Meta-Analysis of Variations in Prevalence and Distribution of Polycystic Ovarian Syndrome (PCOS) Phenotypes: Incorporating Data From the Indian Council of Medical Research TASK FORCE and Global Studies’
Roya Rozati1*, Mohd Ashraf Ganie2, Haroon Rashid2, Imtiyaz Wani2, Amlin Shukla2, Beena Nitin Joshi3, Vanita Suri4 , PKK Jabbar5, Prasanta Kumar Bhattacharya6, Subhankar Chowdhury7, Sarita Agarwal8, Neena Malhotra9, Rakesh Sahay10, Bharati Kulkarni2, Taruna Katyal Arora2, Abhilash Nair5, Rohina Bashir5, Vikram Aiman Ayapati1, Gautam Mehdi Ayapati1, Naila Mohiuddin1, Talia Nazeer Ahmed1, Wajeeda Tabasum1, Nayela Sumaiya1, KKL Prasad11, Saroosh Ahmed12, Aleem Ahmed Khan13 and Rajesh Neeluri11
1Medical and Research Director, Maternal Health and Research Trust (MHRT), India
2Sheri Kashmir Institute of Medical Sciences, Srinagar, Jammu and Kashmir, India
3All India Institute of Medical Sciences, New Delhi, India
4National Institute for Research in Reproductive and Child Health, Mumbai, Maharashtra, India
5Postgraduate Institute of Medical Education and Research, Chandigarh, India
6Government Medical College, Thiruvananthapuram, Kerala, India
7North Eastern Indira Gandhi Regional Institute of Health and Medical Sciences, Shillong, Meghalaya, India
8Institute of Post Graduate Medical Education and Research, Kolkata, West Bengal, India
9Indian Council of Medical Research, India
10AIIMS, Raipur, Chhattisgarh, India
11Andhra Medical College, India
12Deccan College of Medical Sciences, India
13CMH Research and Innovation, Bolarum, Secunderabad Hyderabad, India
Submission: July 22, 2023; Published: July 25, 2023
*Corresponding author: Roya Rozati, Medical and Research Director, Maternal Health and Research Trust (MHRT), India
How to cite this article: Roya R, Mohd Ashraf G, Haroon R, Imtiyaz W, Amlin S, et al. Meta-Analysis of Variations in Prevalence and Distribution of Polycystic Ovarian Syndrome (PCOS) Phenotypes: Incorporating Data From the Indian Council of Medical Research TASK FORCE and Global Studies’. J Gynecol Women’s Health 2023: 25(4): 556167. DOI: 10.19080/JGWH.2023.25.556167
Abstract
Background: Polycystic Ovarian Syndrome (PCOS) is a prevalent endocrine disorder that predominantly affects women in their reproductive years. It is distinguished by disturbances in hormonal equilibrium, the presence of cysts within the ovaries, and the manifestation of various symptoms including irregularities in menstrual cycles, excessive hair growth, acne, and weight gain. The prevalence and distribution of PCOS phenotypes may vary across different populations and geographic regions.
Methods: The study involved a thorough examination and synthesis of relevant literature using a systematic review and meta-analysis methodology. Out of an initial pool of 1261 articles, nine articles were chosen for analysis.
Results: The findings revealed that the pooled prevalence of Polycystic Ovary Syndrome (PCOS) was estimated to be 13.851%. Furthermore, the analysis demonstrated significant heterogeneity in the various PCOS phenotypes. Notably, Phenotype C emerged as the most prevalent, accounting for 32.9% of cases, followed by phenotypes B (27.6%), D (17.9%), and A (16.6%). Our study also highlights the diverse nature of PCOS in Indian women whose prevalence is 19.2% according to the Rotterdam criteria.
Conclusion: Our study elucidates variations in its prevalence and distribution among individuals. These findings contribute to a better understanding of PCOS’s multifaceted manifestations considering different patient populations.
Keywords: Polycystic Ovarian Syndrome; PCOS; Prevalence; PCOS Phenotypes; Epidemiology
Introduction
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder that primarily affects women of childbearing age, and it is recognized as the leading cause of hyperandrogenism and ovulation disorders [1,2]. Its estimated prevalence ranges from 9% to 18% in women, depending on the diagnostic criteria employed, such as those proposed by the National Institutes of Health (NIH), Rotterdam, and the Androgen Excess and PCOS Society (AES). Dewailly et al. [3], Kandaraki et al. [4], and Lizneva et al. [5] have reported that PCOS tends to have the highest prevalence rates in Western societies.
Polycystic ovary syndrome (PCOS) patients commonly exhibit a range of metabolic abnormalities, including insulin resistance, abnormal lipid levels, and increased inflammation [6,7]. Women with PCOS are prone to developing metabolic syndrome, type 2 diabetes, and cardiovascular disease due to elevated insulin resistance and inflammatory markers, such as C-reactive protein (CRP) [8,9]. PCOS is a heterogeneous condition characterized by symptoms of androgen excess and ovarian dysfunction in the absence of an alternative diagnosis. While the exact etiology of PCOS remains unclear, evidence suggests it is a multifactorial condition with significant contributions from genetic, epigenetic, and environmental factors, including nutrition and lifestyle choices. PCOS often leads to menstrual abnormalities and reproductive dysfunction, resulting in infertility among affected women [10,11].
The clinical features of PCOS include oligo-ovulation, hyperandrogenism (manifesting as acne and hirsutism), and the presence of multicystic ovaries in many cases.This meta-analysis focuses on the reported prevalence of PCOS and its phenotypes based on Rotterdam diagnostic criteria across diverse population groups. PCOS appears to represent an evolutionary paradox; much of our understanding of the evolutionary origins of this pervasive disorder will arise from its presentation in the general population [12] Understanding the prevalence and distribution of PCOS phenotypes is crucial for developing effective prevention programs and interventions. However, to date, there is a lack of comprehensive cross-cultural analyses that compare PCOS phenotypes across diverse ethnic groups. Such analyses can provide valuable insights into the variations in the occurrence and distribution of PCOS phenotypes among different ethnicities.
The objective of this study is to conduct a systematic review and meta-analysis of existing literature focusing on the phenotypic features of polycystic ovary syndrome (PCOS) across diverse ethnic groups. By synthesizing data from multiple studies, our aim is to identify and compare the prevalence of PCOS phenotypes, shedding light on the diverse presentations within the PCOS population. This comprehensive study addresses current knowledge gaps and contributes to a better understanding of the complex nature of PCOS, ultimately paving the way for more targeted approaches to the prevention, diagnosis, and management of this prevalent disorder.
In our analysis, we have incorporated data from the population-based Indian TASK FORCE study, which was conducted across ten nationwide participating sites, including our center, MHRT. This study focused on determining the prevalence rate of PCOS among women of Indian ethnicity. Gathering information on various pertinent parameters, this pivotal study established a PCOS prevalence rate among Indian women (Ganie et al., 2023) in addition to yielding valuable insights into the phenotypic distribution of PCOS, providing a crucial basis for comparison in our meta-analysis.
Materials and Methods
In this study, we performed a systematic review and metaanalysis of the existing literature to assess the overall prevalence of PCOS phenotypes according to the Rotterdam criteria. The prevalence of each of these four PCOS characteristics was reported in all relevant research, or at the very least, if there was enough information to determine the prevalence of each phenotype, they were included in our study. This study was conducted and reported in accordance with the PRISMA statement for systematic reviews and meta-analyses.
Search Strategy
A comprehensive literature search was conducted using electronic databases, including PubMed, ProQuest, and manual searches of relevant journals and reference lists to identify relevant studies published between January 2000 and February 2023. The keywords and search terms used were carefully selected to ensure a comprehensive search of the literature related to PCOS prevalence and phenotypic characteristics across different cultures and ethnicities. The following keywords and search terms were used: “PCOS”, “Polycystic Ovarian Syndrome”, “Prevalence”, “Rotterdam Criteria” and “Phenotypes”. To decrease bias, two authors (WT and NM) searched, selected papers, and extracted data from the paper independently.
Selection Criteria
Studies were included if they met the following criteria:
a) They investigated the prevalence and/or phenotypic characteristics of PCOS in human populations;
b) They reported Prevalence in Rotterdam criteria
c) They were published in English, and
d) They were conducted in any country or region around the world.
Studies were excluded if they were review articles, case reports, or animal studies. Studies with a sample size of less than 50, studies with incomplete or inadequate data, and studies published in languages other than English were also excluded.
Data Extraction
Data were extracted from the included studies using a standardized data extraction form. The following data were extracted: author, year of publication, country of origin, sample size, the age range of participants, the prevalence of PCOS according to the Rotterdam Criteria [13,14] and its phenotypic distribution (in percentages), and any relevant study characteristics (Table 1).


Data Analysis
Pooled prevalence rates and odds ratios were calculated using random-effects models. Heterogeneity and Publication Bias were assessed using the I2 statistics across studies, with values greater than 50% indicating substantial heterogeneity. Publication bias was assessed using funnel plots and Egger’s test using MedCalc®Statistical Software version 22.003 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2023).
Result
In the initial phase of our research, a comprehensive search yielded a total of 2717 studies. After eliminating duplicate records, we were left with 1261 unique articles, which were then screened to determine their suitability for inclusion in the meta-analysis. Following a thorough evaluation process, 48 articles were deemed eligible for further analysis. Ultimately, our meta-analysis incorporated nine studies that satisfied the predefined inclusion criteria (as depicted in Figure 1). By examining these studies, we aimed to assess the prevalence of Polycystic Ovary Syndrome (PCOS) in various populations.
The reported rates of PCOS in the studies included in our analysis exhibited a range from 5.5% to 19.8%. To provide a more comprehensive overview of PCOS prevalence, we calculated a pooled prevalence rate of 13.85% (95% confidence interval: 8.950 to 19.619, p = 0.3491). Notably, we did not observe any significant heterogeneity among the studies across the examined populations (as depicted in Figure 2).
In China, two cross-sectional studies (Ma et al. [15] Li R et al.[16]) [15,16] conducted in the years 2010 and 2013 in different communities reported a PCOS prevalence of 5.6% and 6.1%, respectively. In Iran, Tehrani et al. [17] reported a PCOS prevalence rate of 14.6% in their study, while a more recent study by Farhadi-Azar et al. [18] indicated a prevalence range between 13.6% and 17.8%. The studies conducted in urban areas of four randomly selected provinces and among Khouzestani women [19] reported a higher prevalence of PCOS (13.6%) than the population-based cross-sectional study (14.6%) indicating that the prevalence of PCOS is relatively high in urban areas of Iran compared to the general population. This observation warrants further investigation to obtain a deeper understanding In Turkey, the prevalence of PCOS was reported to be 19.9% in the year 2012. A referral-based study conducted in a hospital-based OPD by Yildiz et al. [20] reported a relatively higher prevalence of PCOS (46.2%), whereas a study conducted among women working in a government-based institute reported a lower prevalence of PCOS (5.1%).
In Australia, a retrospective birth cohort study reported a PCOS prevalence of 8.7% ± 2.0% [21]. The study was conducted among women aged 27–34 years, which suggests that PCOS may be common in this age group. Similarly, in Denmark, a prospective cross-sectional study conducted among employees of Copenhagen University Hospital reported a PCOS prevalence of 16.6% [22]. Overall, the prevalence of PCOS seems to be relatively high in China and Turkey, and lower in Iran compared to the other studies discussed so far.

In India, the overall prevalence of Polycystic Ovary Syndrome (PCOS) was determined to be 19.2%. Among the PCOS phenotypes, Phenotype C was the most commonly observed, accounting for 42.1% of cases. This was followed by Phenotype D, Phenotype A, and Phenotype B, representing 22.8%, 20.6%, and 14.5% of cases, respectively. Notably, there were significant differences in the distribution of these phenotypes across different geographic zones within India, indicating noteworthy regional variations in the expression of PCOS phenotypes. However, it is important to note that a detailed discussion of these regional variations falls outside the scope of this paper.
The analysis in our study had the intercept value of 6.7116 in Egger’s test indicating the estimated asymmetry in the funnel plot (Figure 3), but it is not significantly different from zero based on the 95% confidence interval (-8.8514 to 22.2745). The significance level (P-value) of 0.3491 suggests that there is no strong evidence of publication bias. Kendall’s Tau value of 0.1111 represents the correlation coefficient of bias, but the significance level (P-value) of 0.6547 indicated no significant correlation and evidence of publication bias.

Our meta-analysis on the phenotypic distribution of PCOS yielded robust results that provide valuable insights into the prevalence of different phenotypes. Phenotype A exhibited a significant difference in the pooled prevalence estimates among the categorized studies (16.608%, 95% CI: -76703 to 1.6899, P = 0.1789) (Figure 3). Notably, our analysis revealed that Phenotype A had the lowest prevalence among the reported studies, with a pooled prevalence of 16.6%. It is worth mentioning that Gabrielle et al. [23] reported an unusually small percentage of the population with Phenotype A, indicating the potential impact of smaller studies in reporting larger effect sizes (Kendall’s Tau value: -0.4045, P = 0.1035). In terms of Phenotype B, a significant difference in the pooled prevalence estimates was observed among the studies that classified patients into this phenotype (27.647%, 95% CI: -3.9686 to 13.8578, P = 0.2367). Our analysis also revealed no significant evidence of publication bias based on the Egger’s test (intercept value = 4.9446, p = 0.2367) (Figure 4) [24].
Similarly, for Phenotype C, our analysis showed a significant difference in the pooled prevalence estimates among studies that categorized patients into this phenotype (32.963%, 95% CI: -9.2724 to 3.7868, P = 0.3611). Notably, there was no discernible evidence of publication bias according to the Egger’s test (intercept value = -2.7428, p = 0.3611) (Figure 5). This particular phenotype demonstrated the highest prevalence, with an estimated 32.9% of individuals with PCOS expected to exhibit this specific presentation.
Regarding Phenotype D, there was a significant difference in the pooled prevalence estimates between studies that categorized patients into this category (17.965%, 95% CI: -5.5542 to 3.4941, P = 0.6138). Importantly, our analysis did not reveal any significant publication bias based on Egger’s test in this analysis either (intercept value = -1.0301, p = 0.6138) (Figure 6).
Discussion
Our meta-analysis showed that the prevalence rates of PCOS among the included studies varied significantly. The lack of significant variability among the studies, however, suggests that study design or methodology cannot account for all variations in prevalence rates. Obesity, race, and ethnicity are only a few of the variables that are said to be connected to phenotypic variability [25]. According to the Rotterdam criterion, our results show a 13.85% total PCOS prevalence. The prevalence of PCOS varied significantly across different populations and countries, as evidenced by the wide range of rates reported in the included studies, ranging from 5.5% to 19.8%.
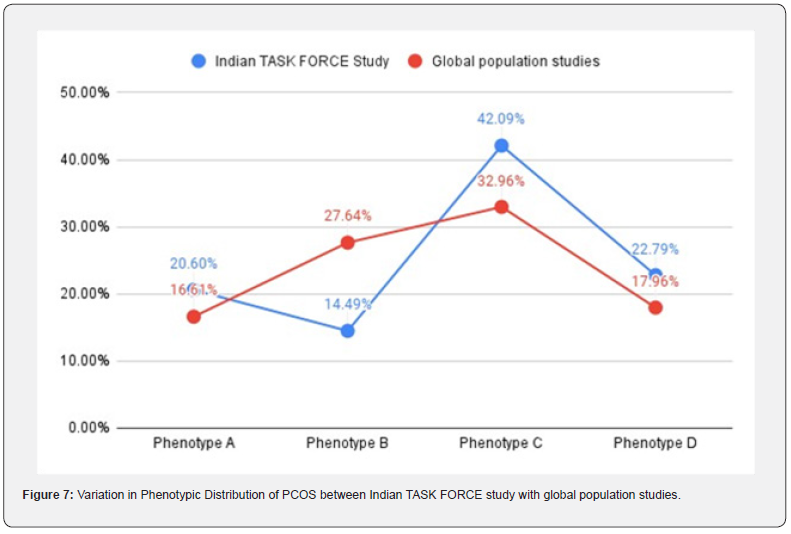
Notably, our meta-analysis included the National PCOS Taskforce project, one of the largest studies in Asia reporting PCOS prevalence. In this study, a total of 9,841 women underwent thorough biochemical and hormonal evaluation, of which 1,891 were diagnosed with PCOS. In India, the prevalence rate was found to be 19.2% according to the Rotterdam Criteria, with Phenotype A accounting for 20.6% of cases, Phenotype B for 14.49%, Phenotype C for 42.09%, and Phenotype D for 22.79%. Consistent with the global distribution of PCOS phenotypes, the Indian population exhibited a higher prevalence of Phenotype C (42.09%) (Figure 7).
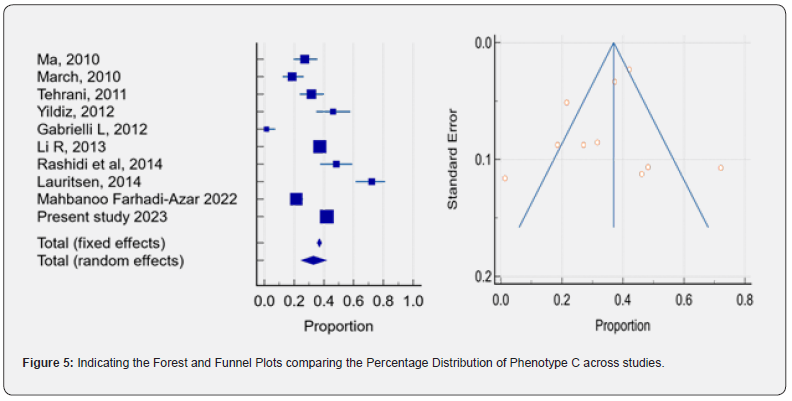

A similar prevalence rate was reported in a study conducted by Yildiz et al. [20]. in 2012, although the sample size was relatively small. This cross-sectional study in Turkey, conducted at a government-based institute, reported a prevalence rate of 19.9%. The study recruited 392 female employees from a single institute, of which 78 were diagnosed with PCOS. The phenotypic distribution in these women was similar, with Phenotype A accounting for approximately 25.6%, Phenotype B for 5.1%, Phenotype C for 46.2%, and Phenotype D for 23.1%.”
When the studies evaluated prevalence using Rotterdam criteria by Li. R et al. [16] reported a 5.6% prevalence rate, and another group reported by Ma et al 201015 reported a 6.1% similar result from China (In Table 1). Women with PCOS identified by referral were significantly more likely to have phenotype A (classical PCOS) than patients identified by unselected populations and had a small proportion of overlapped subjects.
In Iran, several studies reported by different groups, like Tehrani FR et al.[17], Rashidi H et al.[19], Farhadi-Azar M et al.[18], PCOS prevalence ranging from 8.2% to 14.6%. The studies conducted in urban areas of four randomly selected provinces and Khouzestani women reported a higher prevalence of PCOS (14.6% and 13.6%, respectively) than the population-based cross-sectional study (8.2%) by Farhadi-Azar M et al., 2022. These results suggest that the prevalence of PCOS is relatively high in urban areas of Iran compared to the general population.
In Australia, a study by March et al. [21] a retrospective birth cohort study reported a PCOS prevalence of 17.8%The study was conducted among women aged 27-34 years, which suggests that PCOS may be common in this age group. Phenotype A is 21.2%, Phenotype B-27.5%, Phenotype C is 18.9%, and Phenotype D-32.5%. Similarly, in Denmark a study by research group Lauritsen et al., 201422, a prospective cross-sectional study conducted among employees of Copenhagen University Hospital reported a PCOS prevalence of 18.6%. This finding suggests that the prevalence of PCOS may be relatively high among this population where Phenotype A is 4.65%, Phenotype B-4.65%, Phenotype C is 72.09%, and Phenotype D-18.6%.
One of the studies reported in Brazil by Gabrielli et al. [23] has an unselected population where a cross-sectional two-phase study was conducted and reported prevalence of 8.5%. where Phenotype A is 94.5%, Phenotype B-1.36%, Phenotype C is 4.1%, and phenotype D-8.5%. According to Gabrielle et al. [23] and Lauritsen M.P et al. [22], women with PCOS detected in unselected populations had more phenotypes B and C. As reported by March et al. (2010), phenotype D is found to be more prevalent in crosssectional populations of individuals with PCOS, regardless of geographic location. Unselected populations are most likely to have phenotype C, which affects 1 in 3 women. Compared with patients identified in unselected populations, patients in clinics have more severe phenotypes, more severe clinical and biochemical HA, and a higher body mass index (BMI), as well as a higher metabolic risk.
Nonetheless, it is essential to acknowledge the potential limitations associated with the heterogeneity of the included studies. Our data is derived from moderate-quality studies; however, indirect evidence suggests that quality did not significantly influence the results of analyses, and the results of only the high-quality studies would have remained the same if the quality of the studies had not been taken into account. Firstly, it is important to note that all eligible studies were not designed to assess PCOS prevalence, and PCOS characteristics varied widely across studies. The Rotterdam 2003 criteria for PCOM may lead to an overestimation of its prevalence among studies.

Conclusion
In conclusion, our meta-analysis provides valuable insights into the prevalence and distribution of PCOS across different populations and countries. The observed variations in prevalence rates underscore the influence of genetic, environmental, and cultural factors on PCOS. These findings highlight the importance of further research to better understand the underlying factors contributing to the heterogeneity in PCOS prevalence.A comprehensive understanding of PCOS prevalence and its phenotypic distribution is crucial for improving clinical management worldwide.
Authors’ Roles
R.R, M.A.G, H.R, I.W, A.S, B.N.J, V.S, P.K.K.J, P.K.B, S.C, S.A, N.M, R.S, B.K, T.K.A., A.N and R.B. contributed to data collection and analysis and the drafting and revision of the manuscript. R.R, N.M, WT, V.A.A., G.M.A, T.N.A, N.S, A.A.K contributed to study design, data analysis, and the drafting and revision of the manuscript. H.B.W. contributed to study design, data analysis, and the revision of the manuscript. M.S.A, K.K.L.P, R.N contributed to data analysis and the revision of the manuscript.
Funding
The work was supported by Indian Council of Medical Research (ICMR grant RFC No.: RBMH/NTF/1/2017-2018).
References
- Kahsar-Miller MD, Nixon C, Boots LR, Go RC, Azziz R, et al. (2001) Prevalence of polycystic ovary syndrome (PCOS) in first-degree relatives of patients with PCOS. Fertil Steril 75(1): 53-58.
- Rosenfield RL, Ehrmann DA (2016) The pathogenesis of polycystic ovary syndrome (PCOS): the hypothesis of PCOS as functional ovarian hyperandrogenism revisited. Endocr Rev 37(5): 467-520.
- Dewailly D, Hieronimus S, Mirakian P, Hugues JN (2010) Polycystic ovary syndrome (PCOS). InAnnales D'endocrinologie 71(1): 8-13.
- Kandaraki E, Chatzigeorgiou A, Livadas S, Palioura E, Economou F, et al. (2011) Endocrine disruptors and polycystic ovary syndrome (PCOS): elevated serum levels of bisphenol A in women with PCOS. J Clin Endocrinol Metab 96(3): E480-E484.
- Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, et al. (2016) Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril 106(1): 6-15.
- Fauser BC, Tarlatzis BC, Rebar RW, Legro RS, Balen AH, et al. (2012) Consensus on women's health aspects of polycystic ovary syndrome (PCOS). Hum Reprod 27(1): 14-24.
- Pasquali R (2018) Contemporary approaches to the management of polycystic ovary syndrome. Ther Adv Endocrinol Metab 9(4): 123-134.
- Keskin KR, Okyay AG, Hakverdi AU, Gungoren A, Dolapcioglu KS, et al. (2014) The effect of obesity on inflammatory markers in patients with PCOS: a BMI-matched case–control study. Arch Gynecol Obstet 290(2): 315-319.
- Boulman N, Levy Y, Leiba R, Shachar S, Linn R, et al. (2004) Increased C-reactive protein levels in the polycystic ovary syndrome: a marker of cardiovascular disease. J Clin Endocrinol Metab 89(5): 2160-2165.
- Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, et al. (2004) The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab 89(6): 2745-2749.
- Giviziez CR, Sanchez EG, Approbato MS, Maia MC, Fleury EA, et al. (2016) Obesity and anovulatory infertility: a review. JBRA assisted reproduction 20(4): 240-245.
- Azziz R, Dumesic DA, Goodarzi MO (2011) Polycystic ovary syndrome: an ancient disorder? Fertil Steril 95(5): 1544-1548.
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group ((2004a)) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 81: 19-25.
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group (2004b) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod;19: 41–47.
- Ma YM, Li R, Qiao J, Zhang XW, Wang SY, et al. (2010) Characteristics of abnormal menstrual cycle and polycystic ovary syndrome in community and hospital populations. Chinese medical journal 123(16): 2185-2189.
- Li R, Zhang Q, Yang D, Li S, Lu S, et al. (2013) Prevalence of polycystic ovary syndrome in women in China: a large community-based study. Hum Reprod 28(9): 2562-2569.
- Tehrani FR, Simbar M, Tohidi M, Hosseinpanah F, Azizi F, et al. (2011) The prevalence of polycystic ovary syndrome in a community sample of Iranian population: Iranian PCOS prevalence study. Reprod Biol Endocrinol 9(1): 1-7.
- Farhadi-Azar M, Behboudi-Gandevani S, Rahmati M, Mahboobifard F, Khalili Pouya E, et al. (2022) The prevalence of polycystic ovary syndrome, its phenotypes and cardio-metabolic features in a community sample of Iranian population: Tehran lipid and glucose study. Front Endocrinol 13: 134.
- Rashidi H, Tehrani FR, Khomami MB, Tohidi M, Azizi F, et al. (2014) To what extent does the use of the Rotterdam criteria affect the prevalence of polycystic ovary syndrome? A community-based study from the Southwest of Iran. Eur J Obstet Gynecol Reprod Biol 174: 100-105.
- Yildiz BO, Bozdag G, Yapici Z, Esinler I, Yarali H, et al. (2012) Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum Reprod 27(10): 3067-3073.
- March WA, Moore VM, Willson KJ, Phillips DI, Norman RJ, et al. (2010) The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod 25(2): 544-551.
- Lauritsen MP, Bentzen JG, Pinborg A, Loft A, Forman JL, et al. (2014) The prevalence of polycystic ovary syndrome in a normal population according to the Rotterdam criteria versus revised criteria including anti-Mullerian hormone. Hum Reprod 29(4): 791-801.
- Gabrielli L, Aquino EM (2012) Polycystic ovary syndrome in Salvador, Brazil: a prevalence study in primary healthcare. Reprod Biol Endocrinol 10(1): 96.
- Mohd AG, Haroon R, Imtiyaz W, Amlin S, Beena NJ, et al. (2023) Variation in normative values of major clinical biochemistry analytes in healthy reproductiveage women in India: A subset of data from a National Indian Council of Medical ResearchPolycystic Ovary Syndrome task force study. Indian J Pharmacol 55(2): 76-88.
- Dumesic DA, Oberfield SE, Stener-Victorin E, Marshall JC, Laven JS, et al. (2015) Scientific statement on the diagnostic criteria, epidemiology, pathophysiology, and molecular genetics of polycystic ovary syndrome. Endocr Rev 36(5): 487-525.






























