Primary Perianal Paget Disease: Case Report and Review of Literature
Marta Serra1*, Cláudia Mendes1, Joana Romano2, Rita Sampaio3 and Miguel Trigo1
1Department of Plastic Reconstructive Surgery, Egas Moniz Hospital, Lisbon, Portugal
2Department of General Surgery, Egas Moniz Hospital, Lisbon, Portugal
3Department of Anatomic Pathology, Egas Moniz Hospital, Lisbon, Portugal
Submission: January 20, 2021;Published: February 08, 2020
*Corresponding author: Marta Serra, Department of Plastic Reconstructive Surgery, Egas Moniz Hospital, Lisbon, Portugal
How to cite this article: Marta S, Cláudia M, Joana R, Rita S, Miguel T. Primary Perianal Paget Disease: Case Report and Review of Literature. JOJ Dermatol & Cosmet. 2021; 3(4): 555623. DOI: 10.19080/JOJDC.2021.03.555623
Abstract
Mammary Paget disease (MPD) and extramammary Paget disease (EMPD) are both characterized by epidermal Paget cells and their differences lay in anatomical location and histopathology. MPD is known for its involvement of the breast (nipple, areola, skin) and EMPD for its occurrence in apocrine gland bearing areas (perineum, buttocks, axilla, eyelids). EMPD can be primary due to its skin origin or secondary to other neoplasms [1]. Perianal Paget disease (PPD) is a rare occurrence with less than 60 published cases until 2012 [2]. This article presents a clinical case of primary PPD and review of literature.
Keywords: Extramammary Paget Disease; Perianal Paget Disease; Radiotherapy; Dermatovenerology
Abbreviations: CK: Cytokeratin; EMPD: Extramammary Paget Disease; GCDFP: Gross Cystic Disease Fluid Protein; MPD: Mammary Paget Disease; PD: Paget Disease; PPD: Perianal Paget Disease; RT: Radiotherapy; SLNB: Sentinel Lymph Node Biopsy
Introduction
Paget disease (PD) is a rare cutaneous disorder described as an apocrine gland tumor occurring in both a benign and a malignant form, with metastatic potential, which was first reported in the breast by Sir James Paget in 1874. Since then, EMPD has been reported in several sites, including axilla, thigh and perineum [3]. The first case of PPD was reported by Darrier and Couillaud in 1893 [4]. PPD is a variant of EMPD and represents a rare intraepidermal adenocarcinoma characterized by the presence of typical Paget cells. It can be either primary or secondary to carcinoma from an adjacent internal organ. PPD with underlying colorectal adenocarcinoma is usually looked upon as a secondary disease (40-60% of patients). Besides clinical history, patient evaluation and diagnostic work-up exams, skin biopsy with the detection of immunohistochemical markers, such as cytokeratin 7 (CK7), CK20 and gross cystic disease fluid protein 15 (GCDFP-15), can distinguish primary from secondary forms of PPD [5]. We report a case of primary PPD and its multidisciplinary approach in our hospital.
Case Report
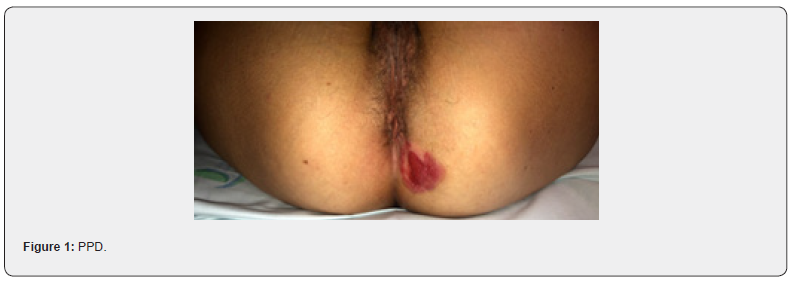
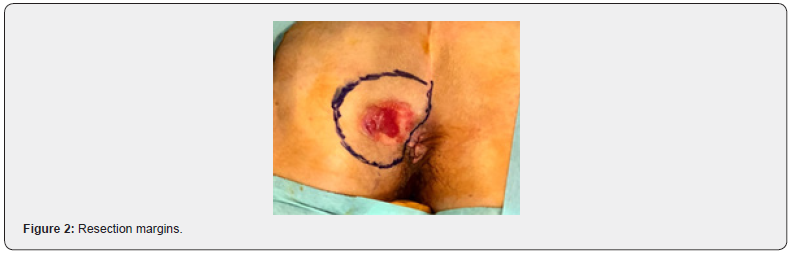
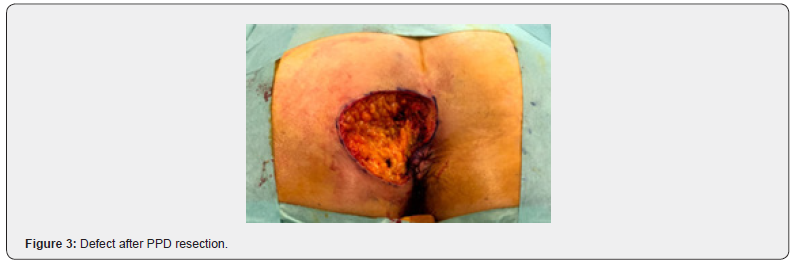
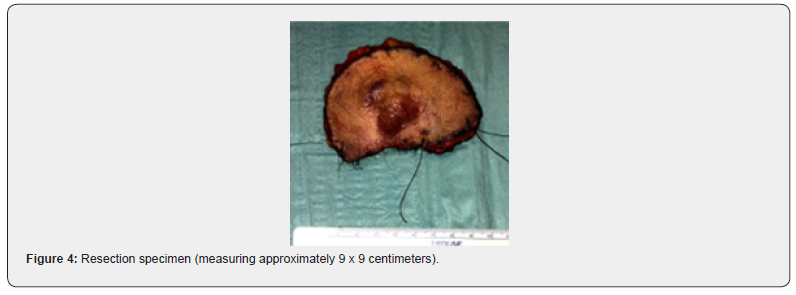
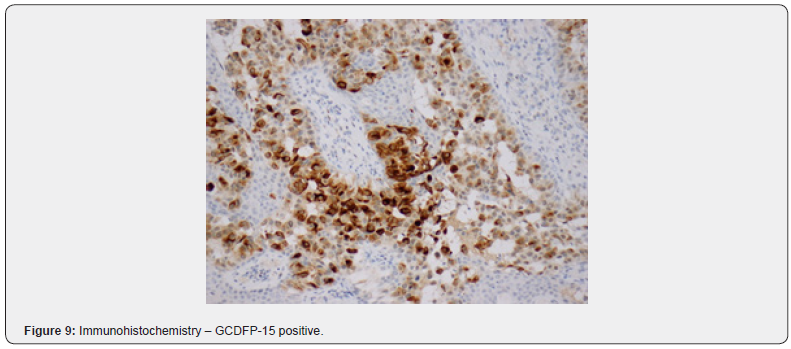
A 52-year-old female was referred to our Dermatovenerology department with a more than 3-year history of perianal skin lesion. She complained of an erythematous lesion with increasing dimensions associated with pruritus and mucous discharge, despite the topical and systemic application of antivirals and antifungals. She had no weight loss or any other gastrointestinal symptoms. She had no family history of colon cancer, but her father died of a primary carcinoma of the chest wall at the age of 87. She had hypertension and was controlled with one tablet of Olsar plus® per day. Also she suffered from recurrent episodes of infected bartholin cysts. Physical examination showed well-defined eritematous plaque-like lesions with hypopigmentation involving 4 to 6 hour anal margin (in clock hour) and left buttock (approximately 4 x 4 centimeters) (Figure 1). Digital rectal examination of the anus and rectum did not reveal any palpable mass. There was no evidence of inguinal lymphadenopathy. The punch biopsy evidenced EMPD. In the preoperative setting, a colonoscopy was done and showed a sessile polyp of 5 millimeters, 30 centimeters from the anal margin, which histology was benign. Pelvic magnetic resonance exhibited no alterations besides the perianal skin lesion. Mammogram, thorax x-ray, abdominal ultrasound, suprapubic and endocavitary gynecological ultrasound did not evidence any synchronous tumor or metastasis. The case was then referred to the Department of Plastic Surgery and we performed wide local excision (with macroscopic margins of 2 centimeters except near the anal margin) and reconstruction with transposition flap (Figures 2-6). The histological examination of the resected specimen revealed apoorly differentiated primary intraepithelial PPD (immunohistochemistry: CK7+, GCDFP-15+, CK20-, CDX2-, S100-, p40-) (Figures 7-9). The margin near the anus had tumor. We directed the case towards the Department of General Surgery who proceeded to the widening of the inner margin. The tumor distanced 1 milimeter from the resection margin and the colleagues proposed abdominoperineal resection for definitive treatment, which procedure the patient had refused. The multidisciplinary team then proposed local adjuvant radiotherapy (RT).
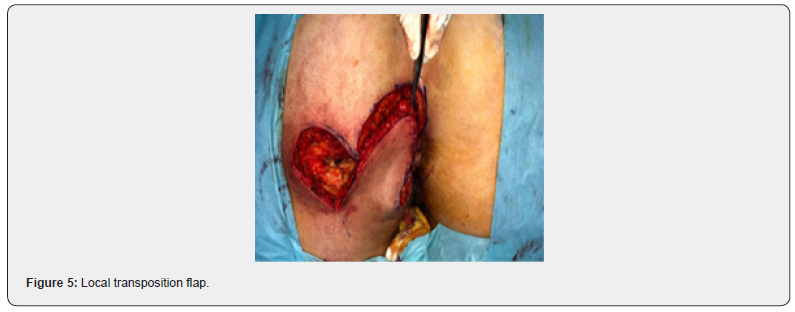
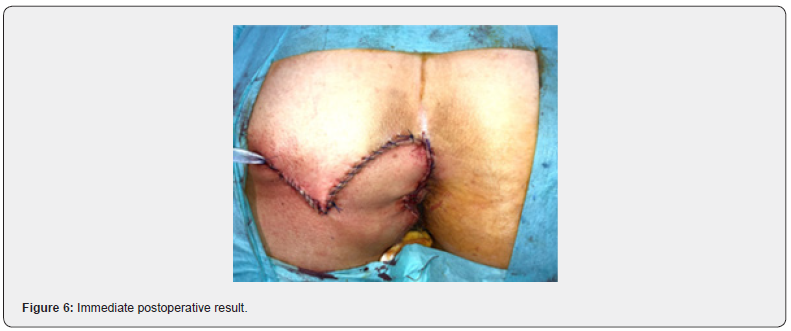
Results
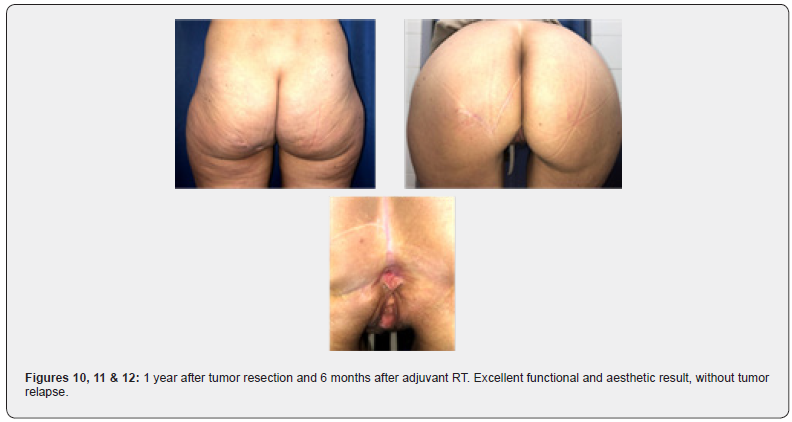
The patient concluded RT 6 months ago. She doesn’t have any complaints and the tumor didn’t relapse. Also the functional and aesthetic result is excellent (Figures 10-12).
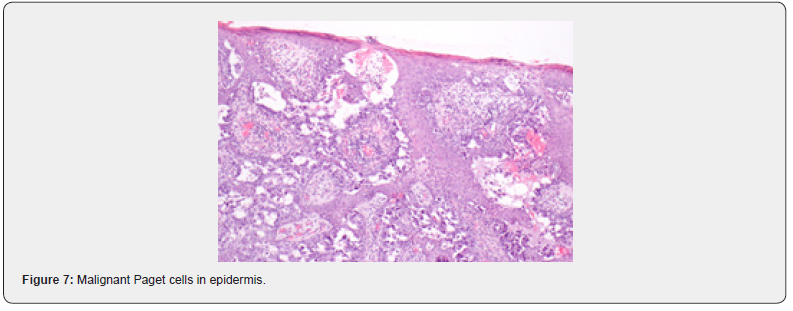
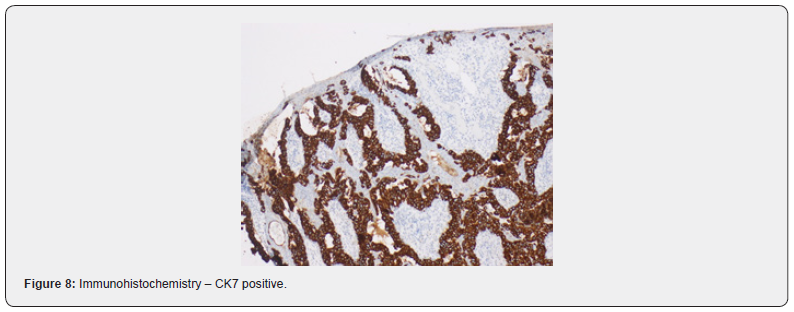
Discussion
EMPD is a rare intraepithelial carcinoma that affects mostly the vulva (65%), the perianal region (20%) and the male genitalia, including scrotum and penis (14%). Nevertheless, it has been reported in other parts of the body, such as axilla, buttocks, eyelids and other apocrine gland rich areas [1,6]. Although it is a disease of the epidermis, it can be invasive and metastasize to distant organs via the lymphatic system [7]. Clinically, lesions of EMPD are insidious and nonspecific and may have many different characteristics. Most frequently, it presents as a wellcircumscribed erythematous plaque or macule with a scaly surface and occasional hyperpigmentation or hypopigmentation. Intense itchiness is very frequent (70% of the cases), and may result in erosion, excoriation and lichenification [1,7]. Despite this, 10% of the patients remain asymptomatic [8]. Differential diagnosis includes many benign conditions, such as eczema, psoriasis and contact dermatitis, but also malignant ones, like malignant melanoma [1]. Because the symptoms are so diverse and the condition being so rare, the diagnosis is often delayed [9,10], like what happened in the reported case. The definitive diagnosis requires a punch biopsy and the specimen should be evaluated with histochemical stains and immunohistochemical markers. The last ones are useful to distinguish primary from secondary EMPD [6]. EMPD lesions have been reported to be associated with internal malignancy (i.e. secondary EMPD) in up to 29% of the cases [11]. That said, after the diagnosis of EMPD, it is recommended to do a thorough review of systems, including a complete physical examination and some specific diagnostic investigations. In the face of a PPD, those diagnostic investigations should include an upper and lower endoscopy with or without a CT scan, a cystoscopy with the possibility of adding an urogram and, if the patient is a woman, a mammogram [1]. Having all the investigation done, it’s time to delineate the treatment course. Because EMPD is so rare, there are no guidelines for its treatment. However, we can base our decision in the likelihood of local or distant recurrence.
Cohen, J. M. et al (2015) defined a series of prognostic factors for patients with EMPD, being the most important ones the depth of invasion and the presence of extracutaneous involvement. That means, invasive lesions (those that get into de reticular dermis) and lesions associated with lymph node or distant metastases have a poor prognosis and require a more aggressive management. To study the lymph node involvement, the sentinel lymph node biopsy (SLNB) is one possibility. Although there are no specific guidelines in this matter, it seems that individuals with clinical involvement of the lymphatic system or in those in whom the disease affects the dermis may benefit from SLNB. There is no evidence that noninvasive lesions benefit from SLNB. In recent years, genetic characteristics of the tumor have become increasingly important as prognostic factors. Studies have shown that lesions with expression of CK20, Ki-67 and cyclin D1 and loss of expression of GCDFP-15 and E-cadherin are more likely to be invasive and associated with metastatic disease [11]. The lesion on the reported case was confined to the epidermis, wasn’t associated with lymph node or distant metastasis and was GCDFP-15+ and CD20-, which means it was a low-risk lesion. Despite the risk stratification of the tumor, surgical excision of the lesion is the standard of treatment. Moreover, RT and chemotherapy can be used as additional adjuvant and neo-adjuvant therapy to provide local control and prevent or treat metastasis [1]. In our case study, and since the lesion was a low-risk one, we tried a surgical approach. Since the tumor distanced 1 millimeter from the inner margin and the patient refused further surgical margin widening, we opted for RT to provide an adequate local control of the disease and relapse prevention. With this approach, as far as the results go, we have been well succeeded.
Acknowledgment
To the Department of Dermatology of the Egas Moniz Hospital, especially to Dr. Cristina Amaro, as assistant doctor of the patient, for the expeditious diagnosis and referral of the case. Thank you Dr. Cristina Amaro and Dr. Leandro Silva for reviewing the article and supporting the collaboration between the different departments.
References
- Davis UC, Claire S, St K, Mba C, Ba AH, et al. (2019) Extramammary Paget disease. Dermatol Online J 25(4): 1-12.
- Pellino G, Sciaudone G, Canonico S, Selvaggi F, Pellino G, et al. (2012) Perianal Paget’s Disease and Malignancies of Lower Hindgut and Anal Canal. J Gastroenterol Hepatol Res 1(1): 1-4.
- Crocker HR (1888) Paget’s disease affecting the scrotum and penis. Trans Pathol Soc L 40: 187-191.
- Darrier J, Couillaud P (1893) Sur un cas de maladie de Paget de la région périnéo-anale et scrotale. Ann Dermatol Syphil 4: 25-31.
- Liao X, Mao W (2014) Perianal Paget’s Disease Co-Associated with Anorectal Adenocarcinoma: Primary or Secondary Disease? Case Rep Gastroenterol 8(2): 186-192.
- Lam C, Funaro D (2010) Extramammary Paget’s Disease: Summary of Current Knowledge. Dermatol Clin 28(4): 807-826.
- Li JW, Ng CM (2014) More than skin deep: Paget’s disease of the perineum. Hong Kong Med J 20(3): 264.e1-e2.
- Shepherd V, Davidson EJ (2005) Extramammary Paget’s disease. BJOG 112(3): 273-279.
- Ito T, Kaku-ito Y, Furue M (2018) Expert Review of Anticancer Therapy The diagnosis and management of extramammary Paget’s disease. Expert Rev Anticancer Ther 18(6): 543-553.
- Kang Z, Zhang Q, Zhang Q, Li X, Hu T, et al. (2015) Clinical and pathological characteristics of extramammary Paget’s disease: report of 246 Chinese male patients. Int J Clin Exp Pathol 8(10): 13233-13240.
- Cohen JM, Granter SR, Werchniak AE (2015) Risk stratification in extramammary Paget disease. Clin Exp Dermatol. 40(5): 473-478.






























