Skin Penetration Ability of 12 Hyaluronic Acids with Different Molecular Weights After Topical Application
Silvana Giardina* and Andrea Poggi
Complife Italia srl, Via G. Rossa, Milan, Italy
Submission: July 04, 2023;Published: July 19, 2023
*Corresponding author: Silvana Giardina, Complife Italia srl, Via G. Rossa, 1, 20024 Garbagnate Milanese MI
How to cite this article: Giardina S, Poggi A. Skin Penetration Ability of 12 Hyaluronic Acids with Different Molecular Weights After Topical Application. JOJ Dermatol & Cosmet. 2023; 5(3): 555665. DOI: 10.19080/JOJDC.2023.05.555665
Abstract
Objective: Hyaluronic acid (HA) is a fundamental component of the extracellular matrix, regulating numerous biological processes. In humans, almost half of the total HA concentrates in the skin, but its availability decreases with age. Therefore, topical exogenous supplies of HA can reduce the aging process of the skin and can represent a valid alternative to the costly and invasive treatments based on injectable HA-based fillers. However, for cosmetically effective topical preparations, it is crucial to determine the properties favouring percutaneous HA penetration, such as HA molecular weight (MW). Therefore, the ability of 12 HA molecules with different MWs to penetrate the skin using an in vitro model was examined.
Materials and Methods: The Franz diffusion cell experimental system was used to estimate the penetration of the HA molecules across three epidermal tissues. The relationship between MW and penetration was inspected by applying a Pearson’s correlation test.
Results: HA molecules with different molecular weights applied topically were all capable of penetrating both the epidermis and dermis and the efficiency of penetration was proportional to the molecular weight of HA.
Conclusion: Altogether, the data suggested that even when applied topically, HA is useful for skin rejuvenation when combined into a single product.
Keywords: Aging Process; Hyaluronic Acid; Percutaneous Penetration; Porcine Ear Skin; Topical Preparation
Introduction
Skin aging is a biological phenomenon driven by both intrinsic and extrinsic factors. Intrinsic or innate aging begins at birth and is influenced by internal physiological factors that are largely inevitable consequences of time, which manifests as fine lines, a thinned epidermis, and gradual atrophy of the skin [1,2]. Extrinsic aging is caused by external aggressors, such as ultraviolet (UV) light, pollution, and cigarette smoke, which lead to the development of coarse wrinkles, loss of elasticity, and rough texture [3,4]. Such aging processes are accompanied by phenotypic changes in skin cells as well as structural and functional changes in extracellular matrix components, such as collagen, elastin, and proteoglycans, which are required for maintaining healthy skin [1,4]. Skin aging is also associated with a loss of skin moisture, and the key molecule involved in this process is hyaluronic acid (HA), which is a glycosaminoglycan with a unique capacity to bind and retain water molecules [5].
As people age, skin turgor, elasticity, and suppleness are reduced, most likely due to dwindling concentrations of HA. HA is a non-sulphated glycosaminoglycan composed of repeating units of polymeric disaccharides of D-glucuronic acid and N-acetyl-D-glucosamine, linked by a glucuronide β (1→3) bond [6,7]. HA is one of the main components of the extracellular matrix and has been implicated in numerous biological processes. Because of its macromolecular size, hygroscopicity, and viscoelasticity, HA modulates tissue hydration, osmotic balance, and the physical properties of the extracellular matrix, providing a hydrated and stable extracellular space in which cells, collagen, and elastin fibres are firmly anchored [8,9]. In addition to this structural role, HA functions as a signalling molecule. Its interactions with cell receptors and other extracellular binding partners play a major role in cell and organ development, responding to tissue injury and inflammation, cell migration, cancer formation and resistance, and cell differentiation and proliferation [10-14].
HA is widely distributed in both prokaryotic and eukaryotic cells [15,16]; in humans, it is most found in the skin, which contains approximately 50% of the HA contained in the entire body [5,17,18]. Furthermore, within the skin, the content of HA is significantly higher in the dermis than in the epidermis [19]. Owing to its unique physicochemical and biological properties, such as viscoelasticity, biocompatibility, biodegradability, and non-immunogenicity [20], HA is widely used for its numerous skin benefits, which include anti-aging, anti-wrinkle, anti-nasolabial fold, tissue regenerative, skin rejuvenating and replenishing, and moisturizing properties [21-26]. Thus, the multifunctional activity of HA is vital for maintaining healthy young skin. However, with age, two main factors affect the amount of HA in the skin: a decrease in its synthesis and an increase in the activity of enzymes, such as hyaluronidases, which degrade HA molecules.
The ensuing loss of HA leads to dehydration of the skin, making it susceptible to oxidation and damaging glycation reactions [27]. Therefore, an exogenous supply of HA can improve the condition and reduce the aging process of the skin [25]. For this reason, HA is the most important bioactive molecule for aesthetic purposes aimed at maintaining a youthful appearance and delaying the aging process by reducing wrinkles and plumping the skin [28]. Although HA-based fillers, which are usually administered by injection, are considered safe, they are relatively costly, invasive, and may cause unwanted side effects. To replenish aging, wrinkled, and hollow skin, it is important to develop HA products that feature efficient topical delivery, which is a system that does not possess the drawbacks or cost of injections and may be more readily accepted by a wider number of users. However, for cosmetically effective topical preparations, it is crucial to determine the properties that favor percutaneous HA penetration.
One important parameter is thought to be molecular weight. Indeed, there is some clinical evidence that the lower the molecular weight of this biopolymer, the easier the penetration via the skin barrier into the dermis, where the substance can more effectively exert its skin rejuvenating, plumping, and filling effects [29]. However, comprehensive examinations of the relationship between the molecular weight of HA and its absorption by the skin remain scarce. Therefore, we examined the ability of topically applied HA molecules with different molecular weights (expressed in Daltons) to penetrate the skin using an in vitro model of the epidermal barrier based on porcine ear skin. This tissue model has features analogous to human skin in terms of general structure, thickness, hair follicle content, pigmentation, collagen, and lipid composition, and thus represents a suitable animal model for in vitro tests in a wide spectrum of research fields [30,31].
Methods
Experimental System
We used the Franz diffusion cell experimental system, which founds wide applications to evaluate the permeation rate of topical drugs, on which the treated skin was anchored. The skin samples were prepared and tested according to Organization for Economic Co-operation and Development guidelines (Test No. 428: Skin Absorption: In Vitro Method). The porcine skin samples included the epidermis and dermis. The Franz diffusion cell consists of three components, usually in a vertical arrangement: donor cell, membrane, and receptor cell. The upper donor cell enables the direct application of a test formulation on the surface of the skin, which is placed on the top of the receptor. Once applied, the test ingredient(s) in the formulation permeates through the skin and eventually diffuses into the fluid contained in the receptor cell, which can be analysed for the presence of the ingredient.
Skin Sample Treatment
We applied 0.4 ml of the solution containing hyaluronic acid (HA) to the skin with an application area of 4 cm2. The concentrations of the tested solutions with different molecular weight species were as follows: hydrolyzed glycosaminoglycans 400 Da, 2.0%; sodium hyaluronate 800 Da, 1.5%; HA 1 kDa, 2.4%; hydrolyzed sodium hyaluronate 5 kDa, 0.012%; sodium hyaluronate 20 kDa, 95.0%; hydrolyzed HA 50 kDa, 0.01%; sodium hyaluronate 80 kDa, 97.0%; sodium hyaluronate 100 kDa, 1.667%; sodium hyaluronate 200 kDa, 0.06%; sodium hyaluronate 500 kDa, 96.4%; sodium hyaluronate 2000 kDa, 1.1%; sodium hyaluronate cross-polymer, 0.12% (this is a reticulated HA, for which the particle dimension cannot be expressed as Da) (Table 1). At the end of each experimental period, the tissue samples were collected and mechanically homogenized for subsequent analysis. Untreated tissue was used to determine the control value for the molecule under study. The solution in the receptor chamber was analysed for the presence of the tested molecule. The tissue was treated at room temperature, and the experiments were performed in duplicate.
Tissue Analysis
Tissue homogenates were prepared either from the epidermis alone or from the epidermis and dermis, following the application of the various HA species. The epidermis was obtained from the whole skin via mild heating (60°C for 45 minutes), which softens the tissue for easier manipulation, followed by chemical peeling with 80% lactic acid until “frosting” (white patches on the skin representing the site of separation between the epidermis and dermis) appears. The stratum corneum was not removed, and the tissue thickness was 200–400 mm. The epidermis and dermis were obtained from the whole skin by surgical removal of the hypodermis. To determine the amount of HA, tissue homogenates and the solution in the receptor chamber were analysed by enzyme-linked immunoassay (ELISA) using primary anti-HA antibodies and horseradish peroxidase (HRP)-conjugated secondary antibodies. The amount of HA contained in the tissues at a given experimental time point is expressed as a percentage of the amount retained in the tissue versus the amount applied. The percentage of penetration through the dermis was calculated by subtracting the value for the epidermis alone from the value for the epidermis and dermis. To control for endogenous HA, each measurement was normalized to the corresponding blank value obtained from the homogenate of the epidermis alone or the epidermis and dermis of untreated skin.
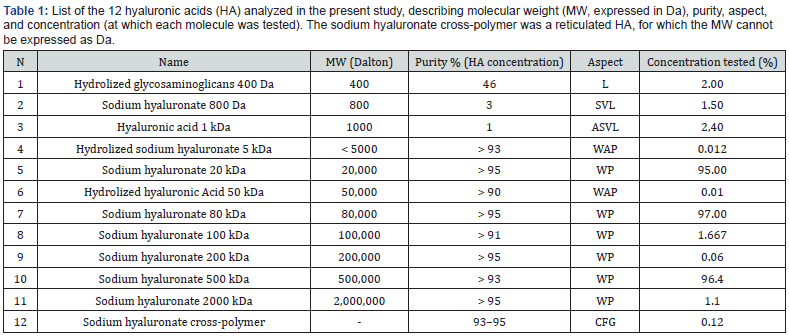
Abbreviations: ASVL = Amber Semi-Viscous Liquid; CFG = Colorless Fluid Gel; HA = Hyaluronic Acid; L = Liquid; MW = Molecular Weight; SVL = Semi-Viscous Liquid; WAP = White-Amber Powder; WP = White Powder
Statistical Analysis
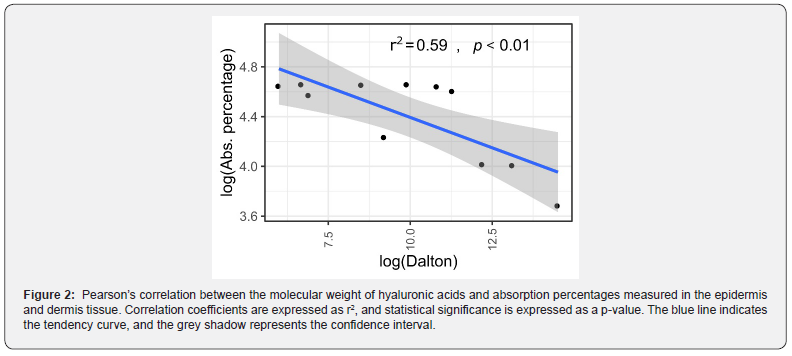
Pearson’s correlation analysis was conducted to investigate whether the absorption percentage was linearly correlated with the molecular weight of the HA species. We used the absorption values in the epidermis and dermis tissues observed at 24 hours. This time point was selected according to the purpose of the test (i.e., penetration was inspected at the end of the experiment). Only data from the epidermis and dermis were included as they showed cumulative information of each tissue considered separately. Absorption measured for the sodium hyaluronate cross-polymer was not considered because the molecular weight of this molecule was not expressible as Da. Data were log-transformed before analysis. Correlation coefficients are expressed as r2, and statistical significance was set at p-value < 0.05.
Results
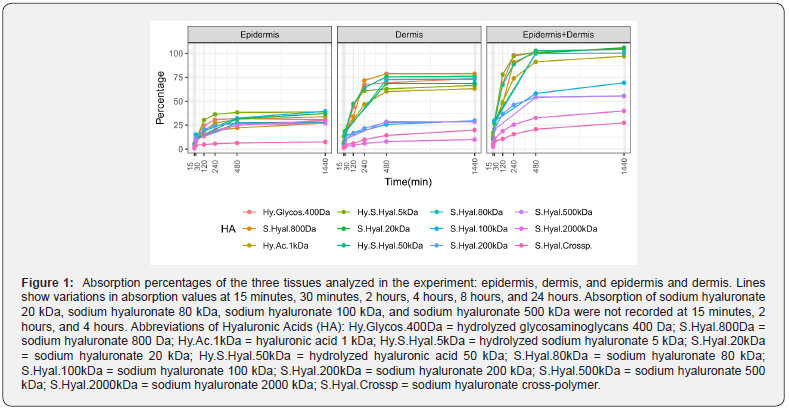
Given the importance of hyaluronic acid in the cosmetic treatment of wrinkled, aging, and hollow skin, we sought to determine the relationship between the ability of HA to penetrate the skin and its molecular weight. HA with 12 different molecular weights was tested in the experimental system (hydrolyzed glycosaminoglycans 400 Da, sodium hyaluronate 800 Da, HA 1 kDa, hydrolyzed sodium hyaluronate 5 kDa, sodium hyaluronate 20 kDa, hydrolyzed HA 50 kDa, sodium hyaluronate 80 kDa, sodium hyaluronate 100 kDa, sodium hyaluronate 200 kDa, sodium hyaluronate 500 kDa, sodium hyaluronate 2000 kDa, and sodium hyaluronate cross-polymer). After the application of HA to the skin, the epidermis and dermis were analyzed separately. The experimental outputs are summarized in Figure 1.
Analysis of the epidermis showed that all HA molecules tested in our experiment began to penetrate after 30 minutes, with rates ranging from approximately 8% to 11%, except for the sodium hyaluronate cross-polymer, which penetrated slightly less efficiently (4%). At 24 hours, the penetration rate was comparable for all molecules, ranging from 27% to 39%; however, the sodium hyaluronate cross-polymer was less efficient (7%). Absorption through the epidermis increased over the experimental period, where the highest percentages were generally recorded at the last time point (24 hours). A slight discrepancy to this pattern was observed for the hydrolyzed glycosaminoglycans 400 Da, which showed lower penetration at 24 hours (30.56%) than at 8 hours (31.82%). Interestingly, absorption from 8 to 24 hours increased at variable rates (between 0.05% and 6.2%) depending on the HA species analysed. Analysis of the dermis showed that, like penetration in the epidermis, penetration of the various forms of HA occurred after 30 minutes.
The efficiency of penetration was greater for the lower molecular weight molecules, with hydrolyzed glycosaminoglycans 400 Da, sodium hyaluronate 800 Da, HA 1 kDa, hydrolyzed sodium hyaluronate 5 kDa, sodium hyaluronate 20 kDa, hydrolyzed HA 50 kDa, sodium hyaluronate 80 kDa, and sodium hyaluronate 100 kDa showing penetration of 14% to 19% and larger molecules displaying penetration of 2.73% to 10.2%. The same trend was observed across all time points. The highest penetration through the dermis was achieved at 24 hours. Slight variations in absorption efficiency were observed between 8 and 24 hours for most of the analysed HA species. Importantly, our data demonstrated that lower molecular weight species, which comprised hydrolyzed glycosaminoglycans 400 Da, sodium hyaluronate 800 Da, HA 1 kDa, hydrolyzed sodium hyaluronate 5 kDa, sodium hyaluronate 20 kDa, hydrolyzed HA 50 kDa, and sodium hyaluronate 80 kDa, penetrated the dermis with high efficiency (63%–78%).
HA molecules with lower molecular weight were able to penetrate completely into the skin (epidermis and dermis) following superficial application, with an efficacy of 100% for hydrolyzed glycosaminoglycans 400 Da, sodium hyaluronate 800 Da, HA 1 kDa, hydrolyzed sodium hyaluronate 5 kDa, sodium hyaluronate 20 kDa, hydrolyzed HA 50 kDa, sodium hyaluronate 80 kDa, and sodium hyaluronate 100 kDa. Penetration through the epidermis and dermis of the HA species significantly correlated with the molecular weight of the species, where the larger the molecule, the lower the absorbance (r2 = 0.59, p = 0.0055; Figure 2). Although penetration of the higher molecular weight species was less efficient, absorption rates remained satisfactory, ranging from 27% to 69%. Finally, HA was not found in the receptor chamber solution even 24 hours after application, which indicated that the tested molecules did not cross the dermis but remained trapped within the skin layers.
Discussion
HA has become increasingly popular for both cosmetics and facial aesthetic medicine because of its water-retaining properties that can plump and fill in wrinkled, hollow, and aging facial skin. Approximately half of the body’s HA is distributed in the skin, where HA molecules of varying molecular weights (between 50 and 3000 kDa) are synthesized by different cell types. Because the level of HA decreases with age, it is important to replenish the skin with HA to maintain a younger and healthier appearance and counteract the effects of aging. One of the most important factors that contribute to skin aging is the reduction in HA and collagen in the dermal matrix [32]. Delivery of exogenous HA to the dermis of aging skin has been shown to induce the morphological stretching of fibroblasts, the production of extracellular matrix components, including new collagen, and stimulate the synthesis of HA, which further enhances the skin-plumping and rejuvenating effect of HA. Although successful HA-based fillers have been developed, they are usually administered by injection to reach the dermis rapidly and directly. However, to avoid the potential drawbacks and high cost of injections, developing HA products that can be applied topically while achieving adequate aesthetic results is crucial.
The ability of HA to penetrate the skin depends on its chemicalphysical properties, with molecular weight playing a potentially crucial role in penetration and thus efficacy. Indeed, we showed using an in vitro porcine skin model that HA molecules with different molecular weights applied topically are all capable of penetrating both the epidermis and dermis and that the efficiency of penetration is proportional to the molecular weight of HA. Our correlation analysis demonstrated that almost 60% of absorbance variability was related to the molecular weight of the HA molecules. The remaining variability was associated with other variables that were not evaluated in the experimental setup, including random events. Furthermore, our data showed that smaller molecules were completely absorbed into the skin in a time-dependent manner, reaching peak absorption 24 hours after application. The observation that all HA molecules could distribute throughout the skin is particularly important because HA works differently in the various layers of the skin.
In the epidermis, HA forms a coating that-owing to its interaction with the molecules present in the epidermis, such as proteoglycans, glycoproteins, elastin, and collagen-limits evaporation and improves hydration. In the dermis, HA stimulates the production of collagen, which is responsible for maintaining the density and compactness of the skin, promotes endogenous HA synthesis, and provides deeper moisturizing, plumping, and filling effects. Together, these properties can help maintain the softness, smoothness, and hydration of the skin, reduce skin imperfections, and fill expression and age lines. Additionally, although penetration is largely complete 24 hours after application, it is notable that it begins very early at 15-30 minutes after application, which suggests that the aesthetic effects of topical HA application can be achieved rapidly. These findings are also supported by preliminary clinical evidence demonstrating that the combination of HA molecules with different molecular weights is effective in inducing a filling, replenishing, and plumping effect, decreasing the clinical signs of skin aging, and improving facial volume [33]. Nevertheless, as the main limitation of our study, our findings should be interpreted in the context of preclinical applications.
Conclusion
In summary, the tested HA molecules were able to efficiently penetrate through the different layers of the porcine ear skin, suggesting that, even when applied topically, HA might be useful for skin rejuvenation.
Acknowledgments:
The work was carried out and patented by Labo Cosprophar AG.
References
5Baumann L (2007) Skin ageing and its treatment. J Pathol 211: 241-251.
13Hussain Z, Thu HE, Katas H, Bukhari SNA (2017) Hyaluronic Acid-Based Biomaterials: A Versatile and Smart Approach to Tissue Regeneration and Treating Traumatic, Surgical, and Chronic Wounds. Polymer Reviews 57: 594-630.
15Lowther DA, Rogers HJ (1955) Biosynthesis of hyaluronate. Nature 175: 435.
16Prehm P (1990) Release of hyaluronate from eukaryotic cells. Biochem J 267: 185-189.
17Juhlin L (1997) Hyaluronan in skin. J Intern Med 242: 61-66.
24Jegasothy SM, Zabolotniaia V, Bielfeldt S (2014) Efficacy of a New Topical Nano-hyaluronic Acid in Humans. J Clin Aesthet Dermatol 7: 27-29.
26Tokudome Y, Komi T, Omata A, Sekita M (2018) A New strategy for the passive skin delivery of nanoparticulate, high molecular weight hyaluronic acid prepared by a polyion complex method. Sci Rep 8: 2336.






























