The Circumambient Sore-Subcutaneous Panniculitis-like T cell Lymphoma
Anubha Bajaj*
Department of Pathology, Panjab University, India
Submission: November 15, 2022;Published: November 28, 2022
*Corresponding author: Anubha Bajaj, Department of Pathology, Panjab University, India
How to cite this article: Anubha B. The Circumambient Sore-Subcutaneous Panniculitis-like T cell Lymphoma. JOJ Dermatol & Cosmet. 2022; 5(1): 555651. DOI: 10.19080/JOJDC.2022.05.555651
Editorial
Subcutaneous panniculitis-like T cell lymphoma is an exceptional, primary, peripheral, adipotropic cutaneous T cell lymphoma derived from mature, post-thymic, cytotoxic αβ T cells. The lymphoma configures subcutaneous plaques and nodules wherein tumour dissemination beyond subcutaneous tissue is uncommonly encountered. Majority of lesions depict an indolent clinical course although hemophagocytic lymphohistiocytosis (HLH) may infrequently ensure.
The adipotropic, peripheral T cell lymphoma composed of cytotoxic αβ T cells immune reactive to CD8+ comprises <1% of cutaneous T cell lymphomas and is associated with excellent overall survival. Subsets of lymphoma accompanied by hemophagocytic lymphohistiocytosis(HLH) demonstrate an inferior survival.
Subcutaneous panniculitis-like T cell lymphoma may be demarcated from lupus erythematosus panniculitis or associated aggressive lymphomas incriminating mature adipose tissue with cogent histology. Although devoid of precise microscopic manifestations indicative of emergent hemophagocytic lymphohistiocytosis, clinical and microscopic concordance of subcutaneous panniculitis-like T cell lymphoma with lupus erythematosus panniculitis may exemplify possible occurrence of hemophagocytic lymphohistiocytosis (HLH) [1,2].
Median age of disease discernment is within thirties although no age of disease emergence is exempt. The lymphoma frequently incriminates subjects < 20 years of age. A female preponderance is observed. Subcutaneous panniculitis-like lymphoma commonly implicates the trunk, upper and lower extremities or gluteal region although diverse sites may be incriminated. In contrast to lupus erythematosus panniculitis, the lymphoma commonly involves gluteal region or lower extremities whereas lesions confined to facial surfaces are uncommon.
Of obscure aetiology, evolution of subcutaneous panniculitis –like T cell lymphoma in concordance with lupus erythematosus panniculitis remains debatable. Concurrent clinical and histological manifestations may exceptionally ensue wherein incriminated subjects may possibly manifest hemophagocytic lymphohistiocytosis [1,2].
T cell genetic rearrangement accompanies the monoclonal lymphocytic infiltrate. Clonal rearrangement of T cell receptor (TCR) may be indicative of subcutaneous panniculitis-like lymphoma, especially within clinically suspected lesions associated with non-specific histologic features.
Clonal T cell receptor genetic rearrangement detected within multiple tissue specimens obtained from diverse sites or varying intervals appears indicative of lymphoma, in contrast to singular tissue specimen manifesting identical cellular clones. Cellular clones may persist in lesions such as atypical lymphocytic lobular panniculitis or diverse T cell dyscrasias non concurrent with lymphoma [1,2]. Subcutaneous panniculitis-like lymphoma may arise subsequent to benign, indolent panniculitis. The condition manifests variably tender, erythematous, subcutaneous plaques and nodules several centimetres in magnitude. Lesions may persist or may undergo resolution along with lipoatrophy, simulating lupus erythematosus panniculitis.
Neoplastic dissemination beyond cutaneous surfaces or regional lymph node enlargement may exceptionally arise. Frequently associated with systemic lupus erythematosus, around ~20% subjects depict antecedent or concomitant autoimmune disorders.
Commonly, symptoms such as fatigue, pyrexia, weight loss or diverse systemic symptoms may be represented [1,2]. Hemophagocytic lymphohistiocytosis (HLH) is variably encountered in ~20% instances although paediatric subjects are infrequently implicated. Upon low power examination, lobular panniculitis is observed. Superimposed epidermis and dermis are minimally involved. Mature adipose tissue lobules appear predominantly circumscribed by an intense inflammatory infiltrate whereas mild extrusion of inflammatory cells may ensue within the septal panniculus. Configuration of germinal centres is exceptional. Inflammatory infiltrate is preponderantly lymphocytic and appears admixed with variable quantities of histiocytes. The macrophages appear to engulf nuclear debris, thereby configuring ‘bean bag’ cells or lipid, denominated as ‘lipophages’. Characteristically, infiltrating lymphocytes circumscribe the adipocytes and demonstrate nuclear atypia [1,2].
Upon microscopy, an inflammatory infiltrate reminiscent of lobular panniculitis appears commingled with focal adipose tissue necrosis and karyorrhexis. Macrophages which engulf karyorrhectic debris or lipid globules may occasionally exhibit erythrophagocytosis.
Mature lymphocytes appear to circumscribe mature adipocytes, characteristically rupture cellular membranes of adipocytes and subsequently invade adipocytic lobules [1,2].
Lymphocytes appear minimally enlarged and exemplify hyperchromatic nuclei with irregular nuclear contours. In contrast to lupus erythematosus panniculitis, features such as plasma cell aggregates, configuration of lymphoid follicles or hyaline lipomembranous adipose tissue necrosis are inconspicuous or absent (Figure 1).
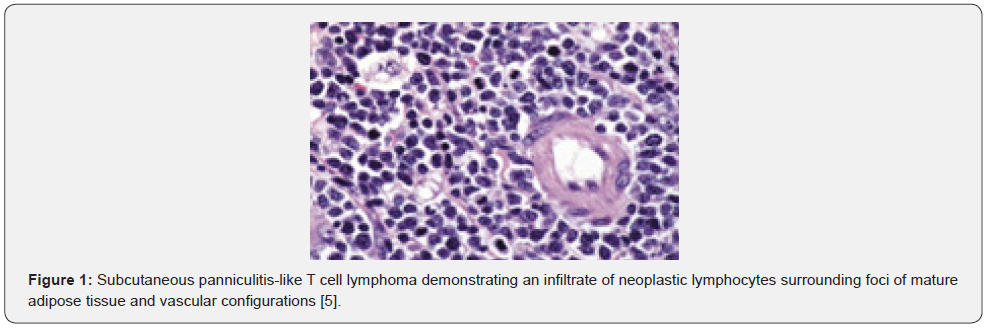
Surgical tissue sampling of ancient lesions enunciates morphological features as granulomatous inflammation and lipo-membranous adipose tissue necrosis or fibrosis, instead of adipotropic lymphoma. Epidermotropism is exceptional and intra-epidermal cellular exudate is minimal wherein configuration of Pautrier’s micro-abscesses is absent [1,2].
Subcutaneous panniculitis-like T cell lymphoma enunciates Ki67 hotspots as denominated by focal, enhanced expression intermingled with mature adipocytes surrounded by CD8 + cytotoxic T cells. Elevated Ki67 proliferation index encountered within > 20% of intra-lobular lymphoid population appears indicative of the lymphoma. Adipotropic lymphocytes appear immune reactive to T cell receptor alpha/ beta (TCRα/β), T cell receptor beta (TCRβF1), CD8 and cytotoxic markers as T cell intracellular antigen 1(TIA1) or granzyme. Exceptionally, lymphocytic infiltrate is immune reactive to CD30 and CD56.
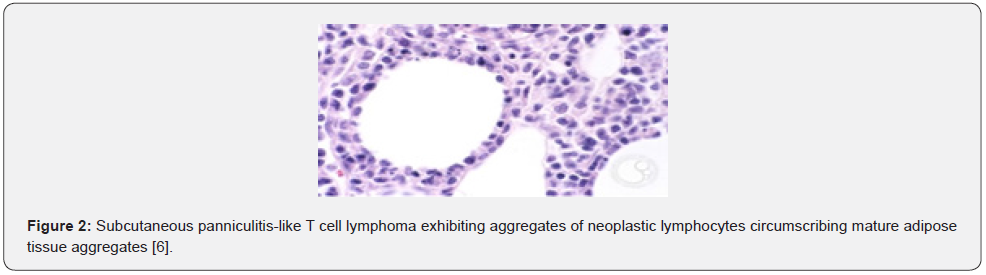
Subcutaneous panniculitis-like T cell lymphoma is immune non-reactive to T cell receptor gamma (TCRγ), Epstein Barr encoded RNAs (EBER), CD4, CD30, CD56 or CD123 [3,4]. Subcutaneous panniculitis-like T cell lymphoma requires segregation from neoplasms such as atypical lymphocytic lobular panniculitis, extra-nodal NK/T cell lymphoma, nasal type, lupus erythematosus panniculitis, mycosis fungoides, peripheral T cell lymphoma not otherwise specified (NOS), primary cutaneous anaplastic large cell lymphoma or primary cutaneous gamma delta T cell lymphoma (Figure 2).
Appropriate discernment of subcutaneous panniculitislike T cell lymphoma mandates intense clinical concurrence and exclusion of lupus erythematosus panniculitis or diverse lymphomas with cutaneous manifestations.
Examination of bone marrow, spleen or lymph node is optimal for discerning hemophagocytosis. Occasionally, subcutaneous panniculitis-like T cell lymphoma manifests reactivity to antinuclear antibodies (ANA), a feature which is non indicative of lupus erythematosus panniculitis. Besides, lupus panniculitis is infrequently associated with systemic lupus erythematosus [3,4]. Hemophagocytic lymphohistiocytosis denominates features such as ~pyrexia ~splenomegaly ~pancytopenia or bicytopenia involving ≥ 2 cellular lineages ~hypertriglyceridemia or hypofibrinogenemia ~minimal or absent activity of NK cells ~serum ferritin ≥ 500 μg/Litre ~soluble CD25 ≥ 2,400 Units/ millilitre~evidence of hemophagocytosis as encountered within bone marrow, spleen or lymph node. An estimated five of aforesaid criteria are mandated for establishing hemophagocytic lymphohistiocytosis, especially in the absence of genetic mutations accompanying the syndrome [3,4].
Subcutaneous panniculitis-like lymphoma can be managed conservatively, especially instances devoid of hemophagocytic lymphohistocytois (HLH). A plethora of therapeutic strategies can be employed and specific treatment pertains to each individual.
Localized therapies as radiotherapy and intralesional steroids or systemic therapies as steroids, antineoplastic agents as bexarotene or denileukin diftitox and histone deacetylase (HDAC) inhibitors can be advantageously utilized. Aggressive disease can be treated with intensive regimen of biologic agents, HDAC inhibitors or single agent chemotherapy.
Subcutaneous panniculitis-like T cell lymphoma devoid of HLH demonstrates an excellent 5-year overall survival of ~90%. Occurrence of hemophagocytic lymphohistiocytosis is associated with decimated 5-year overall survival of ~ 45% [3-6].
References
- Musick SR, Lynch DT (2022) Subcutaneous Panniculitis Like T-cell Lymphoma. Stat Pearls International Treasure Island,
- Kreher MA, Ahn J, Werbel T, Motaparthi K (2022) Subcutaneous panniculitis-like T-cell lymphoma after COVID-19 vaccination. JAAD Case Rep 28: 18-20.
- Xu J, Li J, Ya-Juan S, Quan W, Liu L, et al (2022) CD20-positive subcutaneous panniculitis-like T-cell lymphoma presenting as polycranial neuropathy: A Care-compliant case report and literature review. Medicine (Baltimore) 101(35): e30233.
- Giza A, Gałązka K, Jońca M, Raźny M, Zimowska-Curyło D et al. (2022) Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) with probable mesentery involvement with associated hemophagocytic syndrome (HPS) - how to treat it? J Dermatolog Treat 33(5): 2674-2676.
- Image 1 Courtesy: Plastic surgery key
- Image 2 Courtesy: ASH.com






























