Immunohistochemical Features of the Basal Layer of the Endometrium: Morphological Aspects
Khaskhachykh Dmytro*, Potapov Valentin and Poslavska Oleksandra
Dnipro State Medical University, Dnipro, Ukraine
Submission: July 27, 2023; Published: August 03, 2023
*Corresponding author: Khaskhachykh Dmytro, Dnipro State Medical University, Dnipro, Ukraine
How to cite this article: Khaskhachykh D, Potapov V, Poslavska O. Immunohistochemical Features of the Basal Layer of the Endometrium: Morphological Aspects. J Gynecol Women’s Health 2023: 25(4): 556168. DOI: 10.19080/JGWH.2023.25.556168
Abstract
Background: The article examines the morphological and immunohistochemical characteristics of all layers of the endometrium in women of reproductive age with endometrial hyperplasia without atypia in comparison with secretory endometrium, who had abnormal uterine bleeding (AMB).
Purpose: To study the morphological and immunohistochemical features of the structure of all layers of the endometrium, divided into functional and non-functional zones, which may have an impact on the development of endometrial hyperplasia.
Patients and Methods: The study was performed on endometrial morphological material obtained by diagnostic biopsy from 21 women with abnormal uterine bleeding (ABB) in the gynecological department of the Dnipro Clinical Hospital No. 9 in Dnipro during 2022-2023. The criteria for inclusion in the studied cohort of patients were: the age of women from 32 to 45 years (38.4±2.55), the presence of endometrial hyperplasia without atypia, or secretory changes of the endometrium according to the results of histological examination, the exclusion criteria were: the presence of inflammatory diseases of the pelvic organs , tumor pathology of the uterus and ovaries, endometriosis of the uterus, severe somatic pathology, any form of endocrinopathy and metabolic syndrome. The average body mass index was 27.83±1.96 kg/cm2. The study included morphological and immunohistochemical (IHC) research methods. Endometrial tissue was obtained by endometrial curettage, which was performed in women with abnormal uterine bleeding. Primary antibodies to ER (sp1, RTU), PgR (YR85, 1:200), CK 7 (sp1, RTU), CK8 (sp1, RTU), p53 (E247, RTU) and the UltraVision Quanto imaging system (LabVision). Statistical processing of the obtained results was carried out using the Office 365 A1 for faculty software No. 1003BFFD8C8E8B0D. Arithmetic mean (M) and standard deviation (SD) values were calculated. The non-parametric test χ² was used to compare the distribution of features in groups. The difference was considered statistically significant at p<0.05 (95% significance level).
Results: Analysis of the expression of the biomarkers ER, PgR, Ki67, CK7 and CK8 in women with endometrial hyperplasia and secretory endometrium is important for understanding the physiological and pathological processes that occur in women. The expression of these biomarkers can help establish the diagnosis of endometrial hyperplasia and classify it into subtypes depending on the characteristics of the cells and their activity. The expression of these biomarkers may be a prognostic factor, helping to assess the prognosis for patients with endometrial hyperplasia. For example, high expression of ER and PgR may indicate a more favorable prognosis and a more sensitive response to hormone therapy, which may help in planning treatment and monitoring its effectiveness. Ki67 expression is an indicator of cell proliferation and activity. High expression of Ki67 may indicate increased cellular activity and rapid cell division, which may be characteristic of endometrial hyperplasia. While the expression of Ki67 decreases in the secretory phase of the unchanged normal endometrium, but sometimes such increased expression can reveal the presence of active proliferative processes. In addition, the expression of CK7 and CK8 may be associated with an increased number of epithelial cells. They are found in epithelial cells of various organs, including the endometrium. Studying the expression of these cytokeratins helps reveal the epithelial nature of the cells and to distinguish them from other cells. Thus, analysis of the expression of ER, PgR, Ki67, CK7 and CK8 in endometrial hyperplasia and normal endometrium allows a deeper understanding of the processes of proliferation, differentiation and function of endometrial cells. These data have important diagnostic value for endometrial hyperplasia, the degree of determination of its atypia and the prediction of treatment results.
Conclusion: 1. Analysis of the expression of steroid hormone receptors in different layers of the endometrium of the secretory phase and endometrium with hyperplasia without atypia found a statistically significant difference only in the expression of ER of all layers (surface, functional and basal) with the advantage of the number of samples with high expression of ER in the stroma of samples of endometrial hyperplasia without atypia (p<0.05), on the other hand, such a difference was not found for PgR, despite the trend of a decrease in the basal parts of the endometrium with hyperplasia without atypia and secretory endometrium in the percentage of PgR positive immunoreactive cells to the complete absence in the epithelium of the glands (p>0, 05).
2. The proliferative activity of endometrial cells according to the Ki-67 marker increased from superficial to basal parts and showed a statistically significant difference between endometrial hyperplasia samples without atypia and secretory endometrium (p<0.05).
3. Cytokeratin markers CK8 and CK7 showed a significant difference in expression between themselves: SC8 remained at a constant level of expression, regardless of the depth of the endometrial glands - revealed an organ-specific expression for the endometrium, while SC7, on the contrary, was most expressed in the luminal highly differentiated epithelial of the surface layers and was lost in the basal/stem cells of the deep layers of the endometrium, but a statistically significant difference between secretory endometrium and endometrial hyperplasia without atypia was not found (p>0.05).
Keywords: Endometrium; Receptors; Endometrial hyperplasia; Immunohistochemistry; Estrogen receptors; Progesterone receptors; ER; PgR; Ki67; CK7; CK8
Abbreviations: AUB: Abnormal Uterine Bleeding; HE: Endometrial Hyperplasia; IHC: Immunohistochemically
Introduction
The human endometrium is a dynamic tissue that has a high regenerative capacity after cyclic shedding, namely menstruation. Its changes depending on the phases of the menstrual cycle (i.e. proliferative phase, secretory phase, and desquamation) play a crucial role in the implantation of fertilized eggs. In addition, “endometrial-related diseases” such as adenomyosis, endometriosis, endometrial hyperplasia, and endometrial cancer arise due to its high intrinsic regenerative capacity and affect women’s lives from puberty to menopause [1].
Endometrial hyperplasia (HE) without atypia is a benign pathology without significant somatic genetic changes, caused by extensive estrogen exposure, which is not balanced by the protective effect of progestins. When physiological progesterone levels are restored or therapeutic progestins are used, hyperplastic changes regress, and in most cases, the endometrium becomes healthy again [2]. Spontaneous regression can occur if the hormonal background is corrected to a normal state (correction of obesity, restoration of ovulation, etc.); however, progestagen therapy has a higher cure rate (89-96%) than observation alone (74.2-81%) [3,4].
Histologically, EH defines abnormal proliferation of endometrial glands as having a greater ratio of glands to stroma than in healthy proliferative endometrium, but without invasion of the endometrial stroma. The diagnosis should be based on the histological evaluation of a tissue sample obtained during endometrial biopsy, scraping, or hysterectomy. This distinction is particularly important because the clinical management of these two conditions differs depending on the presence or absence of nuclear atypia. It is this approach to diagnostics that forms the basis for the proper treatment of this disease [5].
A very significant issue for assessing spontaneous regression, further proliferation, and adequate response to pathogenic therapy, often with the use of progestins, is the study of cellular biomarkers and the expression of sex hormone receptors. Because the morphological diagnosis does not make it possible to solve these tasks. At present, the expression of numerous various biomarkers, which are potential candidates for solving issues related to EH, has been studied, but the problem of predicting the course of endometrial hyperplasia remains quite debatable and relevant [6].
Studying the expression of steroid hormone receptors, organ-specific cytokeratin markers, and the proliferation index in different layers of the endometrium is significant for understanding the mechanisms of proliferation and regeneration in the normal menstrual cycle, as well as in its hyperplasia. The need to use biomarkers is not so much for diagnosing types of hyperplasia as for predicting the possibility of spontaneous regression of the disease or an adequate response to pathogenic therapy, depending on the characteristics of cells and their activity [7-9].
Goal
To investigate the morphological and immunohistochemical features of the structure of all layers of the endometrium with the study of the expression of markers ER, PgR, Ki67, CK7 and CK8, with a division into functional and non-functional zones, which may have an impact on the development of endometrial hyperplasia.
Materials and Methods
The study was performed on endometrial morphological material obtained by diagnostic biopsy by curettage from 21 women with abnormal uterine bleeding (ABB) in the gynecological department of the Dnipro Clinical Hospital No. 9 in Dnipro during 2022–2023. The criteria for inclusion in the studied cohort of patients were: the age of women from 32 to 45 years (38.4±2.55), the presence of endometrial hyperplasia without atypia (14 observations), or secretory changes of the endometrium (7 observations) according to the results of histological examination, Exclusion criteria were: the presence of inflammatory diseases of the pelvic organs, tumor pathology of the uterus and ovaries, endometriosis of the uterus, severe somatic pathology, any form of endocrinopathy and metabolic syndrome. The average body mass index was 27.83±1.96 kg/cm2.
Morphological research method. Endometrial samples, fixed in 10% buffered formalin and embedded in paraffin, were taken from the archives of the Dnipro Clinical Hospital No. 9. Paraffin sections of 4-5 μm were obtained on a Microm HM-340 microtome and stained according to the standard method with hematoxylin and eosin (G-E) [2]. Microscopy was carried out using a ZEISS “Primo Star” light microscope (objectives ×10, ×20, ×40).
Immunohistochemical research method (IGH). Paraffin sections were placed on Super Frost Plus adhesive slides. After deparaffinization, rehydration, temperature unmasking of antigens, and inhibition of endogenous peroxidase activity, sections were incubated with primary antibodies in moist chambers at a temperature of 23-250C for 30 minutes. Primary monoclonal antibodies ER (sp1, RTU), PgR (YR85, 1:200), CK 7 (sp1, RTU), CK8 (sp1, RTU), p53 (E247, RTU) and Ultra Vision Quanto imaging system (Lab Vision) were used. To identify the reaction, a solution of the chromogen 3-diaminobenzidine tetrachloride (DAB) (Quanto, Lab Vision) was applied under the control of a microscope for 20 seconds to 3 minutes, with a manifestation in the form of a brown color, then it was additionally stained with Mayer’s hematoxylin for 1-3 minutes. The subsequent dehydration and inclusion in the balm was carried out according to standard methods [10].
According to the recommendations of Antunes A. et al. (2014), ER and PR expression was evaluated in the stroma and glandular epithelium of endometrial tissues using a semiquantitative method of nuclear reaction through analysis of the percentage of stained cells, intensity of nuclear staining, and final score. Counting was carried out in 100 cells of different fields of view under a × 40 objective. The percentage of stained cells was assessed visually and classified as follows: grade 0, no staining; grade 1, <1% staining; degree 2, 1-10% staining; grade 3, 11-33% coloration; 4th degree, 34-66% coloring; and grade 5, >66% staining. Regarding nuclear staining intensity, staining was classified as follows: grade 0, negative; 1st degree, weak reaction; 2 degree, reaction of medium degree; and grade 3, intense reaction. The sum of positivity and intensity resulted in a final score ranging from 0 to 8 [10].
According to the recommendations of Shevra C. R. et al. (2015) Ki-67 proliferation index, was divided into three groups, including low (≤15%), medium (16-30%) and high (>30%) [6]. Evaluation of cytoplasmic staining by cytokeratins CK7 and CK8 was carried out on a semi-quantitative scale according to the number of stained cells: 0 - negative reaction, 1+ - up to 30% of stained cells, 2+ - 31-60% of stained cells, 3+ - more than 60% of stained cells, any what intensity A Zeiss Primo Star microscope camera - Axiocam ERC 5s with licensed ZEN 2 blue edition software was used for digital microscopy.
Statistical processing of the obtained results was carried out using Office 365 A1 for faculty software No. 1003BFFD8C8E8B0D. Arithmetic mean (M) and standard deviation (SD) values were calculated. To compare the distribution of features in groups, the non-parametric test χ² (xi-square) was used. The difference was considered statistically significant at p<0.05 (95% significance level).
Research Results
Analysis of samples of the secretory endometrium showed that the deep, or basal, layer, which covers the deepest part of the endometrial glands, responds only weakly to estrogenic stimulation during the menstrual cycle and is completely insensitive to progesterone. This is very clearly visible on the secretory endometrium during immunohistochemical examination (IGH) with markers for estrogen and progesterone receptors (Figure 1, h-i). Some of these glands showed a tendency to invade the adjacent myometrium, the so-called phenomenon of adenomyosis, but retained the features of the basal layer. The stromal of the basal layer of the endometrium was dense and consisted of small cells often positive for ER and PgR with poor cytoplasm, contrasting with the functional secretory endometrium (Figure 1, g-l). This layer formed a non-functional zone of the endometrium adjacent to the myometrium (Figure 1, g, j), from which regeneration occurs after each menstrual bleeding, and is typically called the “area of reserve cells” in the literature. The surface (compact) layer and the middle (spongy) layer together form the so-called functional zone, which occupies approximately 2/3 of the height of the endometrium (Figure 1, a-f). As shown by immunohistochemical studies, during the luteal phase, the epithelial of the glands in these layers react slightly differently to hormonal stimulation. The surface layer, which includes the necks of the endometrial glands and the luminal epithelium, did not have such convoluted glands as in the spongy layer, demonstrating the number of positive intranuclear IHC reactions with PgR significantly more in stromal cells, compared to the glandular epithelium, a ratio of 8:1 (Figure 1, b), but the ratio of IGH reactions with ER in the stroma and glands is different (1:2), that is, the glands can respond to estrogen stimulation more actively than the stroma (Figure 1, c). In the middle layer, which has a developed vascular system and the most tortuous glands in the secretion phase, the hormonal activity of the stroma and glands was slightly equalized, for PgR this ratio became 5:1, and for ER it generally approached 1:1 (Figure 1, e, f).
Endometrial hyperplasia without atypia showed higher levels of estrogen receptor expression in the functional layer compared to secretory endometrium, not lower than categories 7-8, especially for the stroma, although the core glands often showed >66% stained cells (grade 5) with high staining intensity (degree 3) a total of 8 (Table 1).
For basal sections, increased ER expression also favored hyperplasia, and a statistically significant difference in ER expression was found (p<0.05). Surface sections of EH and secretory endometrium maintained the same trend of expression of estrogen receptors with a functional layer, but with a smaller difference in expression in glands and luminal epithelium (Figure 1).
From the data in table 1, it can be seen that the expression of estrogen receptors (ER) in different layers of the endometrium differs depending on the type of endometrium. It should be noted that in the basal and functional layers of the endometrium with hyperplasia without atypia, a rather high expression of ER was observed in the stroma, while the expression of ER was much lower in the epithelial cells. This may indicate that the stroma is significantly more active in response to the hormonal signal of estrogen, regardless of the level of location, while the decrease in the degree of differentiation of the epithelial cells of the basal layer (endometrial stem cells) is primarily associated with a change in the hormonal status.
In the secretory endometrium, higher ER expression was also observed in the stroma compared to ER expression in epithelial cells. This may indicate that the stroma responds to the hormonal signal of estrogen and may be active during the secretory phase of the cycle, epithelial cells may be less sensitive to estrogen, which is characteristic of normal cycle functions.
The expression of progesterone receptors significantly decreased depending on the level of location - the highest percentage of positively stained stromal nuclei demonstrated the functional layer of the secretory endometrium (categories 7-8), hyperplastic endometrium had lower rates of expression of stromal cells (categories 5-6), and even more epithelial cells (categories 4-5), but no statistically significant difference was found for the functional layer in the expression of PgR (p>0.05). The surface sections of the EH and the secretory endometrium maintained the same trend of progesterone receptor expression with the functional layer, but with a smaller difference in the glands and luminal epithelium (Figure 1).


Note. EH - endometrial hyperplasia, ER - estrogen receptors, p - establishing the reliability of intergroup ties according to the quantitative distribution was carried out by the non-parametric χ² test.
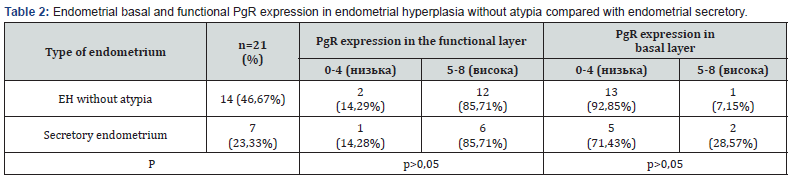
In the basal parts of the EH and the secretory endometrium, the intensity and percentage of immunoreactive cells significantly decreased to complete absence in the glands (categories 2-3) and slightly remained in the stroma (categories 4-5), the majority of PgR expression was observed towards the secretory endometrium, but there was a statistically significant difference in the expression of PgR was not found (p>0.05), (table 2).
Note. EH - endometrial hyperplasia, ER - estrogen receptors, p - establishing the reliability of intergroup ties according to the quantitative distribution was carried out by the non-parametric χ² test.
From the data in table 1, it can be seen that the expression of estrogen receptors (ER) in different layers of the endometrium differs depending on the type of endometrium. It should be noted that in the basal and functional layers of the endometrium with hyperplasia without atypia, a rather high expression of ER was observed in the stroma, while the expression of ER was much lower in the epithelial cells. This may indicate that the stroma is significantly more active in response to the hormonal signal of estrogen, regardless of the level of location, while the decrease in the degree of differentiation of the epithelial cells of the basal layer (endometrial stem cells) is primarily associated with a change in the hormonal status.
In the secretory endometrium, higher ER expression was also observed in the stroma compared to ER expression in epithelial cells. This may indicate that the stroma responds to the hormonal signal of estrogen and may be active during the secretory phase of the cycle, while epithelial cells may be less sensitive to estrogen, which is characteristic of normal cycle functions.
The expression of progesterone receptors significantly decreased depending on the level of location - the highest percentage of positively stained stromal nuclei demonstrated the functional layer of the secretory endometrium (categories 7-8), hyperplastic endometrium had lower rates of expression of stromal cells (categories 5-6), and even more epithelial cells (categories 4-5), but no statistically significant difference was found for the functional layer in the expression of PgR (p>0.05). The surface sections of the EH and the secretory endometrium maintained the same trend of progesterone receptor expression as the functional layer, but with a smaller difference in the glands and luminal epithelium (Figure 1).
In the basal parts of the EH and the secretory endometrium, the intensity and percentage of immunoreactive cells significantly decreased to a complete absence in the glands (categories 2-3) and slightly remained in the stroma (categories 4-5). The majority of PgR expression was observed in the secretory endometrium, but a statistically significant difference in the expression of PgR was not found (p>0.05), (table 2).
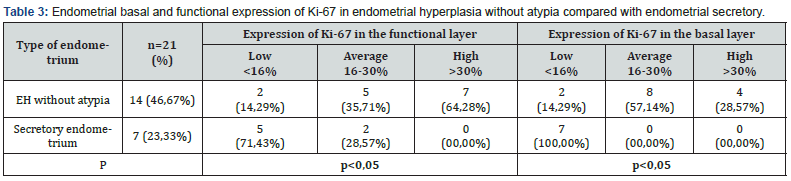
Analysis of the proliferation index by Ki-67 in different layers of the endometrium showed a significant difference between EH samples and secretory endometrium, towards a hyperplastic process (Figure 2). From the data in Table 2, it can be seen that the functional layer of the secretory endometrium only in 28.57% of cases demonstrated Ki-67 expression at the level of 16-30%, compared to EH, where half of 7 out of 14 cases had a high Ki- 67 expression of more than 30% positive immunoreactive cells (p<0.05). In the basal layer, this difference was even greater, as a result of the absence of proliferative activity in the secretory before desquamative stage of the cycle, and the constantly active proliferation of the basal parts during the hyperplastic process without atypia (table 3).
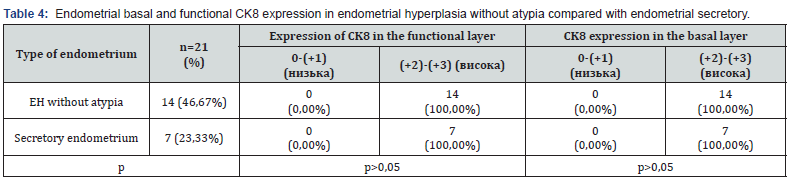
The expression of the marker CK8 demonstrated organspecific expression of high intensity for the epithelium of the endometrial glands in all layers of the endometrium (Figure 2, table 4), regardless of the stage of proliferation or secretion (all p>0.05). With the loss of hormonal status in the basal/stem cells, the specific cytokeratin skeleton with SC8 inherent in this localization still remains. This fact allows the marker CK8 to be used as diagnostic in case of transformation of EH without atypia into atypical EH and subsequent endometrial carcinoma with possible extratumoral spread for verification of the primary source.

Note. EH – endometrial hyperplasia, ER – estrogen receptors, p – reliability of intergroup relationships by quantitative distribution was established by a nonparametric criterion χ².
Note. EH – endometrial hyperplasia, ER – estrogen receptors, p – reliability of intergroup relationships by quantitative distribution was established by nonparametric criterion χ².
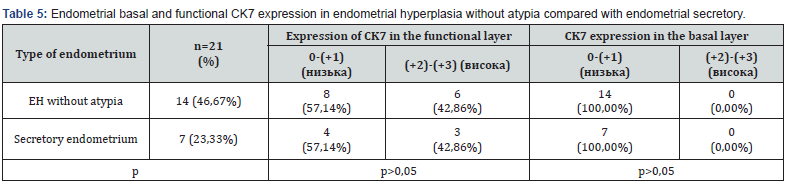
Unlike CK8, another cytokeratin, ck7, showed its positivity mostly in the highly differentiated luminal epithelium of the surface layers and glands of the functional layer, instead, it was lost in the direction from the surface to the basal parts, which was most characteristic of EH (Figure 2). Thus, the lowest rate of CK7 positive cells was found in the basal layer of EH without atypia, but no statistically significant difference was found between the secretory endometrium and EH without atypia in the expression of SK7 (all p>0.05) (table 5).
Note. EH – endometrial hyperplasia, ER – estrogen receptors, p – reliability of intergroup relationships by quantitative distribution was established by nonparametric criterion χ²
Discussion of Research Results
Expression (ER) in different layers of the endometrium in EH without atypia manifested itself in the following way. In the basal and functional layers of the endometrium with EH without atypia, the expression of ER in the stroma spontaneously increases (100.00%); that is, no expression of ER was detected in the epithelial cells (0.00%). This may indicate that the stroma actively responds to the hormonal signal of estrogen, while epithelial cells do not show sensitivity to estrogen. In the secretory endometrium, ER expression in the stroma spontaneously increases (71.43%), compared to ER expression in epithelial cells (28.57%).
This may indicate that the stroma reflects the hormonal signal of estrogen and may be active during the secretion phase. On the other hand, epithelial cells may be less sensitive to estrogen, which is characteristic of a normal menstrual cycle. The results of the analysis show that ER expression can be seen in different types of endometrium. The high expression of ER in the stroma may be associated with the active effect of estrogen on stromal cells, which may be involved in its functional characteristics. On the other hand, the absence of ER expression in epithelial cells may be specific for EH without atypia, which may indicate changes in cellular regulation and endometrial functions in that condition p < 0.05 indicates statistical significance of differences between groups. From the above data, it is clear that the expression of progesterone receptors (PgR) also differs in different endometrial spheres and depends on the endometrium. With EH without atypia in the basal and functional layers of the endometrium, with EH without atypia, the expression of PgR in the glands spontaneously increases (85.71%), which may indicate the activity of progesterone receptors in epithelial cells. In the stroma of the endometrium, the expression of PgR is low (14.29%). This may indicate that stromal cells may be less sensitive to progesterone. With secretory endometrium, the expression of PgR in the glands spontaneously increases (100.00%), which makes it possible to detect the proactivity of progesterone receptors in epithelial cells. At the same time, the expression of PgR in the stroma is low (14.29%), which may be related to the fact that stromal cells are less sensitive to progesterone. The obtained results confirmed that the expression of PgR can be changed in different types of endometrium. High expression of PgR in glands with EH without atypia and secretory endometrium may be associated with the proactivity of progesterone receptors in epithelial cells, which is characteristic of the functionally active layer of the endometrium. On the contrary, low expression of PgR in the stroma may indicate a lower sensitivity of stromal cells to progesterone compared to epithelial cells of the functional layer. The value of p < 0.05 indicates the static significance of the values between the groups, which will confirm the statistical weight of the obtained results.
Analysis of Ki67 biomarker expression in endometrial hyperplasia without atypia and in normal secretory endometrium allows studying the level of cell proliferation in the conditions and identifying possible differences. In EH without atypia, an increased level of Ki67 expression is found. The biomarker Ki67 is an indicator of cell proliferation and cell division activity. Increased expression of Ki67 correlates with increased proliferation of endometrial cells in a proper state. This may be due to increased endometrial screening after menstruation and a disturbance in the balance of cell proliferation and apoptosis. In normal secretory endometrium, expression of Ki67 usually decreases during the secretory phase of the menstrual cycle. This is due to the fact that cell proliferation in this state is limited and occurs in accordance with processes such as the preparation of the endometrium for the implantation of a fertilized egg. In a normal state, the endometrium goes through different phases of the cycle, and the expression of Ki67 corresponds to the level of proliferation according to the phases of the menstrual cycle.
Thus, analysis of Ki67 expression allows us to detect a difference in cell proliferation between endometrial hyperplasia without atypia and normal secretory endometrium. High expression of Ki67 in endometrial hyperplasia causes active processes of cell proliferation, while low expression of Ki67 in normal endometrium indicates its physiological state. These data can be useful for diagnosis and detection of pathological changes of the endometrium. SC7 (cytokeratin 7) is a protein that belongs to the family of cytokeratins, which include intermediate filaments in the cellular cytoskeleton. Cytokeratins are important structural proteins that cause mechanical injury and damage to the cytoskeleton. CK7 is typical for epithelial cells, in particular, it is found in cells of some epithelial tissues, such as the epithelium of the lungs, liver, kidneys, and gall bladder.
CK7 (cytokeratin 7) is a protein that belongs to the family of cytokeratins, which include intermediate filaments in the cellular cytoskeleton. Cytokeratins are important structural proteins that cause mechanical injury and damage to the cytoskeleton. CK7 is typical of epithelial cells; in particular, it is found in the cells of some epithelial tissues, such as the epithelium of the lung, liver, kidney, gall bladder, and endometrium. The role of SK7 includes what can be considered a marker for the detection and characterization of different types of cancer tumors. For example, CK7 expression may aid in tumor differentiation, progression, and metastasis. In endometrial hyperplasia and in normal conditions, SK7 is expressed in endometrial epithelial cells. SK7 expression refers to the typical characteristics of epithelial cells and can be used for the differential diagnosis of endometrial hyperplasia and normal endometrium. In endometrial hyperplasia, which is a precancerous condition, there is an increase in the number of endometrial epithelial cells. SK7 is found in epithelial cells, even with increased numbers. Normally, during functional observations of the endometrium, the expression of SK7 in epithelial cells is also observed. The functional zone is a layer of the endometrium that undergoes cyclical changes depending on the menstrual cycle of a woman. In this case, there is a regular renewal of cells expressing CK7, which remains one of the typical markers of epithelial cells. The basal layer of the endometrium responds weakly to estrogen stimulation and is insensitive to progesterone, which is clearly visible in an immunohistochemical study with markers for estrogen and progesterone receptors. In endometrial hyperplasia and in normal conditions, CK7 continues to be expressed in the basal osseous endometrium. This suggests that even with increased cellular activity and changes in tissue architecture, typical epithelial cells with the presence of SK7 are present in the basal cells. It is also important to note that SK7 is also expressed in functioning endometrial disease, both in hyperplasia and normal. CK7 expression is a characteristic feature of epithet 1. Endometrial hyperplasia without atypia: in the basal dimension of the endometrium with hyperplasia, CK7 is expressed in all samples (100%), which is a massive increase in the number of epithelial cells. At the same time, the functional state of SC7 is also observed in all cases (100%), of which 71.43% have high expression, and 28.57% have a decrease. This can be an observation of structural changes in the endometrium, which are carried out through the processes of cell proliferation and differentiation, which are characteristic of this phase of the menstrual cycle. D B In the secretory endometrium, in its basal layer, SK7 expression was also observed in all samples (100%). In the manifestation of SK7, it was also observed in all samples (100%), in 85.71% of manifestations, and in 14.29% - high. Such an indicator can cause a higher level of cell proliferation during the functional examination of the normal endometrium compared to hyperplasia without atypia. Thus, the analysis of SK7 expression allows us to detect differences in the processes of cell proliferation and differentiation between endometrial hyperplasia without atypia and normal endometrium. These data coincide with the findings of other authors, are important for a deeper identification of pathophysiological mechanisms associated with endometrial hyperplasia, and also include additional markers for the diagnosis and prognosis of this condition [2].
In the functional layer of the endometrium with GE, SK7 expression was observed in all cases (100%), but of them, 71.43% had high expression, and 28.57% - low expression. This may be due to structural changes in the endometrium, which are carried out by the processes of cell proliferation. Examination of the secretory endometrium showed that CK7 expression was also observed in all samples (100%). In the manifestations of CK7, it is also observed in all samples (100%), and in 85.71% it was average, and in 14.29% it was high. Such an indicator can cause a higher level of cell proliferation during the functional examination of a normal endometrium compared to GE without atypia. Thus, the analysis of CK7 expression allows us to detect differences in the processes of cell proliferation and differentiation between endometrial hyperplasia without atypia and normal endometrium. These data are important for the deeper identification of pathophysiological mechanisms associated with endometrial hyperplasia, and also include additional markers for the diagnosis and prognosis of this condition.
Analysis of the expression of the biomarkers ER, PgR, Ki67, CK7 and CK8 in women with endometrial hyperplasia and secretory endometrium is important for understanding the physiological and pathological processes that occur in women. The expression of these biomarkers can help establish the diagnosis of endometrial hyperplasia and classify it into subtypes depending on the characteristics of the cells and their activity. The expression of these biomarkers may be a prognostic factor, helping to assess the prognosis for patients with endometrial hyperplasia. For example, high expression of ER and PgR may indicate a more favorable prognosis and a more sensitive response to hormone therapy, which may help in planning treatment and monitoring its effectiveness. Ki67 expression is an indicator of cell proliferation and activity. High expression of Ki67 may indicate increased cellular activity and rapid cell division, which may be characteristic of endometrial hyperplasia. While the expression of Ki67 decreases in the secretory phase of the unchanged normal endometrium, but sometimes such increased expression can reveal the presence of active proliferative processes. In addition, the expression of SC7 and SC8 may be associated with an increased number of epithelial cells. They are found in epithelial cells of various organs, including the endometrium. Studying the expression of these cytokeratins helps reveal the epithelial nature of the cells and to distinguish them from other cells. Thus, analysis of the expression of ER, PgR, Ki67, CK7 and CK8 in endometrial hyperplasia and normal endometrium allows a deeper understanding of the processes of proliferation, differentiation, and function of endometrial cells. These data have important diagnostic value for endometrial hyperplasia, the degree of determination of its atypia and the prediction of treatment results.
Conclusion
Analysis of the expression of steroid hormone receptors in different layers of the endometrium of the secretory phase and endometrium with hyperplasia without atypia found a statistically significant difference only in the expression of ER of all layers (surface, functional and basal) with the advantage of the number of samples with high expression of ER in the stroma of samples of endometrial hyperplasia without atypia (all p<0.05), on the other hand, such a difference was not found for PgR, despite the trend of a decrease in the basal parts of the endometrium with hyperplasia without atypia and secretory endometrium in the percentage of PgR positive immunoreactive cells to a complete absence in the epithelium of the glands (all p> 0.05).
Proliferative activity of endometrial cells according to the Ki- 67 marker increased from superficial to basal parts and showed a statistically significant difference between samples of endometrial hyperplasia without atypia and secretory endometrium (all p<0.05).
Cytokeratin markers SC8 and SC7 showed a significant difference in expression between themselves: SC8 remained at a constant level of expression, regardless of the depth of the endometrial glands - revealed an organ-specific expression for the endometrium, while SC7, on the contrary, was most expressed in the luminal highly differentiated epithelium surface layers and was lost in basal/stem cells of deep endometrial layers, but no statistically significant difference was found between secretory endometrium and endometrial hyperplasia without atypia (all p>0.05).
Acknowledgement
The authors of this study possess no acknowledgments to make with regards to this study and this manuscript’s content.
Ethical Approval
The study was approved at the meeting of the bioethics commission of the State Medical University №1 on January 16, 2017. All women who participated in the study received the necessary information about the goals and possible consequences of the study and gave written informed consent.
Author Contributions
Khaskhachykh DmytroAs. Prof, , Phd. – https://orcid. org/0000-0001-5097-6667
Dnipro State Medical University
Potapov Valentin, Professor, doctor of medical science - https://orcid.org/0000-0001-7498-7416
Dnipro State Medical University
Poslavska Oleksandra, As. prof, Doctor of medical science - https://orcid.org/0000-0002-3133-8413
Dnipro State Medical University
Authors’ Contribution Statement
Khaskhachikh D. - conceptualization, writing the initial project, research, wrote the manuscript.
Potapov V. - administration of verification, review and editing, management, finding financial support.
Poslavska O. - research, methodology.
References
- Sanderson PA, Critchley HO, Williams AR, Arends MJ, Saunders PT, et al. (2017) New concepts for an old problem: the diagnosis of endometrial hyperplasia. Human reproduction update 23(2): 232-
- Schmidt D (2009) Endometriale Karzinome und Vorstufen--Neue Aspekte [Endometrial carcinomas and precursor lesions--new aspects]. Der Pathologe 30(4): 261–267.
- Dall'Agnol MA, Dias JA (2019) Endometrial hyperplasia: a review for clinical practice. Clinics 74:
- Khaskhachikh D, Potapov V, Kukina G (2019) A differentiated approach to the treatment of endometrial hyperplasia without atypia in women of reproductive age. Current issues of pediatrics, obstetrics and gynecology 2(24): 149-154.
- Khaskhachikh DA, Potapov VO, Poslavska OV (2023) Factors of resistance to progestin therapy in endometrial hyperplasia in women. Morphologia 17(1): 56-62.
- Shevra CR, Ghosh A, Kumar M (2015) Cyclin D1 and Ki-67 expression in normal, hyperplastic and neoplastic endometrium. J Postgrad Med 61(1): 15-
- Bendifallah S, Genin AS, Naoura I, Chabbert-Buffet N, Bolze PA, et al. (2018) The impact of endometrial thickness changes in patients with simple hyperplasia without atypia. European journal of obstetrics, gynecology, and reproductive biology 224: 75-81.
- Owings RA, Quick CM (2014) Endometrial intraepithelial neoplasia. Arch Pathol Lab Med 138(4): 484–491.
- Antunes A, Vassallo J, Pinheiro A, Leão R, Neto AM, et al. (2014) Immunohistochemical expression of estrogen and progesterone receptors in endometrial polyps: A comparison between benign and malignant polyps in postmenopausal patients. Oncology letters 7(6): 1944-
- Ramos-Vara JA (2017) Principles and Methods of Immunohistochemistry. Methods in molecular biology (Clifton, N.J.), 1641: 115-128.






























