Ehlers Danlos Syndrome with COL5A1 Variant c.3257C>T
Zoe Feld, Sandra Dreike and Robert Wallerstein*
Hawai'i Community Genetics, Kapiolani Medical Center for Women and Children, US
Submission: March 23, 2018; Published: May 11, 2018
*Corresponding author: Robert Wallerstein, Hawai'i Community Genetics, Kapiolani Medical Center for Women and Children, 1319 Punahou Street, Honolulu, HI 98626, USA, Email: robert.wallerstein@kapiolani.org
How to cite this article: Zoe Feld, Sandra Dreike, Robert Wallerstein. Ehlers Danlos Syndrome with COL5A1 Variant c.3257C>T. JOJ Case Stud. 2018; 6(5): 555699. DOI: 10.19080/JOJCS.2018.06.555699
Abstract
Ehlers Danlos Syndrome (EDS) is a connective tissue disorder characterized by genotypic and phenotypic heterogeneity, with joint hypermobility and hyperelasticity of the skin being two of the most common manifestations. An estimated 90% of Classical EDS cases are associated with genetic mutations in COL5A1 or COL5A2. The genetic cause of EDS Hypermobility Type, on the other hand, has been widely regarded as unknown. We present a case of a patient diagnosed clinically with Classical EDS, in whom genetic testing reveals a COL5A1 variant that has been previously reported in the literature only once, in association with EDS Hypermobility Type.
Keywords: Ehlers Danlos; EDS; Cclassical type; Hypermobility type; Genotype-phenotype association; Next Generation Sequencing
Introduction
Ehlers Danlos Syndrome (EDS) is a connective tissue disorder characterized by genotypic and phenotypic heterogeneity, with joint hypermobility and hyperelasticity of the skin being two of the most common manifestations. Over the years, the diverse presentations of EDS have been segregated into descriptive categories, with the Villefranche nomenclature commonly used to distinguish six unique subtypes. Each subtype is delineated by specific sets of manifestations, with major and minor criteria. Classical EDS (cEDS) is implicated by the presence of 3 major criteria: skin hyperextensibility, widened atrophic scars, and joint hypermobility. Associated minor criteria for cEDS include smooth velvety skin, muscle hypotonia, easy bruising, and surgical complications. The hypermobility type (EDS-HT) is associated with 2 major criteria: hyperextensible, smooth, velvety skin and generalized joint hypermobility. EDS-HT minor criteria include recurring joint dislocations and chronic joint or limb pain. While the various EDS subtypes, cEDS and EDS-HT in particular, overlap clinically with one another, the Villefranche nomenclature is still used to guide genetic testing [1]. An estimated 90% of cEDS cases are associated with genetic mutations in COL5A1 or COL5A2. The genetic cause of EDS-HT, on the other hand, has been widely regarded as unknown. Next Generation Sequencing (NGS) is a method of genetic sequencing that captures a broader spectrum of mutations than previously used methods and it is rapidly being incorporated into clinical practice. As NGS increases the identification of gene mutations associated with EDS, reconsideration of the classification of EDS subtypes may be warranted and this may enhance our understanding of this complex and multifaceted disorder.
Case Presentation
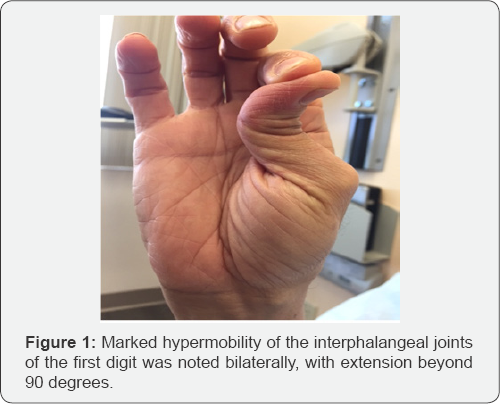
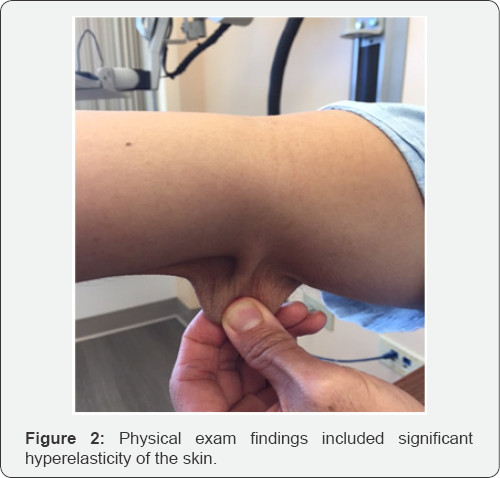
We present a case of a 53 year old japanese American man with a medical history significant for fibromyalgia, chronic fatigue syndrome, borderline lupus laboratory testing, and alopecia areata. He presented for genetics evaluation seeking an underlying cause for his autoimmune concerns, citing that his family history is significant for multiple autoimmune conditions. Review of his records showed a dilated aortic root detected by echocardiogram two years prior. While no genetic testing was offered for his autoimmune concerns, further assessment for aortopathy was undertaken, with consideration given to connective tissue disorders. In depth interview revealed additional history of branch retinal vein occlusion when he was in his thirties, as well as family history significant for branch retinal vein occlusion in his mother and a maternal uncle. History included chronic bilateral foot pain, back pain, and shoulder pain, with distant history of multiple shoulder dislocations and two ACL tears. On physical exam, skin was hyperelastic and joints, particularly thumbs, were hypermobile. His Beighton hypermobility score was 6/9.
Based on these findings, genetic testing for Ehlers-Danlos Syndrome and other conditions that may present with joint hypermobility, connective tissue or bone fragility, or aortopathy was performed. Results of a panel using sequencing and deletion/duplication analysis of 14 genes (ADAMTS2, ATP7A, CHST14, COL1A1, COL1A2, COL3A1, COL5A1, COL5A2, CRTAP, FKBP14, FLNA, P3H1, PLOD1, SLC39A13) revealed a heterozygous variant of uncertain significance in COL5A1 (c.3257C>T:p.Ala1086Val) (Figure 1 & 2).
Discussion
The COL5A1 variant (c.3257C>T) identified in our patient has been previously reported in association with EDS, Hypermobility Type phenotype. Weerakkody et al. [2] initially recognized this as a novel pathogenic variant in a 35 year old female with a Beighton score of 6/9 who fulfilled 2 major criteria of the Villefranche criteria for a classification of Hypermobility Type. While she was noted to fulfill 3 major Classical Type criteria to a mild degree, her pres
Hypermobility Type phenotype. Weerakkody et al. [2] initially recognized this as a novel pathogenic variant in a 35 year old female with a Beighton score of 6/9 who fulfilled 2 major criteria of the Villefranche criteria for a classification of Hypermobility Type. While she was noted to fulfill 3 major Classical Type criteria to a mild degree, her presentation of EDS was categorized as Hypermobility type.
Mutations in COL5A1 and COL5A2 comprise the etiology of an estimated 90% of cases of Classical Ehlers Danlos Syndrome (cEDS) [3]. Yet the underlying genetic changes responsible for EDS-HT have been reported as unknown and not yet classified. Before the availability of Next Generation Sequencing, patients with a clinical diagnosis of EDS-HT may not have undergone genetic testing. Now, genotyping has shed more light on the limitations of the Villefranche classification. The c.3257C>T mutation underlying this patient's EDS has been reported in association with similar phenotypic presentation in two patients who were ultimately each clinically diagnosed with distinct EDS types. While clinical diagnosis and segregation by phenotypic presentation continues to yield useful information for prognostication and management, as NGS becomes more widespread and a greater number of novel variants are reported in the literature with their associated presentation and sequelae, we can look forward to the potential for review and revision of EDS classification with consideration of genotype. Revising the framework for EDS classification can enrich the geneticist's ability to provide enlightening anticipatory guidance and screening recommendations for patients living with EDS.
References
- Castori M (2012) Ehlers-danlos syndrome, hypermobility type: an under diagnosed hereditary connective tissue disorder with mucocutaneous, articular, and systemic manifestations. ISRN Dermatol pp. 1-22.
- Weerakkody RA, Vandrovcova J, Kanonidou C, Mueller M, Gampawar P, et al. (2016) Targeted next-generation sequencing makes new molecular diagnoses and expands genotype-phenotype relationship in Ehlers-Danlos syndrome. Genet Med 18(11): 1119-1127.
- Symoens S, Syx D, Malfait F, Callewaert B, De Backer J, et al. (2012) Comprehensive molecular analysis demonstrates type V collagen mutations in over 90% of patients with classic EDS and allows to refine diagnostic criteria. Hum Mutat 33(10): 1485-1493.






























