- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Review on Leishmaniasis: A Neglected Global Disease
Irfan C1, Kharal F2, Zia S3, Das A4, Karangula S5 Razi S6*, Shamail F7 and Khalid S8
1Department of Pharmacy, Ziauddin University, Pakistan
2Department of Medicine, Combined Military Hospital, Pakistan
3Department of Pathology, Jinnah Sindh Medical University, Pakistan
4Department of Medicine, Nilratan Sircar Medical College, India
5Department of Medicine, Malla Reddy Institute of Medical Sciences, India
6Department of Pathology, Jinnah Medical and Dental College, Pakistan
7Department of Pathology, Ziauddin University, Pakistan
8Department of Dermatology, Baqai Medical University, Pakistan
Submission: June 20, 2022; Published: August 12, 2022
*Corresponding author: Razi S, Department of Pathology, Jinnah Medical and Dental College, Innah Medical and Dental College, Pakistan
How to cite this article: Irfan C, Kharal F, Zia S, Arghadip D, Karangula S, Razi S, et al. Review on Leishmaniasis: A Neglected Global Disease. JOJ Dermatol & Cosmet. 2022; 4(4): 555644. DOI: 10.19080/JOJDC.2020.04.555644
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Abstract
As per World Health Organization, Leishmaniasis is a neglected, tropical disease. It is initiated by intracellular transmission of a parasite to humans, by sand fly bite. Factors like population variability, climate change etc. lead to the expansion of this disease. This article provides review of epidemiology and management of leishmaniasis i.e., cutaneous, mucosal, and visceral. Literature was studied by systematic exploration in PubMed including various articles from 2012-2022 without setting any exclusion criteria. There have been reports of development of strains, resistant to traditional therapy of leishmaniasi, along with simultaneous infections like HIV/Leishmania spp., and other targeted pharmaceutical resources i.e., amphotericin B and formulations, miltefosine, pentavalent antimonials. Since long, only few drugs are available for the management of the disease, having adverse effects and toxicity due to which the treatment control program is put in check globally. New strategies are required to manage this neglected disease adequately.
Keywords: Leishmaniasis; Tropical disease; Leishmania spp; Visceral leishmaniasis; Cutaneous leishmaniasis Abbreviations: MCL: Mucocutaneous Leishmaniasis; VL: Visceral Leishmaniasis; PKDL: Post Kala-azar Dermal Leishmaniasis; HAT: Human African Trypanosomiasis; CR: Complete Remission; DCL: Diffuse Cutaneous Leishmaniasis; CL: Cutaneous: LCL: Localised Cutaneous Leishmaniasis; IDSA: Infectious Diseases Society of America; ASTMH: American Society of Tropical Medicine and Hygiene; SSG: Sodium Stibogluconate; MA: Meglumine Antimoniate; MPL-SE: Monophosphoryl Lipid A Plus Squalene: WHO: World Health Organization; qPCR: quantitative PCR; HRM: High Resolution Melt; kDNA: Kinetoplast DNA; SSG: Sodium Stibogluconate; MA: Meglumine Antimoniate; OWCL: Old World CL; NWCL: New World CL
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Introduction
Leishmaniasis is initiated by intracellular transmission of a parasite to humans, by sand fly bite [1], mostly Lutzomyia and Phlebotomus and in North Africa, Asia, Middle Eastern countries, Europe and certain South American countries; exceptional spread can also be due to a mishandling of specimen in the laboratory [2]. Leishmaniasis is considered as a tropical and subtropical ailment [1]. The World Health Organization (WHO) considers leishmaniasis among one of the seven grave tropical ailments that portends a poor prognosis. This disease has a possible fatal consequence due to a broad range of clinical manifestations.
Leishmaniasis is being observed worldwide except Oceania [3] and is endemic in confined geographical zones of Southern Europe, Southeastern Mexico, the Middle East, Central and South America and Northeastern Africa [4]. The cases are rising globally due to various reasons like urbanization, deforestation [5], resistance to therapy, enhanced diagnosis and also for the reason of inadequate coverage of health services [6,7]; vector control strategies are influenced by armed encounters and masses shift in regions [8,9].
It is reported that 98 species of the genera Lutzomyia and Phlebotomus are recognized or assumed vectors for human leishmaniasis, spread via infected phlebotomine sand flies [10,11]. Mammals are attacked by female sand flies only for blood meals necessary to complete egg development cycle. Apart from mammals, certain sand flies have wide-ranging hosts that include rodents, marsupials canids and hyraxes. Consequently, human leishmaniasis may have zoonotic or anthroponotic spread forms [12,13]. Leishmania parasites reside and grow in phagocytic cells intracellularly (within phagolysosomes) in the mammals. More than 20 diverse pathogenic Leishmania species are defined for humans at present [9,14-16] among which the 3 chief types are visceral, cutaneous, and mucocutaneous leishmaniasis [9]. The clinical presentation of this ailment is related to the specie of the parasite, tropism, its infectivity and immune system of the host [17].
Different Leishmania species are much alike morphologically.
Depending on the types of invaded phagocytic cells, these species cause 2 chief forms of clinical leishmaniasis i.e., cutaneous (CL) and visceral leishmaniasis (VL), [18]. In case of CL, macrophages in the skin are infected by the parasites. The host cell bursts when it is full of parasites, releasing amastigotes that then infect adjacent macrophages. On the contrary, the free amastigotes are transmitted via blood circulation in VL and contaminate the mononuclear phagocytic cells of reticulo-endothelial system of spleen, liver, lymph nodes, intestine, and bone marro [12].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Methodology for Review
Only English language articles are included in this review article. Literature was studied in PubMed using regular keywords (Table 1). The articles are included from 2012-2022 without setting any exclusion criteria (Figure 1).
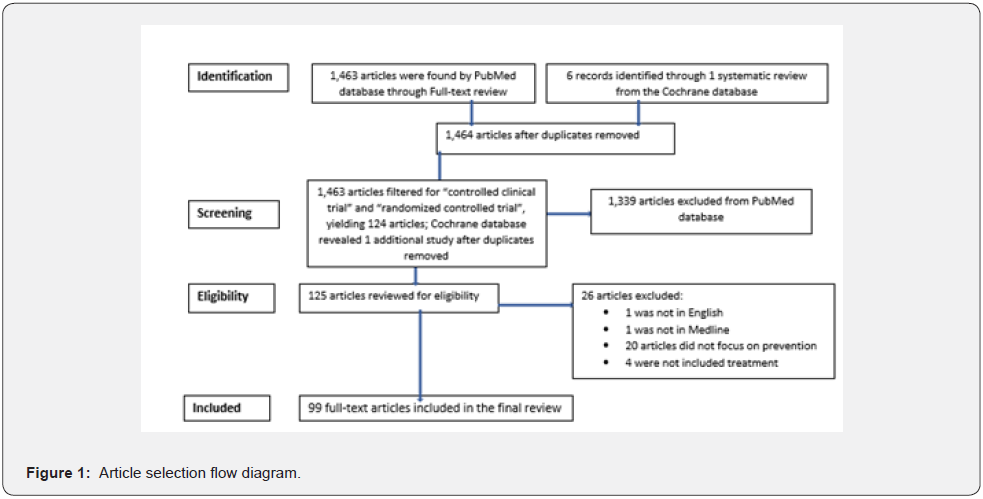
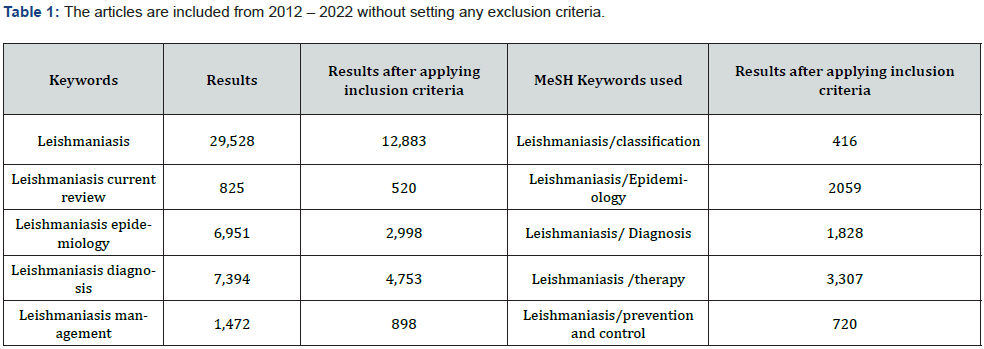
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Results
By systematic exploration in PubMed, 31,389 articles were found with the keyword Leishmaniasis of the skin. From 2012 to 2022, 12,884 articles were found out of which 99 are included in this review article.
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Discussion
Leishmaniasis is the chief endemic health burden in many regions globally. In the 14 high-burden VL states, total of annually reported cases of VL has fallen from 60,000 (2006) to 30,000 (2014) which is chiefly owing to a drop of five-fold in Indian cases of visceral leishmaniasis [19]. In 12 CL high-burden countries, total of annually reported cases of CL is unchanged which is about 150,000 over the same retro [20]. Several factors have contributed to the rise in leishmaniasis cases during the former 25 years worldwide. In non-endemic areas, globalization and changes in climate are important factors for the transmission of leishmaniasis [21]. There has been a rise in the cases of Leishmaniasis over the last several years in international travellers [22]. Furthermore, Leishmania infections have ensued in patients who never went to leishmaniasis endemic areas due to the international movement of blood products [21]. It is observed that there is no blood bank screening to check the existence of anti-leishmanial antibodies in the preserved blood products. Also, there is an increased dispersal of sand flies towards north due to global warming which could consequently transmit leishmaniasis in hitherto non-endemic area [21,23].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Epidemiology of Leishmaniasis
There is a global distribution of Leishmaniasis disease with its existence in approximately 89 countries in the world [24,25]. The endemic regions include the Mediterranean, Africa, the Americas, and Asia. It is mostly a jungle zoonosis in the America transmitted by mainly genera Phlebotomus and Lutzomyia of sand flies; yet leishmaniasis can be acquired in hot semi-desert like geographical regions or cold regions too. From southern parts of United States to northern part of Argentina shires, there is 0.17% seroprevalence of CL [26] except Uruguay, El Salvador, and Chile [25,27]. Worldwide 12 to 15 million individuals are infected while there is risk of disease acquisition in 350 million people. There is incidence of about 1.5 - 2 million cases of leishmaniasis annually due to which 70,000 mortalities per year occur [25,28]. WHO made reports in 2012 regarding the burden and distribution of VL and CL in over 100 parts of and the data acquired until 2010 showed that globally about 90% VL cases happened in India, South Sudan, Brazil, Ethiopia, Bangladesh, and Sudan. Approximately 70% CL cases were found in Syrian Arab Republic, Brazil, Algeria, Colombia, Afghanistan, Iran, Ethiopia, Sudan, and Costa Rica [29].
Due to the Syrian civil war, about 3 million expats were received by Turkey who were placed at the camps or houses in the southern parts of Turkey. Epidemiological research was done from 2009 to 2015 in Turkey employing semi-nested polymerase chain reactions in real-time which discovered the presence of 263 patients positive for leishmaniasis; among them there were, 33.46% Syrian, 66.15% Turkish, and 0.38% Afghani people. Species of Leishmania identified in Turkish and Syrian patients were Leishmania infantum and Leishmania tropica [30]. A 13 years report from Algeria confirmed 4,813 CL cases showing the influence of disease in all municipalities and entire age groups. Males were more affected (65%) and the age group among the most affected were 10 to 19 years (31.41%) and children less than 9 years (25.70%) [31]. Geographic and seasonal distribution of CL was found in Iran with 589,913 CL cases. The yearly incidence was around 30.9 per 100,000 with majority occurrence in the central parts of Iran [32] and in autumn the greatest prevalence rate (35.14%) of lesions was reported [33].
VL clinical type is endemic in Bangladesh, India, and Nepal. All VL cases (> 60%) globally relate to South Asia, predominantly in rural zones. Related to this, around 5.7 million individuals are at risk in Nepal confined chiefly to Terai state, bordering the VLendemic areas of the Indian Bihar state. Between 1980 and 2007, 23,368 cases were reported together with 311 demises. The Department of Health Services of Nepal in 2006–2007 presented the annual health report showing 2.67 per 10,000 people at risk for VL incidence [34]. It is estimated in Latin America that about 60,000 incidences of all types of leishmanisis happen yearly. Leishmanisis is characteristic of environments having temperatures higher than 20°C, an altitude of 0 to 1,500 m beyond sea level and rainfall of 1,500 to 3,000 mm yearly. Still, some “Uta” cases have been identified in the cold and humid highlands of Peru where this ailment is spread by a tiny bug [35,36].
Reports show that Leishmaniasis affects men commonly; the high risks individuals include farmers, hunters, timber exploiters, military workers, biologists, and those practicing ecological tourism, etc. in Mexico while in Peru and Brazil, it influences the gatherers of rubber, tea, banana, coffee, and coca [2,37]. There is risk of contact in endemic sectors to women, children, and elderly. Leishmanisis has been recognized since the pre-Hispanic period in Mexico [37], reporting all the clinical forms of the ailment [38]. The very common are pure cutaneous and the cutaneous-chondral types which cause the typical chiclero’s ulcer (gum tree harvester’s ulcer) when affecting ears. The foremost endemic region in Mexico is in the Neotropical zone from the southeast [2,37,39,40]; new cases of 5.08 per 100,000 people has been detected in the Yucatán Peninsula annually [41,42]. Antibodies in 17% of the overall populace of southern state of Yucatán, Becanchén, have been acknowledged after serlogical investigations. Tabasco is regarded as high-endemic zone due to incidence and prevalence rate of 2.35 and 9.41 per 100,000 populace, correspondingly [37]. Positive serology of 20.42% military persons operational in this zone was observed. Lutzomyia olmeca and Lutzomyia cruciate are the two major vectors of the ailment in the Yucatán Peninsula [43].
Leishmania braziliensis and mexicana are prevailant in Belize and Guatemala, while only Leishmania mexicana has been reported in Campeche and Yucatán in Mexico [4]. According to data of 2016, WHO reported 200,918 CL incidences and 22,233 VL incidences. Brazil, Ethiopia, India, Kenya, Somalia, South Sudan, and Sudan represented 90% VL cases globally, whereas Afghanistan, Algeria, Brazil, Colombia, Pakistan, and the Syrian Arab Republic, Morocco, Nicaragua, Peru, Sudan, Tunisia, and Yemen, are accounted for 90% CL cases. Information to WHO is still awaited from Iran and Iraq, having >10,000 of CL cases as per 2015 report [44].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Clinical Presentation of Leishmaniasis
CL is the commonest type of leishmaniasis with incidence of 0.7–1.3 million every year globally. There are 3 different forms of CL: localised cutaneous leishmaniasis (LCL), diffuse cutaneous leishmaniasis (DCL) and mucocutaneous leishmaniasis (MCL). Skin wounds and ulcers on bare body parts are characteristic of LCL that leave lasting scars on body. DCL is notable due to the growth of numerous, gradually developing nodules encompassing the full body without ulceration and is less common. MCL is constrained to Latin America. Next to healing of primary skin lesion, the disease extends to the mucous membranes of the nose, mouth and throat. Afterwards, the mucosal ulcers destroy the nasal septum, lips and palate causing facial mutilating extensively. VL is considered as the utmost severe type of leishmaniasis with each year likely 0.2–0.4 million incidences worldwide [45]. VL is lethal in more than 95% of patients if left untreated. The indications of VL are asymmetrical fever, loss of weight, splenomegaly, hepatomegaly, hepatosplenomegaly at times and anemia [12]. Other signs of VL can be alienated into disease-specific wounds at infected parts (i.e., papules, nodules, ulcers) and nonspecific wounds (i.e. purpura, kwashiorkor like hair discoloration, xerosis) [46].
An immunologically mediated outcome of VL is Post–kala-azar dermal leishmaniasis (PKDL) mostly noticed in Sudan and India. The onset of disease is typically 6 months - 1 or more years post therapy but it can happen later up to 20 years [46]. The hallmark injuries of PKDL can be generally described as erythematous or hypopigmented macules, papules, skin-colored nodules, and malar erythema, though variances are present in cutaneous findings as per geographic situation and existence or level of immune suppression [46,47]. The lesions appear typically in the perioral region at first and then become widespread. Maintenance of feeling in the lesioned skin supports to differentiate PKDL from leprosy [9].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Diagnosis of Leishmaniasis
The diagnosis depends on clinical signs, epidemiological and laboratory data. The clinical evidence for CL diagnosis include: travel history to or accommodation in endemic areas, a small number of lesions (1-3), lesions without pain, localization on bare body parts, patients having history of a number of months or slow variation over weeks, unresponsive persons to former systemic or topical antibiotics, rubber like feeling on palpation [9]. There is no gold-standard available for human patients or animals for the laboratory methods [48], which impairs precise epidemiological data assembly. Besides, false-negative results can cause deferment of therapy resulting in reservoirs maintenance. Due to non-availability of gold-standard parasitologic diagnostic test, it is suggested to associate histopathology, culture, and DNA amplification methods to escalate sensitivity to identify the disease on a species basis. It is suggested to connect with a reference laboratory in advance to attain specimen as nearly all specimen collection methods and laboratory diagnostic techniques require greatly specific information for Leishmaniasis; concurrent diagnostic methodology for other possible etiologies (like sporotrichosis, blastomycosis, mycobacterial infections, etc.) should be considered too [9,49].
Recently, numerous immunological and molecular diagnostic tools to diagnose leishmaniasis have been established [50]. Due to their greater sensitivity, specificity, and potential use to different types of clinical samples, the usage of molecular procedures particularly has turned out to be ever more applicable. The realtime PCR or quantitative PCR (qPCR) has recently become very common as it is fast with extensive dynamic range and crosscontamination is significantly diminished as the reaction tubes for post-PCR investigation are not required to be open. High resolution melt (HRM) study can be employed to distinguish amplicons built on sequence variations for genotyping [51]. The use of probes permits multiplexing) and gives additional specificity to the assay but is more expensive typically, moderately decreasing the cost per assay. This methodology offers less false positives than the intercalating dye procedure [52]. Bretagne et al. [53] and Nicolas et al. [54] defined 2 qPCR-based procedures to spot and quantify Leishmanial parasites by use of DNA polymerase gene and minicircles kinetoplast DNA (kDNA) as a target, correspondingly. Subsequently, various qPCR-based assays have been instituted on diverse molecular targets for recognition, quantification, and genotyping of Leishmania species [55,56]. No uniformity is noted in published qPCR assays for sensitivity and specificity reports. For example, qPCR assays made for VL diagnosis in individuals indicated a specificity difference between 29.6–100% and sensitivity between 91.3–100%, demonstrating qPCR practicality for pivotal sensitive tool [50,57].
Dermatoscopy
Different dermatoscopic characteristics have been described like white starburst like form, tear drop like patterns, yellow tears, and salmon-colored ovoid forms. The specificity of these outcomes may necessitate additional analysis [58,59].
Leishmania Smear
A smear is regarded an economical, simple, and swift means to diagnose CL. It is commended to attain a smear sample in the initial assessment, pursued by direct inspection with Giemsa stain. There are 4 methods to take samples: slit-skin, scraping, touch (imprint) smear, and fine-needle aspiration [17]. The recognition of Leishmania amastigotes via microscope is adequate for diagnosis yet it involves ample proficiency [49]. Recently, a diagnostic algorithm has been recommended in which a positive direct microscopic investigation by means of smear is followed by anti-Leishmania therapy, while a negative preliminary investigation should be operated with a skin biopsy [17].
Culture
The isolation of the parasite in culture media should be done as it permits verification of the diagnosis and the isolates can be employed for additional analysis. The specimen gotten for culture can be assessed for further probable causative agents also. requires A sterile method is needed in sampling for parasitologic culture, with evasion of residual iodine and alcohol which may influence the growth of parasite on culture media [49].
Other Diagnostic Techniques
For VL diagnosis, serologic testing can be supportive though the current serologic assays are not adequately sensitive or specific to be employed [49]. The Leishmanin skin test (Montenegro test or Leishman reaction) is a delayed hypersensitivity assessment, which is positive in about 90% of CL or ML patients with a period of more than 3 months [46].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Existing and Evolving Antileishmanial Agents
Pentavalent antimonials (SbV)
SbV is present as sodium stibogluconate (SSG) and meglumine antimoniate (MA). For many decades, the standard treatment of VL was Antimonials with the dosage of 20 mg/kg body weight for 28–30 days till it became unsuccessful in India due to development of resistance [60,61]. While it is efficacious in other regions globally particularly Africa, its use is limited due to greater toxicity specifically cardiotoxicity in terms of cardiac arrhythmias, extended QTc interval, ventricular premature beats, ventricular tachycardia, ventricular fibrillation, and torsades de pointes [19]. The patients coinfected with HIV get marked side effects during SSG therapy with reduced efficacy and more deaths [62,63].
Amphotericin b (AmB)
Amphotericin B deoxycholate is a polyene drug which was widely used in India for VL noncompliant to antimonials. The cure rates were ~ 100% at a dosage of 0.75–1.0 mg/kg for 15–20 i.v infusions [64,65]. With dose of 1 mg/kg/ day, up to 60–80 doses for more than 4 months, it is also suggested to treat PKDL in the India [66]. The adverse effects of this therapy include infusion reactions, nephrotoxicity, hypokalemia, and myocarditis due to which the patient should be monitored and hospitalized hence increasing therapy cost. To reduce such adverse effects, lipid preparations of the drug were developed replacing deoxycholate with further lipids. These preparations are quickly taken up into organs like liver and spleen and concentrate in the site of VL disease i.e. reticuloendothelial tissue. The drug stays there for an extended time permitting provision of big doses of the agent in short time [19]. Three lipid preparations have been widely tried for leishmaniasis principally VL: liposomal amphotericin B (AmBisome; Gilead Sciences; L-AmB), amphotericin B lipid complex (ABLC; Abelcet, Enzon pharmaceuticals), and amphotericin B cholesterol dispersion (Amphotericin B colloidal dispersion (ABCD); Amphotec_, InterMune Corp.). among these, only L-AmB is US FDA permitted. For treatment of VL, the total dose requirements of lipid preparations differ by regions. In India, one dose of 10 - 30 mg/kg is the treatment of choice [65,67].
Miltefosine (MIL)
It is the single oral medication accepted for leishmaniasis which is an alkyl phospholipid. It was registered for VL in 2002 for use in India, ensuing a Phase III clinical trial in which 50–100 mg/day dose for 28 days provided complete remission (CR) of 94% [65,68]. A Phase IV study in India similarly exhibited CR of 82%. The availability for oral use, comfort of consumption and effectiveness made MIL the medication of choice in India, Nepal, and Bangladesh for VL eradication [44].
Paromomycin (PM, aminosidine)
It is an aminoglycoside-aminocyclitol drug [19] which has been employed to treat VL in parenteral form and for CL in both topical and parenteral forms. It was used for treating dysentery in VL patients in southern Sudan in the 1989 VL epidemic. This drug reduced the dysentery incidence and showed synergism with SbV in VL management [44]. Then it was used in Africa in combination with SbV. The dose is 15mg/day for 21 days [65].
Pentamidine
It is an aromatic diamidine which was used for treating antimony resistant VL in India in the early 1980s [19]. But its effectiveness failed, and it was abandoned due to its severe toxicities i.e. pancreatitis causing insulin-dependent diabetes, hypoglycemia, hypotension, QT elongation and hyperkalemia. In recent times it has been employed as secondary prophylaxis with a dose of 4 mg/kg monthly for 12 months; to prevent VL relapse there can be 6-month extension if cluster of differentiation 4 (CD4) count was ≤ 200 cells/μl in Ethiopian HIV-VL coinfected individuals [44, 69].
Sitamaquine
It is an oral agent which is 8-aminoquinoline; it has undergone Phase II trials for VL in India and Kenya. At the dosage of 1.75 and 2 mg/kg/day for 28 days, the CRs were 89% and 100%, correspondingly while CR was 85% at 2 mg/kg/day for 21 days in India. The CRs were 80%, 82%, and 91% at doses of 2, 2.5, and 3 mg/kg/day respectively in Kenya. At doses > 2 mg/kg, nephrotoxicity was mainly observed, and it exhibits low efficacy in low doses [44].
Azoles
Ergosterol synthesis of Leishmania parasites are blocked by these drugs. For CL management, Ketoconazole, itraconazole, and fluconazole have been widely used. For L. major, fluconazole in dose of 200 mg per day orally for 6 weeks had CR of 44–59% in old world CL (OWCL). Gastrointestinal symptoms and hepatotoxicity were the main adverse effects observed. On escalating fluconazole dose to 400 mg, CR amplified to 81%. For L. braziliensis, fluconazole with dosage of 5–8 mg/kg/day for 4–12 weeks displayed 75-100% CR in new world CL (NWCL). A randomized controlled trial in Brazil for L. braziliensis infection exhibited 22% CR when associated to 53% for standard SbV with fluconazole 6.5–8 mg/kg/day for 28 days [44].
Nitroimidazole
Nitroimidazole is an anti-protozoal agent and Fexinidazole was revealed by Hoechst AG in the 1970s (now Sanofi-Aventis); it was reinvented as a favorable agent by the Drugs for Neglected Diseases initiative (DNDi) to treat Human African trypanosomiasis (HAT). Oral fexinidazole was administered once a day as 1800 mg for day 1–4, 1200 mg for day 5– 10 for the late stage of HAT and showed 91% CR with safety. Anti-leishmanial activity of fexinidazole and its principal metabolites namely fexinidazole sulfoxide and sulfone has been established in the in vitro studies [70] which was followed by DNDi in Sudan in a Phase II trial to evaluate safety and efficacy of fexinidazole to treat VL. 14 patients were enrolled in the trial and at the end of treatment majority showed parasite clearance however only 3 patients were healed at 6-month follow-up, hence terminating this study. Due to the safety and oral administration of the drug, DNDi is investigating to utilize fexinidazole and miltefosine combination in eastern Africa to treat VL [44].
Another potent compound of nitroimidazole group called pretomanid (PA-824) has been found potent against both replicating and non-replicating Mycobacterium tuberculosis. PA-824 was found to be tolerated well and safe showing higher bactericidal action in drug susceptible tuberculosis in a Phase IIb trial in combination with moxifloxacin and pyrazinamide, during 8 weeks of therapy [71]. Antileishmanial activity has been exhibited by both the S and R enantiomers of PA-824, yet the R enantiomer having insignificant activity against M. tuberculosis proved to be a 5 fold more potent inhibitor of L. donovani as compared to the S-enantiomer in vitro. The combined medication research in vitro pointed out that fexinidazole and (R)-PA-824 have additive effects [72].
Delaminid
This oral antitubercular agent is a very effective inhibitor of Leishmania donovani in vitro and in Expert Opinion on Pharmacotherapy 5 vivo research also. This drug was found to be more effective than miltefosine and fexinidazole drugs [73].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Local therapy
Thermotherapy
Thermotherapy is built on in vitro laboratory investigation that Leishmanial parasites do not increase in number at temperatures > 39°C. Thermotherapy with radio-frequency waves has been done for Old and NWCL both. Mostly investigations have employed a battery-operated, localized current field radio frequency (RF) generator (ThermoMed) or RF heat generator (Ellman International Inc., NY, USA). A single use of thermotherapy at 50°C for 30 seconds provided the CR between 69.4% - 82.5% in Afghanistan [74], 98% in India [75] for lesion by L. tropica, whereas for L. major infection, the CR was 48% in Iraq and Afghanistan [76]. For L. Mexicana infections, thermotherapy with a single therapy of 50°C for 30 seconds at 8 weeks exhibited 90% CR [77]. The CRs were found to be 58–59% as compared to 72% for MA and 59% for miltefosine in Columbia [78,79]. A 60% CR was achieved when a low-cost heat pack (HECT-CL) was used at preliminary temperature of 52°C ± 2°C for 3 minutes to every wound and repeated for 7 days daily in Peru [80]. The efficacy of thermotherapy was found to be 73% as compared to 70% with SbV in a meta-analysis [81]. The disadvantages observed were pain, local anesthesia requirement, probe sterilization and likelihood of producing burns. Overall, thermotherapy is effective with least scarring, fewer treatments, no laboratory observations, increases patient compliance and is economical comparatively. It is preferred for CL treatment when there is low prevalence of mucocutaneous type and in patients contraindicated for systemic therapy [65].
Cryotherapy
Owing to smaller risk of mucosal involvement, cryotherapy (liquid nitrogen at −195°C) has been generally used for OWCL. This therapy involves intracellular ice formation producing cell destruction and leading to localized ischemic necrosis. Cryotherapy is applied to the lesion once or twice weekly up to 6 weeks and it was found > 90% efficacious in many countries for L. aethiopica and L. donovani infections except Turkey having less than 80% efficacy [82,83]. For single lesion NWCL cryotherapy against L. braziliensis infection, a CR of only 20% was seen in Bolivia [84]. Recently a meta-analysis showed that cryotherapy had similar efficacy as SbV but it was linked with adverse effects like erythema, hypo- or hyperpigmentation edema onsite, secondary infections and burning at times [65].
Carbon dioxide (CO2) laser
This procedure performs by explicit thermolysis of infested tissue with slight adverse effects in normal tissue [85]. CO2 laser therapy showed complete response in Iran between 93% - 93.7% and it was found to be more effective than MA showing 83.8% results. A single use of this therapy in Cuba healed all 10 patients without reverts in more than 2-year follow-up time. The adverse effects of this therapy most commonly were found to be hyperpigmentation, continuous erythema and hypertrophic scarring [44].
Combination Therapy
Multidrug therapy offers synergistic or additive action at diverse sites; it also reduces therapy duration and dosing requisite thus decreasing risks of toxicities, cost, and prevention of drug resistance [19]. In the 1980s, multidrug treatment with SSG and PM was accidentally revealed during VL epidemic in southern Sudan. Ever since, many African investigations and very few Indian studies have exhibited the advantage of 17 days therapy of PM with SbV. This combination was not adopted in India due to growing resistance to SbV. Yet, it was employed widely initially in relapse patients using conventional SbV and also as first-line VL treatment in Africa by Me´decins Sans Frontie’res (MSF) since 2002, which was permitted by WHO in 2010. A big pharmacovigilance study i.e. with (n = 3126) in Africa included individuals from Sudan, Kenya, Ethiopia, and Uganda and found an initial CR of 95%. It was observed to be safe and effective in the patients except those with co-infection HIV/VL having 56% CR or patients of more than 50 years of age showing 81.4% CR [86].
For Indian VL, a short course of amphotericin B with miltefosine due to the easy administration, efficacy and tolerance is preferred. In addition to protection of miltefosine from drug resistant parasites, the decrease in time and quantity of drug utilized contributes to decreased toxicity and cost [87]. Pentoxifylline is an inhibitor of tumour necrosis factor alpha; its use with pentavalent antimonial has proven to be effective against aggressive types of leishmaniasis like mucocutaneous leishmaniasis [88, 89]. This combination was also observed to be effective in Iranian CL patients [90]. For The use of topical or local therapy is not recommended in severe types of CL, nevertheless, combining a topical drug with a systemic agent may denote a stimulating choice and should be studied additionally [87].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Current Treatment Recommendations
The efficacy and essential dose of the antileishmanial therapies differ in various regions, therefore WHO issued the management recommendations in 2010 based on these variances [91]. In 2016 the guidelines were issued by the Infectious Diseases Society of America (IDSA) and the s. PAHO and WHO issued the treatment guidelines for leishmaniasis in 2018 in America [44].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Leishmaniasis Control: Future Directions
There is a strong need to invest in basic research focusing leishmaniasis control against its emergence and expansion. Effective vaccines against human leishmaniasis are required as they are not easily achievable due to long-life immunity convened by specific healed infections caused by Leishmania species [92]. The features of CL and VL are distinct as they are caused by diverse parasites. L. donovani infections require treatment if clinically obvious and relapse cases are common while a cured infection by L. major generally convenes immunity. This steered to investigation of cross-protection concept from a severe leishmaniasis type via immunity to a more benign type; in an animal infection model, safety from VL was accomplished by generation of a robust immunity to CL [93]. It logically supports for live vaccines and proposes that only one antigen vaccine against leishmaniasis may not be adequate to convene immunity. To date, the identified vector-derived virulence causes include promastigote secretory gel, exosomes, and saliva components. It is well-known that these vector-derived virulence factors are integral part of disease pathogenesis as they give rise to creation of parasites ensuing sand fly spread of Leishmania [94, 95].
Apart from vaccine exploration, new medications and insecticides are needed to avoid emergence of resistance in parasites and vectors both while enhancing the existing usage of diagnostic procedures and multidrug treatments [96]. Five vaccines have been approved and licensed to counter Leishmanial species so far. Among these 2 had been approved for human use; one is used in Brazil for immunotherapy of CL using killed L. amazonensis parasites and another in using live L. major parasites in Uzbekistan. In Brazil, recombinant proteins for prophylactic immunization of dogs are employed [97,98]. Licensed vaccines for human CL are unavailable currently and the ones forwarded to clinical trials are: (a) 2 killed L. amazonensis vaccines which have produced varying results between vaccine and placebo groups, and (b) defined polyprotein-containing subunit vaccine LEISHF1+ monophosphoryl lipid A plus squalene (MPL-SE) which was proven to be safe while producing an antigen-specific T helper 1(Th1) response [99].
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
Conclusion
Leishmaniasis is a neglected, vector-borne, complex disease that has been offering formidable challenges worldwide. Factors like population variability, climate change and globalization are lead to the expansion of this disease. Research advancements with combined commitments of governments and non-profit groups are continuously working for leishmaniasis control and the commitment of endemic states is essential for the success in long term. Development of effective vaccines is the ideal solution for leishmaniasis abolition from the world. It is possible to eliminate this disease in a few decades with new investigations discovering the realms of improved therapy of this ailment.
- Review Article
- Abstract
- Introduction
- Methodology for Review
- Results
- Discussion
- Epidemiology of Leishmaniasis
- Clinical Presentation of Leishmaniasis
- Diagnosis of Leishmaniasis
- Existing and Evolving Antileishmanial Agents
- Local therapy
- Current Treatment Recommendations
- Leishmaniasis Control: Future Directions
- Conclusion
- References
References
- Kammona O, Tsanaktsidou E (2021) Nanotechnology-aided diagnosis, treatment and prevention of leishmaniasis. International Journal of Pharmaceutics 605: 120761.
- Vera-Izaguirre DS, Vega-Memije E, Quintanilla-Cedillo MR, Arenas R (2006) Leishmaniasis. A review. Dermatología Cosmética, Médica Y Quirúrgica 4(4): 252-260.
- Andrade-Narvaez FJ, Canto Lara SB, Van Wynsberghe NR, Rebollar-Tellez EA, Vargas-Gon Azalez, et al. (2003) Seasonal transmission of Leishmania (Leishmania) mexicana in the state of Campeche, Yucatan Peninsula, Mexico. Memórias do Instituto Oswaldo Cruz 98: 995-998.
- Torres-Guerrero E, Ferreira CS, Branquinha MH, Santos ALS, Chaud MV et al. (2017) Leishmaniasis: A review. F1000Research p. 6.
- Ferreira CS, Branquinha MH, Santos ALS, Chaud MV, Santos ALS, et al. (2021) Overcoming multi‐resistant leishmania treatment by nanoencapsulation of potent antimicrobials. Journal of Chemical Technology & Biotechnology 96(8): 2123-2140.
- Khursheed Z, Hano C, Anjum I, Anjum S (2019) Applications of nanomaterials in leishmaniasis: a focus on recent advances and challenges. Nanomaterials 9(12): 1749.
- Volpedo G, Costa L, Ryan N, Halsey G, Satoskar A, et al. (2019) Nanoparticulate drug delivery systems for the treatment of neglected tropical protozoan diseases. Journal of Venomous Animals and Toxins including Tropical Diseases 25: e144118.
- Muhjazi G, Francesco Gabrielli A, Ruiz-Postigo JA, Atta H, Osman M, et al. (2019) Cutaneous leishmaniasis in Syria: A review of available data during the war years: 2011–2018. PLoS neglected tropical diseases 13(12): e0007827.
- Gurel MS, Tekin B, Uzun S, Sakib B, Croft Simon L, et al. (2018) Leishmaniasis. The Lancet 392(10151): 951-970.
- Maroli M, Feliciangeli MD, Bichaud L, Charrel RN, Gradoniet L et al. (2013) Phlebotomine sandflies and the spreading of leishmaniases and other diseases of public health concern. Medical and veterinary entomology 27(2): 123-147.
- Steverding D (2007) The history of leishmaniasis. Parasites & vectors 10(1): 1-10.
- Heras‐Mosteiro J, Monge-Maillo B, Pinart M, Lopez Pereira P, Reveiz L, et al. (2017) Interventions for Old World cutaneous leishmaniasis. Cochrane Database Syst Rev 11(11): CD005067.
- Akhoundi M, Kuhls K, Cannet A, Votýpka J, Marty P et al. (2016) A historical overview of the classification, evolution, and dispersion of Leishmania parasites and sandflies. PLoS neglected tropical diseases 10(3): e0004349.
- Shaw J, Pratlong F, Floeter-Winter L, Ishikawa E, El Baidouri F, et al. (2015) Characterization of Leishmania (Leishmania) waltoni n. sp.(Kinetoplastida: Trypanosomatidae), the parasite responsible for diffuse cutaneous leishmaniasis in the Dominican Republic. The American journal of tropical medicine and hygiene 93(3): 552-558.
- Espinosa O, Serrano MG, Camargo EP, Teixeira MMG, Shaw JJ, et al. (2018) An appraisal of the taxonomy and nomenclature of trypanosomatids presently classified as Leishmania and Endotrypanum. Parasitology 145(4): 430-442.
- Uzun S, Gürel MS, Durdu M, Akyol M, Karaman BF, et al. (2018) Clinical practice guidelines for the diagnosis and treatment of cutaneous leishmaniasis in Turkey. Int J Dermatol 57(8): 973-982.
- Vandeputte M, Van Henten S, Van Griensven J, Huits R, Van Esbroeck M, et al. (2020) Epidemiology, clinical pattern and impact of species-specific molecular diagnosis on management of leishmaniasis in Belgium, 2010–2018: A retrospective study. Travel Med Infect Dis 38: 101885.
- Sasidharan, S, Saudagar P (2021) Leishmaniasis: where are we and where are we heading? Parasitol Res 120(5): 1541-1554.
- Mondiale de la Santé O, W.H. Organization (2016) Leishmaniasis in high-burden countries: an epidemiological update based on data reported in 2014. Weekly Epidemiological Record= Relevé épidémiologique hebdomadaire 91(22): 286-296.
- Shaw J (2007) The leishmaniases-survival and expansion in a changing world: a mini-review. Memórias do Instituto Oswaldo Cruz 102(5): 541-547.
- Mansueto P, Seidita A, Vitale G, Cascio A (2014) Leishmaniasis in travelers: a literature review. Travel medicine and infectious disease 12(6): 563-581.
- Aspöck, H, Gerersdorfer T, Formayer H Walochniket J, et al. (2018) Sandflies and sandfly-borne infections of humans in Central Europe in the light of climate change. Wien Klin Wochenschr 120(4): 24-29.
- Manna P, YAkhter Y, Alaidarous M, Rub A (2017) Screening of novel inhibitors against Leishmania donovani calcium ion channel to fight leishmaniasis. Infect Disord Drug Targets 17(2): 120-129.
- Reithinger R, Dujardin JC, Louzir H, Pirmez C, Alexander B, et al. (2007) Cutaneous leishmaniasis. The Lancet infectious diseases (9): 581-596.
- Hoyos CL, Cajal SP, Juarez M, Marco JD, Alberti D'Amato AM et al. (2016) Epidemiology of American tegumentary leishmaniasis and Trypanosoma cruzi infection in the Northwestern Argentina. Biomed Res Int 2016: 645603.
- Miranda O, Gonzále I (2007) Leishmaniasis cutá Presentación de casos. Rev Cub Med Mil 36(4): 51-54.
- Bravo F, Sanchez MR (2003) New and re-emerging cutaneous infectious diseases in Latin America and other geographic areas. Dermatologic Clinics 21(4): 655-668.
- Organization WH (2006) World Health Organization Weekly Epidemiologic Record. World Health Organization (WHO)(2007). Schistomiasis. WHO Fact Sheet (115): 145-164.
- Özkeklikçi A (2017) The new situation of cutaneous leishmaniasis after Syrian civil war in Gaziantep city, Southeastern region of Turkey. Acta Tropica 166: 35-38.
- Khezzani B, Bouchemal S (2017) Demographic and spatio-temporal distribution of cutaneous leishmaniasis in the Souf oasis (Eastern South of Algeria): Results of 13 years. Acta Tropica 166: 74-80.
- Holakouie-Naieni K, Mostafavi E, Boloorani AD, Mohebali M,Pakzad R, et al. (2017) Spatial modeling of cutaneous leishmaniasis in Iran from 1983 to 2013. Acta tropic 166: 67-73.
- Khosravani, M, Moemenbellah-Fard MD, Sharafi M, Azam Rafat-Panah A, et al. (2016) Epidemiologic profile of oriental sore caused by Leishmania parasites in a new endemic focus of cutaneous leishmaniasis, southern Iran. Journal of Parasitic Diseases 40(3): 1077-1081.
- Pandey BD, Bahadur Pun S, Kaneko O, Pandey K, Hirayamaet K, et al. (2011) Case report: Expansion of visceral leishmaniasis to the western hilly part of Nepal. The American journal of tropical medicine and hygiene 84(1): 107.
- Cruz-Ruiz AL and M.d.R. García-Miss, Incriminación del" Sand-fly" como vector de leishmania.
- Inchaustegui, A., De la leishmaniosis americana y de la úlcera de los cjicleros en Mé 1918: Talleres Gráficos de la Escuela Industrial de Huérfanos.
- Córdova C (1993) Leishmaniasis: estudio epidemiológico preliminar en una localidad de la zona endémica del estado de Tabasco. Salud Pública de México 35(4): 345-350.
- Albertos-Alpuche N (1990) Vectores de la leishmaniasis cutánea en Mé Rev Biomed 1(2): 92-102.
- Albertos Alpuche NE, Patrón B, Pérez V (1996) Leishmaniasis cutánea localizadaíndice alérgico en la comunidad de Becanchén, Tekax, Yucatán, Mé
- Canto-Lara SB, Cardenas-Maruffo MF, Vargas-Gonzalez A, Andrade-Narvaez F et al. (1998) Isoenzyme characterization of Leishmania isolated from human cases with localized cutaneous leishmaniasis from the State of Campeche, Yucatan Peninsula, Mexico. The American journal of tropical medicine and hygiene 58(4): 444-447.
- Andrade-Narvaez FJ, Simmonds-Diaz E, Rico-AguilarS, Andrade-Narvaez M, et al. (1990) Incidence of localized cutaneous leishmaniasis (chiclero's ulcer) in Mexico. Transactions of the Royal Society of Tropical Medicine and Hygiene 84(2): 219-220.
- Van Wynsberghe N, Canto-Lara SB, Sosa-Bibiano EI, Rivero-Cárdenas NA, et al. (2009) Comparison of small mammal prevalence of Leishmania (Leishmania) mexicana in five foci of cutaneous leishmaniasis in the state of Campeche, Mexico. Revista do Instituto de Medicina Tropical de São Paulo 51: 87-94.
- Rebollar-Téllez EA, Ramírez-Fraire A, Andrade-Narvaez FJ (1996) A two years study on vectors of cutaneous leishmaniasis: Evidence for sylvatic transmission cycle in the State of Campeche, Mexico. Mem Inst Oswaldo Cruz 91(5): 555-560.
- Chakravarty J, Sundar S (2019) Current and emerging medications for the treatment of leishmaniasis. Expert opinion on pharmacotherapy 20(10): 1251-1265.
- Assis TSMde, Pinheiro Rosa DC, Eliane de Morais Teixeira Ede, G Cota G, Azeredo-da-Silva ALF, et al. (2017) The direct costs of treating human visceral leishmaniasis in Brazil. Revista da Sociedade Brasileira de Medicina Tropical 50(4): 478-482.
- Bravo F (2018) Protozoa and worms. Dermatology. Fourth edition. Beijing: Elsevier 1470-1502.
- Zijlstra EE (2016) The immunology of post-kala-azar dermal leishmaniasis (PKDL). Parasites & vectors 9(1): 1-9.
- Rodríguez-Cortés A (2010) Leishmania infection: laboratory diagnosing in the absence of a “gold standard”. The American journal of tropical medicine and hygiene 82(2): 251.
- Aronson N, Herwaldt BL, l Libman M, Pearson R, Lopez-Velez R, et al. (2016) Diagnosis and treatment of leishmaniasis: clinical practice guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Clinical infectious diseases 63(12): 202-264.
- de Paiva-Cavalcanti M (2015), Leishmaniases diagnosis: an update on the use of immunological and molecular tools. Cell & bioscience 5(1): 1-10.
- Reed GH, Kent JO, Wittwer CT (2007) High-Resolution DNA Melting Analysis for Simple and Efficient Molecular Diagnostics. Pharmacogenomics 8(6).
- Galluzzi L, Marcello Ceccarelli, Aurora Diotallevi, Michele Menotta, Mauro Magnani (2018) Real-time PCR applications for diagnosis of leishmaniasis. Parasites & vectors 11(1): 1-13.
- Bretagne S, Durand R, Olivi M, Garin JF, Sulahian A, et al. (2001) Real-time PCR as a New Tool for Quantifying Leishmania infantum in Liver in Infected Mice. Clinical Diagnostic Laboratory Immunology 8(4): 828-831.
- Nicolas L, Prina E, Lang T, Milon G (2002) Real-time PCR for Detection and Quantitation of Leishmania in Mouse Tissues. Journal of Clinical Microbiology 40(5): 1666-1669.
- Nath Chowdhury M, Sangaralingam M, Bastien P, Ravel C, Pratlong F, et al. (2016) Real-time PCR using FRET technology for Old World cutaneous leishmaniasis species differentiation. Parasites & Vectors 9(1): 1-11.
- Tsukayama P, et al. (2013) A FRET-Based Real-Time PCR Assay to Identify the Main Causal Agents of New World Tegumentary Leishmaniasis. PLoS Neglected Tropical Diseases 7(1): e1956.
- Thakur S, Joshi J, Kaur S (2020) Leishmaniasis diagnosis: An update on the use of parasitological, immunological, and molecular methods. Journal of Parasitic Diseases 44(2): 253-272.
- Fernandez Crehuet P, Ruiz Villaverde R (2017) White starburst-like pattern as a dermoscopic clue in Old World cutaneous leishmaniasis. An Bras Dermatol 92(2): 266-267.
- Bustamante MLP (2017) Dermatoscopic signs in cutaneous leishmaniasis. An Brasileiros de Dermatologia 92: 844-846.
- Husein ElAhmed H, Gieler U, Steinhoff M (2020) Evidence supporting the enhanced efficacy of pentavalent antimonials with adjuvant therapy for cutaneous leishmaniasis: a systematic review and meta‐analysis. JEADV 34(10): 2216-2228.
- Rijal S, Chappuis F, Singh R, P A Bovier PA, Acharyaet P, et al. (2003) Treatment of visceral leishmaniasis in south-eastern Nepal: decreasing efficacy of sodium stibogluconate and need for a policy to limit further decline. Transactions of the Royal Society of Tropical Medicine and Hygiene 97(3): 350-354.
- Roatt BM, Cardoso JMO, Brito RCF, Coura Vital W, Aguiar Soares RDO, et al. (2020) Recent advances and new strategies on leishmaniasis treatment. Applied Microbiology and Biotechnology 104(21): 8965-8977.
- Diro E, Lynen L, Mohammed R, Boelaert M, Hailu A, et al. (2014) High parasitological failure rate of visceral leishmaniasis to sodium stibogluconate among HIV co-infected adults in Ethiopia. PLoS Neglected Tropical Diseases 8(5): e2875.
- Thakur CP, Singh RK, Hassan SM, Kumar R, Narain S, et al. (1999) Amphotericin B deoxycholate treatment of visceral leishmaniasis with newer modes of administration and precautions: a study of 938 cases. Transactions of the Royal Society of Tropical Medicine and Hygiene 93(3): 319-323.
- Pradhan S, Schwartz RA, Patil A, Grabbe S, Goldust M (2022) Treatment options for leishmaniasis. Clinical and experimental dermatology 47(3): 516-521.
- Meeting WECotCotL (2010) W.H. Organization, Control of the Leishmaniases: Report of a Meeting of the WHO Expert Committee on the Control of Leishmaniases p. 22-26.
- Sundar S, Chakravarty J, Agarwal D, Rai M, Murray HW (2010) Single-dose liposomal amphotericin B for visceral leishmaniasis in India. N Engl J Med 362(6): 504-512.
- Sundar S, Jha TK, Thakur CP, Engel J, Sindermannet H (2002) Oral miltefosine for Indian visceral leishmaniasis. N Engl J Med 347(22): 1739-1746.
- Diro E, Ritmeijer K, Boelaert M, Alves F, Mohammed R, et al. (2018) Long-term clinical outcomes in visceral leishmaniasis/human immunodeficiency virus–coinfected patients during and after pentamidine secondary prophylaxis in ethiopia: a single-arm clinical trial. Clin Infect Dis 66(3): 444-451.
- Wyllie S, Stephen Patterson, Laste Stojanovski, Suzanne Norval, Robert Kime, et al. (2012) The Anti-Trypanosome Drug Fexinidazole Shows Potential for Treating Visceral Leishmaniasis. Science Translational Medicine 4(119): 119re1.
- Dawson R, Diacon AH, Everitt D, van Niekerk C, Donald PR et al. (2015) Efficiency and safety of the combination of moxifloxacin, pretomanid (PA-824), and pyrazinamide during the first 8 weeks of antituberculosis treatment: a phase 2b, open-label, partly randomised trial in patients with drug-susceptible or drug-resistant pulmonary tuberculosis. The Lancet 385(9979): 1738-1747.
- Patterson S, Wyllie S, Stojanovski L, Perry MR, Norval S, et al. (2013) The R Enantiomer of the Antitubercular Drug PA-824 as a Potential Oral Treatment for Visceral Leishmaniasis. Antimicrobial Agents and Chemotherapy 57(10): 4699-4706.
- Patterson S, Wyllie S, Norval S, Stojanovski L, Rc Simeons F, et al. (2016) The anti-tubercular drug delamanid as a potential oral treatment for visceral leishmaniasis. Elife 5: e09744.
- Safi N, Davis GD, Nadir M, Hamid H, Robert LL (2012) Evaluation of thermotherapy for the treatment of cutaneous Leishmaniasis in Kabul, Afghanistan: A randomized controlled trial. Military medicine 177(3): 345-351.
- Bumb R, Prasad N, Khandelwal K, Aara N, Mehta RD, et al. (2013) Long‐term efficacy of single‐dose radiofrequency‐induced heat therapy vs. intralesional antimonials for cutaneous leishmaniasis in India. British Journal of Dermatology 168(5): 1114-1119.
- Aronson NE, Wortmann GW, Byrne WR, Howard RS. Bernstein WB et al. (2010) A randomized controlled trial of local heat therapy versus intravenous sodium stibogluconate for the treatment of cutaneous Leishmania major infection. PLoS neglected tropical diseases 4(3): e628.
- Velasco-Castrejon O, Walton C, Rivas-Sanchez B, Garcia MF, et al. (1997) Treatment of Cutaneous Leishmaniasis witth Localized current field. Am J Trop Med Hyg 57(3): 309-312.
- López L, Robayo M, Vargas M, Vélezet ID, et al (2012) Thermotherapy. An alternative for the treatment of American cutaneous leishmaniasis. Trials 13: 58.
- López L, Cruz C, Godoy Sara M G, Vélez RD (2013) Thermoterapy effective and safer than miltefosine in the treatment of cutaneous leishmaniasis in Colombia. Revista do Instituto de Medicina Tropical de São Paulo 55: 197-204.
- Valencia BM, Miller D, Witzig RS, Boggild AK, Llanos-Cuentas A, et al. (2013) Novel low-cost thermotherapy for cutaneous leishmaniasis in Peru. PLoS neglected tropical diseases 7(5): e2196.
- Cardona-Arias JA, Vélez IDLopez-Carvajal L (2015) Efficacy of thermotherapy to treat cutaneous leishmaniasis: a meta-analysis of controlled clinical trials. PLoS One 10(5): e0122569.
- Negera E, Gadisa E, Hussein J, Engers H, Kuru T, et al. (2012) Treatment response of cutaneous leishmaniasis due to Leishmania aethiopica to cryotherapy and generic sodium stibogluconate from patients in Silti, Ethiopia. Trans R Soc Trop Med Hyg 106(8): 496-503.
- Ranawaka R, Weerakoon HS, Opathella N (2011) Liquid nitrogen cryotherapy on Leishmania donovani cutaneous leishmaniasis. Journal of dermatological treatment 22(4): 241-245.
- Soto J, Rojas E, Guzman M, Verduguez A, Nena W, et al. (2013) Intralesional antimony for single lesions of bolivian cutaneous leishmaniasis. Clinical infectious diseases 56(9): 1255-1260.
- Nassif PW, Perles DE Mello TF, Navasconi TR, Mota CA, Demarchi IG, et al. (2017) Safety and efficacy of current alternatives in the topical treatment of cutaneous leishmaniasis: a systematic review. Parasitology 144(8): 995-1004.
- Kimutai R, Musa AM, Njoroge S, Omollo R, Alves F, et al. (2017) Safety and effectiveness of sodium stibogluconate and paromomycin combination for the treatment of visceral leishmaniasis in eastern Africa: results from a pharmacovigilance programme. Clinical drug investigation 37(3): 259-272.
- Uliana SR, Trinconi CT, Coelho AC (2018) Chemotherapy of leishmaniasis: present challenges. Parasitology 145(4): 464-480.
- Lessa HA, Machado P, Lima F, Cruz AA, Bacellaret O, et al. (2001) Successful treatment of refractory mucosal leishmaniasis with pentoxifylline plus antimony. The American journal of tropical medicine and hygiene 65(2): 87-89.
- Machado PR, Lessa H, Lessa M, Guimarães LH, Bang H, et al. (2007) Oral pentoxifylline combined with pentavalent antimony: a randomized trial for mucosal leishmaniasis. Clin Infect Dis 44(6): 788-793.
- Sadeghian, G, Nilforoushzadeh M (2006) Effect of combination therapy with systemic glucantime and pentoxifylline in the treatment of cutaneous leishmaniasis. Int J Dermatol 45(7): 819-821.
- Sundar S, Chakravarty J (2013) Leishmaniasis: an update of current pharmacotherapy. Expert Opin Pharmacother 14(1): 53-63.
- Gillespie PM, Beaumier CM, Strych U, Hayward T, Hotezet PJ et al. (2016) Status of vaccine research and development of vaccines for leishmaniasis. Vaccine 34(26): 2992-2995.
- Romano A, Doria NA, Mendez J, Sacks DL, Peters NC, et al. (2015) Cutaneous infection with Leishmania major mediates heterologous protection against visceral infection with Leishmania infantum. J Immunol 195(8): 3816-3827.
- Abdeladhim M, Kamhawi S, Valenzuela JG (2014) What’s behind a sand fly bite? The profound effect of sand fly saliva on host hemostasis, inflammation and immunity. Infect Genet Evol 28: 691-703.
- Atayde VD, Aslan H, Townsend S, Hassani K, Kamhawi S et al. (2015) Exosome secretion by the parasitic protozoan Leishmania within the sand fly midgut. Cell reports 13(5): 957-967.
- Mo A, Pesce J, Hall BF (2016) Visceral leishmaniasis control and elimination: is there a role for vaccines in achieving regional and global goals? Am J Trop Med Hyg 95(3): 514.
- Marcondes M, Day MJ (2019) Current status and management of canine leishmaniasis in Latin America. Research in veterinary science 123: 261-272.
- Starita C, Gavazza A, Lubas G (2016) Hematological, biochemical, and serological findings in healthy canine blood donors after the administration of CaniLeish® Vet Med Int 2016: 4601893.
- Coutinho De Oliveira B, Duthie MS, Alves Pereira VR (2020) Vaccines for leishmaniasis and the implications of their development for American tegumentary leishmaniasis. Hum Vaccin Immunother 16(4): 919-930.






























