuvenation Centers together with the Nanofiber patches. In total, 24 Nanofiber patches are needed annually to apply the serum with the patch (two times per month).
Nanofiber Patch
The here described and used nanofiber patch was designed for versatile nanoencapsulation of cosmetic ingredients. The technology is based on electrostatic spinning technology (electrospinning), using a set proprietary so called NEUNEX production unit (Respilon, Austria, www.respilon.com). The NEUNEX technology enables continuous formation of high-quality core/shell nanofibers, which may combine in them both water-soluble and oil-soluble molecules. The technology is based on the formation of emulsions and dispersions with high stability, which guarantees homogeneity of the nanofibers produced. In typical case, the water-soluble active polymers (i.e., collagen and pullulan) and water-soluble active molecules create the continuous phase of the final nanofiber. The oil soluble molecules (i.e., essential oils, vitamins, oil soluble drugs) are dispersed in the emulsion as droplets, which results in the formation of emulsions. During the electrospinning process, the polymeric liquid is stretched by using electrostatic forces to form nanofibers. Due to a high surface area of nanofibers, the solvent (typically water) is evaporated in the production process resulting in the creation of a dry nanofiber web. Thanks to the NUENEX technology, these formulations can be created with an almost indefinite number of combinations of polymers and small molecules with different chemical properties (i.e., water- and oil-soluble) in high concentrations. The secretome can be attached to the patch to allow it to migrate into deeper skin layers where collagen and elastine synthesis will occur. We recommend applying the nanofiber patch with Avancell Stem Cell SecretomeTM for 30 minutes in each body region. Body regions can be facial skin, thighs, hands or any region (Figure 4,5).
Discussion
Senescence of the skin is an inevitable process and is a combination of cell death, limited cell turns over and loss of interstitial fluid and collagen and elastic fibres [57]. The wish for young-looking skin has unleashed a billion-dollar business in the past decades [6]. All strategies, be it conservative or operative, have two major goals: 1. Less wrinkles and 2. Less sagging of the skin. Rejuvenation of the ageing skin has been a matter of research during past years. To the best of our knowledge, the technique described herein has never been described before. It combines the technological advances of nanofiber technology with the use of ASCs-derived secretome for skin rejuvenation to allow for secretome infiltration to deeper skin layers. The secretome is a bioactive complex that contains many growths factors, cytokines, mRNA, exosomes, and endosomes that can stimulate various biological reactions mainly to modulate new tissue formation. The relevance of stem cell-derived secretomes first was described for wound healing by Shabbir et al. and by Hu et al. [58,59]. Also, Raik et al. gives a very good overview of a cell-free therapeutic approach using supernatants [60]. Kim et al. reported on exosomes from umbilical cord blood stem cells and that they stimulate rejuvenation of the human skin [61]. The use of secretomes for skin rejuvenation has been described recently [62]. In this context, Liu et al. also highlighted the regenerative potency of ASCs-derived secretomes for skin rejuvenation [63]. The regenerative efficacy of stem cell derived secretomes was also pointed out for patients suffering from psoriasis and vitiligo. Bellei et al. [64] found that what they called “adipose tissue extracellular fraction” could be useful as a natural approach to improve treatment of vitiligo. Also, Bellei et al [41]. described the relevance of the secretome in complex conditions such as atopic dermatitis, acne, lichen sclerosus, non-resolving wounds and alopecia.
The role of nanofiber materials as scaffold for medical applications is well known. Kistha et al [65]. found nanofiber scaffolds may be a helpful tool in myocardial regeneration when combined with adipose-derived stem cells. Lin et al. [49] were able to show that epidermal stem cells maintain stemness via a biomimetic micro/nanofiber scaffold that promotes wound healing. Same results were found for liver regeneration and bone healing [66,67]. Also, neuronal regeneration was matter of research in the past and it was shown that nanofiber materials are also able to promote neuronal regeneration [68]. The combination of nanofibers and ASCs was recently pointed out for chronic wounds [53,69]. Also, it has recently been pointed out that stem cell-derived supernatants seem to be promising for skin regeneration and that they may preclude the need for a more expensive and difficult cell therapy approach [70]. In the method described here, the combination of nanofibers and ASCs-derived secretome was described for the first time. It is a reproducible and relatively easy way to produce and to process a fat graft to generate the ASCs-derived secretome which can be used for skin rejuvenation. The same technique fat graft harvesting was recently described by the first author who treats patients with cartilage defects and osteoarthritis [54]. In this context, we state that only a few patients had mild hematoma at the abdominal harvesting site and therefore we can conclude that the procedure is safe. The product can be used as an in-office or as an at-home application. From a marketing point of view, it is the first and only product now that uses autologous cell-free ASCs secretomes as a skincare product. Other skincare products that promote “stem cells” or “secretomes” or “exosomes” do not use them at all and only use fancy names that suggest their use.
Conclusion
This is the first and now the only personalized skincare product using autologous ASCs-derived secretomes in combination with nanofiber technology patches. The first applications were encouraging and the harvesting procedure as well as the patching procedure are safe. We are currently conducting a large study to evaluate the effect of this treatment method.
References
- Zhang S, Duan E (2018) Fighting against skin aging: The way from bench to bedside. Cell Transplant 27(5): 729-738.
- Zouboulis CC, Hoenig LJ (2019) Skin aging revisited. Clin Dermatol 37(4): 293-295.
- Bonte F, Girard D, Archambault JC, Alexis D (2019) Skin changes during aging. Sub cell Biochem 91: 249-280.
- Lowry WE (2020) It’s written all over your face: the molecular and physiological consequences of skin aging. Mech Ageing Dev 190: 111315.
- Zouboulis CC, Ganceviciene R, Liakou AI, Athanasios T, Rana E, et al. (2019) Aesthetic aspects of skin aging, prevention, and local treatment. Clin Dermatol 37(4): 365-372.
- Cosmetic Market Size and Share Analysis - Growth Trends and Forecasts (2024 - 2029).
- Maisel-Campbell AL, Ismail A, Reynolds KA, Emily P, Linda S, et al. (2020) A systematic review of the safety and effectiveness of platelet-rich plasma (PRP) for skin aging. Arch Dermatol Res 312(5): 301-315.
- Peng GL (2019) Platelet-rich plasma for skin rejuvenation: Facts, fiction, and pearls for practice. Facial Plast Surg Clin North Am 27(3): 405-411.
- Hesseler MJ, Shyam N (2019) Platelet-rich plasma and its utility in medical dermatology: A systematic review. J Am Acad Dermatol 81(3): 834-846.
- Mazini L, Rochette L, Admou B, Said A, Gabriel M (2020) Hopes and limits of adipose-derived stem cells (ADSCs) in wound healing. Int J Mol Sci 21(4): 1306.
- Bacakova L, Zarubova J, Travnickova M, Jana M, Julia P, et al. (2018) Stem cells: their source, potency and use in regenerative therapies with focus on adipose-derived stem cells - a review. Biotechnol Adv 36(4): 1111-1126.
- Sheykhhasan M, Wong JKL, Seifalian AM (2019) Human adipose-derived stem cells with great therapeutic potential. Curr Stem Cell Res 14(7): 532-548.
- Strioga M, Viswanathan S, Darinskas A, Ondrej S, Jaroslav M, et al. (2012) Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Dev 21(14): 2724-2752.
- Dufrane D (2017) Impact of age on human adipose stem cells for bone tissue engineering. Cell Transplant 26(9): 1496-1504.
- Si Z, Wang X, Sun C, Yuchun K, Jiakun X, et al. (2019) Adipose-derived stem cells: Sources, potency, and implications for regenerative therapies. Biomed Pharmacother 114: 108765.
- Barrera JA, Trotsyuk AA, Maan ZN, Clark AB, Madelyn RL, et al. (2021) Adipose-derived stromal cells seeded in pullulan-collagen hydrogels improve healing in murine burns. Tissue Eng Part A 27: 844-856.
- Levi B, James AW, Nelson ER, Dean V, Min Lee, et al. (2010) Human adipose derived stromal cells heal critical size mouse calvarial defects. PLoS One 5(6): 11177.
- Tajima S, Tobita M, Mizuno H (2018) Bone regeneration with a combination of adipose-derived stem cells and platelet-rich plasma. Methods Mol Biol 1773: 261-272.
- Toyserkani NM, Quaade ML, Sorensen JA (2016) Cell-assisted lipotransfer: a systematic review of its efficacy. Aesthetic Plast Surg 40(2): 309-318.
- Zhang B, Wu Y, Mori M, Kotaro Y (2022) Adipose-derived stem cell conditioned medium and wound healing: a systematic review. Tissue Eng Part B Rev 28(4): 830-847.
- Toyserkani MM, Christensen ML, Sheikh SP, Jems Ahm S (2015) Adipose-derived stem cells: New treatment for wound healing? Ann Plast Surg 75(1): 117-123.
- Kuo YR, Wang CT, Cheng JT, Gour-Shenq K, Yuan-Cheng C, et al. (2016) Adipose-derived stem cells accelerate diabetic wound healing through the induction of autocrine and paracrine effects. Cell Transplant 25(1): 71-81.
- Rodrigues M, Kosaric N, Bonham CA, Geoffrey CG (2019) Wound healing: a cellular perspective. Physiol Rev 99(1): 665-706.
- Zomer HD, Trentin AG (2018) Skin wound healing in humans and mice: Challenges in translational research. J Dermatol Sci 90(1): 3-12.
- Khavkin J, Ellis DA (2011) Aging skin: histology, physiology, and pathology. Facial Plast Surg North Am 19(2): 229-234.
- Zhang S, Duan E (2018) Fighting against skin aging: the way from bench to bedside. Cell Transplant 27(5): 729-738.
- Lee H, Hong Y, Kim M (2021) Structural and functional changes and possible molecular mechanisms in aged skin. Int J Mol Sci 22(22): 12489.
- Fisher GJ, Kang S, Varani J, Zsuzsanna B, Yinsheng W, et al. (2002) Mechanisms of photoaging and chronological skin aging. Arch Dermatol 138(11): 1462-1470.
- Park BS, Jang KA, Sung JH, Jeong Soo P, Yong Hyon K, et al. (2008) Adipose-derived stem cells and their secretory factors as promising therapy for skin aging. Dermatol Surg 34(10): 1323-1326.
- Jo H, Brito S, Kwak BM, Sangkyu P, Mi-Gi L, et al. (2021) Applications of mesenchymal stem cells in skin regeneration and rejuvenation. Int J Mol Sci 22(5): 2410.
- Gaur M, Dobke M, Lunyak VV (2017) Mesenchymal stem cells from adipose tissue in clinical applications for dermatological indications and skin aging. Int J Mol Sci 18(1): 208.
- Charles-de-Sa L, Gontijo-de-Amorim NF, Rigotti G, Andrea S, Paolo B, et al. (2020) Photoaged skin therapy with adipose-derived stem cells. Plast Reconstr Surg 145(6): 1037e-1049e.
- Maguire G (2019) The safe and efficacious use of secretome from fibroblasts and adipose-derived (but not bone marrow-derived) mesenchymal stem cells for skin therapeutics. J Clin Aesthet Dermatol 12(8): E57-E69.
- Hani R, Khayat L, Rahman AA, Alaaeddine N (2023) Effect of stem cell secretome in skin rejuvenation: a narrative review. Mol Biol Rep 50(9): 7745-7758.
- Kim H, Lee JW, Han G, Kwangmeyung K, Yoosoo Y, et al. (2021) Extracellular vesicles as potential theranostic platforms for skin diseases and aging 13(5): 760.
- Kalluri R, LeBleu VS (2020) The biology, function, and biomedical applications of exosomes. Science. 367: eaau6977.
- Hade MD, Suire CN, Suo Z (2021) Mesenchymal stem cell-derived exosomes: applications in regenerative medicine. Cells 10: 1959
- An Y, Lin S, Tan X, Zhu S, Nie F (2021) Exosomes from adipose-derived stem cells and application to skin wound healing. Cell Prolif 54: e12993.
- Ha DH, Kim HK, Lee J, Hoon Kwon H, Park GH, et al. (2020) Mesenchymal stem/stromal cell-derived exosomes for immunomodulatory therapeutics and skin regeneration. Cells 9: 1157.
- Hu S, Li Z, Cores J, Ke Huang , Teng Su, et al. (2019) Needle-free injection of exosomes derived from human dermal fibroblast spheroids ameliorates skin photoaging. ACS Nano 13: 11272-11282.
- Bellei B, Migliano E, Picardo M (2022) Therapeutic potential of adipose tissue-derivates in modern dermatology. Exp Dermatol 31: 1837-1852.
- Li Y, Wang G, Wang Q et al. (2022) Exosomes secreted from adipose-derived stem cells are a potential treatment agent for immune-mediated alopecia. J Immunol Res 2022: 7471246.
- Dinh PC, Paudel D, Brochu H, Popowski KD, Gracieux MC et al. (2020) Inhalation of lung spheroid cell secretome and exosomes promotes lung repair in pulmonary fibrosis. Nat Commun 11: 1064
- Liang Y, Duan L, Lu J, Xia J, (2021) Engineering exosomes for targeted drug delivery. Theranostics 11: 3183-3195.
- Dos Santos DM, Correa DS, Medeiros ES, Oliveira JE (2020) Advances in functional polymer nanofibers: from spinning fabrication techniques to recent biomedical applications. ACS Appl Mater Interfaces 12: 45673-45701.
- Kai D, Jin G, Prabhakaran MP, Ramakrishna S (2013) Electrospun synthetic and natural nanofibers for regenerative medicine and stem cells. Biotechnol J 8: 59-72.
- Gholizadeh-Galeh Aziz S, Gholizadeh-Galeh Aziz S, Akbarzadeh A (2016) The potential of nanofibers in tissue engineering and stem cell therapy. Artif Cells Nanomed Biotechnol 44: 1195-1200.
- Ingavle GC, Leach JK (2014) Advancements in electrospinning of polymeric nanofibrous scaffolds for tissue engineering. Tissue Eng Part B Rev. 20: 277-293
- Lin Z, Zhao C, Lei Z et al. (2021) Epidermal stem cells maintain their stemness via a biomimetic micro/nanofiber scaffold that promotes wound healing by activating the Notch signaling pathway. Stem Cell Res Ther 12: 341.
- Mirzaei-Parsa MJ, Ghanbari H, Alipoor B, Tavakoli A, Najafabadi MRH, et al. (2019) Nanofiber-acellular dermal matrix as a bilayer scaffold containing mesenchymal stem cells for healing of full-thickness skin wounds. Cell Tissue Res 375: 709-721
- Duan X, Chen HL, Guo C (2022) Polymeric nanofibers for drug delivery applications: a recent review. J Mater Sci Mater Med 33: 78.
- Mbese Z, Alven S, Aderibigbe BA (2021) Collagen-based nanofibers for skin regeneration and wound dressing applications. Polymers (Basel). 13: 4368
- Liu Y, Liu Y, Zhao Y, Wu M, Mao S et al. (2020) Application of adipose mesenchymal stem cell-derived exosomes-loaded ß-chitin nanofiber hydrogel for wound healing. Folia Histochem Cytobiol 60: 167-178.
- Weninger P, Feichtinger X, Steffel C et al. Arthroscopy with lipoaspirate and plasma infiltration using adipose-derived stem cells plus platelet-rich plasma: harvesting and injection for arthroscopic treatment for cartilage defects of the knee. Arthrosc Techn. 2023. in press
- Weninger P (2023) Joint preservation in patients with grade-IV osteoarthritis of the knee-12-months results after a multimodal approach using radiofrequency ablation and platelet-rich plasma supercharged with stem cell-rich fat graft in 42 consecutive patients. J Surg 8: 1914
- Weninger P. How to avoid knee arthroplasty in patients with grade IV osteoarthritis – combination of adipose-derived stem cells and radiofrequency ablation. A case series of 30 consecutive patients with 12-months follow-up. Ortho & Rheum Open Access J. 2023, 22: 556089
- Franco AC, Aveleira C, Cavadas C (2022) Skin senescence: mechanisms and impact on whole-body aging. Trends Mol Med. 28: 97-109.
- Shabbir A, Cox A, Rodriguez-Menocal L, Salgado M, Van Badiavas E, et al. (2015) Mesenchymal stem cell exosomes induce proliferation and migration of normal and chronic wound fibroblasts and enhance angiogenesis in vitro. Stem Cells Dev 24: 1635-1647.
- Hu Y, Rao SS, Wang ZX, Jia Cao, Yi-Juan Tan, et al. (2018) Exosomes from umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function. Theranostics 8: 169-184.
- Raik S, Kumar A, Bhattacharyya A (2018) Insights into cell-free therapeutic approach: Role of stem cell “soup-ernatant”. Biotechnol Appl Biochem 65: 104-118.
- Kim S, Lee SK, Kim H, Kim TM (2018) Exosomes secreted from induced pluripotent stem cell-derived mesenchymal stem cells accelerate skin cell proliferation. Int J Mol Sci 19: 3119.
- Hani R, Khayat R, Rahman AA et al. (2023) Effect of stem cell secretome in skin rejuvenation: a narrative review. Mol Biol Rep 50: 7745-7758.
- Liu M, Lu F, Feng J (2023) Therapeutic potential of adipose tissue derivatives in skin photoaging. Regenerative Medicine 18 (11): 869-883
- Bellei B, Papaccio F, Filoni A, Caputo S, Lopez G, et al. (2019) Extracellular fraction of adipose tissue as an innovative regenerative approach for vitiligo treatment. Exp Dermatol 28: 695-703.
- Kishta MS, Ahmed HH, Ali MAM, A Aglan H, Ragaa Mohamed M, et al. (2022) Mesenchymal stem cells seeded onto nanofiber scaffold for myocardial regeneration. Biotech Histochem 97: 322-333.
- Morelli S, Piscioneri A, Salerno S, De Bartolo L, et al. (2022) Hollow fiber and nanofiber membranes in bioartificial liver and neuronal tissue engineering. Cell Tissues Organs 211: 447-476.
- Lim DJ (2022) Cross-linking agents for electrospinning-based bone tissue engineering. Int J Mol Sci 23(10): 5444.
- Shi S, Ou X, Cheng D (2023) How advanced is peripheral nerve regeneration using nanofiber scaffolds? A comprehensive review of the literature. Int J Nanomedicine 18: 6763-6779.
- Liu Y, Liu Y, Wu M, Zou R, Mao S, et al. (2022) Adipose-derived mesenchymal stem cell-loaded ß-chitin nanofiber hydrogel promote wound healing in rats. J Mater Sci Mater Med 33: 12.
- Kraskiewicz H, Paprocka M, Bielawska-Pohl A et al. (2020) Can be supernatant from immortalized adipose tissue MSC replace cell therapy? An in vitro study in chronic wounds model. Stem Cell Res Ther 11: 29.






























