Abstract
Background: Congenital pouch colon (CPC) is a rare variant of anorectal malformation (ARM) where a portion of the shortened colon is replaced by a dilated, pouch-like structure. While relatively common in northern India, its occurrence in sub-Saharan Africa has remained undocumented, often leading to potential misdiagnosis as a simple ARM in resource-limited settings.
Case Presentation: A 54-hour-old Nigerian male neonate presented with failure to pass meconium, progressive abdominal distension, and bilious
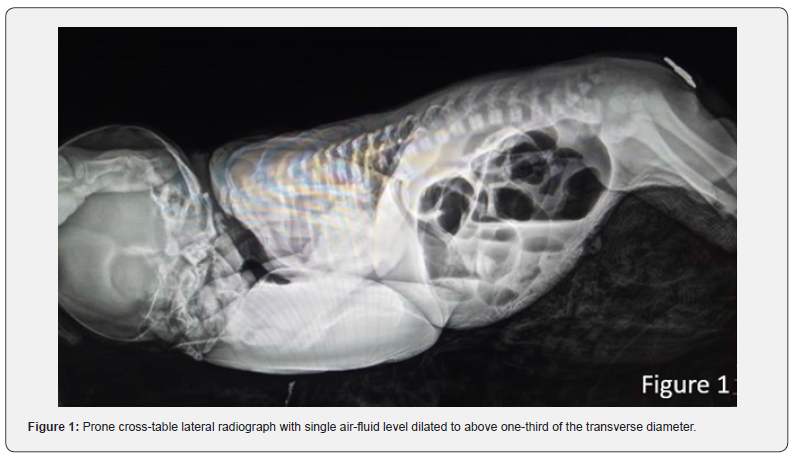
vomiting. Physical examination revealed a tense, distended abdomen and an absent anal opening. Radiographic imaging showed a solitary,
massively dilated bowel loop with a large air-fluid level, raising suspicion of CPC. Exploratory laparotomy confirmed a type 4 CPC, characterized
by a terminal pouch distal to a short segment of normal colon, notably lacking a fistulous connection to the genitourinary tract. The patient
underwent a successful descending colostomy. This represents the first published case of CPC in sub-Saharan Africa.
Conclusion: This case highlights the presence of CPC in the African population and underscores the need for a high index of clinical suspicion when managing high ARMs. Early recognition and surgical intervention are vital for survival, particularly in settings where diagnostic and
specialized surgical resources are constrained.
Keywords: Case report; Congenital pouch colon; Anorectal malformation; Sub-Saharan Africa; Nigeria; Neonatal intestinal obstruction
Introduction
Congenital pouch colon (CPC) is a rare and complex congenital anomaly characterized by a shortened colon that terminates in a dilated, pouch-like structure, typically associated with an anorectal malformation (ARM) [1]. This condition represents a distinct subset of ARM, differing from classical imperforate anus due to the presence of a pouch-like dilatation of the colon, which often communicates with the genitourinary tract via a fistula [2]. First described in 1912 by Spriggs in a specimen at the London Hospital Museum [1,2] CPC has since been recognized as a condition with variable severity and anatomical presentations. Its embryogenesis remains poorly understood, though it is hypothesized to result from a disruption in the caudal migration and separation of the hindgut and urogenital sinus during the 6th to 8th weeks of gestation [3]. Environmental factors, such as maternal iodine or vitamin B deficiency and exposure to pesticides, have been proposed as potential contributors, particularly in regions with high incidence [4].
The anatomical hallmark of CPC is a significantly shortened colon, with the pouch replacing all or part of the normal colonic structure. The pouch typically exhibits abnormal histology, including disorganized muscle layers and absent or rudimentary haustra and taeniae coli [1]. In most cases, the pouch terminates in a fistulous connection to the bladder, urethra, or vagina, though rare variants without fistulae have been reported [5]. The widely accepted classification by Narsimha Rao et al. categorizes CPC into four types based on the length of normal colon proximal to the pouch: type 1 (no normal colon), type 2 (short normal colon), type 3 (normal colon up to the transverse colon), and type 4 (nearnormal colon with a terminal pouch).[3] This classification aids in surgical planning and prognosis, with types 1 and 2 historically more common, though recent trends suggest a shift toward less severe types 3 and 4 [6].
Epidemiologically, CPC is a rare entity with precise incidence and prevalence data lacking due to its sporadic occurrence and underreporting in many regions. The majority of documented cases originate from northern India, where it is considered a regional anomaly, possibly linked to environmental and dietary factors [4]. Sporadic reports have emerged from other countries, including China, the USA, Sweden, Japan, the UK, and Saudi Arabia [1,7,8] but the condition’s global distribution remains poorly mapped. Notably, no cases have been published from sub-Saharan Africa, a gap likely attributable to multiple factors: limited access to diagnostic facilities, scarcity of specialized pediatric surgical care, and low awareness among healthcare providers [5]. In resource-limited settings, CPC may be misdiagnosed as a simple ARM or overlooked entirely, contributing to its apparent rarity in the region.
The clinical significance of CPC lies in its association with neonatal intestinal obstruction, often presenting with failure to pass meconium, abdominal distension, and vomiting. Associated anomalies, including genitourinary, gastrointestinal, vertebral, limb, and cardiac defects, are frequently reported, complicating management [9]. Diagnosis requires a high index of suspicion, as radiographic findings, such as a dilated bowel loop with a large air-fluid level, may be subtle or misinterpreted [6]. Surgical intervention, typically involving colostomy followed by a pullthrough procedure, is the mainstay of treatment, though outcomes depend on the type of CPC, associated anomalies, and available resources [10].
In sub-Saharan Africa, where healthcare infrastructure is often constrained, the burden of rare congenital anomalies like CPC remains largely unquantified. This dearth of data hinders the development of tailored diagnostic and therapeutic strategies. The present case report addresses this knowledge gap by documenting the first known instance of CPC in a sub-Saharan African neonate, highlighting its clinical features, diagnostic challenges, and management in a resource-limited setting. By doing so, it underscores the importance of considering CPC in the differential diagnosis of neonatal ARM across all geographic regions and calls for increased research and awareness in Africa.
This manuscript was prepared following the CARE guidelines (https://www.care-statement.org).
Patient Information
A 54-hour-old term male neonate, born to a 28-year-old primiparous mother of Yoruba ethnicity, was referred to the University of Ilorin Teaching Hospital in Kwara State, Nigeria, for evaluation of suspected congenital anomalies. The infant was delivered vaginally at a local primary health center after an uncomplicated 38-week gestation, with a birth weight of 2.6kg and an Apgar score of 8 and 9 at 1 and 5 minutes, respectively. The mother, a subsistence farmer with no formal education, had attended four antenatal visits at the same facility, where routine ultrasonography at 20 weeks gestation revealed no abnormalities.
There was no maternal history of chronic illness, substance use, or exposure to known teratogens, and the family history was negative for congenital malformations or consanguinity. The neonate was breastfed but had not passed meconium since birth, prompting concern from the family and subsequent referral. On arrival, the infant was noted to have progressive abdominal distension and multiple episodes of bilious vomiting, raising suspicion of an intestinal obstruction.
Clinical Presentation
Upon admission, the neonate appeared irritable but hemodynamically stable, with a heart rate of 140 beats per minute, respiratory rate of 40 breaths per minute, and temperature of 36.8°C. Physical examination revealed a grossly distended abdomen that was shiny and tense, with minimal movement on respiration. Prominent superficial veins and visible peristalsis were observed across the abdominal wall. Palpation was limited by the distension, rendering abdominal organs non-palpable, though no overt masses were appreciated. Percussion elicited a uniformly tympanic note, suggesting significant air accumulation, while auscultation disclosed hyperactive, high-pitched bowel sounds, consistent with an acute obstruction. Examination of the perineum confirmed an absent anal opening, establishing the presence of an anorectal malformation. Additional findings included a palpable left undescended testis, with the right testis descended and normal in size and consistency. No other external congenital anomalies, such as vertebral deformities or limb defects, were evident, and cardiac and respiratory examinations were unremarkable. The constellation of findings—failure to pass meconium, progressive distension, bilious vomiting, and absent anus—pointed toward an anorectal malformation, necessitating urgent diagnostic imaging and intervention.
Diagnosis
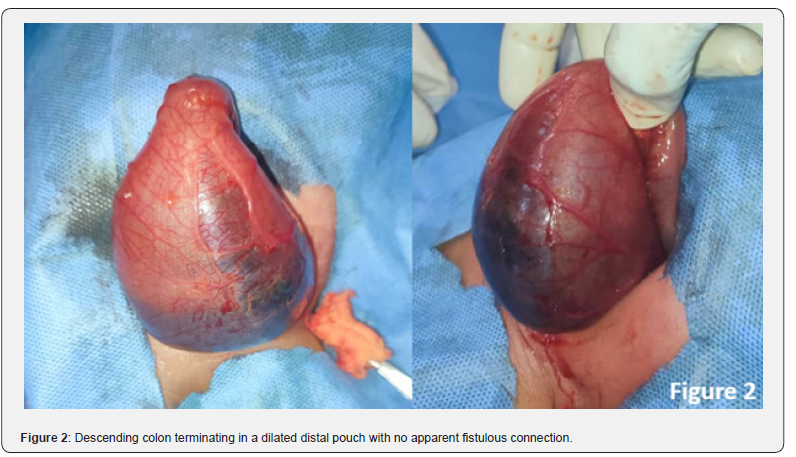
Initial diagnostic workup included a cross-table lateral abdominal X-ray (Figure 1), performed with the infant in the prone position to assess air distribution. The radiograph revealed a high anorectal malformation, characterized by a large, solitary bowel loop occupying the central abdomen, with a single air-fluid level dilated to more than one-third of the transverse abdominal diameter. This finding raised suspicion of congenital pouch colon (CPC), as the extent of dilatation exceeded that typically seen in classical ARM [6]. No free intraperitoneal air or calcifications were noted, ruling out perforation or meconium peritonitis. Following resuscitation with intravenous fluids, nasogastric decompression, and broad-spectrum antibiotics (ampicillin and gentamicin), the neonate was prepared for exploratory laparotomy. Intraoperative exploration confirmed the radiographic suspicion: the descending colon abruptly terminated in a dilated, thick-walled distal pouch, devoid of haustra or taeniae coli, with no fistulous connection to the urinary tract or other structures (Figure 2). The pouch was located distal to a short segment of normal-appearing colon, consistent with type 4 CPC per the Narsimha Rao classification [3]. The bladder and ureters appeared anatomically normal, and no additional intra-abdominal anomalies were identified. The diagnosis of CPC was thus established based on the triad of a shortened colon, pouch-like dilatation, and association with ARM, as delineated by Gupta & Sharma [1] with the absence of a fistula marking this as a rare variant.
Management
The neonate underwent a descending colostomy to relieve the obstruction and allow for growth and further evaluation. The procedure was uneventful, and the postoperative course was stable with no immediate complications. The patient was discharged on postoperative day 7 with a plan for a definitive pull-through procedure at a later date, following nutritional optimization and possible resolution of the undescended testis. The choice of a staged approach was driven by the patient’s age, the complexity of CPC, and resource constraints, including limited access to neonatal intensive care.
Discussion
CPC is a rare entity with an uncertain embryogenesis, often linked to environmental factors such as iodine or vitamin B deficiency, pesticide exposure, and low socioeconomic status, as observed in northern India [4]. Nigeria shares similar environmental and socioeconomic characteristics, including a predominantly agrarian population and widespread pesticide use, suggesting a potential underrecognized burden of CPC in this region.
The Narsimha Rao classification highlights a historical predominance of severe types 1 and 2 CPC, though recent trends indicate a shift toward less severe types 3 and 4, [3] as seen in this case. The absence of a fistula, reported in only 4 cases by Pandey et al. (75% type 2, 25% type 1), [5] underscores the rarity of this presentation. Associated anomalies, such as the undescended testis in our patient, are well-documented, including genitourinary, gastrointestinal, and cardiac defects [1,9].
Preoperative diagnosis of CPC requires a high index of suspicion, as the characteristic dilated bowel loop with an airfluid level occupying over half the abdominal diameter is often overlooked in ARM evaluations [6]. In resource-limited settings like sub-Saharan Africa, diagnostic challenges are compounded by limited imaging modalities and expertise, potentially contributing to underreporting. Management in this case was constrained by the lack of advanced neonatal surgical facilities, necessitating a staged approach rather than a primary pull-through, which is feasible in better-resourced settings [10].
This case contrasts with reports from India, where fistulaassociated CPC predominates, and highlights a unique presentation in Africa. It underscores the need for increased awareness among clinicians and robust epidemiological studies to determine the true prevalence of CPC in this region.
Strengths and Limitations
This case report represents the first documented instance of CPC in sub-Saharan Africa, filling a critical gap in the global epidemiological understanding of this rare condition. The detailed clinical and radiographic documentation provides valuable insights for clinicians in resource-limited settings. However, several limitations warrant acknowledgment. First, the lack of advanced imaging modalities (such as CT or MRI) and histopathological examination of the pouch limits the anatomical characterization. Second, the absence of genetic testing precludes investigation of potential hereditary factors. Third, as a single case report, it cannot establish the true prevalence or incidence of CPC in the African population. Finally, long-term follow-up data are pending, limiting conclusions about outcomes following the planned definitive repair. Despite these constraints, this report underscores the importance of clinical suspicion in diagnosing CPC and highlights the need for multicenter studies in sub-Saharan Africa.
Conclusion
This report documents the first known case of CPC in sub- Saharan Africa, a type 4 variant without fistula in a Nigerian neonate. Early recognition and surgical intervention were critical to the patient’s survival, yet challenges in diagnosis and management reflect broader healthcare limitations. CPC should be considered in all neonates with suspected high ARM, regardless of geographic region, as it may be more prevalent in Africa than previously thought. Further research is essential to elucidate its incidence, risk factors, and optimal management strategies in resource-limited settings.
Statements
Informed consent
Informed consent has been obtained from the patient’s parents or guardian for publication of this case report and accompanying images.
Authorship
All authors attest that they meet the current ICMJE criteria for Authorship.
References
- Gupta DK, Sharma S (2006) Congenital pouch colon. In: Holschneider AM, Hutson JM, (Eds.), Anorectal Malformations in Children: Embryology, Diagnosis, Surgical Treatment, Follow-up. Berlin Heidelberg: Springer-Verlag; pp. 211-222.
- Gupta DK, Sharma S (2007) Congenital pouch colon — Then and now. J Indian Assoc Pediatr Surg 12(1): 5-12.
- Narasimharao KL, Yadav K, Mitra SK, Pathak IC (1984) Congenital short colon with imperforate anus: a definitive surgical procedure. J Pediatr Surg 19: 379-380.
- Singh S, Pathak IC (1972) Short colon associated with imperforate anus. Surgery 71(5): 781-786.
- Pandey V, Panigrahi P, Kumar R (2017) Congenital pouch colon without fistula: our experiences and lessons learned. Asian Journal of Health and Medical Research 3: 1-2.
- Rajiv C, Niyaz AK (2017) Congenital pouch colon. J Indian Assoc Pediatr Surg 22(2): 69-78.
- Arestis NJ, Clarke C, Munro FD, Micallef C, O'Sullivan MJ (2005) Congenital pouch colon (CPC) associated with anorectal agenesis: a case report and review of literature. Pediatr Dev Pathol 8(6): 701-705.
- Chadha R, Bagga D, Mahajan JK, Gupta S (1998) Congenital pouch colon revisited. J Pediatr Surg 33(10): 1510-1515.
- Singh A, Singh R, Singh A (1977) Short colon malformation with imperforate anus. Acta Paediatr Scand 66(5): 589-594.
- Trusler GA, Mestel AL, Stephen CA (1959) Colon malformation with imperforate anus. Surgery 45(2): 328-334.






























