Unusual Orocutaneous Manifestations of Tuberous Sclerosis Complex in 49 Patients: Follow Up Over 25 Years, and Treatment Outcomes
Ahlam Mohammad Alshami1, Mohammad Ali Alshami2*, Mona Jameel Mohana2, Amat Alkhaliq Mohammad Al-Sayaghi1 and Hadeel Mohammad Alshami2
1Department of Conservative Dentistry, Faculty of Dentistry, Sana’a University, Sana’a, Yemen
2Department of Dermatology and Venerology, Faculty of Medicine and Medical sciences, Sana’a University, Sana’a, Yem
Submission: May 20, 2022;Published: August 09, 2022
*Corresponding author: Mohammad Ali Alshami, Department of Dermatology and Venerology, Faculty of Medicine and Medical Sciences, Sana’a University, Sana’a, Yemen
How to cite this article: Alshami AM, Alshami MA, Mohana MJ, Al-Sayaghi AM, Alshami HM. Unusual Orocutaneous Manifestations of Tuberous Sclerosis Complex in 49 Patients: Follow Up Over 25 Years, and Treatment Outcomes. JOJ Dermatol & Cosmet. 2022; 4(4): 555642. DOI: 10.19080/JOJDC.2022.04.555642
Abstract
Background and purpose of the study: Tuberous sclerosis complex (TSC) is an autosomal dominant inherited neurocutaneous genodermatosis characterized by hamartomas in many organs especially the skin, brain, eye, kidney, heart, and lungs. No studies assessing the pattern and frequency of orocutaneous manifestations of TSC has been published from Arabian & Middle East countries. The orocutaneous features play a significant role in early recognition of that syndrome. This study aims: first to assess the frequency and characteristics of the common, and unusual orocutaneous manifestations of TSC in 49 Yemeni patients, second to show the results of follow up of some patients for up to 25 years, and third to present the outcome of electrocautery, CO2 laser, and TCA peel, in treating facial angiofibromas, fibrous cephalic plaques, mollusca fibrosa, and ungual fibromas in Yemeni patients.
Method: A prospective clinical study, conducted over a 25-years period, from November 1997 to April 2022, at the dermatology clinic of Kuwait university hospital in Sana’a city, Yemen. Suspected cases who presented to the clinic during the study period were thoroughly investigated for orocutaneous or systemic features of TSC, by two dermatologists, and two dentists, until the diagnosis of TSC was confirmed according to the updated diagnostic criteria for TSC 2012. Those individuals with confirmed TSC diagnosis regardless of age or sex, were selected for the study after obtaining a formal consent either from the patients or from their caregivers. Diagnoses, and assessments were performed by those dermatologists, and dentists, who also made the records in the medical files, and documented the finding by taking color photos of every patient. Age of diagnosis is included in Table 1. The study variables included age, sex, family history of TSC, and orocutaneous manifestations of TSC. Data analysis was performed using the SPSS version 21.
Results: TSC was diagnosed in 49 patients, 26 were male, and 23 females. The male to female ratio was (1.1:1) and the average age was 17.4 years. Positive family history was found in 6 patients (12.2%). All patients (100%) had one or more of the TSC orocutaneous signs. Facial angiofibromas, shagreen plaques, and hypopigmented macules were seen in all patients (100%), followed by fibrous cephalic plaques, seen in 40 (82%), ungual fibromas in 25 (51%), molluscum pendulum in 22 (45%), and confetti like hypopigmentation in 3 patients (6%). Out of the 49 TSC patients 17 underwent oral examination, that revealed oral fibromas in 15/17 (88%) and Enamel pits in 12/17(70%) patients. Unusual orocutaneous features of TSC, which have been not, or rarely reported in association with TSC, seen in our patients, were pachydermodactyly, macrodactyly, congenital localized sacral hypertrichosis, onychomatricoma, Becker nevus, cutaneous syndactyly of toes, port wine stain, digital fibrokeratoma, agminated zosteriform lentiginosis, and nail pits.
Conclusion: Conspicuous orocutaneous clinical features are particularly common, and easily visible manifestation of TSC, and so they may offer important diagnostic clues for the dermatologist, and for the dentist as well.
Keywords:Tuberous Sclerosis; Dental pits, Oral mucosal fibromas
Abbreviations:FCP: Fibrous Cephalic Plaque, CLS: Confetti Like Skin Lesions, DP: Dental pit, FAF: Facial Angiofibromas, FH: Family History, HM: Hypomelanotic Macule, MD: Macrodactyly, MFP: Molluscum Fibrosum Pendulum, OF: Oral Fibroma, SHP: Shagreen Plaques, UF: Ungual Fibroma
Introduction
Historical Background
Tuberous sclerosis complex (TSC), an autosomal dominant neurocutaneous syndrome with variable clinical expression, is also known as Epiloia or Bourneville-Pringle disease. Hamartomas are found in many organs, most often the skin, kidney, and central nervous system [1]. Some of its various clinical manifestations, were gained from isolated case reports, describing results of a pathology of an unknown disease, more than 150 years ago. Virchow and Von Recklinghausen identified hamartomas in the brain and heart, respectively, during the mid- 19th century [2]. After a postmortem examination of the brains of patients who had seizures and cognitive impairment, the term ‘’Tuberous’’ was coined due to the aspect of the tumors found, which look like tuberous potatoes. Bourneville first reported the discovery of this new syndrome, at the end of the 19th century, and concomitantly described its findings [3]. John James Pringle, a Scottish dermatologist described the facial angiofibromas, for the first time. He mistakenly called it adenoma sebaceum. The eponyms “Pringle’s adenoma sebaceum’, was awarded to him [4].
Orocutaneous manifestations started only in the early 20th century to be related to the disease described by Bourneville, although they had been described previously by other authors. Campbell, in 1905, and Vogt, in 1908, were the first to recognize the relationship between mental retardation, epilepsy, and sebaceous adenoma, which was later established, and formed the Vogt triad characterizing TSC [5].
Edward Sherlock, a barrister-at-law and lecturer in biology, coined the term ‘Epiloia’, a portmanteau of epilepsy, low intelligence (anoia, or mindless), and angiofibroma in 1911 [6] Jan van der Hoeve, a Dutch ophthalmologist described the retinal hamartomas (phakoma), in 1920 and grouped both TSC and neurofibromatosis together as “phakomatosis” (later called neurocutaneous syndromes) [7].
The era of molecular biology started after years of studies limited to, clinical, pathological, and certain genetic aspects of TSC. Signaling pathways of hamartoma growth, are the new objects of interest. Researchers have recently obtained great success recognizing proteins, enzymes and signals associated with the etiopathological process of the syndrome. These findings can help understand diseases with uncontrolled growth of tumor cells as a common causal factor, such as TSC [8]. TSC is a complex condition, due to diverse schedule of the emergence of its manifestations the symptoms and signs they create. The most affected organ systems are the central nervous system (CNS), skin, kidney, lung, and heart are, despite the fact that, not all of these manifestations are specific to TSC [9].
TSC requires attention from various medical specialties, as it is a complex clinical entity with highly variable clinical manifestations. Since its orocutaneous features represents the most prevalent finding, the dermatologist plays an essential role in early diagnosis, and intervention in the natural course of this syndrome [10].
Epidemiology
Tuberous sclerosis complex (TSC) is a rare, autosomal dominant inherited disease manifesting with the development of benign tumors in many organs of the body. It is caused by mutations in either the TSC1 or TSC2 genes, encoding the proteins hamartin and tuberin, respectively, which are detected in approximately 85- 90% of TSC cases [11,12]. A family history is found in only 30% of patients, while the majority of TSC cases occur sporadically [13- 15].
It has an estimated incidence between 1/6000 and 1/10,000 live births and an estimated population prevalence of 1/20,000. A population- based studies in the UK, stated that, approximately 1 in 12,000-14,000 children under 10 years of age have TSC [13,14] TSC seems to affect 1 to 2 million individuals worldwide. In Caucasian population, its estimated prevalence is around one case for every 10000 to 25000individuals. According to a study in Taiwan, its estimated prevalence was stated to be 1:95,136 and the prevalence was 1:14,608, for cases younger than six years of age [15]. According to that study only 15% of patients had a family history of TSC.
The period prevalence rate of tuberous sclerosis complex for children and adolescents younger than 20 years, was determined to be 3.5 per 10,000 for China (on Hong Kong Island excluding the eastern region with 125,100 youngers than 20 years in 2003), as stated in a study which consisted of 44 cases over a 12-month period (January- December 2003) [16].
Epidemiological studies generally underestimate the prevalence of the disease, because of the variation in gene expressivity between affected individuals, both intra- as well as interfamilial.
Different results regarding incidence and prevalence of Tuberous Sclerosis Complex, are gained from different epidemiological studies. Its recognition is sometime impeded, due to its great phenotypic variability [17,18] 1 in 10,000 newborns, are affected and most of patients are diagnosed with the disease within the first 15 months of life [19]. Characteristic clinical signs were overlooked during a previous medical evaluation in about 25% of the individuals diagnosed after that age [20]. An estimated prevalence of 1:6,000 persons in the general population, was calculated, by using new techniques of genetic studies and by defining new clinical manifestations. [20,21].
TSC affects both sexes equally, although women may show more prominent signs. It occurs in all races and ethnic groups. The early- onset seizures most often leads to diagnosis of the syndrome [22]. Characteristic orocutaneous manifestations may help early diagnose the disease; however, they tend to appear at later stages [22-24]. The leading causes of mortality and morbidity are neurological and renal complications, although the incidence of certain organ systems involvements is variable [25,26].
Etiopathogenesis
Inactivating mutations of either TSC1 or TSC2 tumor suppressor genes, located on chromosomes 9q34 and 16p13.3, respectively, is the cause of TSC. The TSC1 gene encodes hamartin and TSC2 encodes tuberin, which form the hamartin-tuberin complex, acting as a tumor growth inhibitor. Its absence stimulates cell proliferation and tumor formation [27-29].
Many types of mutations can be encountered, such as deletions, insertions, nonsense, and missense mutations [30]. Despite verification of such changes, mutations are not identified in 15% of the cases [31-39]. Germline mutations are responsible for the familial forms of TSC, even though their transmission can be hereditary, while somatic mutations are responsible for 70% of the cases, especially TSC2 mutations [40]. Sporadic cases of the disease can be explained by somatic mutations. The highly variable mutations and the involvement of multiple genes explain, partially, the diverse phenotypes of the disease [40,41]. Mosaicism is another factor that could explain, the multifaceted, and capricious clinical manifestations of TSC [41,42].
In sporadic cases, The TSC2 mutations are five times more frequent than that of TSC1, contrary to familial cases, where TSC2, and TSC1 mutations occurs with similar frequency. TSC2 mutations leads to more severe disease, than TSC1 mutations. Familial Cases, associated with TSC1 mutations, present with mild to moderate disease, at times not meeting the various diagnostic criteria [42,43].
Not fully understood is how these mutations lead to cellular hyper-proliferation and development of many hamartomas in any organ of the human body. Scientists began to understand some of the steps of this proliferation pathway. The human cells respond steady to a broad spectrum of coordinated biological stimuli. Malfunctions of this signaling mechanism will lead to development of benign as well as malignant tumors, as seen in TSC [44,45]. The TSC1 and TSC2 genes regulate cell growth, through the phosphoinositide 3-kinase signaling pathway, which inhibits the mechanistic target of rapamycin (mTOR), a major sensor of energy and nutrient availability, therefore regulating cell growth, differentiation, and proliferation. The hamartin-tuberin complex creates a heterodimer that inhibits (Rheb), the signaling protein, known as “RAS homolog enriched in brain”. mTOR, sequentially, regulates the phosphorylation of ribosomal protein S6 kinase 1 and 2 (S6K1 and S6K2) isoforms & of the protein inhibiting translation initiation, 4E-BP1 (also known as PHAS-1) after stimulation by Rheb [46,47].
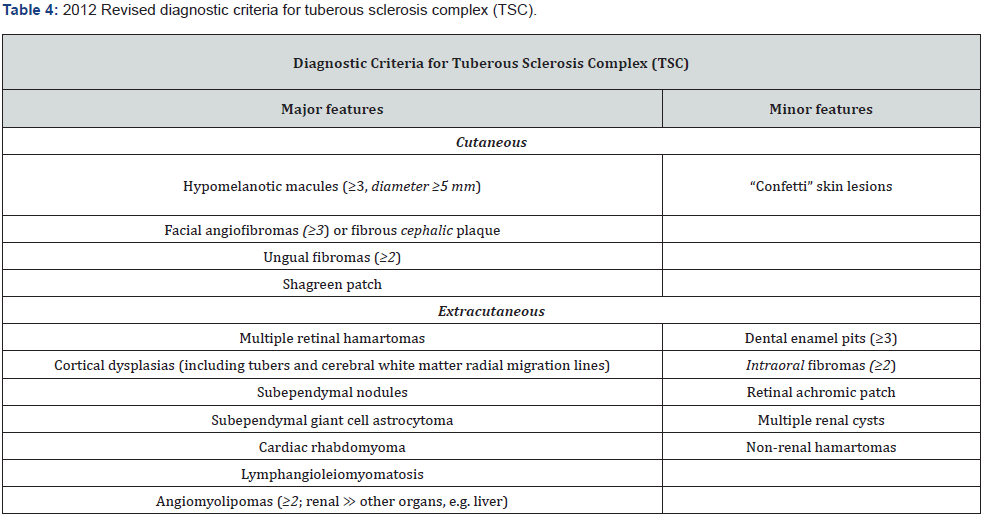
The absence of suppressive effect of the hamartin-tuberin complex on Rheb permits mTOR to stimulate the S6K and 4EBP1 proteins, resulting in an increase in ribosomal biogenesis, and protein synthesis, which finally terminates in unrestricted cell proliferation, and the appearance of tumors seen in tuberous sclerosis [48,49]. Hence, TSC1 or TSC2 mutations result in essential activation of mTOR leading to development of hamartomas in many organs of the body, especially the brain, kidney, heart, and lung [48]. The greatest clinical variability of TSC and the lack of population-based studies assessing the frequency of individual clinical features in the general population complicate establishing definitive diagnostic criteria [49]. However there seems to be an agreement in the literature regarding criteria for clinical diagnosis. Roach first formulated the clinical diagnostic criteria in 1998 at the Tuberous Sclerosis Complex Consensus Conference [49], which were updated in 2012. Inclusion of genetic testing results, along with reducing diagnostic classes from three (possible, probable, and definite) to two (definite, possible), constitute the Major changes compared with 1998 criteria. According to the 2012 International TSC Consensus Group guidelines, clinical features of TSC proceed to be the essential tools of diagnosis, requiring the presence of two major features or one major plus two minor features for a definitive diagnosis while the presence of either one major or two or more isolated minor features are needed for a ‘possible’ clinical diagnosis (Table 4).
In addition, recognition of a pathogenic mutation of TSC1 or TSC2 by genetic testing is considered an independent diagnostic criterion, even though current testing is not able to identify such a mutation in 10-15% of clinically affected patients. Selective organ imaging and laboratory testing, including genetic evaluation support the diagnosis of TSC. Supplementary investigations are used principally to confirm the diagnosis, determine the extent of disease, and to clarify the recurrence risks for sibling [50]. Although no specific symptoms for TSC exist, several common presentations, should evoke consideration of TSC and additional investigation. Identification of a family member with TSC, is the first and paramount step. There is an up to 50% chance for an under evaluation to have TSC, if the family member is a first-degree relative (i.e., a parent or siblings). This 50% risk persists when multiple siblings are afflicted and may occur surprisingly if the affected parent is oblivious of his or her diagnostic condition. A gonadal mosaicism in a clinically unaffected parent, a condition by which the gonads and their affected gametes are the only organ affected with the TSC mutation, is considered an additional, albeit rare, cause of TSC, which permits the potential for an unaffected parent to produce multiple affected children [51]. The various schedule of the onset of manifestations and the symptoms and signs they generate increase the complexity of the condition. Infantile spasms are regularly the initial manifestations to be recognized in TSC patients [52]. Despite that orocutaneous and neurodevelopmental manifestations typically present early in life, they are commonly misdiagnosed. Although renal involvement is discovered at an earlier age (3-5 years) and presumably starts even earlier, the complications they cause, are often first noticed in adolescence or adulthood. The pulmonary Lymphangioleiomyomatosis (LAM), affects almost exclusively female TSC patients, and manifests typically in adults. Abnormalities of the bone and eye have been frequently reported, although they have no diagnostic value. The auditory, endocrine, hepatic, and gastrointestinal systems are infrequently affected by TSC. Some additional tests should be performed earlier at the onset of clinical manifestations of TSC, in an effort to verify the diagnosis and to illuminate the causes of symptoms. Hence, in case of suspected TSC, cranial computed tomography (CT) or magnetic resonance imaging (MRI) must be done; and in case of seizures electroencephalogram must be requested; while renal ultrasonography must be performed in all suspected cases; pulmonary CT must be requested for women; and pulmonary function tests for women with respiratory symptoms. Echocardiography, electrocardiogram, should be requested for children with suspected cardiac involvement. Renal function tests should be requested in cases of children with polycystic kidneys of adults with extensive renal involvement. One should also assess the neuropsychomotor development and behavior to prove the presence of autism, hyperactivity, and mental retardation [53].
The presence of rhabdomyomas, can be seen by echocardiography, prior to neurological and orocutaneous features, and even prenatally. Subependymal nodules, often calcified, present in the walls of the lateral ventricles, which may be protruding into the ventricular cavity, can be visualized by Cranial CT scanning. MRI of the brain using FLAIR sequences proves to be superior to other neuroimaging studies, for evaluation of cortical tubers, which may be associated with white matter abnormalities, such as radial migration lines. Tubers in the central nervous system, can be seen by fetal MRI, from the 26th week of pregnancy [53]. The identification of diagnostic features should prompt further examination of an underlying diagnosis of TSC. The brain, renal, dermatological, respiratory, and circulatory organ systems are most frequently affected, while few of these manifestations are peculiar to TSC. Orocutaneous features are the most often and easily visible manifestation of TSC. One or more orocutaneous lesions, are seen in more than 90% of patients with TSC, which typically evolve early in life. The dermatologist must be able to identify TSC-related skin manifestations to warrant prompt diagnosis, early treatment institution and multifaceted referral for follow-up of another TSC-related outcome.
This study concentrates primarily on orocutaneous TSCassociated features. Aside from positive genetic testing confirming a pathological TSC1 or TSC2 mutation, the clinical diagnosis of TSC depends on a combination of recognizable major and minor characteristics, with orocutaneous findings comprising the greatest of both major (hypomelanotic macules, angiofibromas, ungual fibromas, shagreen plaque) and minor (‘confetti’ skin lesions) features. Orocutaneous manifestations of TSC are easily observable during close physical examination. Besides internal organ evaluation, a detailed orocutaneous examination is advocated upon diagnosis of TSC, superseded by not less than annual orocutaneous examinations [54]. The subtypes of orocutaneous lesions do appear in an age-related way, myriad arising early in life. Some can be subtle, principally in young children, besides that not all are specific for TSC.
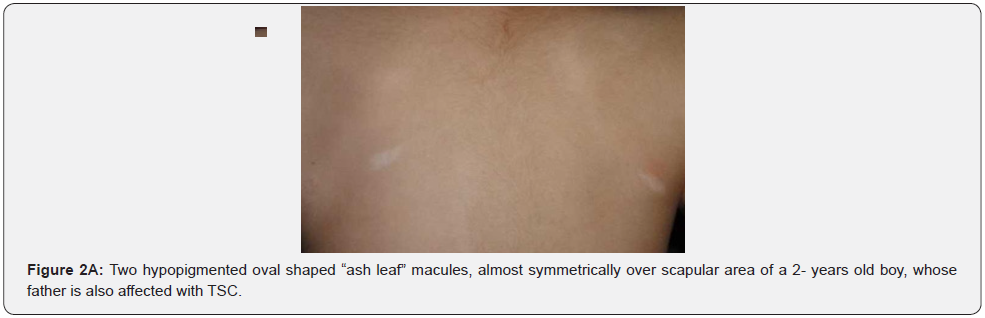
Facial angiofibromas, the most visually apparent TSCassociated orocutaneous feature, were inaccurately named adenoma sebaceum, often start to develop during the first 2– 5 years of life and basically occur in about 75% of patients. They are usually rose to brown, red papulonodules with a flat, glassy surface and are routinely equally bilaterally dispersed over the central face, occasionally misdiagnosed as acne [55,56]. They grow gradually and accelerate during puberty [56]. Fibrous cephalic plaques are considered histologically a larger variant of angiofibromas. They are firm, elevated, tan to yellow-brown colored plaques, routinely seen on the forehead or scalp. They can occur at any age, have different size and shape and can be as large as several centimeters in diameter [55,56]. The earliest and most frequently seen orocutaneous features in TSC are the hypomelanotic macules. They have different sizes, various morphologies and should be differentiated from depigmented patches of vitiligo. In fair-skinned individuals, a Wood lamp, can help recognize hypomelanotic macules by making them more visible [57]. They are one of the earliest visible signs of TSC, found in >50% of children at birth and in almost all patients by the age of 2. They are named ‘ash leaf’ spots due to their resemblance to the leaf of the eastern mountain ash tree, long oval with one tapered and one rounded end. The thumbprint-like shaped, usually (0.5-2 cm in diameter), are the most discovered ones.
Hypomelanotic macules are frequently seen asymmetrically distributed on the trunk and buttocks. Poliosis can signal a rare scalp involvement [57]. Another type of hypomelanotic macules, are small numerous, (approximately 1-3 mm in diameter) ‘Confettilike’ hypomelanotic macules, usually symmetrically distributed over the distal extremities, which are found in 2.8-28% of patients with TSC. They can develop any time from infancy to adulthood. Shagreen plaques, typically developing during the first decade of life, elevated pink to yellow-brown plaques with an orange peellike texture, several millimeters to several centimeters in diameter are found in roughly half the patients with TSC. They usually appear asymmetrically, on the back and the lumbosacral regions, but at times on the chest or the abdomen. Ungual fibromas, previously called periungual, subungual fibromas, or Koenen tumors, red to skin-colored papulonodules, typically develop near the proximal nail fold of the toenails or, less commonly, the fingernails, are found in roughly 15% of patients with TSC and are frequently seen in early adolescence. A nail groove without tumor, can be the initial features of ungual fibroma. Finally, doughy, flesh -colored, stalked, papules and nodules (molluscum pendulum), which simulate large acrochordons, and prefer flexural areas, namely the neck, axillae and groin can be found in up to 23% of patients with TSC [56,57].
Management of TSC orocutaneous Manifestations: According to the international TSC management guidelines, any symptomatic, rapidly changing, or disfiguring TSC-associated skin lesion, must be treated according to clinical setting and type of lesion, using methods including surgery, lasers or topical mTOR inhibitors. The clinical presentation, intensity, and appearance of other TSC manifestations, determines the best approach to the treatment.
The study population consisted of 49 patients, 26 males, and 23 females, aged 2 to 42 years, clinically confirmed cases of (TSC), according to the international diagnostic criteria for TSC proposed by the TSC consensus conference in 2012, who were examined at the dermatology clinic of Kuwait university hospital, in Sana’a city, Yemen, during 1997-2022. All the patients underwent a detailed orocutaneous examination, with the aid of wood lamp. The absence or presence of the following orocutaneous lesions was noted. History was taken with reference to onset of various cutaneous lesions. Family history was taken in all patients, including details of any affected first-degree relative and consanguinity. Every patient was subjected to a thorough clinical examination for orocutaneous manifestations of TSC, like facial angiofibromas, fibrous cephalic plaques, shagreen plaques, hypomelanotic macules (ash-leaf macules, and confetti like macules), ungual fibromas, molluscum fibrosum pendulum, oral fibromas, and dental pits. Imaging and relevant laboratory investigations were performed whenever possible.
Results
During the period of 25years (November 1997 to April 2022), a total of 180000 patients have attended the outpatient dermatology clinic of Kuwait university hospital in Sana’a city, Yemen, out of them only 49 patients, fulfilled the international diagnostic criteria for TSC proposed by the TSC consensus conference in 2012, producing a prevalence of 2.7 for every 10000 individuals. As given in (Table 1), our study sample consisted of 26 (53%) males and 23 (47%) females, giving a male to female ratio of (1.1:1). Their ages ranged from 2 to 42 years, with a mean age of (17.1 ± 4.75) years. Most of our patients (51 %) were aged 10- 20 years, followed by age group of 21-30 years old, with (22.4%), and the age group <10 years old, with nine patients (18%), and the least common was the age group of >30 years old, with (6%). Most of our patients were sporadic TSC cases (N= 43, 87.7%) while for the remaining (N= 6, 12%) a positive familial history of TSC was reported; two of them had a history of sibling afflictions and four had a history of parental afflictions. Our results revealed that all patients (N= 49, 100%) had one or more TSC cutaneous lesions. The identified TSC orocutaneous manifestations among patients of this study were facial angiofibromas, fibrous cephalic plaques, shagreen plaques, hypomelanotic macules (ash-leaf macules), ungual fibromas, and molluscum pendulum. The frequency of the orocutaneous manifestations of TSC in the studied population is illustrated in (Table 2).
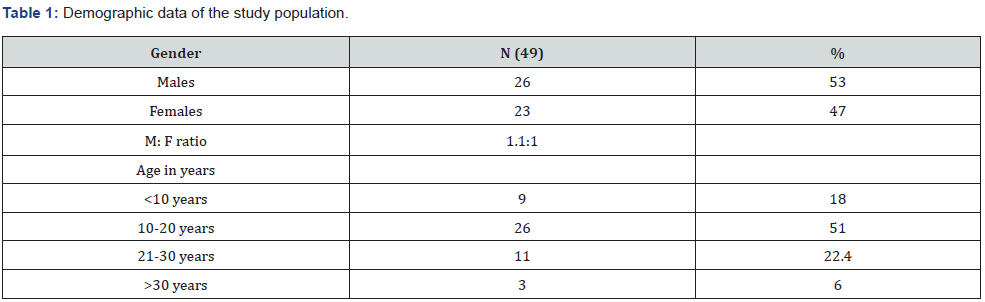
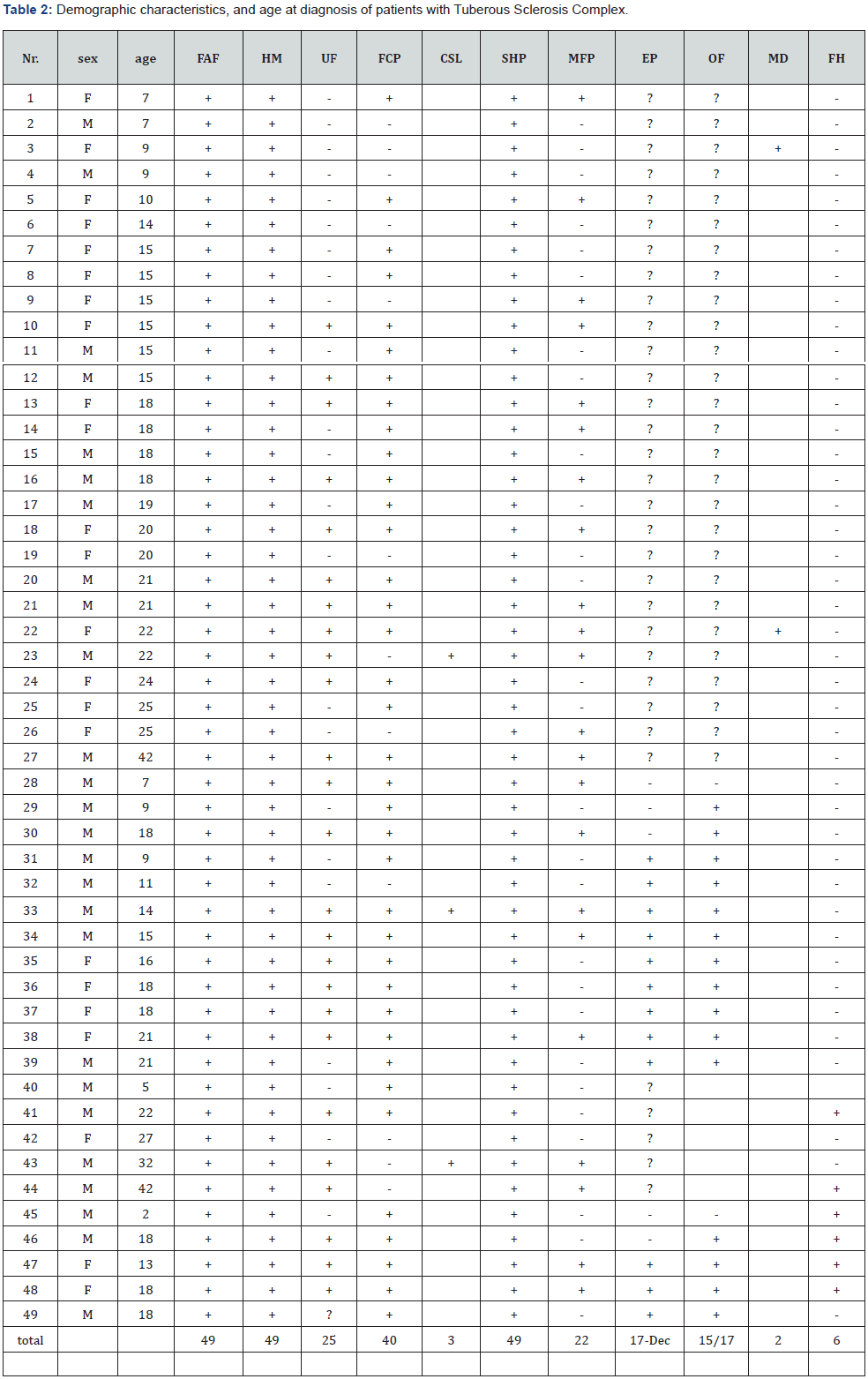
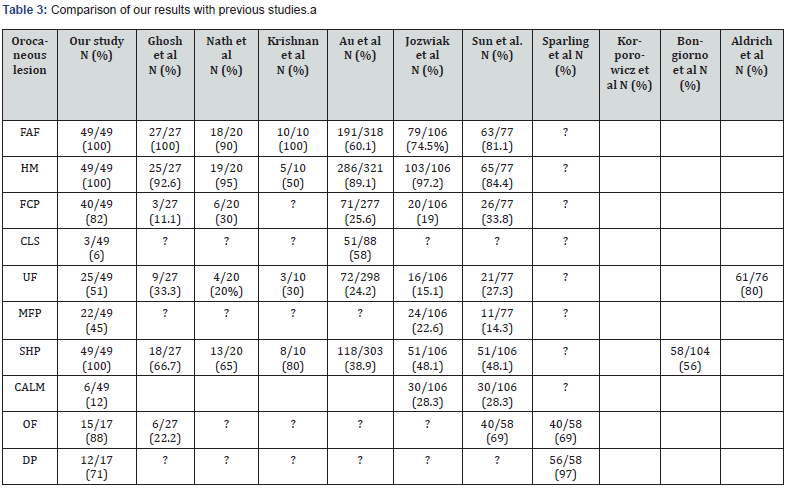
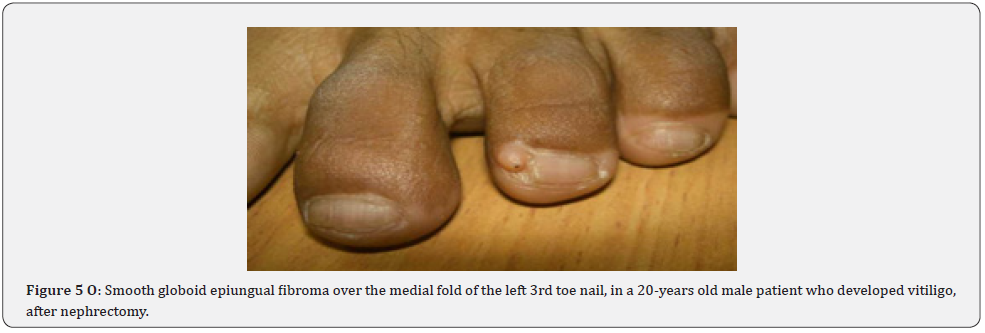
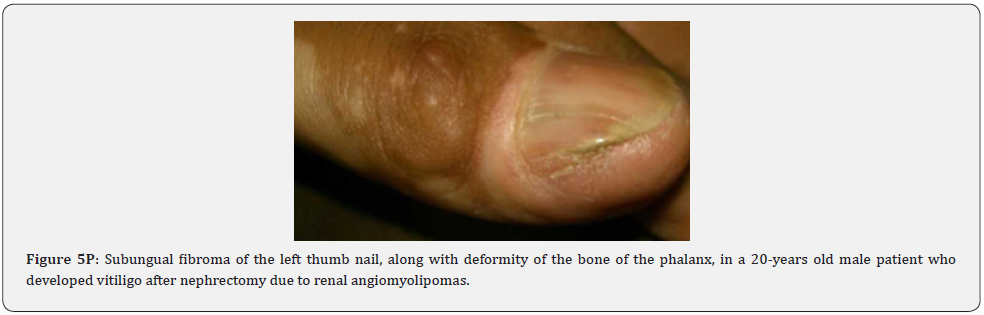
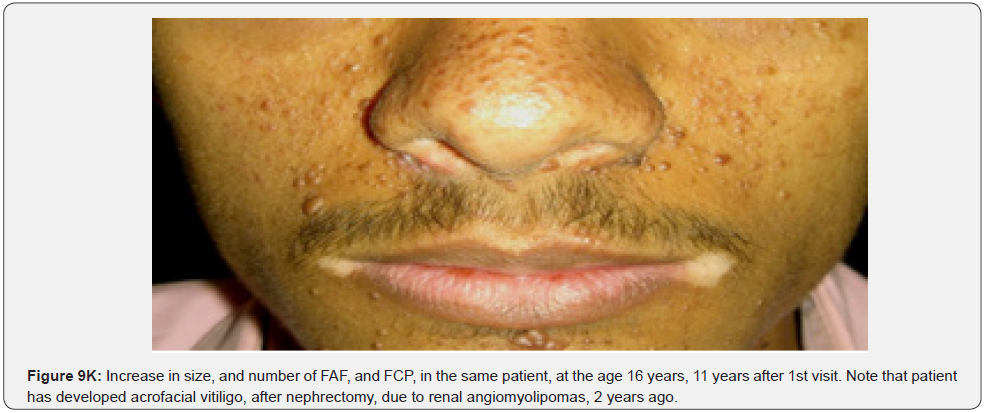
The study findings demonstrate that the most common TSCrelated orocutaneous lesions were facial angiofibromas, shagreen plaques, and hypomelanotic macules which were seen in all cases (N=49, 100%), followed by fibrous cephalic plaques in 40 patients (82%), ungual fibromas in 25 cases (51%), and lastly molluscum pendulum found in only 22 patients (45%) as shown in (Table 3, Table 4). Confetti skin lesions, and café au lait macules were found in 3 patients (6%) each. Unusual orocutaneous finding, which have been either not, or rarely reported in association with TSC, seen in our study included, pachydermodactyly, macrodactyly, congenital localized sacral hypertrichosis, onychomatricoma, Becker nevus, cutaneous syndactyly of toes, port wine stain, digital fibrokeratoma, agminated zosteriform lentiginosis, and nail pits. One young child was operated for cardiac rhabdomyoma. He, and one other patient underwent nephrectomy, at the age of 16years, due to ruptured renal angiomyolipomas. He also developed vitiligo after nephrectomy.
Discussion
Tuberous sclerosis complex (TSC) is a neurocutaneous genodermatosis affecting several organ systems with exceptionally varying clinical manifestations. It has a calculated incidence of 1 in 6000. Mutations in one of the genes TSC-1, or TSC-2, located on chromosomes 9 and 16, respectively, leads to hamartomatous lesions in various organs including the skin, since they act as tumor suppressors. Although, focal or generalized seizures are often the first sign of the disease, vigilant orocutaneous examination can identify hypopigmented macules even earlier, delivering an early diagnostic hint, hence reducing morbidity and mortality. It is a complex condition, due to the diverse schedule of the emergence of its manifestations and the symptoms and signs they create. The central nervous system (CNS), respiratory, dermatological, renal, and circulatory organ systems are most regularly afflicted, while few of these manifestations are typical for TSC. TSC is a complex clinical condition consisting of many clinical manifestations, which require attention from different medical specialties. The dermatologist plays a crucial role in its history, since orocutaneous involvement represents the most frequent finding, permitting early diagnosis of the syndrome and intervention in its natural course.
Epidemiological studies generally underestimate the prevalence of the disease, because of the variation in gene expressivity between affected individuals, both intra- as well as interfamilial. Its recognition is sometime impeded, due to its great phenotypic variability. The incidence of TSC is estimated to be between 1/6000 and 1/10,000 live births and the population prevalence is estimated to be 1/20,000. Approximately 1 in 12,000-14,000 children under 10 years of age have TSC according to population- based studies in the UK. Worldwide, TSC is thought to affect 1 to 2 million individuals. The estimated prevalence of TSC is around one case for every 10000 to 25000 in Caucasian population. A study in Taiwan revealed that the prevalence of TSC was estimated to be 1:95,136 and the prevalence for cases less than six years of age was 1:14,608. In China, a study which consisted of 44 cases over a 12-month period (January-December 2003) determined the period prevalence rate of tuberous sclerosis complex for children and adolescents younger than 20 years to be 3.5 per 10,000. In this study, 10 out of 19200 patients were clinically diagnosed with tuberous sclerosis complex (TSC) throughout the period of 4 years (November 2013 to April 2017). Lack of awareness among the primary health care providers, social and health education status of the affected families and the strong influence of traditional healers in the region makes it very difficult to estimate the prevalence of TSC in many developing countries including Yemen.
TSC can present at any age and is a diverse disease with a highly irregular clinical presentation, which also differs with the age of the patients. The frequency of each manifestation differs relying on the age of the study population [58].
Most of our patients presented in their second decades of life. The gender ratio in the present study was 1.1:1, which is consistent with published literature that shows sex ratio to be almost equal with symptoms being more predominant in females.
Similar reports expressed that both sexes are affected in a similar prevalence, but women may show more eminent signs. There are no reports showing unequal involvement in a certain ethnic group [59]. Other studies, on the other hand, have suggested that more severe neurological morbidity is seen among male individuals.
The majority of our patients (88%) were sporadic TSC cases and only (12%) had a positive familial history. This is consistent with several studies reports showing that the majority of TSC cases occur sporadically, with a family history found in only 30% of patients [60]. A study in Taiwan showed that only 15% of patients had a family history of TSC which is higher than our data. The familial forms of TSC arise from germline mutations and, although their transmission can be hereditary, 70% of the cases are a result of somatic mutations, especially when the mutant gene is TSC2. Somatic mutations are responsible for sporadic cases of the disease. The great variety of mutations and the possibility of involvement of different genes explain, in part, the phenotypic diversity of the disease. Another factor that may explain why clinical manifestations of TSC are unpredictable is the presence of mosaicism.
Studies show that changes in TSC2 are five times more frequent than changes in TSC1 in sporadic cases. This relationship becomes 1:1 in cases of familial transmission. Changes in the TSC2 gene result in more severe pathology, when compared to TSC resulting from mutations in TSC1. Cases of familial transmission, which show higher frequency of changes in the TSC1 gene, result in mild to moderate disease, sometimes not meeting the various diagnostic criteria.
The age reliant appearance of distinctive features of TSC has historically offered challenges for diagnosis in early childhood and necessitates a broad understanding of the wide range of its manifestations. Despite the accessibility of genetic testing for case confirmation in the developed countries, in a developing country like Yemen, detailed cutaneous, neurological, and ocular assessment will remain essential for its diagnosis. With regard to our study patients with 100% orocutaneous involvement, a careful orocutaneous examination of patients at risk proceeds to be the simplest and most available method of diagnosis. All our patients fulfilled the criteria for “definite” TSC, based on the 2012 International TSC Consensus Group guidelines.
The recognition of diagnostic hallmarks should prompt further consideration of an underlying diagnosis of TSC. The central nervous system (CNS), renal, dermatological, respiratory, and circulatory organ systems are most frequently afflicted, even though few of these manifestations are characteristic for TSC. Orocutaneous findings are the most frequent and easily visible manifestation of TSC. More than 90% of patients with TSC have one or more skin lesions, which usually appear early in life.
Aside from positive genetic testing confirming a pathological TSC1 or TSC2 mutation, the clinical diagnosis of TSC depends on a combination of recognizable major and minor characteristics, with orocutaneous findings comprising the greatest of both major (hypomelanotic macules, angiofibromas, ungual fibromas, shagreen plaque) and minor (‘confetti’ skin lesions) features. Tuberous sclerosis complex has a broad variation of orocutaneous manifestations consisting of angiofibromas (misnamed as adenoma sebaceum), connective tissue nevi (shagreen plaques), hypomelanotic macules (ash-leaf macules, and confetti like hypopigmentation), ungual fibromas, and fibrous cephalic plaque (which was named forehead plaques, but can present anywhere on the face, head, and neck), dental pits, and oral mucosal fibromas.
Orocutaneous lesions are the most frequently observed manifestations of TSC [61]. Our results revealed that all patients (100%) had one or more TSC cutaneous lesions. Facial angiofibromas, shagreen plaques, and hypomelanotic macules were present in all our cases (100%). Fibrous cephalic plaques in 82%, ungual fibromas in 51%, molluscum pendulum in 45%, and confetti like hypopigmentation in 6%. Regarding the oral manifestations oral fibromas were found in 88%, and enamel pits in 71% of the cases.
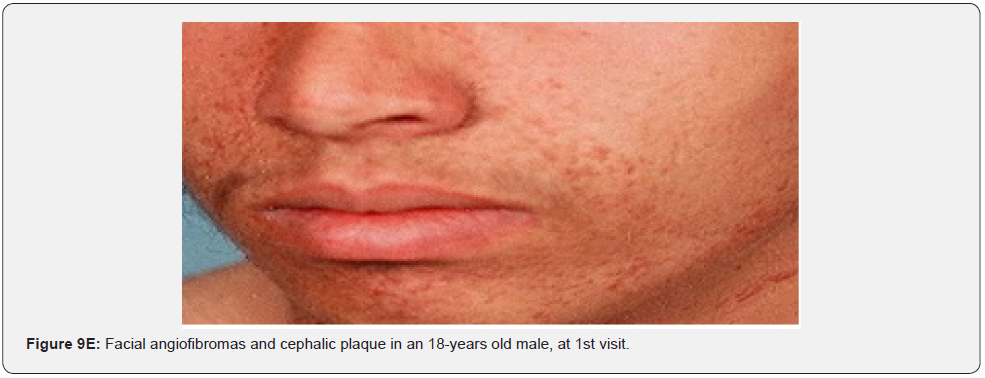
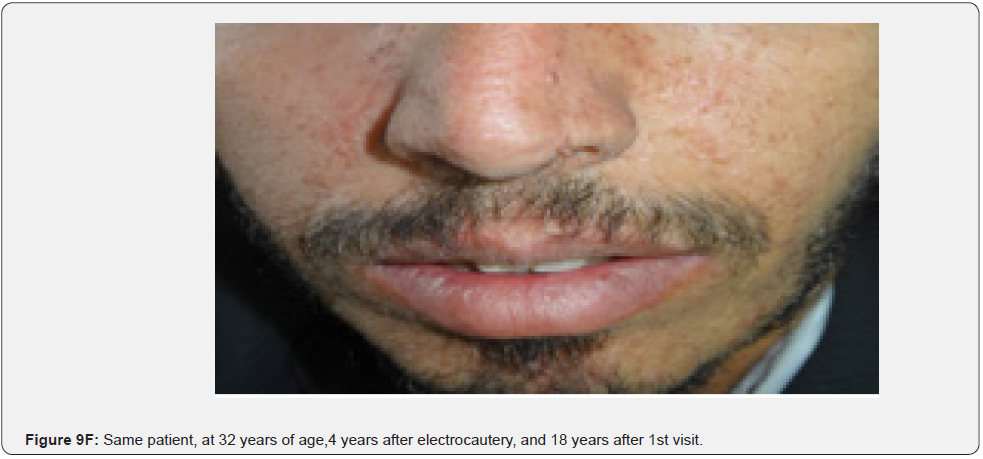
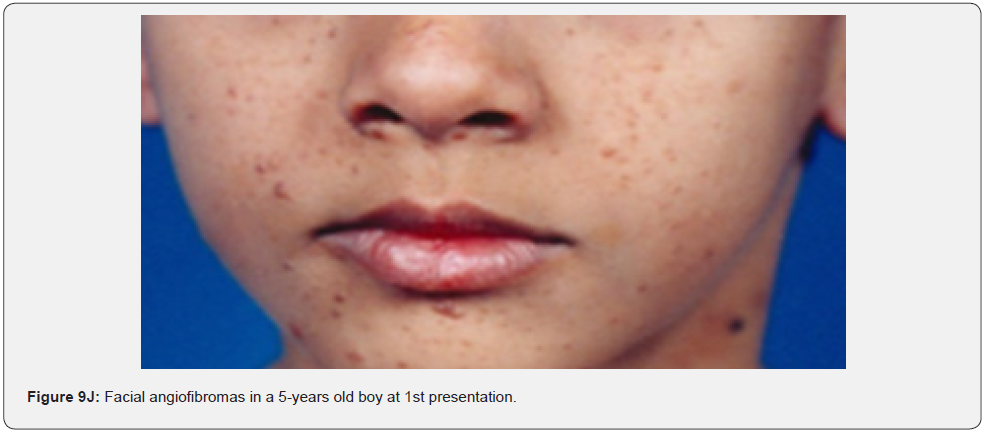
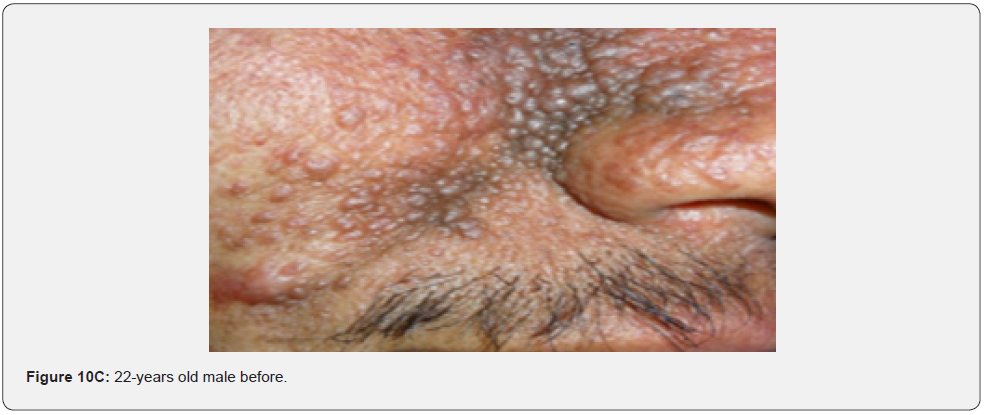
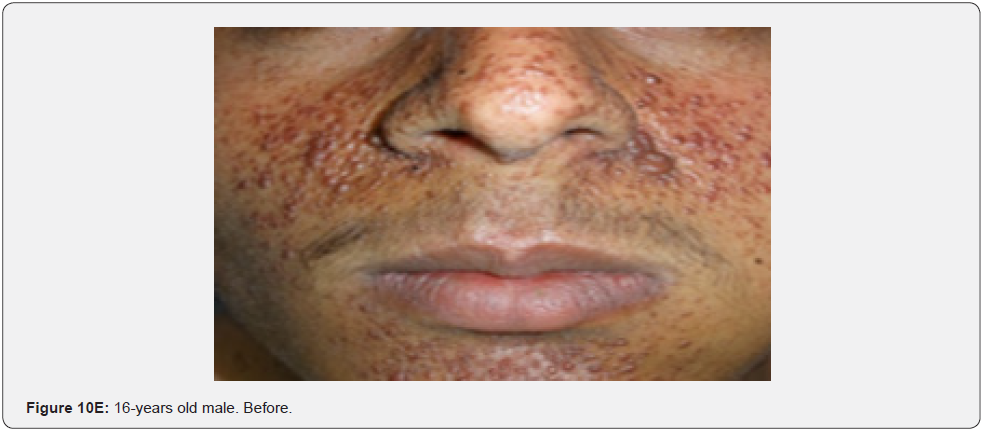
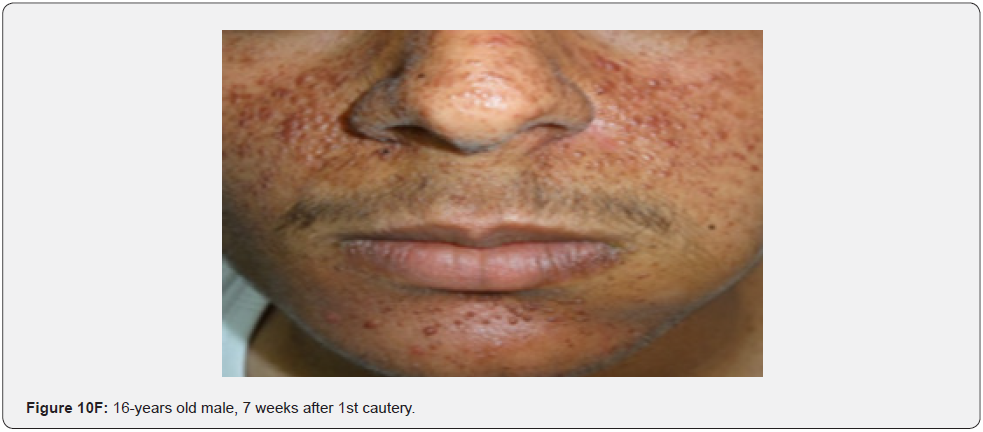
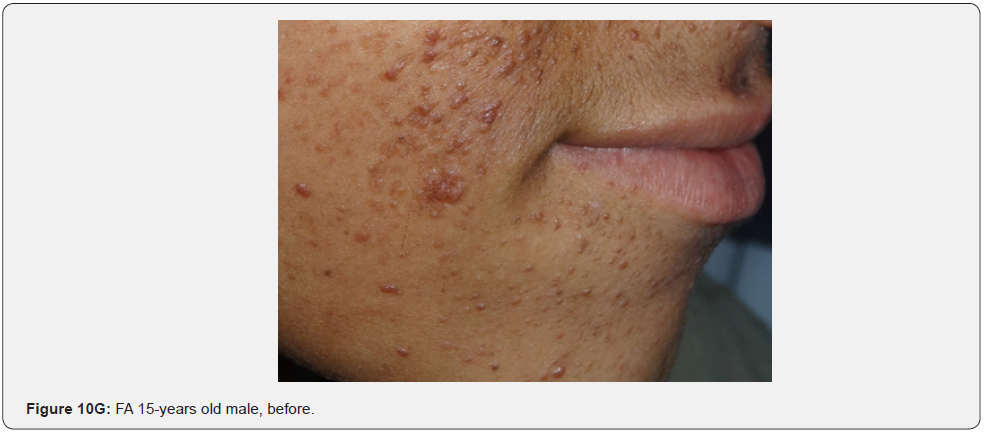
Angiofibromas (AF) are the most symbolic symptom of TSC because of their prominent location on the face. They manifest as multiple, separate, transparent, reddish flexible papules, distributed uniformly on the nasolabial folds and over the cheeks and forehead (Figure 1A-G). They emerge generally between ages 2 and 5 years. Despite that most TSC patients have numerous facial angiofibromas milder cases of TSC with confined facial angiofibromas have been described. By the age of four years, ninety to ninety six percent of the patients may have facial angiofibromas. The existence of not less than three facial angiofibromas is now proposed to meet the major criteria for TSC. This feature was seen in all (100%) the patients in our study, which is similar to the results of an Indian study, but higher than the results of another Indian study stating a much lower result (60.1%) [62].
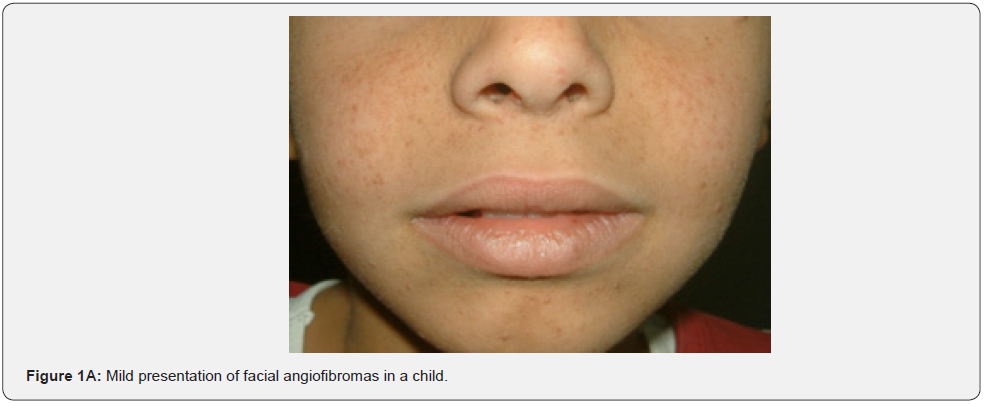
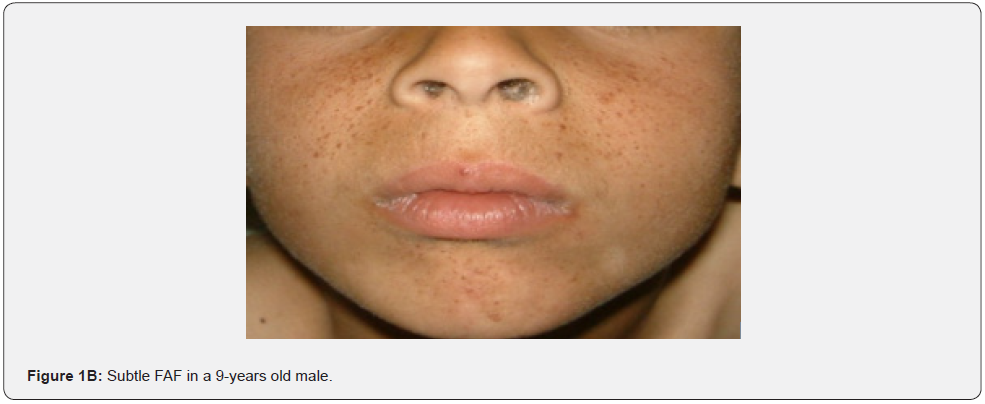
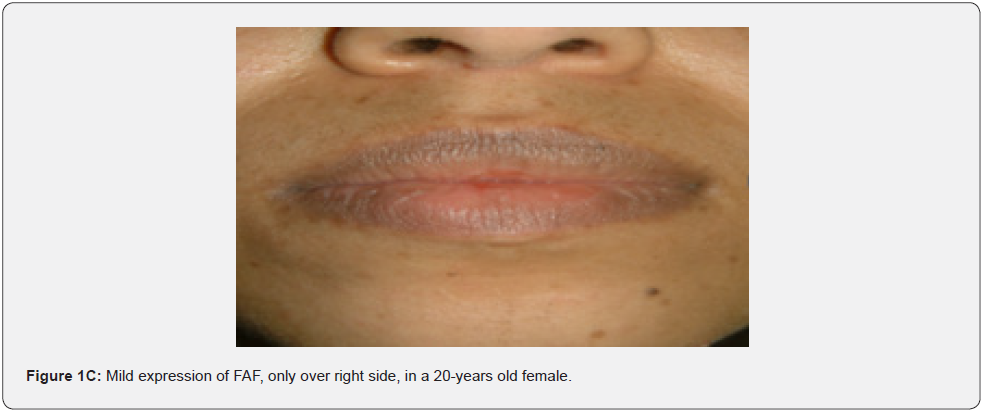
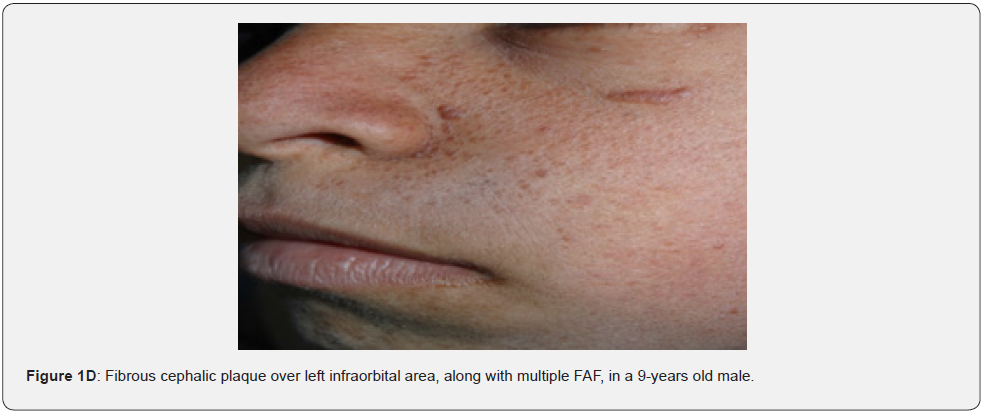
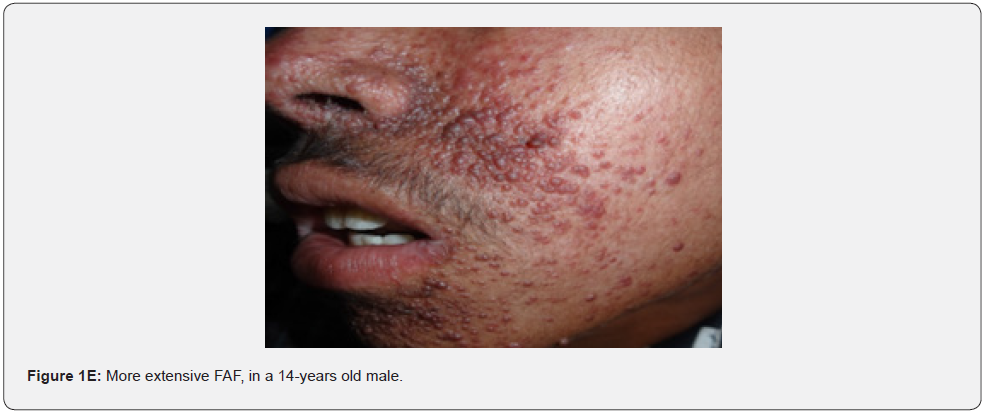
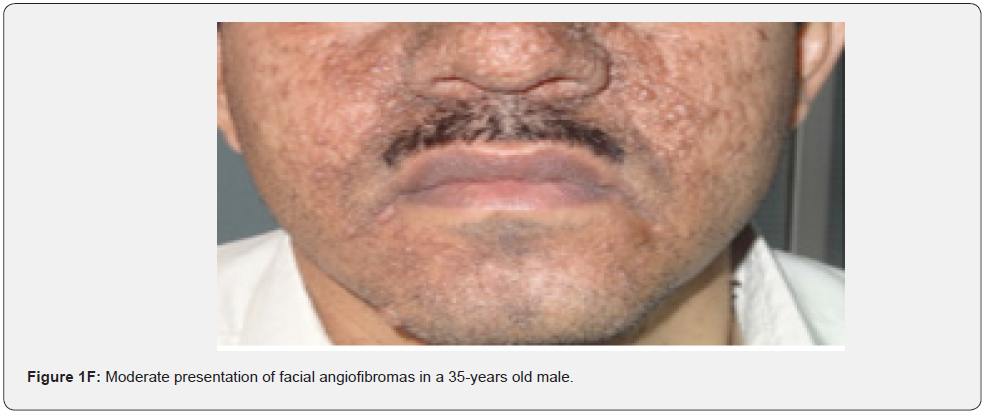
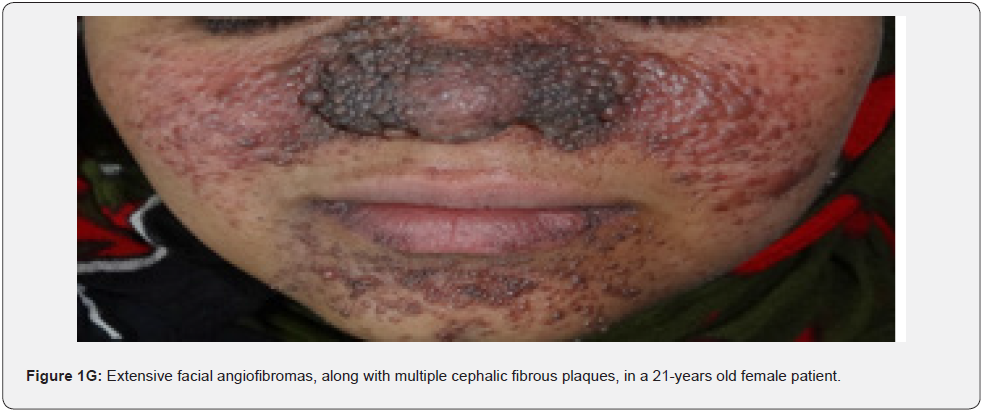
Hypomelanotic macules are an important feature because they are detected in about 90% of patients with TSC, they usually appear at birth or infancy, and they may be a presenting sign of TSC. Hypomelanotic macules may take the form of elongated ashleaf off-white lesions or multiple small confetti-like lesions (Figure 2A-2H) [63]. They were present in 100% of our patients including three patients with confetti like hypopigmented macules, which is consistent with results of previous studies, stating different results.
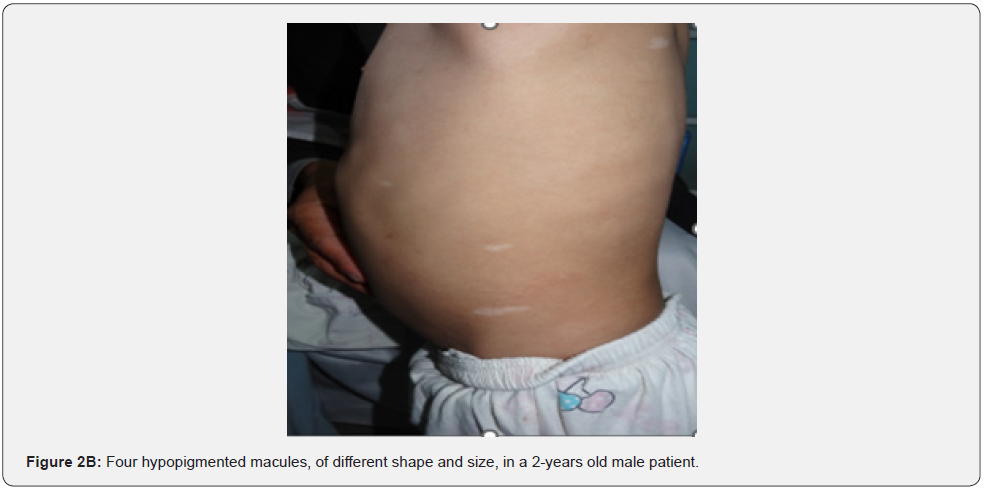
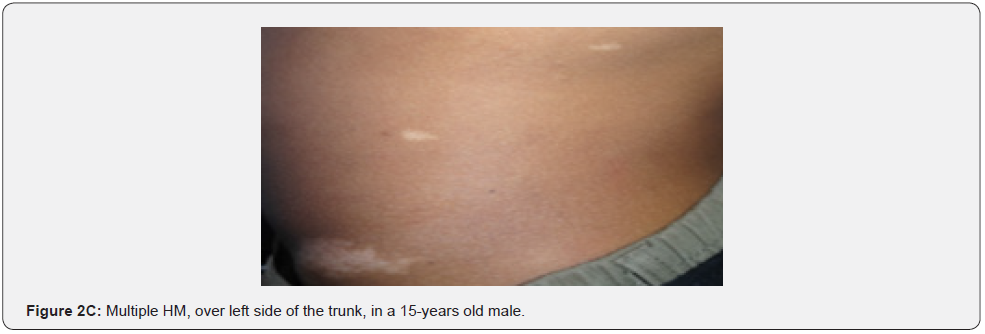
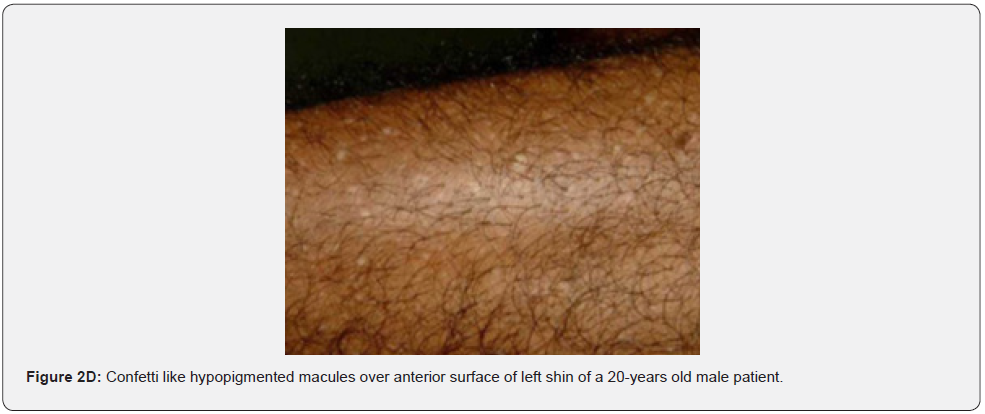
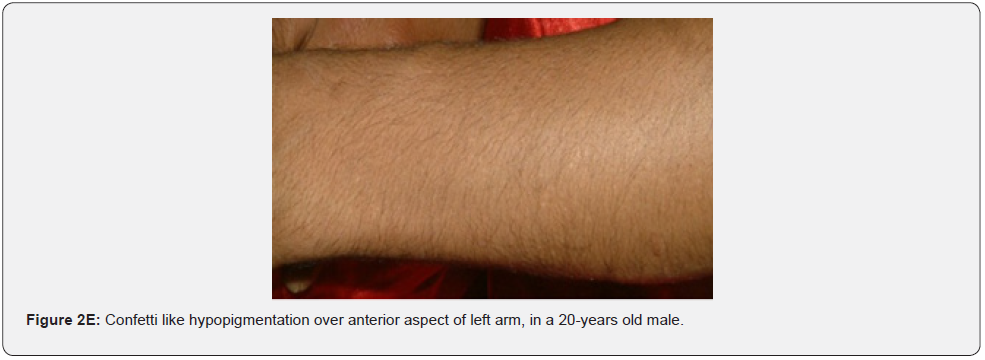
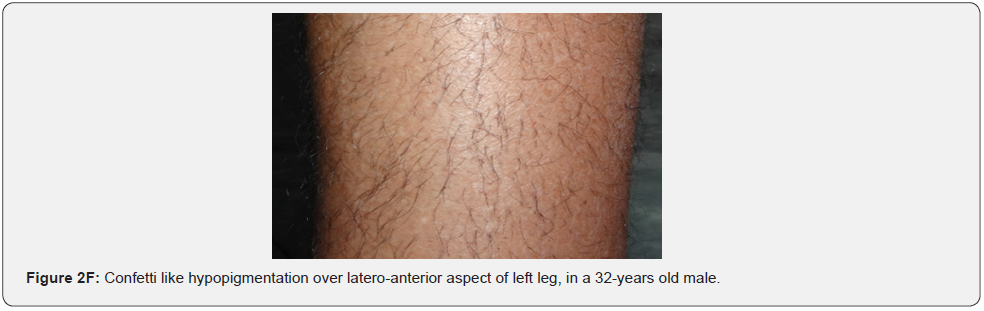
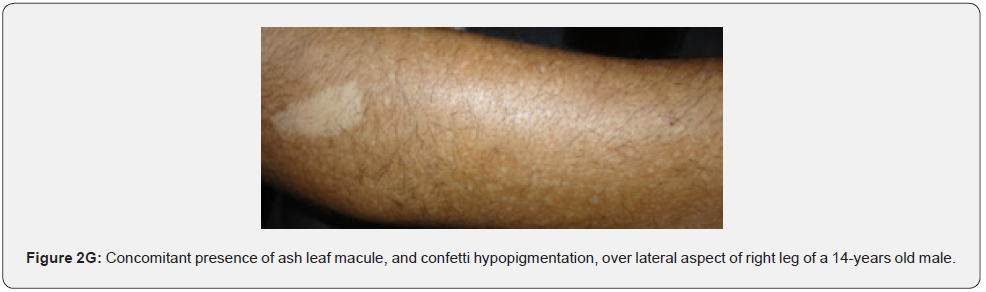
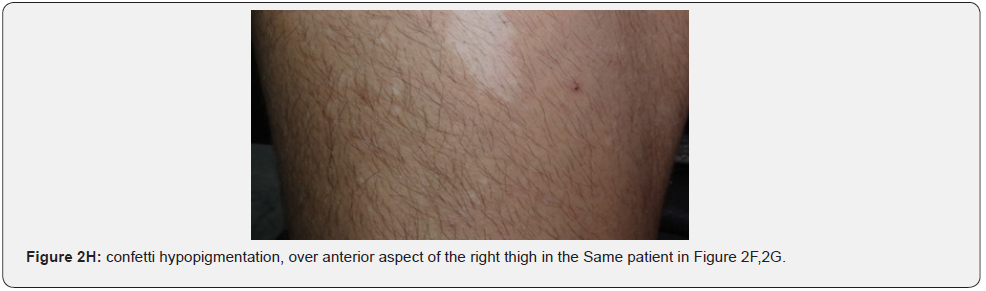
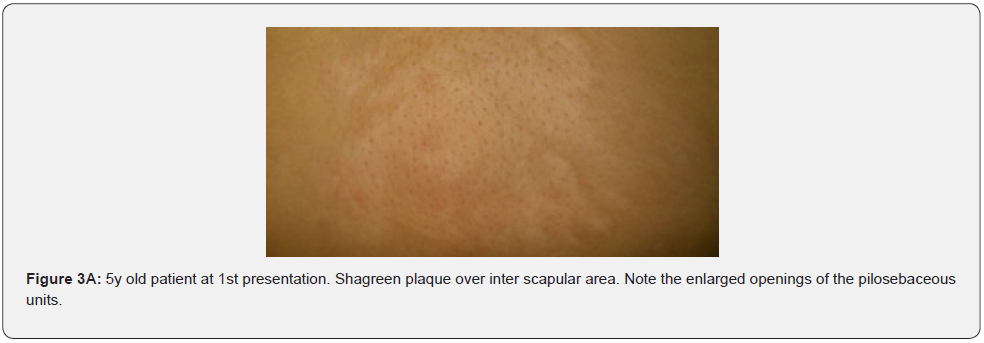
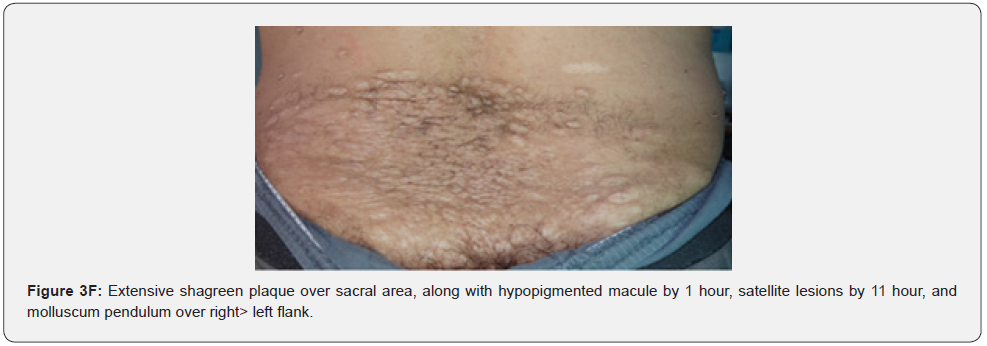
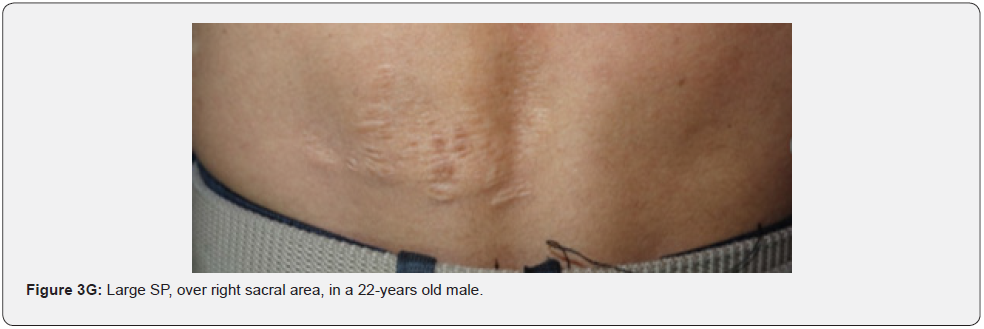
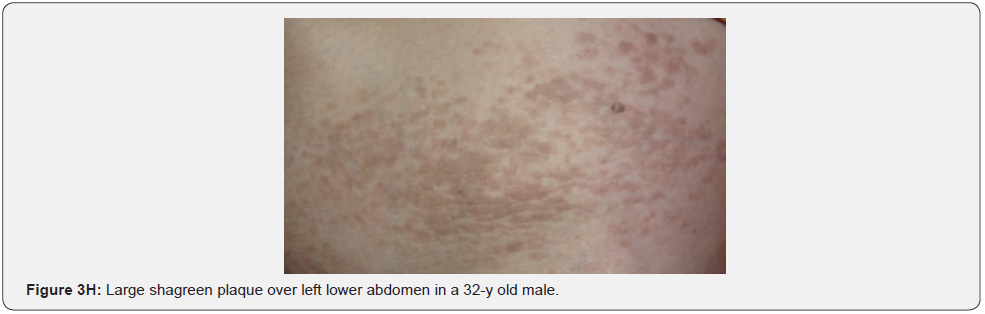
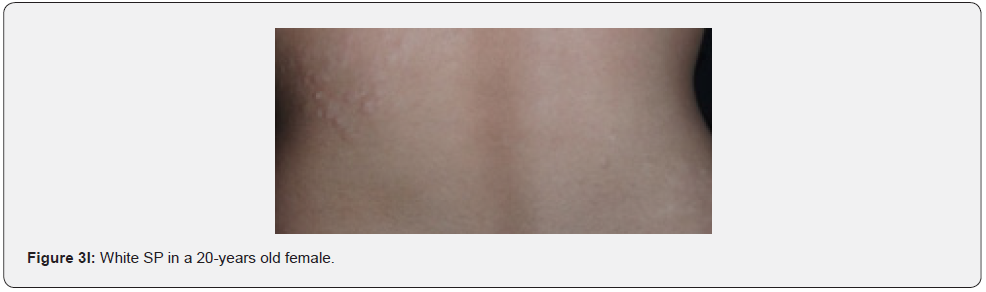
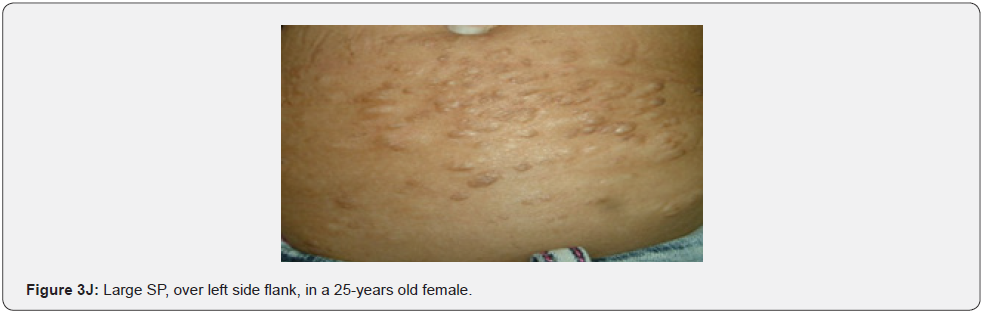
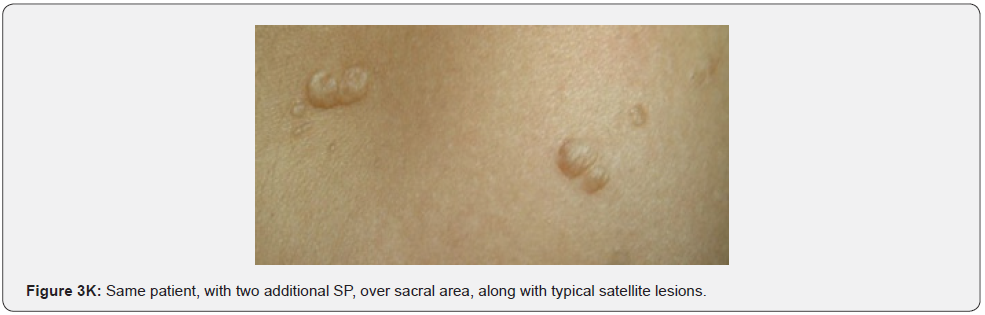
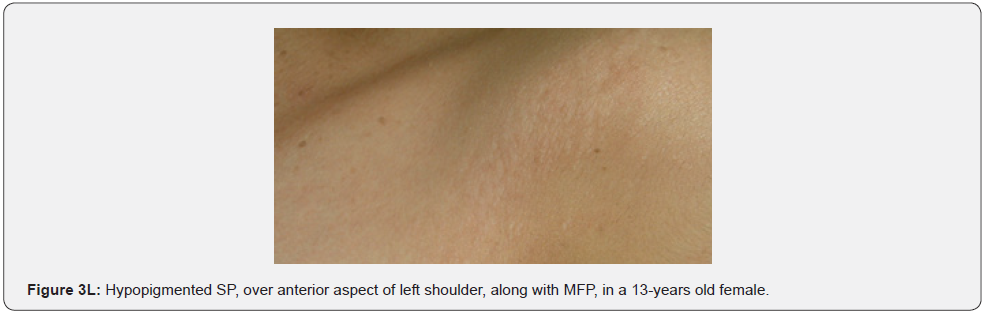
Confetti skin lesions are countless 1-3 mm hypopigmented macules distributed over the arms and legs (Figure 2D-2H). They have variable frequency from 3% in children to about 58% overall [62]. Regardless of their relatively low prevalence, they may still be beneficial, and they were retained as a minor diagnostic feature. Their value in adults is limited because many adults in the general population have similar-appearing lesions (idiopathic guttate hypomelanosis) as a result of chronic sun exposure. The diagnosis of confetti lesions in such cases, may be assisted by their asymmetric distribution, or by a history of initiation in the first decade of life. Shagreen plaques (erroneously called shagreen patch) usually present as large leathery plaques, found mostly on lower back (Figure 3A-3L). They have a distinctive pigskinlike appearance, due to prominent, slightly depressed, follicular openings, and have their onset typically in the first decade of life. They were found in all (100%) our patients, which is much higher than stated to occur in many previous studies (39%-80%) [64].
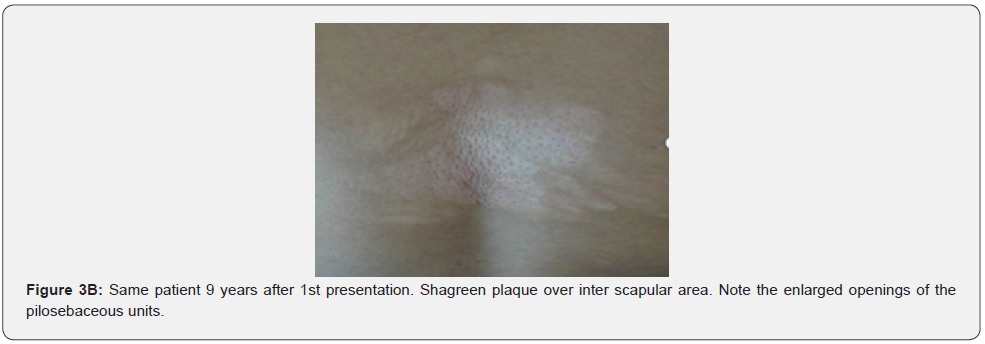
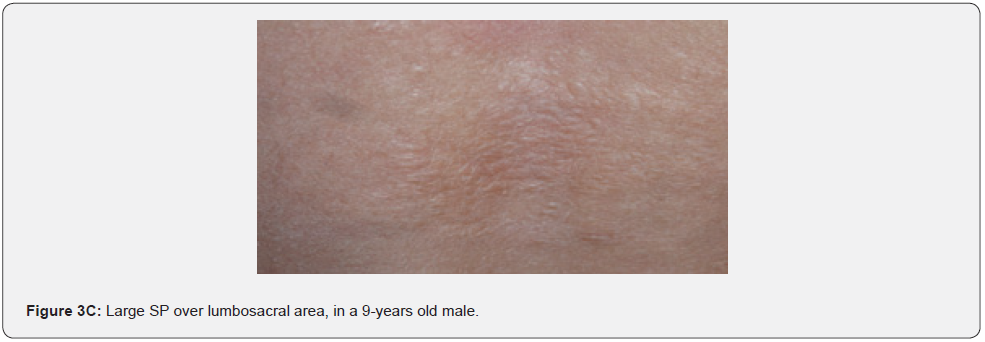
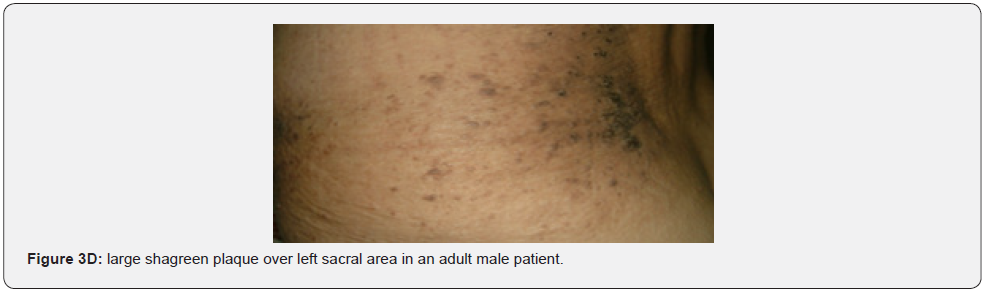
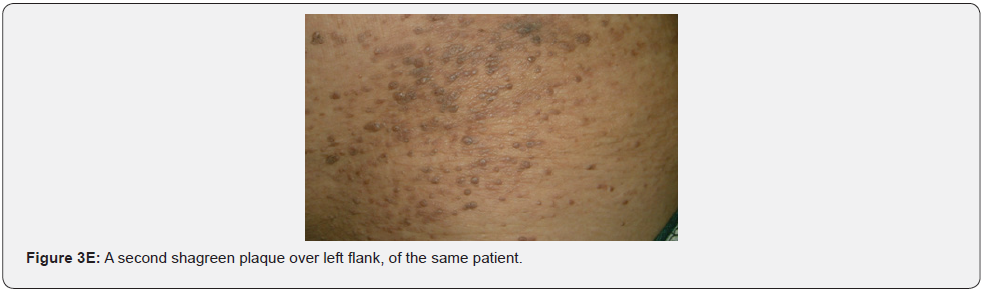
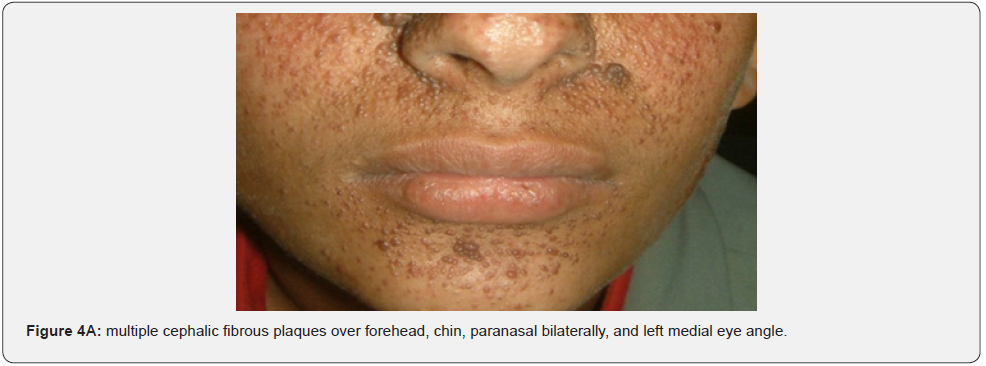
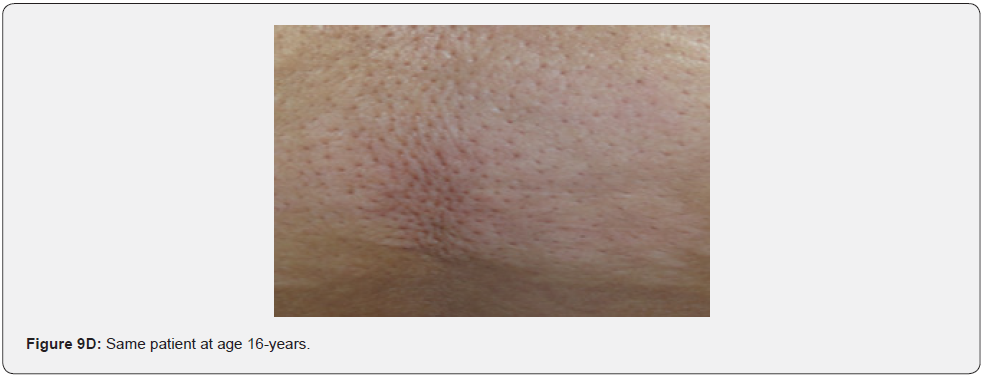
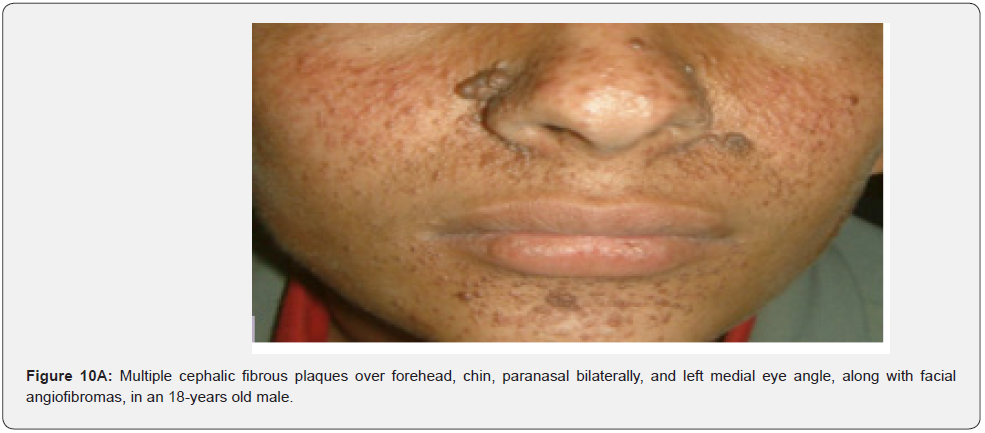
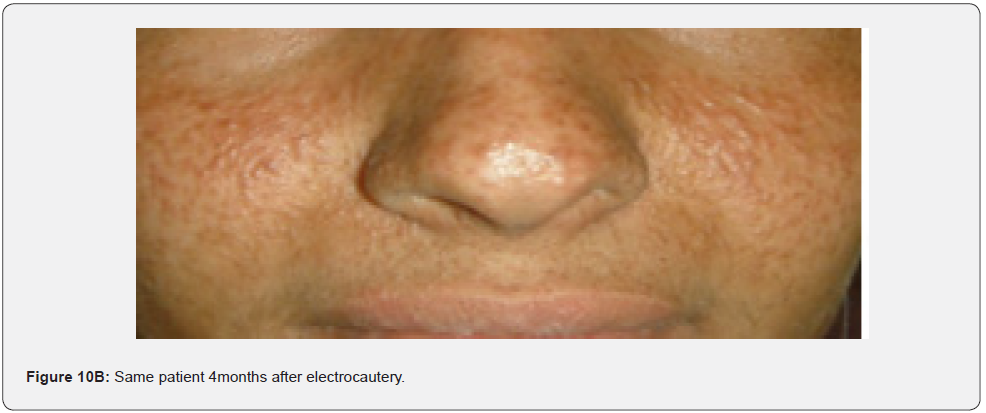
Fibrous cephalic plaques, which have similar histology to angiofibromas, may be the most distinct skin finding for TSC. They are usually firm and yellow brown to pink-tan in color, and grow very slowly over time, up to several centimeters in diameter (Figure 4A-4E). The board of the TSC consensus conference in 2012 advocated changing the nomenclature from forehead plaque to fibrous cephalic plaque. This term was generated to increase attention that these fibrous plaques, whilst often located unilaterally on the forehead, may present on other segments of the face or head. Fibrous Cephalic plaques were found in 82% of our patients compared to a lower percentage (3.8%-3o%) observed in previous studies conducted in different countries. Fibrous cephalic plaques may signal CNS involvement at an early stage of tuberous sclerosis [65].
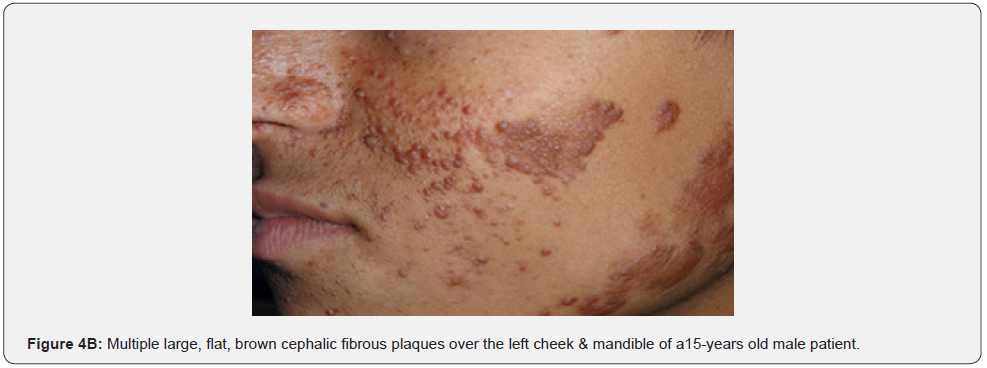
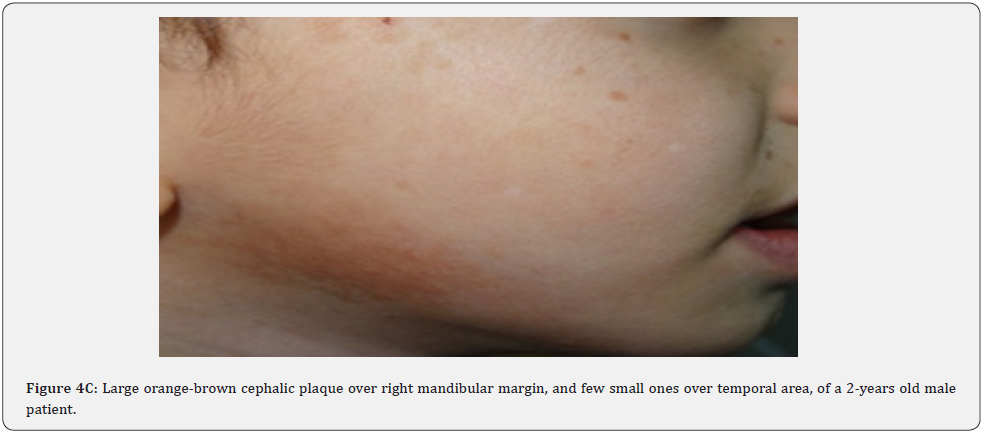
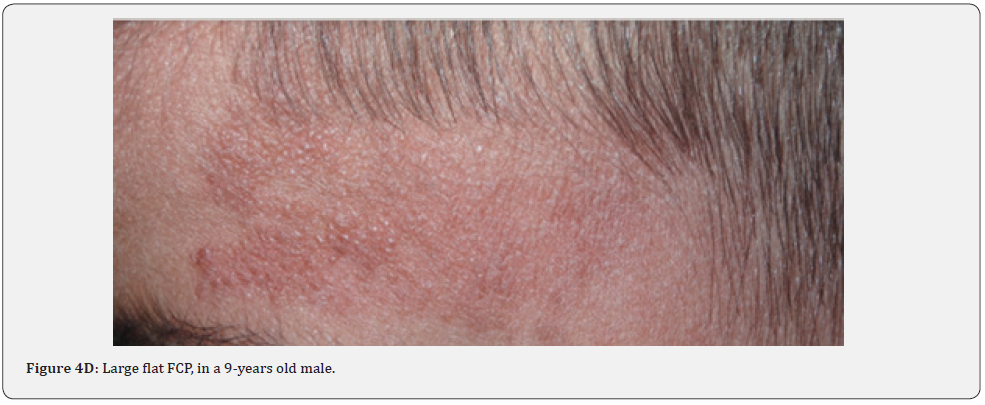
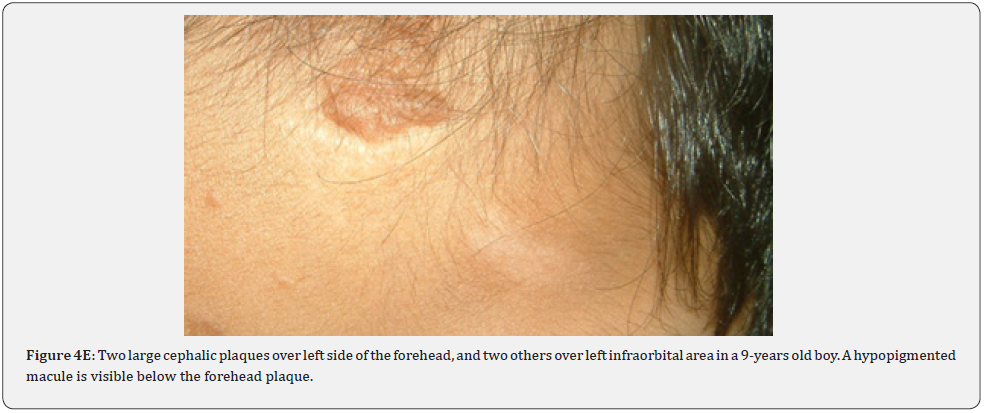
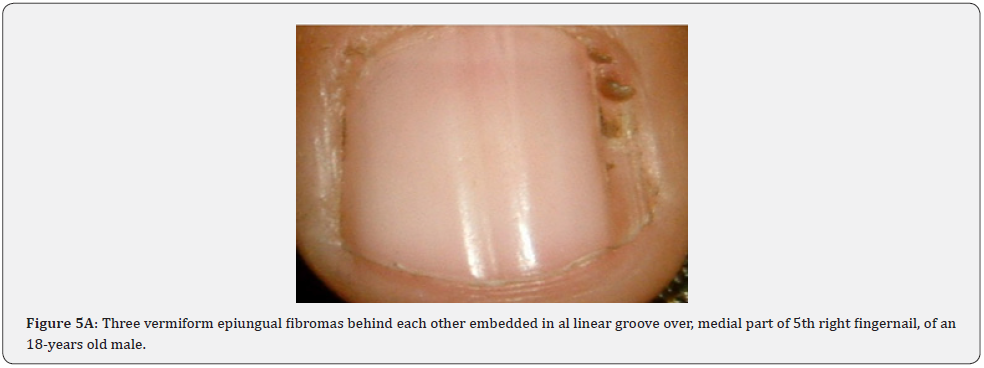
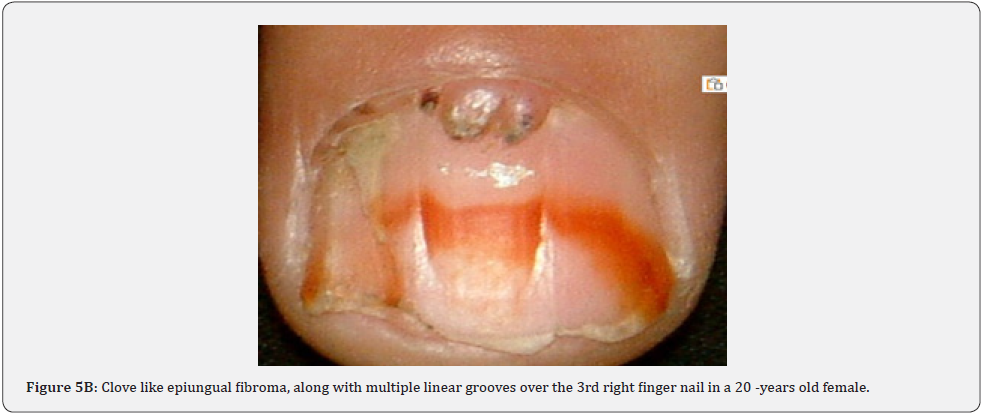
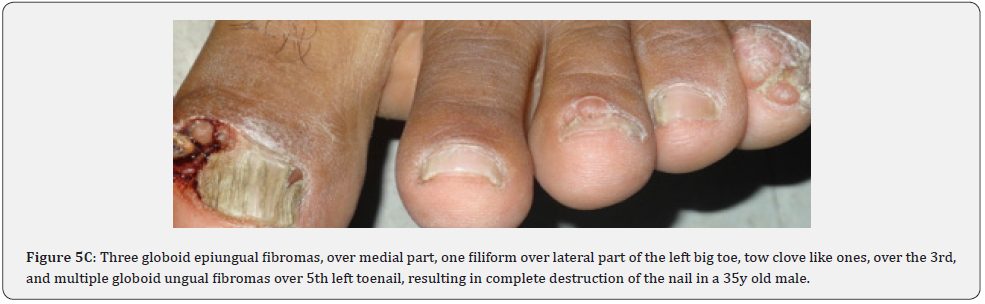
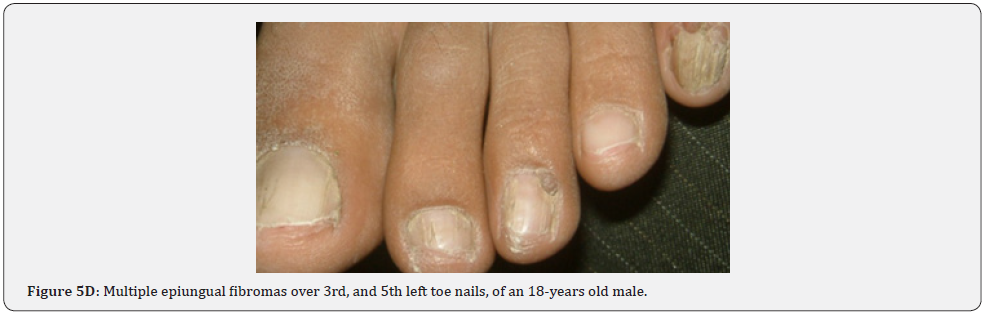
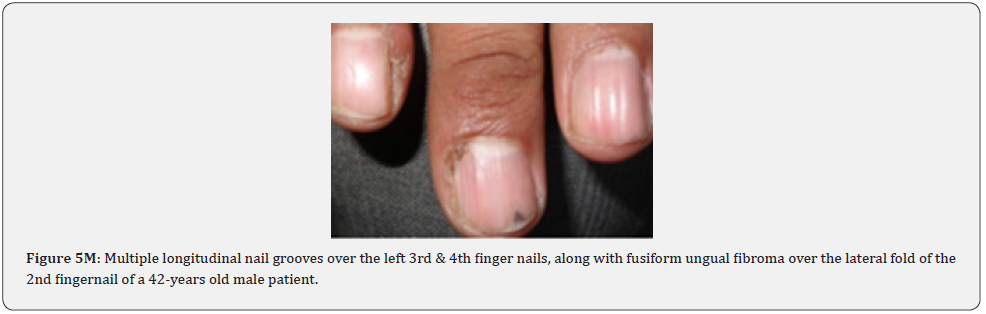
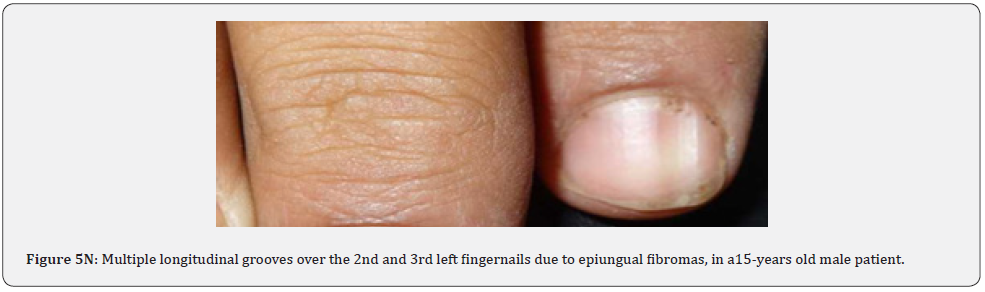
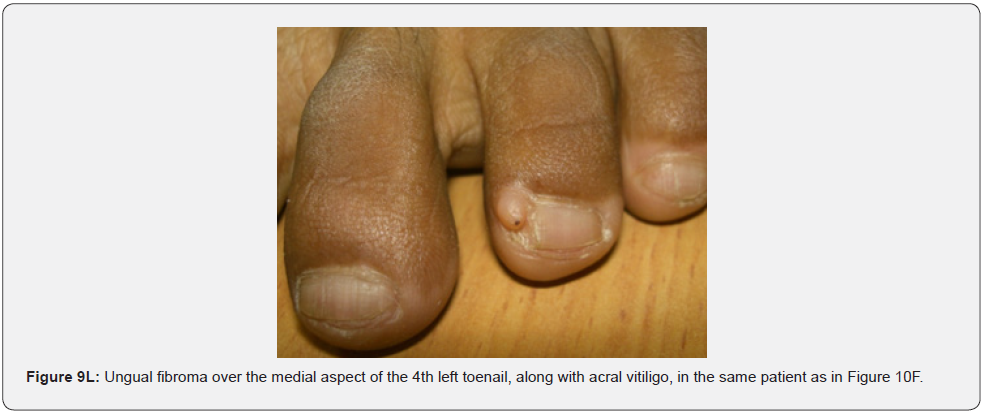
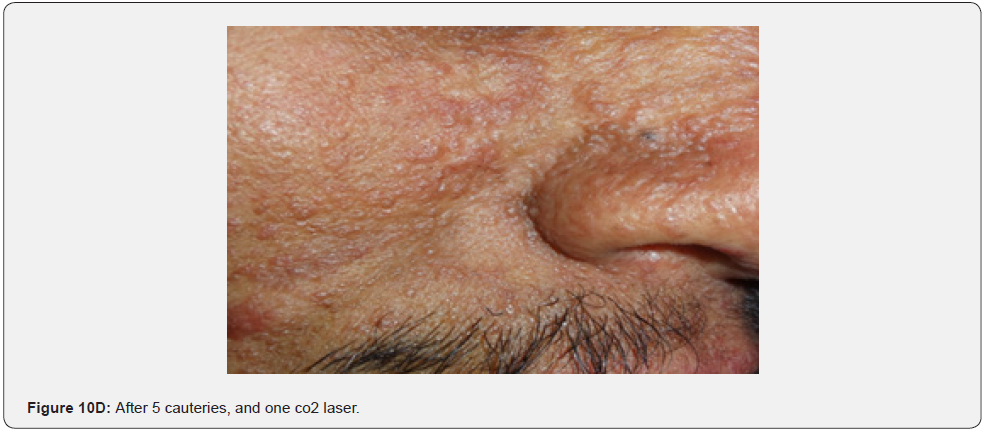
Ungual fibromas (known also as Koenen’s tumors), are small digitate protruding asymptomatic periungual papules or nodules, which begin to develop later in childhood and can continue to form throughout adulthood. They were found in (51%) of our patients, compared to (15.1%-33.3%) in previous studies. These fibromas are seen commonly around toenails than the fingernails, and some lesions lead to distortion of the nail plate (longitudinal groove) without a visible tumor (Figure 5A-5P). Aldrich et al found a higher frequency (80%), than found in our study [66]. He also provided a detailed description of their clinical presentations, and a proposal for their classification into epi, sub, and periungual fibromas [66].
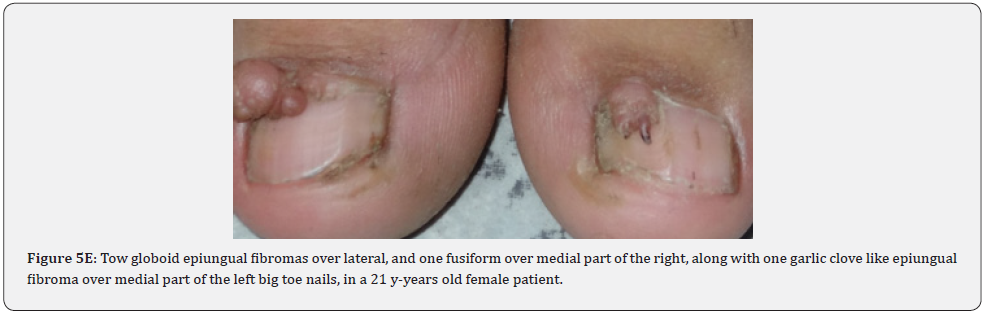
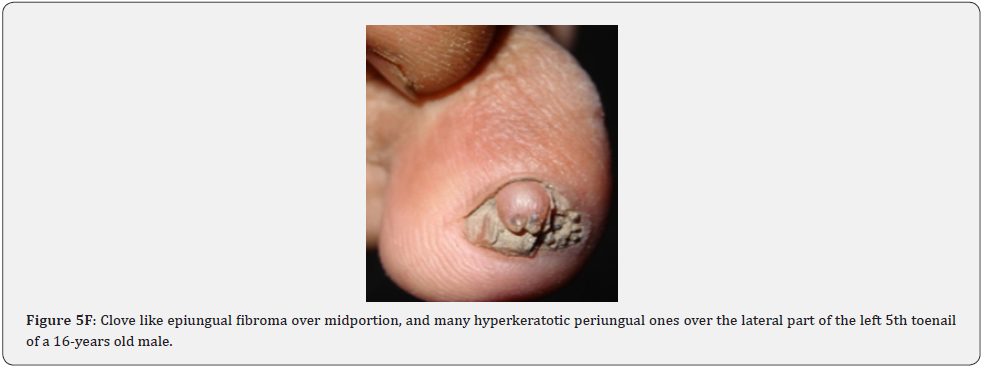
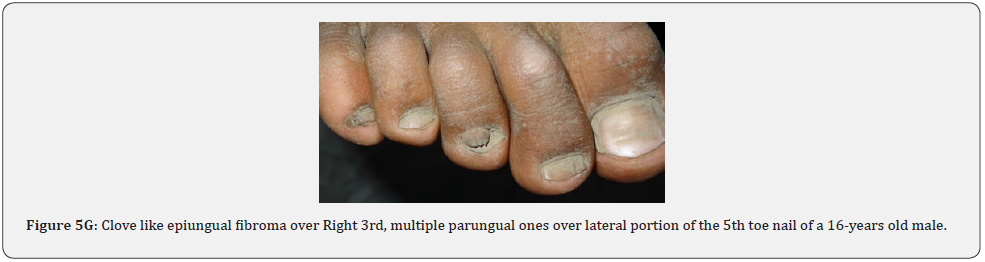
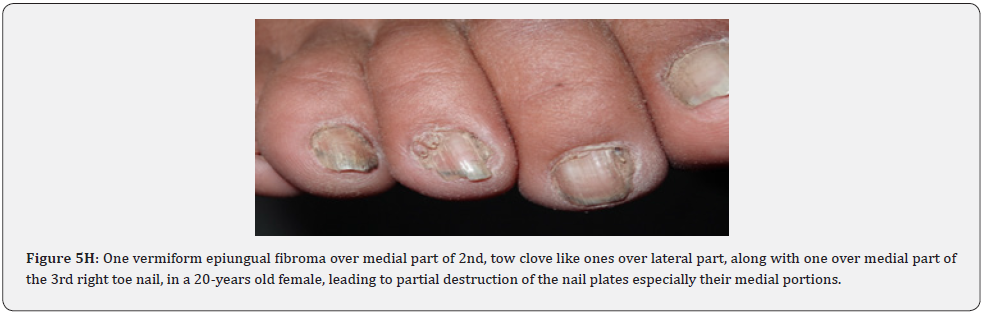
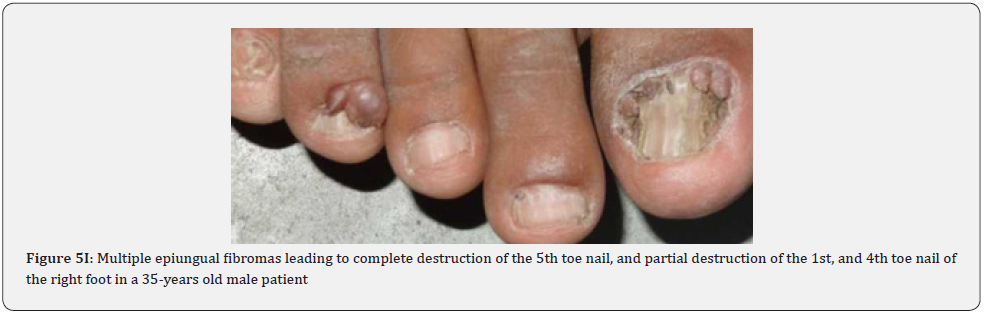
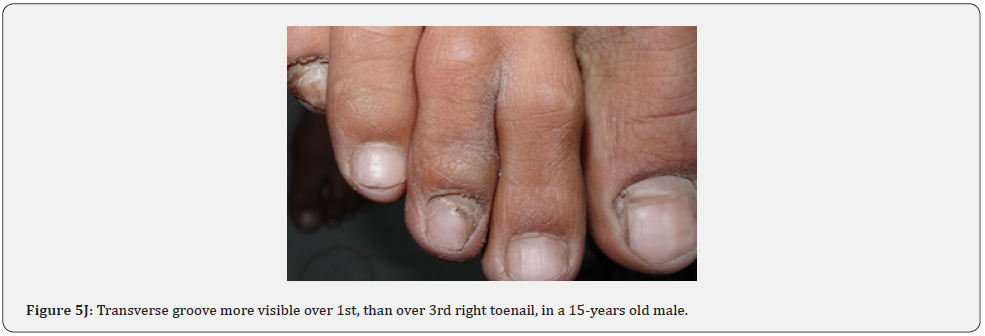
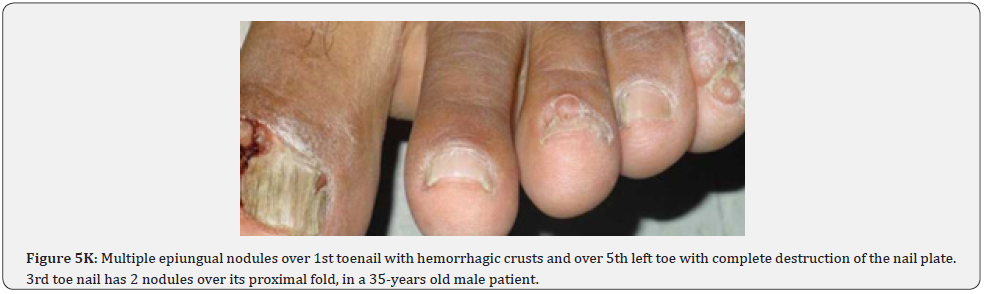
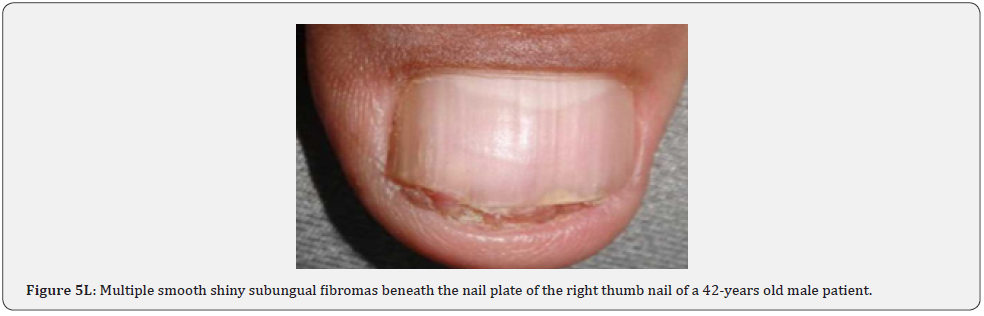
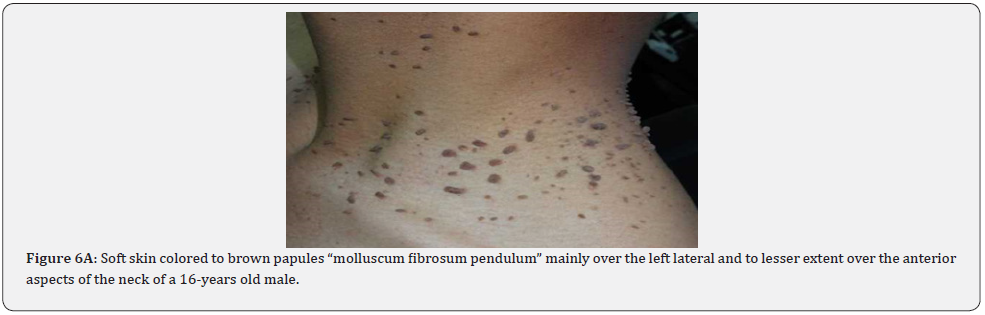
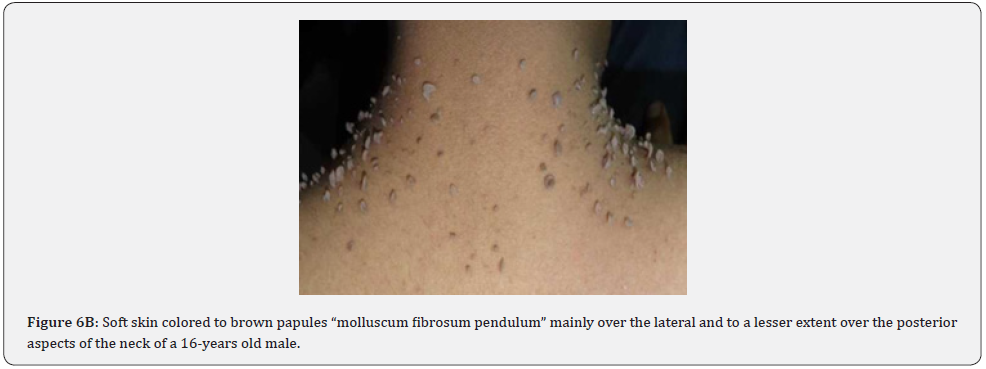
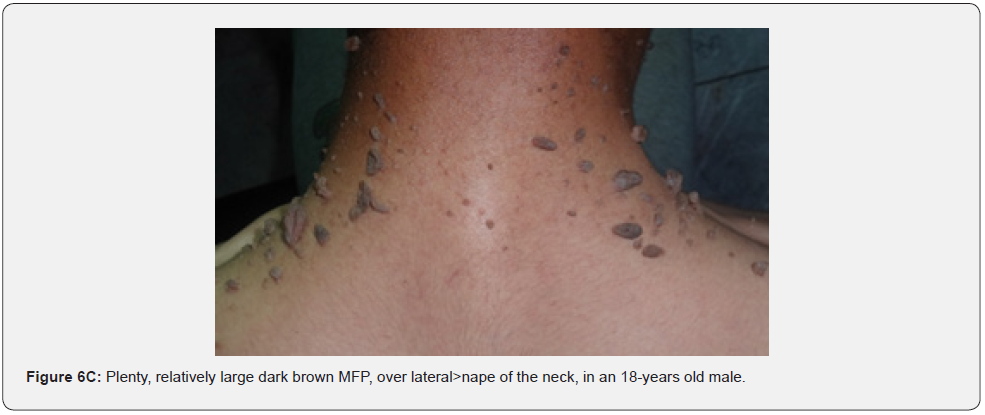
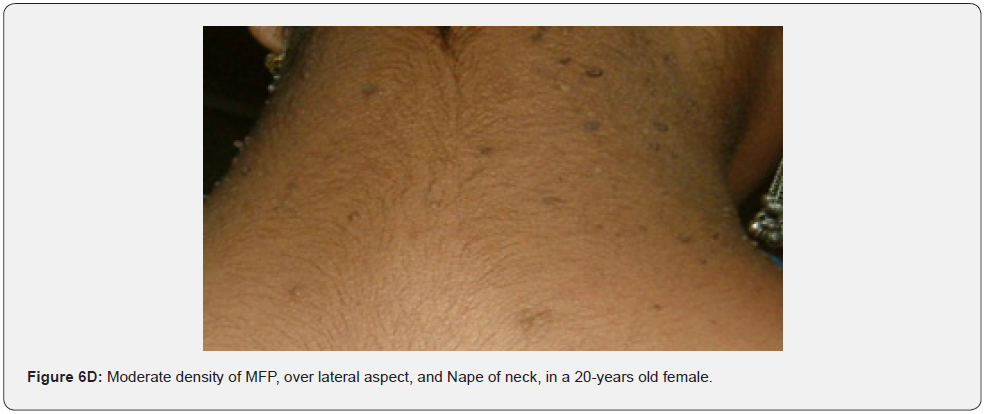
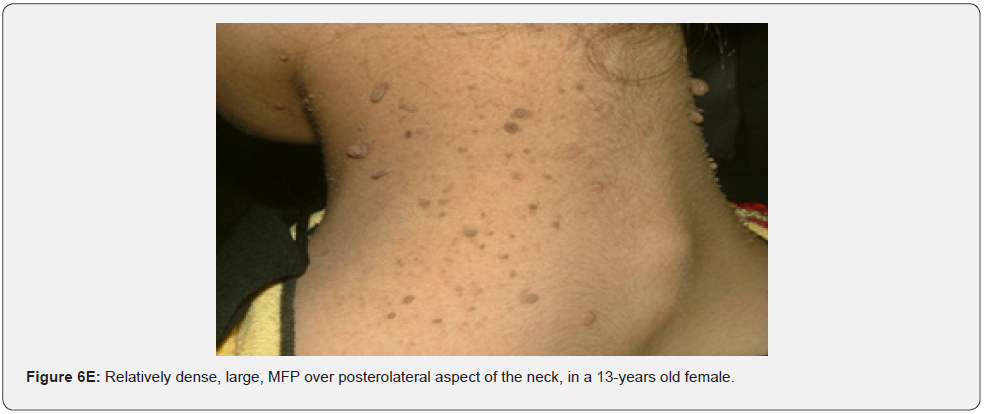
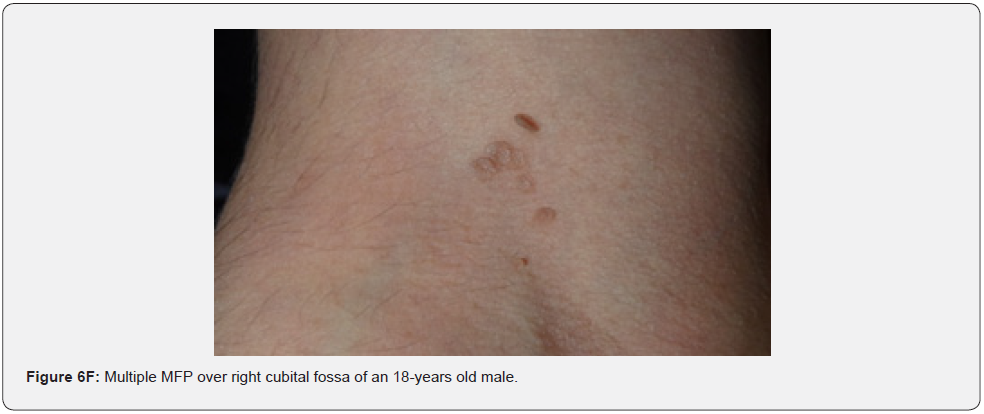
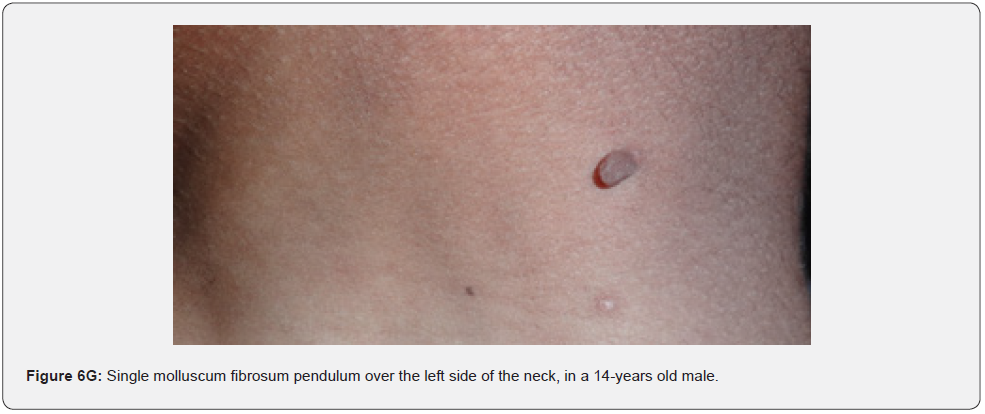
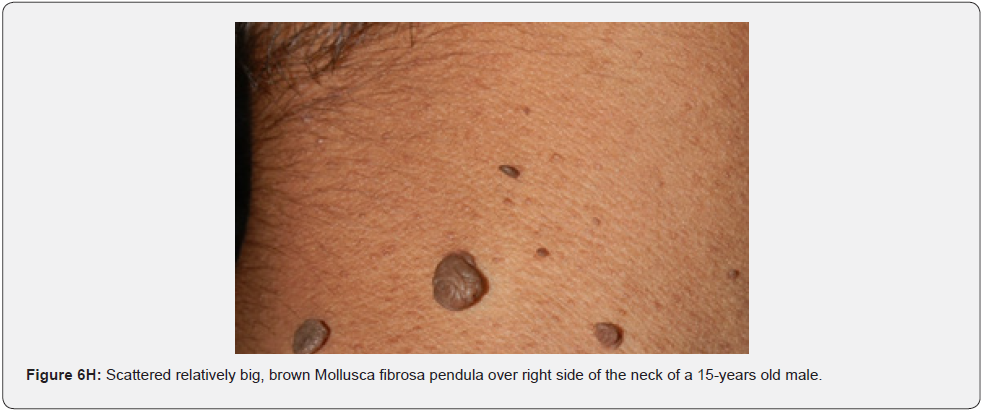
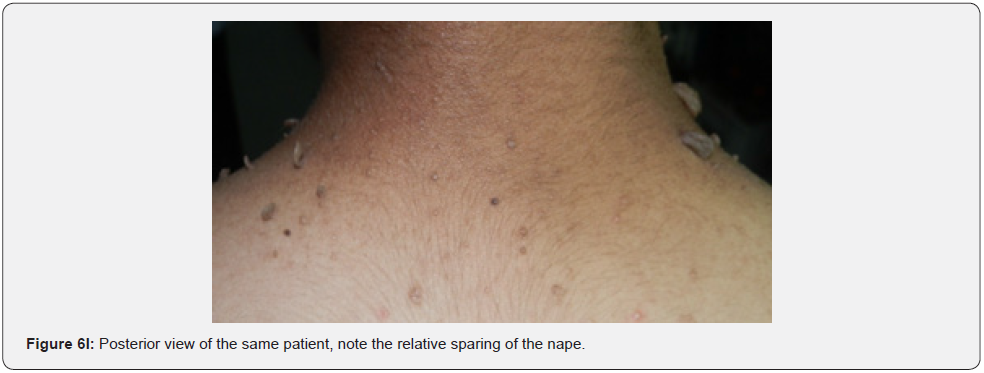
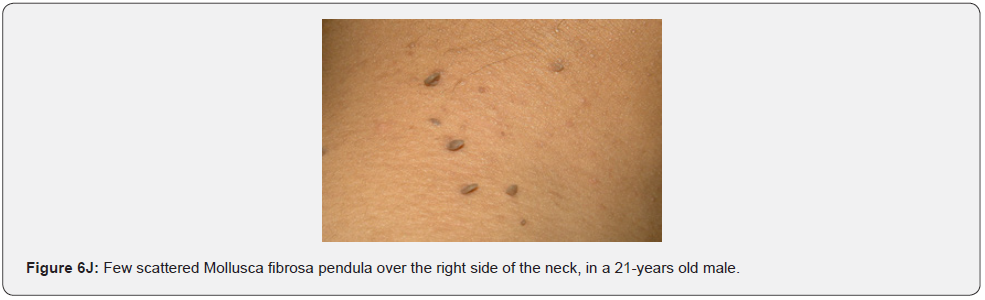
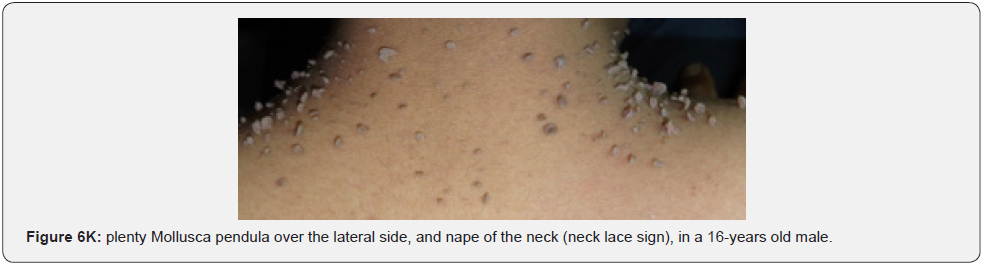
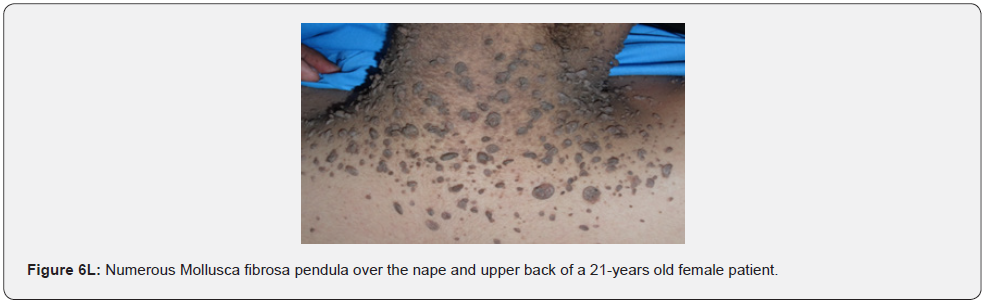
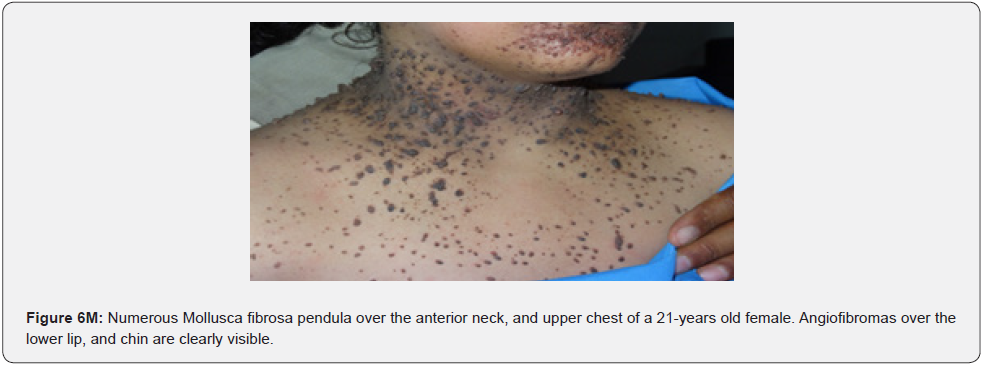
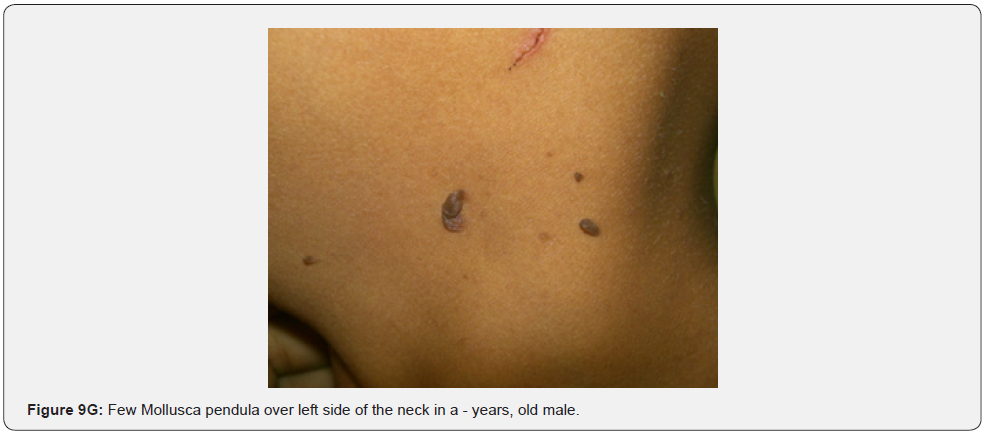
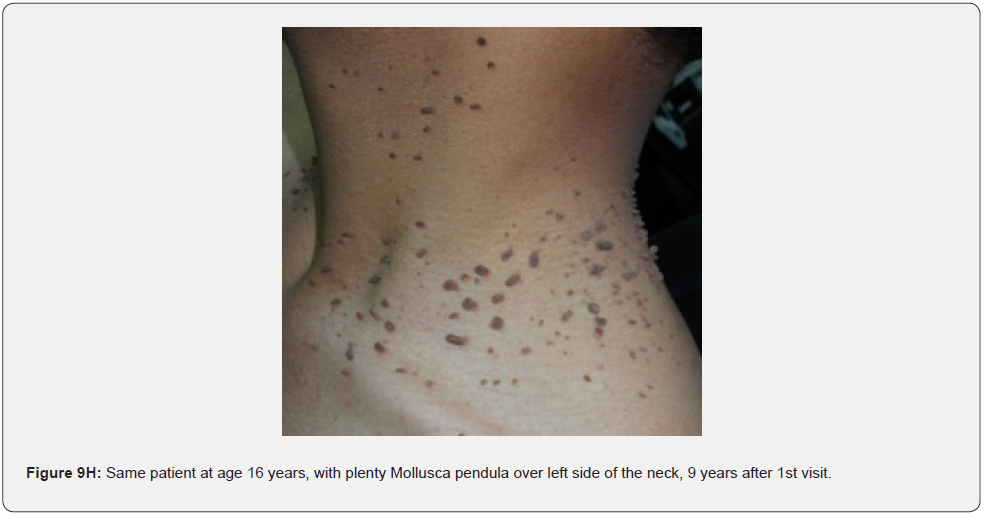
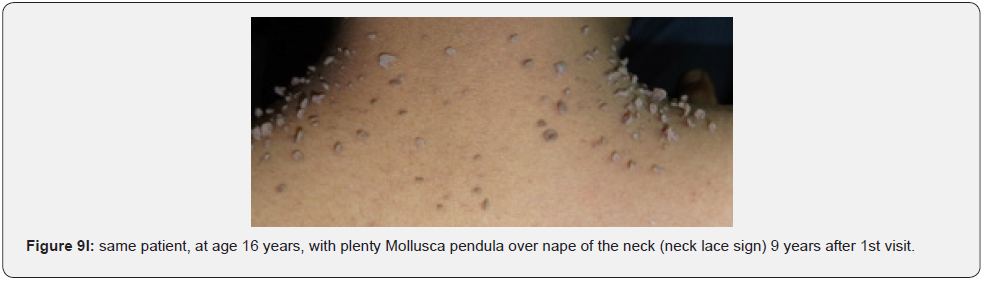
Finally, soft, pedunculated, skin-colored papules and nodules (molluscum pendulum), which resemble large acrochordons, and favor flexural areas, such as the neck, axillae and groin can be found in 23% of patients with TSC (Figure 6A-6M). These acrochordons were seen in 22 (45%) patients in our study while in only (22.6%) children in another study, and in only (14.3%) in another study.
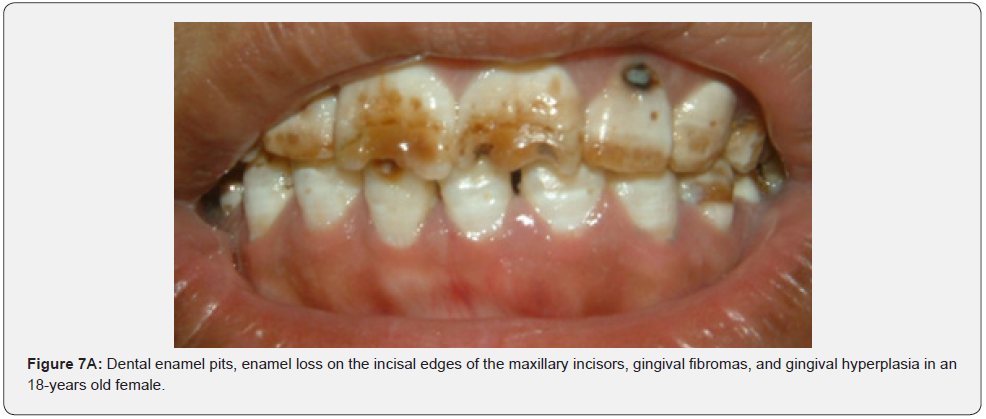
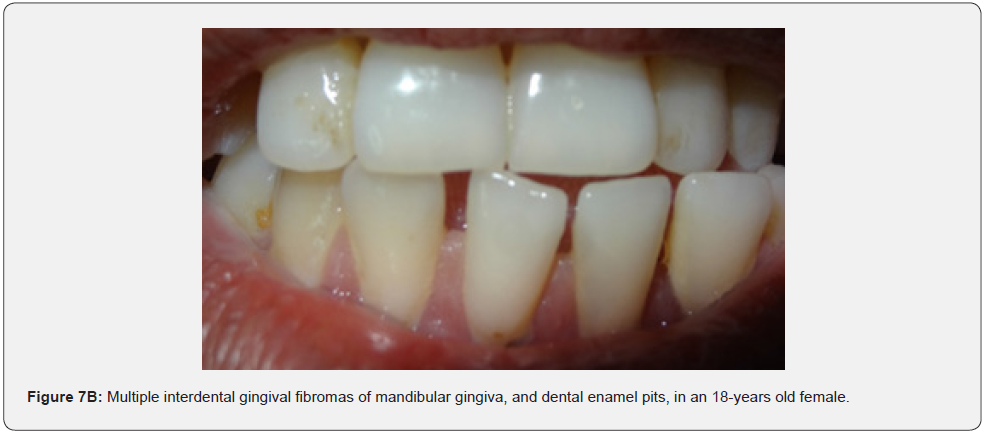
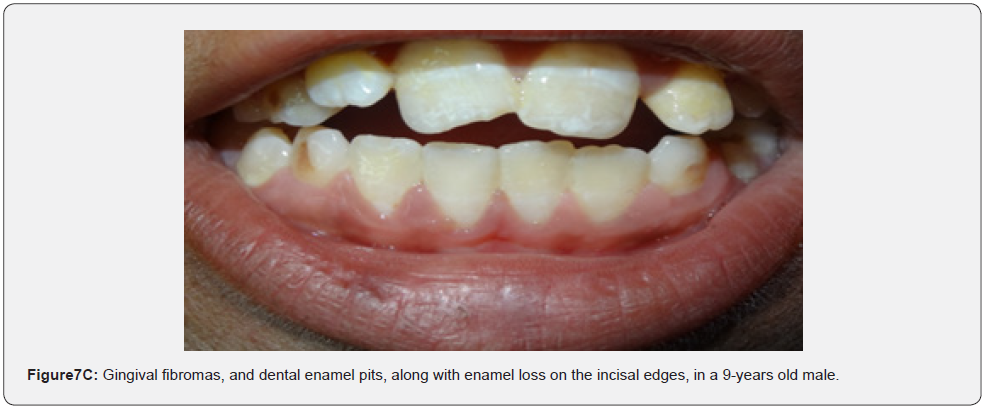
Oral fibromas (Figure 7A-7L) were seen in 15/17 (88%) of our patients, which is higher than the result found by Sparling et al (69%), while enamel dental pits (Figure 7A-7L) were found in only 12/17 (71%) of them, which is much lower than the result found by Sparling (97%) [67].
Oral manifestations of tuberous sclerosis (Figure 7A-7L) are noted in 11%– 56% of the patients[68]. Enamel pits are the most common oral manifestation of the disease and are present in almost all patients [69,70].
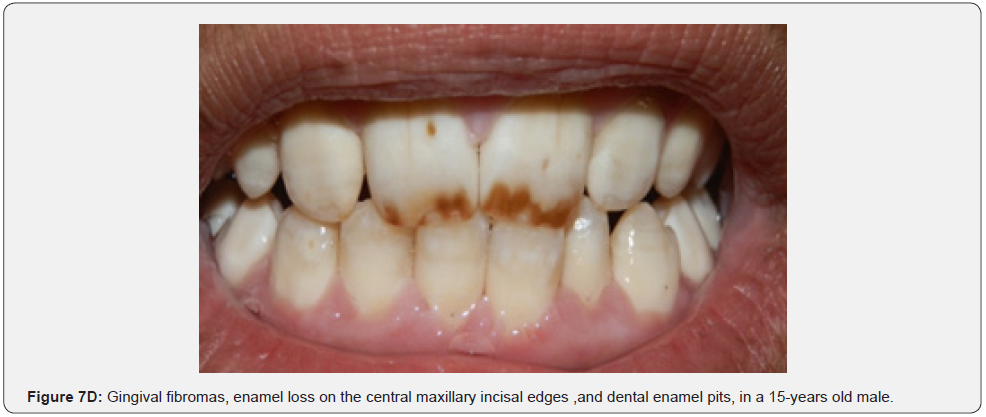
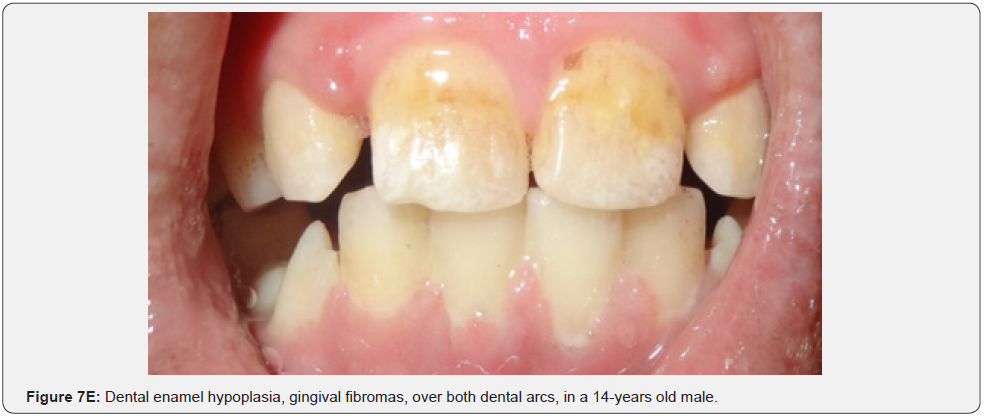
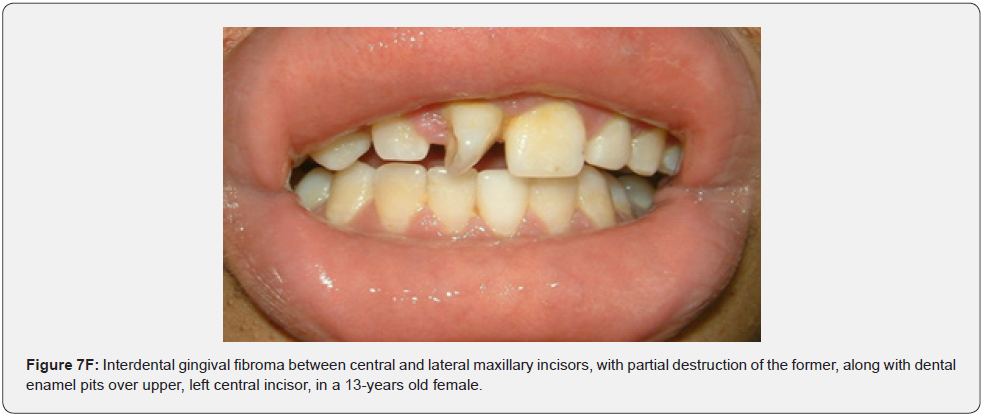
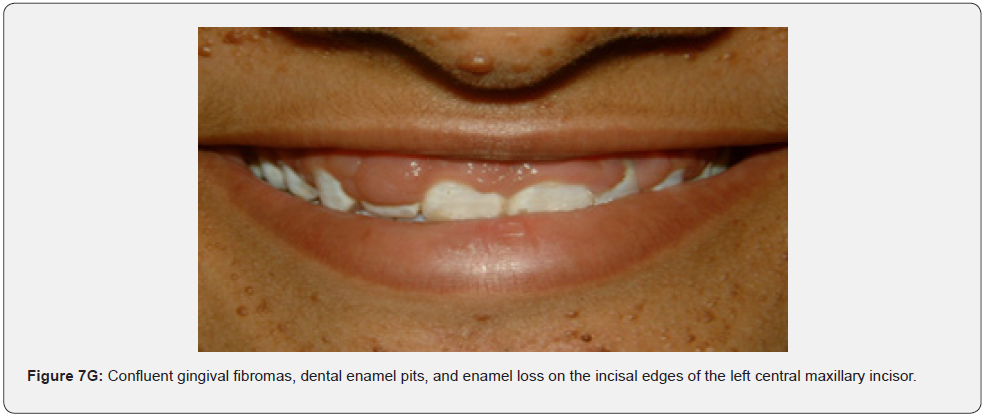
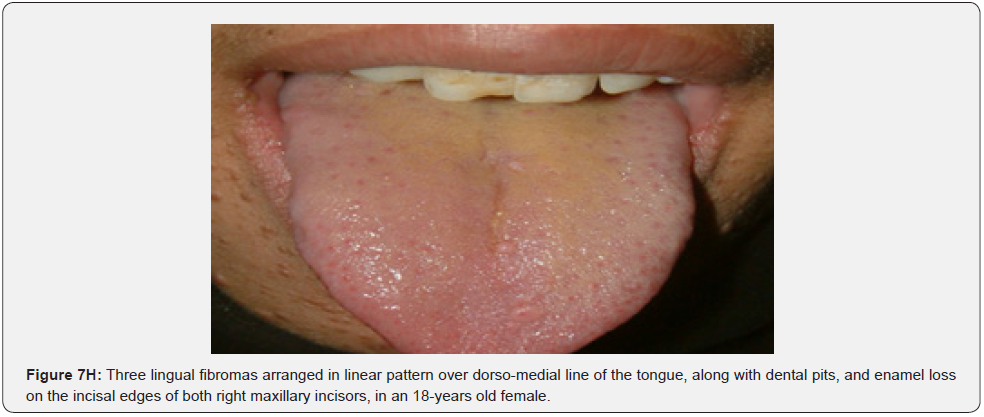
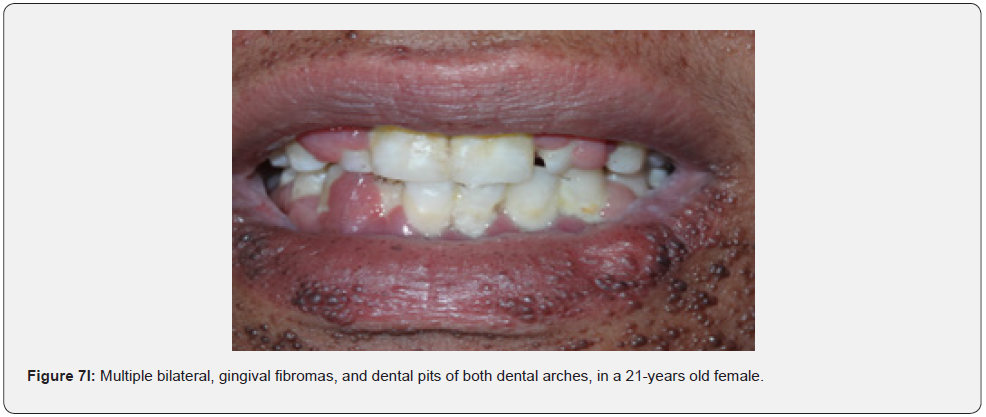
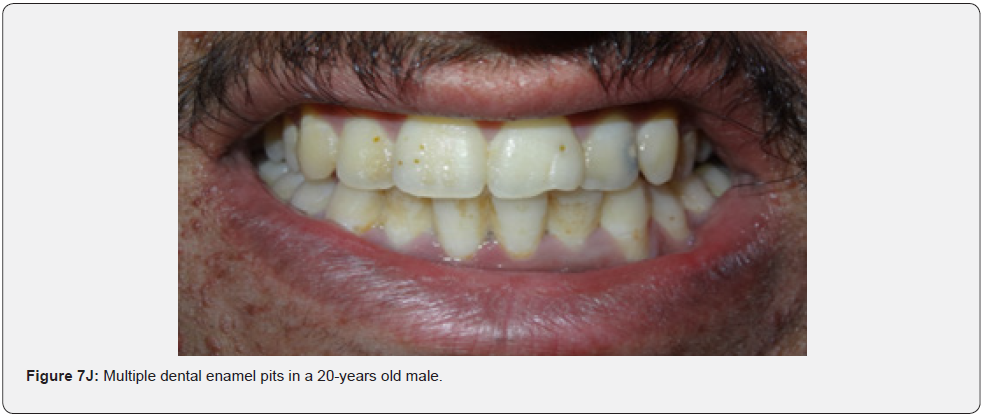
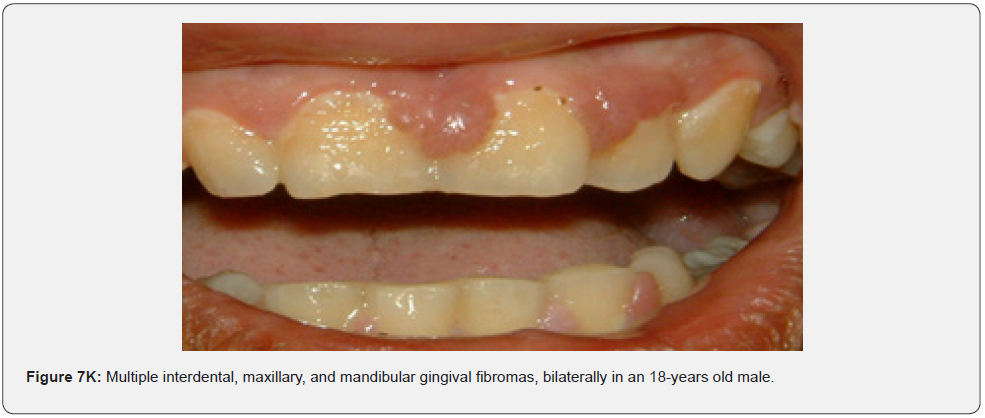
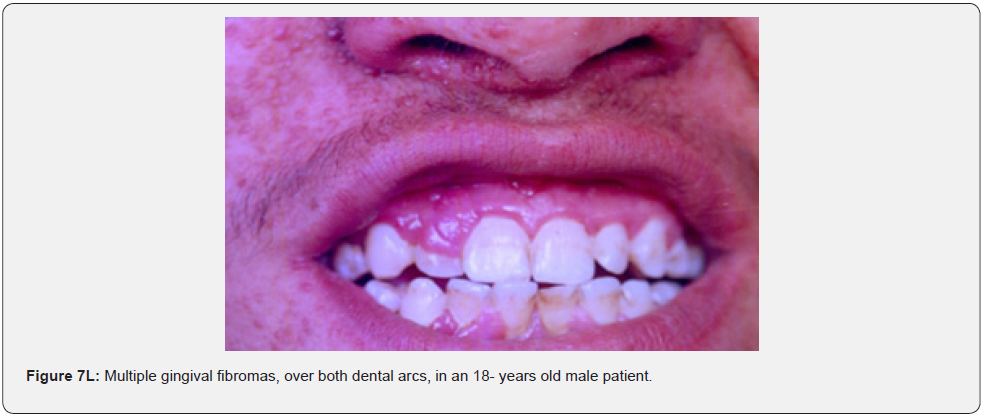
Oral fibromas and enamel pits are regarded typical oral manifestations of tuberous sclerosis. The incidence of oral fibromas in TSC patients can even get as far as 69%. These two oral finding are included as minor diagnostic criteria for TSC since 2012.
Oral fibromas and enamel pits are regarded typical oral Unusual finding in our study included, pachydermodactyly, macrodactyly, congenital localized sacral hypertrichosis, onychomatricoma, Becker nevus, cutaneous syndactyly of toes, port wine stain, digital fibrokeratoma, agminated zosteriform lentiginosis, and nail pits.
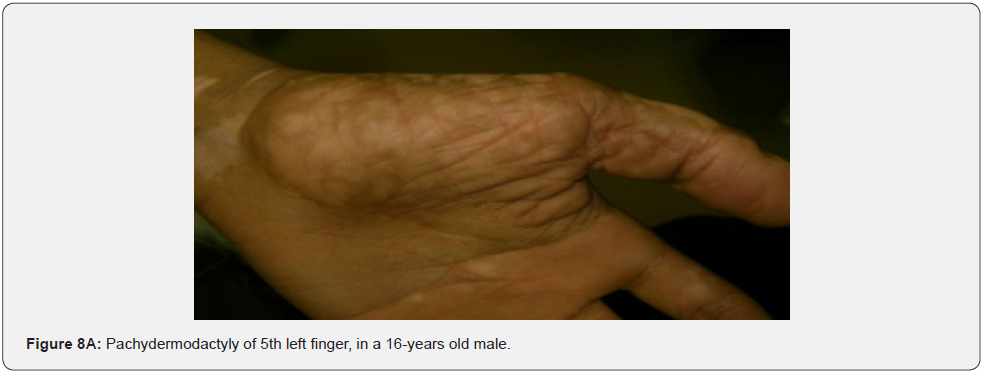
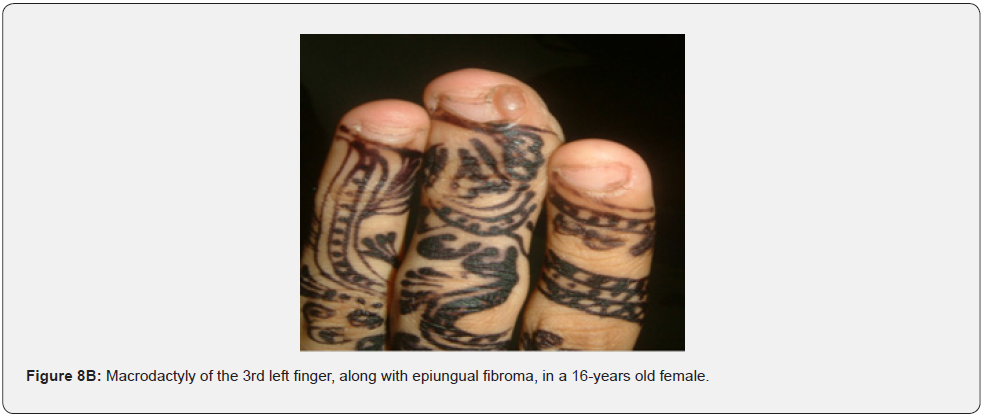
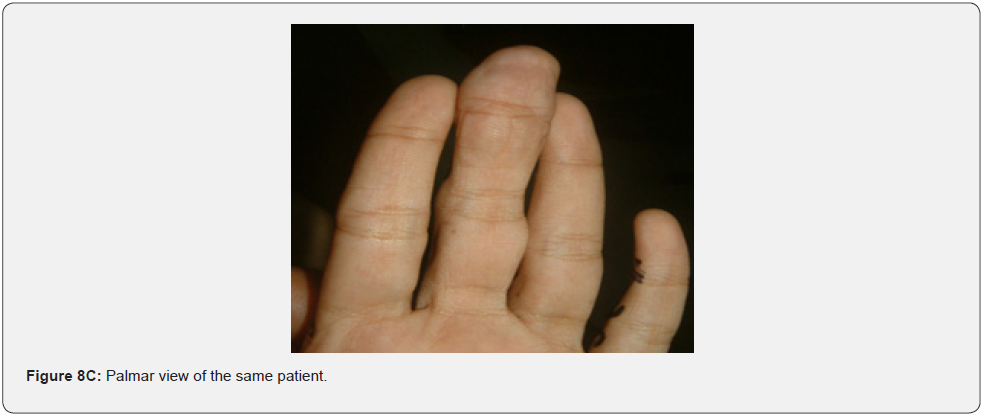
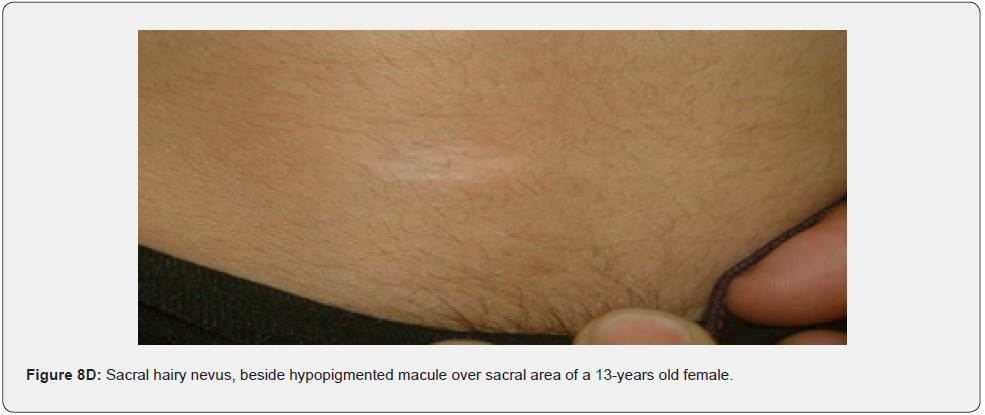
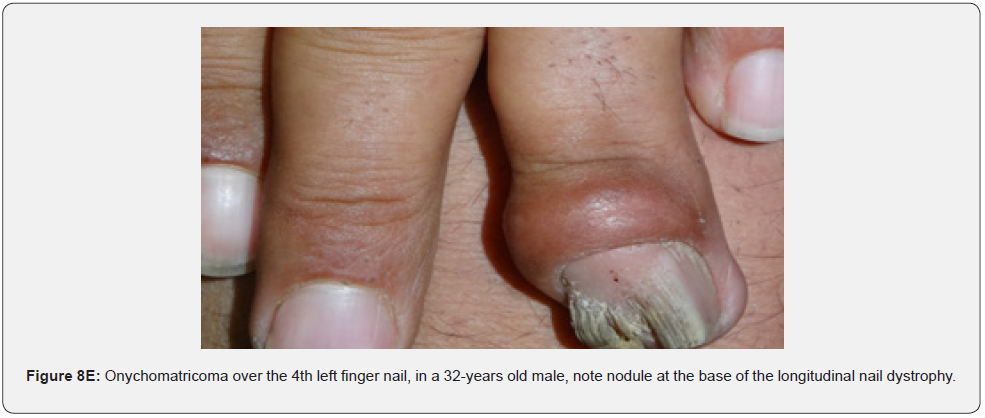
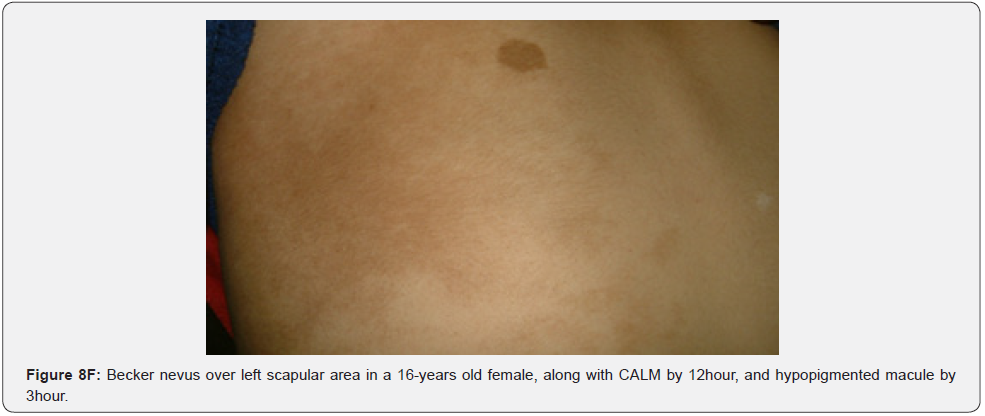
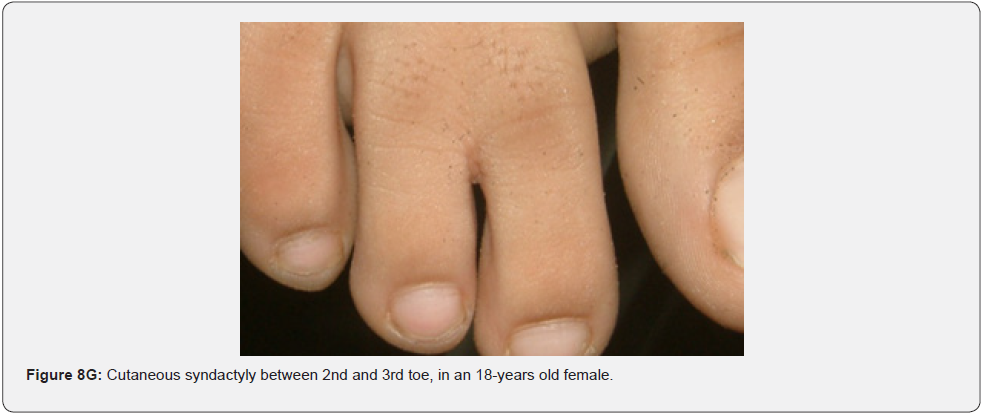
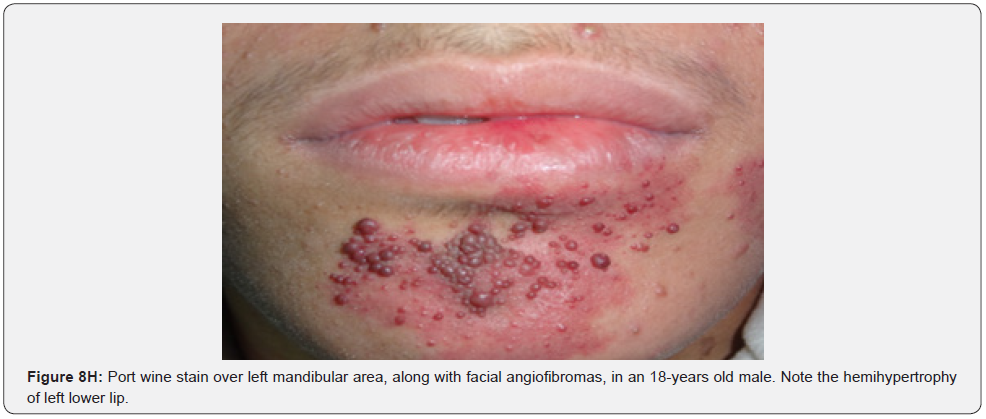
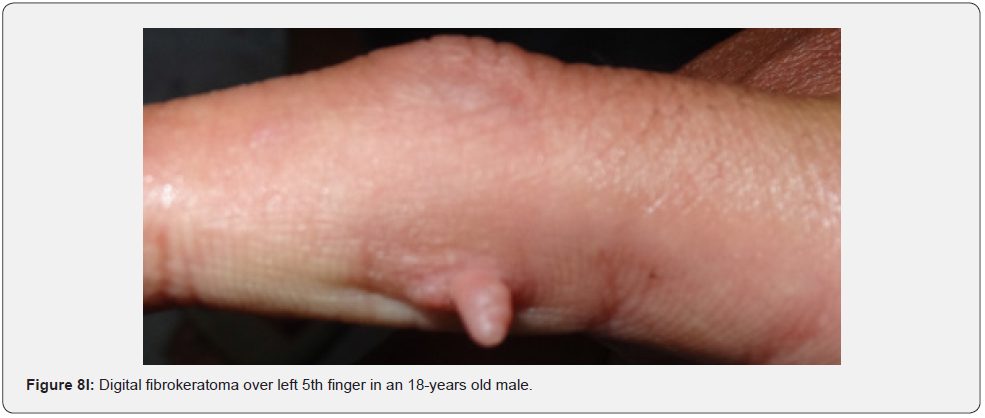
Oral fibromas and enamel pits are regarded typical oral Pachydermodactyly (Figure 8A) means thickened skin of a finger. This was reported once in a 5-year-old Chinese child with TSC in 1993 [70]. Macrodactyly (Figure 8B,8C) means big finger, approximately 10 cases were reported in association with TSC, first time in 1994 [71,72]. Congenital localized sacral hypertrichosis (Figure 8D), or faun tail nevus, characterized by hair tuft with terminal hair on the lumbosacral area, that may be a sign of occult spina bifida, a dysraphism or incomplete closure of spine canal, to the best of our knowledge has not been reported in association with TSC [73]. Onychomatricoma (Figure 8E), a benign tumor originating from nail matrix to the best of our knowledge, has been reported once in association with TSC [74]. Becker’s nevus is (Figure 8F) a benign cutaneous hamartoma, characterized by large hyperpigmented macule with irregular border and hypertrichosis, that mostly appear at puberty, and is usually unilateral, on scapular area. To the best of our knowledge, it has been reported in association with neurofibromatosis type 1, but not in association with TSC, albeit both are neurocutaneous disorders [75]. Cutaneous syndactyly (Figure 8G), a fusion of two fingers, or toes involving only the skin, but not the underlying tissue. To the best of our knowledge, it has not been reported in association with TSC. Capillary hemangioma, or port wine stain (Figure 8H), is a congenital malformation of the vasculature of the skin and mucous membrane, was stated in one study to occur in 12% of TSC patients [76]. Digital fibrokeratomas (Figure 8I) are rare, nonmalignant, fibrous, and hyperkeratotic benign soft tissue tumors of the finger.
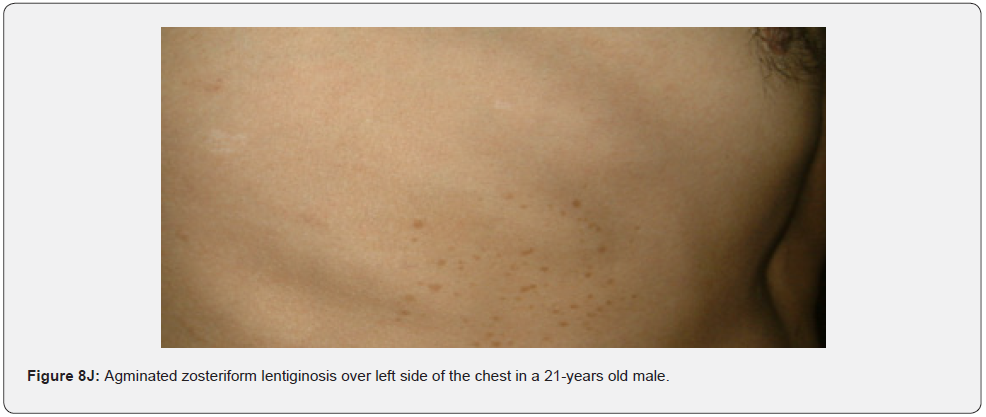
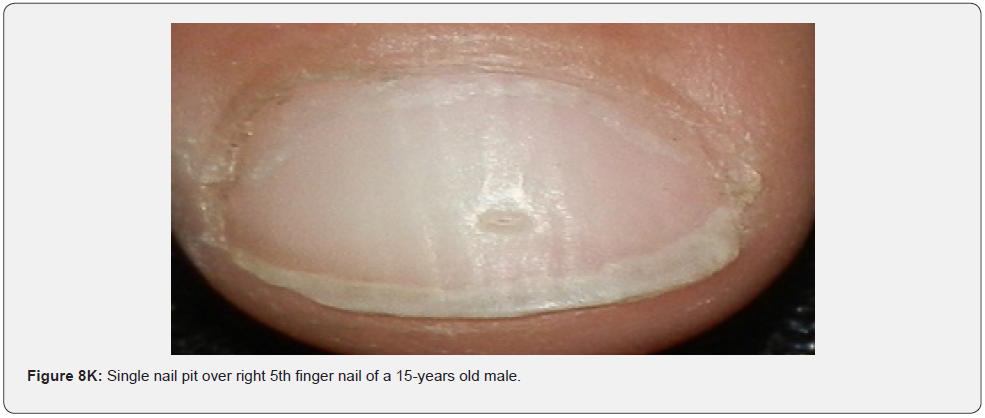
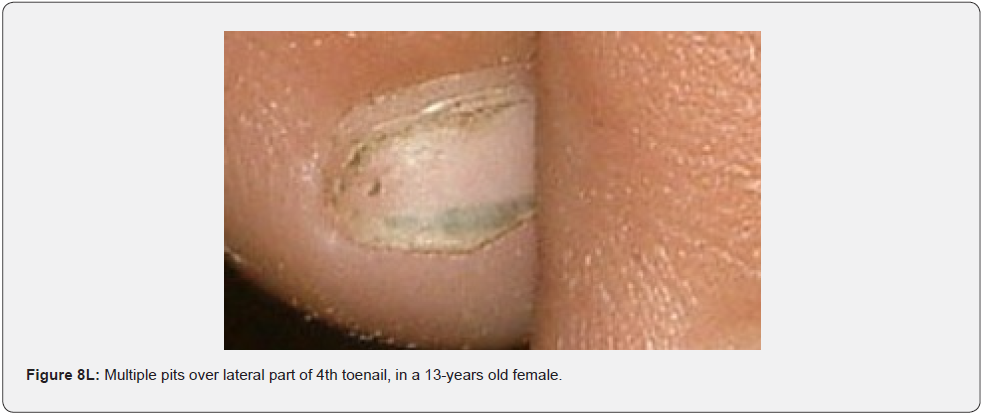
They are firm, hyperkeratotic projections arising out of a collarette of slightly raised skin. Clinically, they resemble rudimentary supernumerary digits. Microscopically, a fibrokeratoma is composed of a core of normal appearing connective tissue covered with an acanthotic epidermis which produces a dense enveloping hyperkeratosis [77-79]. Agminated melanocytic nevi (Figure 8J) are clustered macules or papules, of brown to black color, mostly unilateral, in a dermatomal distribution. To the best of our knowledge, they have not been reported in association with TSC [80]. Nail pits are small crater like depressions of the nail plates (Figure 8K,8L). They are most associated with psoriasis, alopecia areata, and eczema. To the best of our knowledge, they have not been reported in association with TSC. They can be explained by dyskeratosis like dental enamel pits seen in almost 100% of TSC patients.
Conclusion
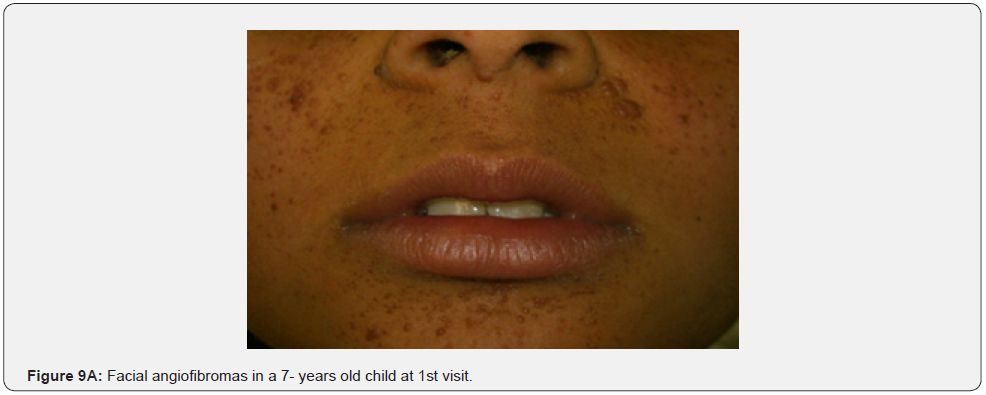
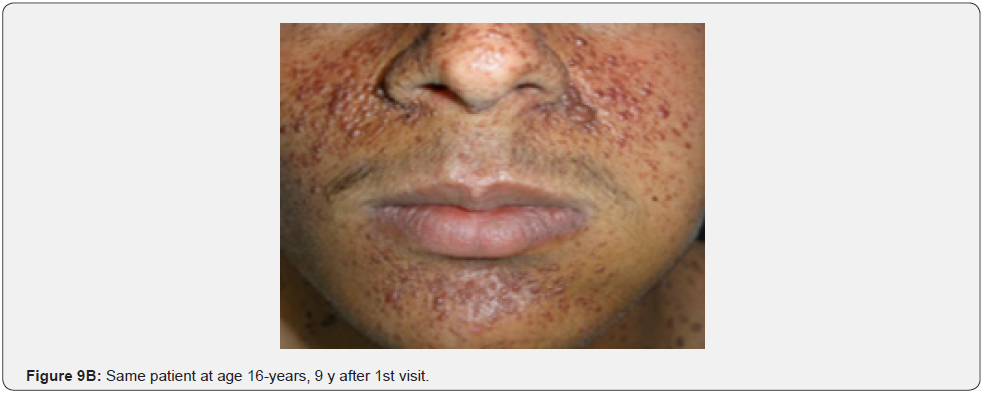
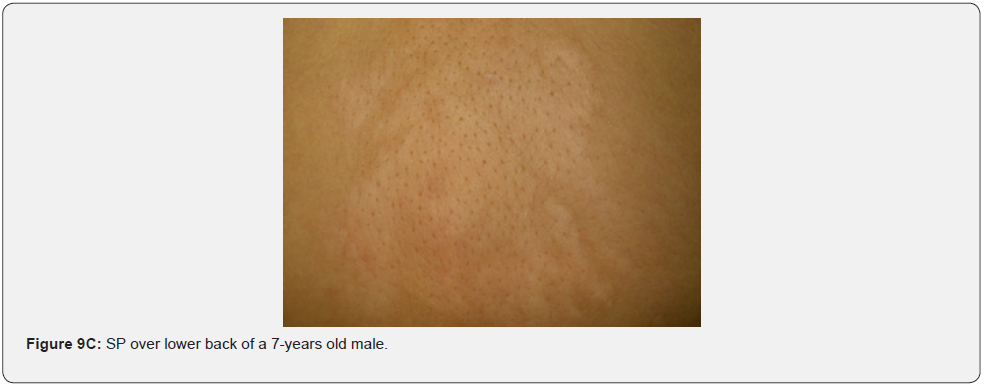
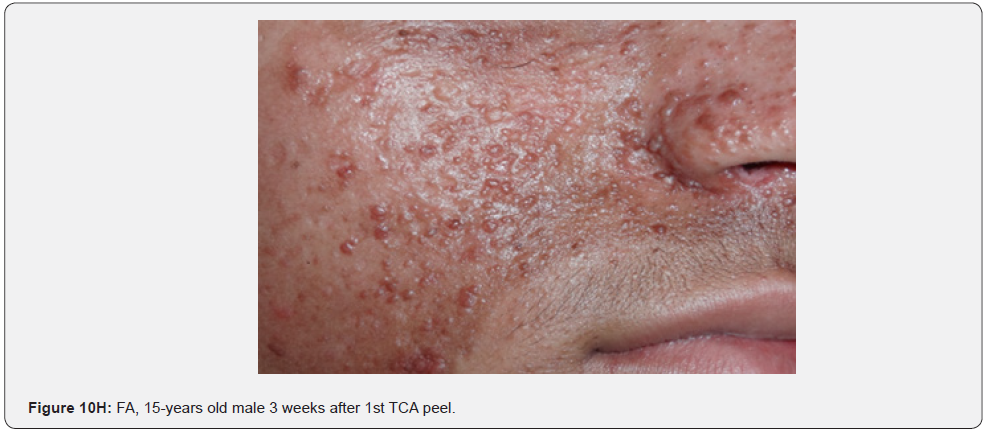
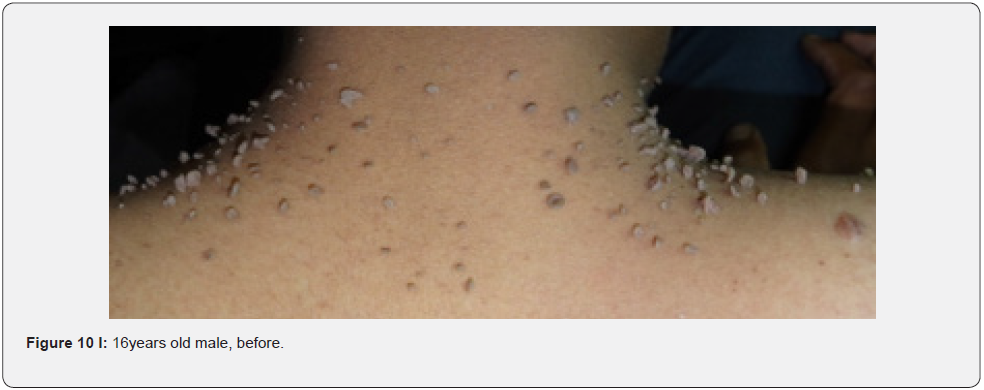
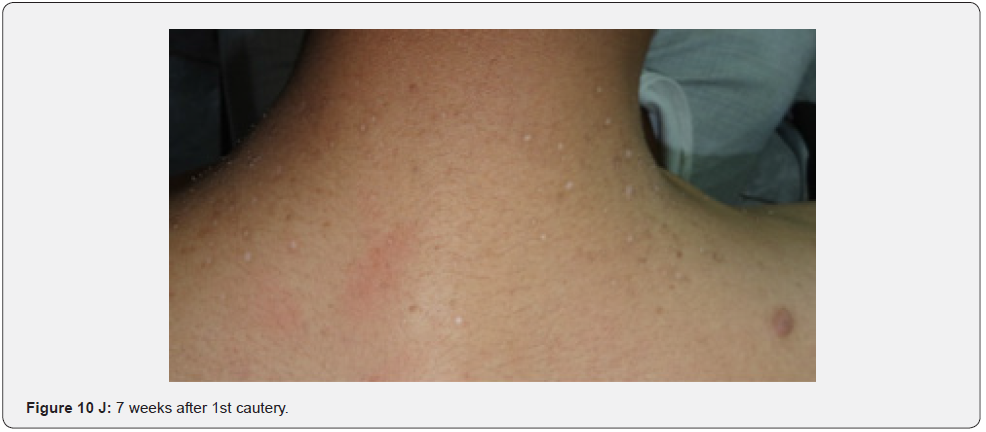
In conclusion, our study emphasizes that awareness and recognition of the simply distinguishable, very frequent, and distinctive orocutaneous features of this syndrome may help dermatologists, as well as dentists to early diagnose this serious neurocutaneous syndrome. Clinicians should be aware of the potential presenting symptoms and signs of TSC as early diagnosis may reduce morbidity and mortality and may prevent fatal complications like renal bleeding due to renal angiomyolipomas. However, a multidisciplinary approach must be undertaken from both diagnostic and therapeutic point of view. Our study is the first to provides photo-documented information about the natural history of the orocutaneous lesions of TSC patients over a period of up to twenty-five years (Figure 9A-9L), in addition to rare, unusual feature, which have been either not, or rarely reported before. Lastly our study gives information about the efficacy of electrocautery, CO2 laser and TCA chemical treatment of orocutaneous lesions, separated or combined, in TSC patients (Figure 10A-10J).
References
- Orlova KA, Crino PB (2010) The tuberous sclerosis complex Ann N Y Acad Sci 1184: 87-105.
- Bolognia JL, Jorizzo JL, Rapini RP (2017) Dermatology 4th Mosby: Elsevier.
- Curatolo P, Bombardieri R, Jozwiak S (2008) Tuberous Sclerosis. Lancet 372: 657-668.
- Pringle JJ (1890) A case of congenital adenoma sebaceum. Br J Dermatol 2: 1-14.
- Lendvay TS, Marshall FF (2003) The tuberous sclerosis complex and its highly variable manifestations J Urol 169: 1635-1642.
- Sherlock E (1911) The Feeble-minded: A Guide to Study and Practice. London: Macmillan & Co.
- Van der Hoeve J (1920) Eye Symptoms in Tuberous Sclerosis of the Brain. Trans Ophthal Soc UK 40: 329-334.
- Roach ES, Gomez MR, Northrup H (1998) Tuberous sclerosis complex consensus conference: revised clinical diagnostic criteria. J Child Neurol 13(12): 624-628.
- Osborne JP, Fryer A, Webb D (1991) Epidemiology of tuberous sclerosis. Ann N Y Acad Sci 615: 125-127.
- Wiederholt WC, Gomez MR, Kurland LT (1985) Incidence and prevalence of tuberous sclerosis in Rochester Minnesota, 1950 to 1982. Neurology 35(4): 600-603.
- Ocallaghan FJ, Shiell AW, Osborne JP, Martyn CN (1998) Prevalence of tuberous sclerosis estimated by capture-recapture analysis. Lancet 351(9114):1490.
- Sampson J, Scahill S, Stephenson J, Mann L, Connor J (1989) Genetic aspects of tuberous sclerosis in the west of Scotland. J Med Genet 26(1): 28-31.
- Webb D, Clarke A, Fryer A, Osborne J (1996) The cutaneous features of tuberous sclerosis: a population study. Br J Dermatol 135(1): 1 -5.
- Devlin LA, Shepherd CH, Crawford H, Morrison PJ (2006) Tuberous sclerosis complex: clinical features, diagnosis, and prevalence within Northern Ireland. Dev Med Child Neurol 48(6): 495-499.
- Hong CH, Darling TN, Lee CH (2009) Prevalence of tuberous sclerosis complex in Taiwan: a national population-based study. Neuroepidemiology 33(4): 335-341.
- Wong V (2006) Study of the relationship between tuberous sclerosis complex and autistic disorder. J Child Neurol 21(3): 199-204.
- Rosser T, Panigrahy A, McClintock W (2006) The diverse clinical manifestations of tuberous sclerosis complex: a review. Semin Pediatr Neurol 13(1): 27-36.
- Roach ES, Gomez MR, Northrup H (1998) Tuberous sclerosis complex consensus conference: revised clinical diagnostic criteria. J Child Neurol 13(12): 624-628.
- Slowinska M, Jozwiak S, Peron A, Borkowska J, Chmielewski D, et al. (2018) Early diagnosis of tuberous sclerosis complex: a race against time. How to make the diagnosis before seizures? Orphanet J Rare Dis 13(1): 25.
- Napolioni V, Curatolo P (2008) Genetics and molecular biology of tuberous sclerosis complex. Curr Genomics 9(7): 475-487.
- Northrup H, Krueger DA (2013) International Tuberous Sclerosis Complex Consensus Group. Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol 49(4): 243-254.
- Portocarrero LKL, Quental KN, Samorano LP, Oliveira ZNP, Rivitti-Machado MCDM (2018) Tuberous sclerosis complex: review based on new diagnostic criteria. An Bras Dermatol 93(3): 323-331.
- Hake S (2011) Cutaneous manifestations of tuberous sclerosis. Ochsner J 10(3): 200-204.
- Cardis MA, DeKlotz CMC (2017) Cutaneous manifestations of tuberous sclerosis complex and the pediatrician’s role. Arch Dis Child 102(9): 858-863.
- European Chromosome 16 Tuberous Sclerosis Consortium (1993) Identification and characterization of the tuberous sclerosis gene on chromosome 16 Cell 75(7): 1305-1315.
- Zollner JP, Franz DN, Hertzberg C, Nabbout R, Rosenow F, et al. (2020) A systematic review on the burden of illness in individuals with tuberous sclerosis complex (TSC). Orphanet J Rare Dis 15(1): 23.
- De Waele L, Lagae L, Mekahli D (2015) Tuberous sclerosis complex: the past and the future. Pediatr Nephrol 30(10): 1771-1780.
- Rosset C, Netto CBO, Ashton-Prolla P (2017) TSC1 and TSC2 gene mutations and their implications for treatment in Tuberous Sclerosis Complex: a review. Genet Mol Biol 40(1): 69-79.
- Kozlowski P, Roberts P, Dabora S, Franz D, J Bissler J, et al. (2007) Identification of 54 large deletions/duplications in TSC1 and TSC2 using MLPA, and genotype/phenotype correlations. Hum Genet 121(3-4): 389-400.
- Bongiorno MA, Nathan N, Oyerinde O, Wang JA, Lee CR, Brown GT, et al. (2017) Clinical Characteristics of Connective Tissue Nevi in Tuberous Sclerosis Complex with Special Emphasis on Shagreen Patches. JAMA Dermatol 153(7): 660-665.
- Martin KR, Zhou W, Bowman MJ, Shih J, Au KS, et al. The genomic landscape of tuberous sclerosis complex. Nat Commun 15(8): 15816.
- Huang J, Dibble C, Matsuzaki M, Manning B (2008) The TSC1-TSC2 complex is required for proper activation of mTOR complex 2. Mol Cell Biol 28(12): 4104-4115.
- Au KS, Williams AT, Roach ES, Batchelor L, Sparagana SP, et al. (2007) Genotype/phenotype correlation in 325 individuals referred for a diagnosis of tuberous sclerosis complex in the United States. Genet Med 9(2): 88-100.
- Kingswood C, Bolton P, Crawford P, Harland C, Johnson SR, et al. (2016) The clinical profile of tuberous sclerosis complex (TSC) in the United Kingdom: A retrospective cohort study in the Clinical Practice Research Datalink (CPRD). Eur J Paediatr Neurol 20(2): 296-308.
- Sun XF, Yan CL, Fang L, Shen FM, Liao KH (2005) Cutaneous lesions and visceral involvement of tuberous sclerosis. Chin Med J 118 (3): 215-219.
- Dabora SL, Jozwiak S, Franz DN, PS Roberts, A Nieto, et al. (2001) Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1, disease in multiple organs. Am J Hum Genet 68(1): 64-80.
- Taveira DaSilva AM, Moss J (2015) Clinical features, epidemiology, and therapy of lymphangioleiomyomatosis. Clin Epidemiol 7: 249-257.
- Van Slegtenhorst M, Verhoef S, Tempelaars A, Bakker L, Wang Q, et al. (1999). Mutational spectrum of the TSC1 gene in a cohort of 225 tuberous sclerosis complex patients: no evidence for genotype-phenotype correlation. J Med Genet 36(4): 285-289.
- Lam HC, Nijmeh J, Henske EP (2017) New developments in the genetics and pathogenesis of tumors in tuberous sclerosis complex. J Pathol 241(2): 219-225.
- Caban C, Khan N, Hasbani DM, Crino PB (2016) Genetics of tuberous sclerosis complex: implications for clinical practice. Appl Clin Genet 10: 1-8.
- Lim JS, Gopalappa R, Kim SH, Ramakrishna S, Lee M, et al. (2017) Somatic Mutations in TSC1 and TSC2 Cause Focal Cortical Dysplasia. Am J Hum Genet 100(3): 454-472.
- Darling TN, Thiele EA, Moss J (2021) TSC1 and TSC2 Genotype in Tuberous Sclerosis Complex: Are Other Manifestations of this Multisystem Disease Affected by Genotype? Ann Am Thorac Soc 18(5): 775-777.
- Reyna-Fabián ME, Hernández-Martínez NL, Alcántara-Ortigoza MA, Ayala-Sumuano JT, Enríquez-Flores S, et al. (2020) First comprehensive TSC1/TSC2 mutational analysis in Mexican patients with Tuberous Sclerosis Complex reveals numerous novel pathogenic variants. Sci Rep 10(1): 6589.
- Han JM, Sahin M (2011) TSC1/TSC2 signaling in the CNS. FEBS Lett. 585(7): 973-980.
- Feliciano DM (2020) The Neurodevelopmental Pathogenesis of Tuberous Sclerosis Complex (TSC). Front Neuroanat 14: 39.
- Saxton RA, Sabatini DM (2007) mTOR Signaling in Growth, Metabolism, and Disease. Cell 68(6): 960-976.
- Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149(2): 274-293.
- Sampson JR (2009) Therapeutic targeting of mTOR in tuberous sclerosis. Biochem Soc Trans 37(1): 259-264.
- Staley BA, Vail EA, Thiele EA (2011) Tuberous sclerosis complex: diagnostic challenges, presenting symptoms, and commonly missed signs. Pediatrics 127(1): 117-125.
- Rodrigues DA, Gomes CM, Costa IMC (2012) Tuberous sclerosis complex. An Bras Dermatol 87(2): 184-196.
- DiMario FJ Jr, Sahin M, Ebrahimi-Fakhari D (2015) Tuberous sclerosis complex. Pediatr Clin North Am. 62(3): 633-648.
- Roach ES, DiMario FJ, Kandt RS, Northrup H (1999) Tuberous Sclerosis Consensus Conference: recommendations for diagnostic evaluation. National Tuberous Sclerosis Association. J Child Neurol 14(6): 401-407.
- Almobarak S, Almuhaizea M, Abukhaled M, Alyamani S, Dabbagh O, et al. (2018) Tuberous Sclerosis Complex: Clinical Spectrum and Epilepsy: A Retrospective Chart Review Study. Transl Neurosci 9: 154-160.
- Yates JR (2006) Tuberous sclerosis. Eur J Hum Genet 14: 1065-1073.
- Krueger DA, Northrup H (2013) Tuberous sclerosis complex surveillance and management: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol 49(4): 255-265.
- Schwartz RA, Fernandez G, Kotulska K, Sergiusz Jóźwiak (2007) Tuberous sclerosis complex: advances in diagnosis, genetics, and management. J Am Acad Dermatol 57(2): 189-202.
- Jozwiak S, Schwartz RA, Janniger CK, Michałowicz R, Chmielik J, et al. (1998) Skin lesions in children with tuberous sclerosis complex: their prevalence, natural course, and diagnostic significance. Int J Dermatol 37(2) 911-917.
- Huggins RH, Schwartz RA, Janniger C (2005) Vitiligo. Acta Dermatovenerol Alp Pannonica Adriat pp. 144-145.
- Wataya-Kaneda M, Tanaka M, Hamasaki T, Katayama I (2013) Trends in the Prevalence of Tuberous Sclerosis Complex Manifestations: An Epidemiological Study of 166 Japanese Patients. PLoS ONE 8(5): e63910.
- Wolff K, Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, et al. (2007) Fitzpatrick´s Dermatology in General Medicine. 7th Mc Graw-Hill, New York, USA.
- Astrinidis A, Henske EP (2005) Tuberous sclerosis complex: linking growth and energy signaling pathways with human disease. Oncogene 24(50): 7475-7481.
- Teng JM, Cowen EW, Wataya-Kaneda M, Gosnell ES, Witman PM et al. (2014) Dermatologic and dental aspects of the 2012 International Tuberous Sclerosis Complex Consensus Statements. JAMA Dermatol 150(10): 1095-1001.
- Krishnan S, Yesudian DP, Jayaraman M (1996) Tuberous sclerosis. Indian J Dermatol Venereol Leprol 62: 239-241.
- Vanderhooft SL, Francis JS, Pagon RA, Smith LT, Sybert VP (1996) Prevalence of hypopigmented macules in a healthy population. J Pediatr 129 (3): 355-361.
- Ghosh SK, Debabrata B, Gobinda C, Ghosh A, Sarkar S, et al. (2009) Mucocutaneous Changes in Tuberous Sclerosis Complex: A Clinical Profile Of 27 Indian Patients. Indian J Dermatol 54 (3): 255-257.
- Nath J, Dubey A, Pavan R (2015) Analysis of twenty pediatric cases of tuberous sclerosis complex: are we doing enough? Indian J Dermatol Venereol Leprol 81(1): 23-28.
- Rama Rao GR, Krishna Rao PV, Gopal K, Kumar YH, Ramachandra BV (2008) Forehead plaque: A cutaneous marker of CNS involvement in tuberous sclerosis. Indian J Dermatol Venereol Leprol 74(1): 28-31.
- Aldrich CS, Hong CH, Groves L, Olsen C, Moss J, et al. (2010) Acral lesions in tuberous sclerosis complex: insights into pathogenesis. J Am Acad Dermatol 63(2): 244-251.
- Sparling JD, Hong CH, Brahim JS, Moss J, Darling TN (2007) Oral findings in 58 adults with tuberous sclerosis complex. J Am Acad Dermatol 56(5): 786-790.
- López E, Escovich L, Vigna A (2003) Tuberous Sclerosis: Presentation of a clinical case with oral manifestations. Med Oral 8(2): 122-128.
- Harutunian K, Figueiredo R, Gay-Escoda C (2011) Tuberous sclerosis complex with oral manifestations: A case report and literature review. Med Oral Patol Oral Cir Bucal 16(4): 478-481.
- Korporowicz E, Olczak-Kowalczyk D, Lipiec M, Słowińska M, Gozdowski D, et al. (2020) Oral Findings in Children, Adolescents and Adults with Tuberous Sclerosis Complex. J Clin Pediatr Dent 44(3): 190-195.
- Lo WL, Wong CK (1993) Localized pachydermodactyly in tuberous sclerosis. Clin Exp Dermatol. 18(2): 146-147.
- Norman-Taylor F, Mayou BJ (1994) Macrodactyly in tuberous sclerosis. J R Soc Med. 87(7): 419-420.
- Sindhuja T, Agarwal S, Gupta V (2021) Macrodactyly in tuberous sclerosis complex. Clin Exp Dermatol 46(5): 929-931.
- Birol A, Bademci G (2004) Faun tail: diagnosis of occult spinal dysraphism with a rare cutaneous marker. J Dermatol 31(3): 251-252.
- Fernández-Sánchez M, Saeb-Lima M, Charli-Joseph Y, Méndez-Flores S, Sánchez-Hernández C, et al. (2012) Onychomatricoma: an infrequent nail tumor. Indian J Dermatol Venereol Leprol 78(3): 382-383.
- Nijhawan M, Agrawal S, N Sehga V, Bagri M, Bishnoi S, et al. (2019) Becker’s Nevus and Shagreen Patch: Extraordinary Cutaneous Association with Neurofibromatosis in Two Successive Generation. JOJ Dermatol and Cosmet 1(5): 555575.
- Ben-Amitai D, Halachmi S, Lapidoth M (2011) Are port wines stains a feature of tuberous sclerosis? J Eur Acad Dermatol Venereol 25(7): 804-807.
- Bart RS, Andrade R, Kopf AW, Leider M (1968) Acquired digital fibrokeratomas. Arch Dermatol. 97(2): 120-129.
- Shin J, Kim YC (2013) Multiple agminated acquired melanocytic nevi. Ann Dermatol 25(2): 251-252.






























