Safety And Efficacy of a Therapeutic Containing Pea Protein, Grape Seed Extract and Lactic Acid as a Vaginal Co-Adjuvant Treatment to Clotrimazole in Vaginal Yeast Infection: A Double Blind, Parallel, Randomized, Multicenter Study
Vasilica Bausic1, Elvira Bratila2, Doru Diculescu3, Mihai Manolache4* and Alina Calin5
1Department of Cell Biology and Histology, “Carol Davila”, University of Medicine and Pharmacy, Unirea Medical Centre, Romania
2Department of Obstetrics and Gynecology, “Carol Davila” University of Medicine and Pharmacy, Unirea Medical Centre, Romania
3Department of Obstetrics-Gynecology, Iuliu Hatieganu University of Medicine and Pharmacy, Clinical Emergency Hospital, ObGyn Department, Romania
4Cebis International, Romania
5Saint Andrew Clinical Emergency Hospital, Romania
Submission: October 14, 2022; Published: October 20, 2022
*Corresponding author: Mihai Manolache, Cebis International, Calea Plevnei, Romania
How to cite this article: Bausic V, Bratila E , Diculescu D, Manolache M, Calin A. Safety And Efficacy of a Therapeutic Containing Pea Protein, Grape Seed Extract and Lactic Acid as a Vaginal Co-Adjuvant Treatment to Clotrimazole in Vaginal Yeast Infection: A Double Blind, Parallel, Randomized, Multicenter Study. J Gynecol Women’s Health 2022: 24(2): 556131. DOI: 10.19080/JGWH.2022.24.556131
Abstract
Aim: Vulvovaginal candidiasis (VVC) is a gynecological condition that strongly affects the physical and psychological wellbeing of patients. VVC and recurrent VVC are characterized by local inflammation and alteration of the vaginal mucosa. The aim of this study is to evaluate the use of a topical formulation of pea protein, grape seed extract and lactic acid (PGL) versus lactic acid alone as add-on therapy to clotrimazole vaginal tablets to relieve symptoms and prevent overgrowth of pathogens that cause recurrent vaginal infections.
Methods: 42 women with vaginal yeast infection participated in the study. Patients were randomly assigned to 7 days of treatment with a topical formulation containing PGL or with lactic acid alone. Both treatments were administered in addition to clotrimazole vaginal tablets for the first 3 days. Safety and efficacy were assessed by monitoring clinical signs and symptoms such as leucorrhea, pruritus, dysuria, dyspareunia, vaginal pH and mycological counts.
Results: 24 and 18 subjects received the formulations containing PGL or lactic acid, respectively. The results showed that PGL was well tolerated and enhances the effect of azole in reducing the mycological count compared to lactic acid after 7 days (p = 0.04). PGL also reinforces the efficacy of clotrimazole in eliminating the severe and moderate cases of pruritus, leucorrhea, dysuria, and dyspareunia and promoting the restoration of an optimal vaginal pH range.
Conclusions: The PGL formulation proved to be a safe and effective co-adjuvant treatment to clotrimazole vaginal tablets to effectively eradicate C. albicans infections.
Keywords: Epithelial barrier; Lactic acid; Muco-protectants; Pea protein; Tannins; Vaginal infection
Abbreviattaion: VVC: Vulvovaginal Candidiasis; BV: Bacterial Vaginosis; SAE: Serious Adverse Events
Introduction
Vaginal infections are the most common genital conditions affecting women and can be generally classified as Bacterial Vaginosis (BV), Vulvovaginal Candidiasis (VVC) and trichomoniasis [1]. Vaginal infections occur when the vaginal ecosystem is altered. In particular, the cervicovaginal microbiota is characterized by a prevalence of Lactobacillus species (e.g., L. crispatus, L. jensenii, L. gasseri and L. iners) but it is also colonized by Candida species, mainly represented by C. albicans, which behaves in most cases as a commensal. Lactobacilli strictly regulate the composition of the vaginal microbiota and, through the secretion of lactic acid, contribute to the maintenance of an optimal vaginal pH (< 4.5) [2]. C. albicans commensalism is regulated by Lactobacilli and the host immune response. Hence, depending on the host homeostasis, an imbalance of C. albicans can lead to the development of vaginal infections called vulvovaginal candidiasis [3].
VVC is recognized as the most prevalent human candida infection, involving 75% of women who get VVC at least once in their lifetime [4]. Recurrent VVC (RVVC), defined as the occurrence of more than 3 episodes per year, arises when the infection is not completely eradicated [5]. RVVC is estimated to affect 8% of women worldwide, with psychological consequences, high economic costs, and negative impact on the quality of life [4-7]. The most common clinical manifestations of VVC are vulvar burning, vaginal soreness, and irritation, leading to dyspareunia and dysuria [8]. VVC and especially RVVC have been also associated with psychological manifestations such as distress, discomfort, altered self-esteem, anxiety, decreased work performance and impaired sexual and affective relationships [8-10]. Since the symptoms and signs of VVC are not specific, the diagnosis requires a correlation between clinical signs and laboratory analyses [11].
Major risk factors for VVC include high estrogen and progesterone levels during pregnancy, use of antibiotics, uncontrolled diabetes, a weakened immune system (use of glucocorticoids, chemotherapy-associated immunodepression, HIV), and elevated estrogen levels due to therapies (e.g., hormone therapy during menopause). Other important contributing factors are the use of oral contraceptives, intrauterine devices, spermicides, hygiene, clothing, and sexual habits [12]. The pathogenicity of C. albicans is determined by events such as adhesion, biofilm formation, production of extracellular hydrolytic enzymes, and tissue damage [13]. Adhesion to vaginal epithelial cells is the most important event in VVC and is mediated by host ligand recognition by adhesins such as agglutinin-like sequence proteins and Hwp1 [14-17]. Remarkably, C. albicans can adhere not only to the vaginal mucosa but also to the surface of implantable devices [17]. Adhesion is an early step in biofilm formation, representing an important virulence factor that confers properties such as resistance to antifungal agents or host defense mechanisms [18].
Once a biofilm has formed, C. albicans secretes hydrolytic enzymes that promote adhesion, tissue penetration, and host tissue destruction, resulting in a change in the barrier properties of the vaginal mucosa [19]. In the treatment of RVVC, special attention must be paid to the integrity of the vaginal mucosa. Indeed, the continued release of hydrolytic enzymes by pathogens can lead to serious mucosal damage, promoting the invasion of microorganisms and chronic tissue inflammation [20,21].
The primary therapeutic approach against VVC is based on the use of antifungal drugs such as azoles, which act as cytostatic agents by altering the cell membrane. In particular, they inhibit the activity of 14-demethylase, resulting in inhibition of ergosterol production [22]. Among the azoles, fluconazole and clotrimazole, administered orally and topically, respectively, are considered the drugs of first choice. Unfortunately, this treatment does not allow the complete eradication of the pathogen and often leads to local or systemic side effects, such as local irritation, gastrointestinal alterations and severe drug-interactions [23]. Furthermore, the extensive use of azoles has led to the selection of azole-resistant species. The high incidence of resistant species reinforces the need to find alternative safe and effective treatments [24].
Beneficial effects on the integrity of vaginal mucosa can be achieved by using proteins from Pisum sativum (pea protein) and grape seed extract. Pea proteins support the barrier effect of the vaginal mucosa through a mucoadhesive effect that can restore the integrity of the epithelium [25]. Grape seed extract, which is rich in polyphenols and flavonoids, supports mucosal wound healing [26]. In addition, topical application of pea protein and grape seed extract in a mouse model of VVC was recently shown to be able to reduce vaginal fungal load, vaginal irritation and maintain the integrity of the vaginal mucosa [27] which is essential to prevent local inflammation, pathogen invasion, and subsequent recurrence [28,29].
The optimal vaginal microbiota of reproductive aged women is typically characterized by dominance of lactic acid producing Lactobacillus species. In vitro studies have shown that lactic acid inactivates BV-associated bacteria [30] and pathogens including Chlamydia trachomatis, Neisseria gonorrhoeae. Lactic acid has also been shown to prevent C. trachomatis infection in cervicovaginal epithelial cells [31], and to help reduce the production of inflammatory cytokines and chemokines from cervicovaginal epithelial cells in vitro [32]. Lactic acid-containing products have been evaluated for BV treatment in clinical trials, and several overthe- counter lactic acid-containing products are marketed to treat BV or support optimal vaginal microbiota.
Given the importance of adjunctive treatment to support standard pharmacological therapies, the aim of the study was to evaluate the effects of a new topical formulation containing pea protein, grape seed extract and lactic acid (PGL), compared to lactic acid alone as an adjunct to topical clotrimazole in a cohort of patients with VVC.
Materials and methods
Aim and Design of the Study
The present study is a multicenter, randomized, controlled trial designed to evaluate the safety and efficacy of a substancebased medical device containing PGL compared with lactic acid, administered vaginally as an adjunct treatment to clotrimazole vaginal tablets in patients affected by VVC.
The study was performed in compliance with the requirements of the National Agency of Medicine and Medical Devices of Romanian and National Ethical Committee for Biomedical Research and gained full regulatory approval from the National Agency of Medicine and Medical Devices of Romania. The study was registered with the following EudraCT Number 2014-005315- 16 and with the following ISCRTN Number ISRCTN12908359.
Inclusion and Exclusion Criteria
Caucasian female patients between 18 and 50 years of age, suffering from vaginal yeast infections were included in the study. To meet the inclusion criteria, patients had to start therapy with clotrimazole vaginal tablets for 3 days. Pregnant and breastfeeding women or patients with known allergy to any of the product components were excluded from the study. All participating patients signed a written informed consent.
Treatments
Patients were randomly assigned to treatment with the PGL formulation or with lactic acid in a 1:1 ratio for 7 consecutive days. During the first 3 days, the topical formulations were administered as an adjunct treatment along with clotrimazole vaginal tablets, according to the approved leaflet. From day 4 to day 7, PGL or lactic acid were administered alone.
Study Endpoints
The primary endpoint of the study was to evaluate the safety of the PGL-based medical device. The parameters monitored to evaluate this endpoint were the occurrence and classification of adverse events (AE), serious adverse events (SAE) and Suspected Unexpected Serious Adverse Reactions (SUSAR). The secondary endpoint of the study was to assess the efficacy of the treatment.
The following parameters were evaluated: mycological count, vaginal discharge, itching, dysuria and superficial dyspareunia. In addition, the efficacy of the treatment was assessed by measuring the vaginal pH.
Statistical Analysis
Sample size was calculated as the percentage of patients in whom itch decreased by 1 point after 7 days, with a power of 80% and a confidence interval of 95% [33]. Assuming 10% of patients were lost to follow-up, the calculated sample size was 44 patients for each group. The percentage of reduction in leucorrhea, itching, irritation of the vagina and surrounding tissues, pain during intercourse, burning sensation during urination and pH values between groups was analyzed at visits 1 and 2. Mycological count and age are given as mean ± standard deviation. Medical history, clinical examination and diagnosis details are given as frequency and percentage.
To evaluate the significance between the effect of treatment on mycological count, the independent samples t-test or Mann Whitney test were used. To assess the decrease in vaginal itching, chi-square or fisher exact tests were used to measure the association between the vaginal itching by using Scott-Huskisson scale leucorrhea, itching, irritation of the vagina and surrounding external tissues, pain during intercourse, burning sensation during urination and pH values within treatment groups. The McNemar’s test was used to determine statistically significant differences between groups in parameters such as leucorrhea, itching, irritation of vagina and surrounding tissues, pain during intercourse, burning sensation during urination and pH values at visit 1, 2 and 3. A p-value less than 0.05 was considered statistically significant.

Results
Population Characteristics
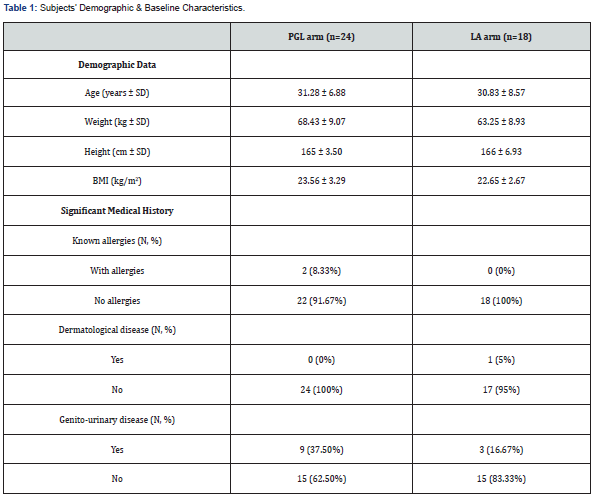
A total number of 42 subjects were included in the analysis. In the interim analysis performed, the decrease in itching symptoms was 5 points when comparing the PGL group with the lactic acid group, indicating that 42 subjects were sufficient to confirm the hypothesis tested. Subjects aged 18 to 45 years (mean age 31), were randomly assigned to the two treatment groups, with 24 patients receiving PGL formulation and 18 patients receiving lactic acid (6 subjects withdrew consent). Detailed population characteristics are reported in Table 1. The flowchart of the study is described in Table 2 and Figure 1.

T.A. - Treatment allocation; T.E. - Treatment Evaluation
Primary Endpoint - Safety
No adverse events were reported in either treatment group (Table 3). Furthermore, no adverse systemic or local effects were reported by either patients or physicians, indicating that the treatments were well tolerated (Table 4).

Secondary Endpoints
Mycological count: Mycological count was evaluated at day 1, 3 and 7 of treatment. PGL significantly increased the efficacy of clotrimazole by reducing the mycological count by 83% from day 1 to day 7 (p=0.00001) (Figure 2). Both treatments were found to be effective, but the decrease in mycological count between day 1 and 7 was significantly more effective with PGL than with lactic acid alone (p = 0.04).

Vaginal itching: Vaginal itching was monitored and selfevaluated by each subject, using Scott-Husskison scale. At baseline, 54.16% and 61.11% of the subjects reported vaginal itching in the group receiving PGL and lactic acid, respectively. In particular, 3 severe cases of vaginal itching were reported in both groups. In the group treated with PGL the symptom resolved after 48 hours in all cases, whereas in the lactic acid group, itching symptoms resolved after 72 hours (Figure 3). 18 cases of moderate itching were also reported in both groups; in the PGL group, 9 cases out of 10 (90%) resolved at day 7 of treatment. In the group receiving lactic acid, 7 cases out of 8 (87.5%) resolved at day 7 (Figure 4).
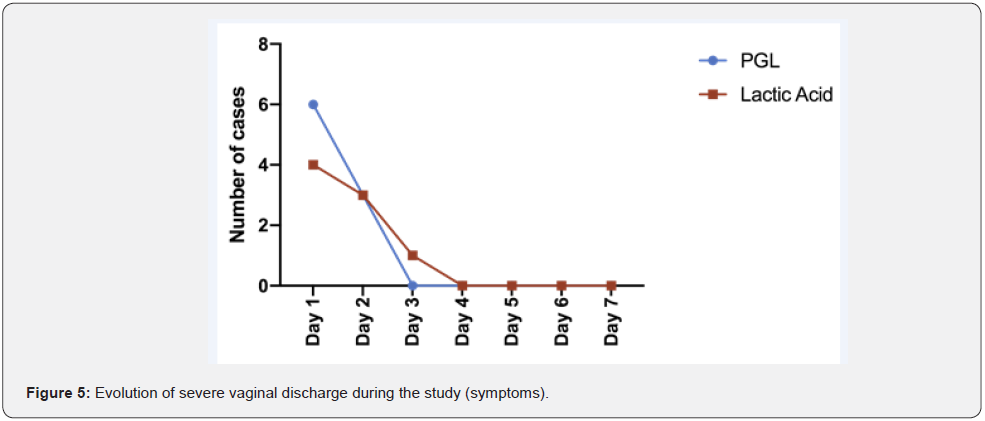
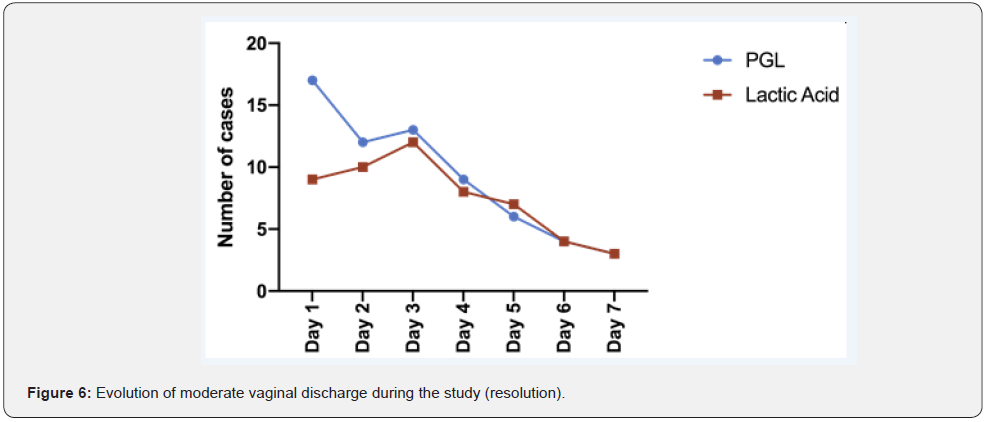
Vaginal discharge: Leucorrhea, a thick, white discharge, often resembling cottage cheese, was reported by all patients. On the second day, 6 and 4 severe cases of leucorrhea were noted in the PGL- and lactic acid-treated groups, respectively. Topical application of PGL promoted resolution of all severe cases after 48 hours. In contrast, in the lactic acid-treated group, symptoms disappeared in all cases after 72 hours (Figure 5). In the PGL group, 82.4% (14 of 17) of the moderate-severe cases had resolved after 7 days, whereas in the lactic acid-treated group, 66.7% (6 of 9) of the moderate-severe cases had resolved (Figure 6).
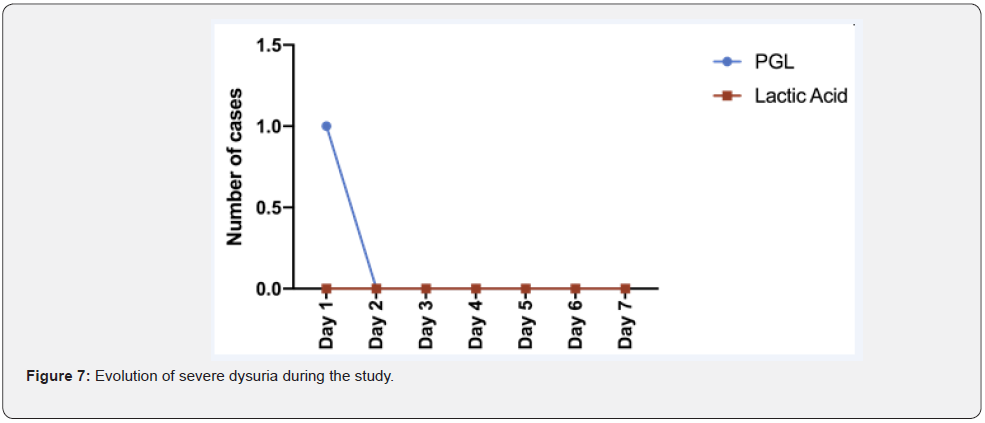
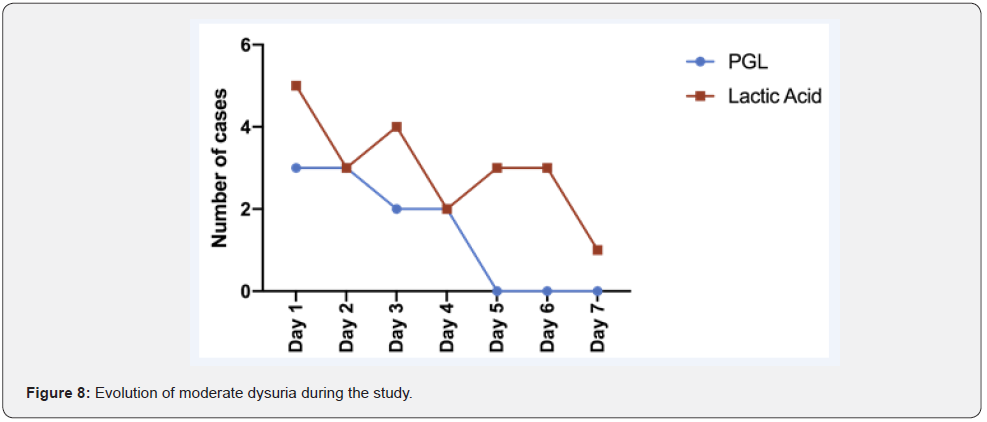
Dysuria: At day 1, all patients experienced dysuria. In the group treated with PGL product, 1 case of severe dysuria was reported, that effectively resolved after 1 day. No severe dysuria cases were reported in the lactic acid treated group (Figure 7). Moderate dysuria occurred in 3 and 5 patients from the PGL and lactic acid arm, respectively. At day 7, all cases were resolved in the PGL group while in the lactic acid group, one case persisted (Figure 8).
Dyspareunia: All patients reported dyspareunia at baseline. One patient in the PGL arm suffered from severe dyspareunia at day 1 and the symptom was treated within the first 24 hours. No severe cases were reported in the lactic acid group (Figure 9). 7 and 5 cases of moderate dyspareunia were reported in the PGL and lactic acid arms, respectively. At the end of the study (day 7), all the moderate cases of dyspareunia were resolved (Figure 10).
Vaginal pH: Local pH is an important parameter for the homeostasis of the vaginal environment. Values ranging between 3.8 and 4.5 are generally considered to be optimal. At the beginning of the study (visit 1), vaginal pH values were higher than 4.5 in both groups. At the time of the last evaluation in the PGL group, vaginal pH ranged between 3.5 and 4.5, and in the lactic acid group, vaginal pH was > 4.5. These results indicate that PGL is more effective than lactic acid alone in restoring vaginal pH.




Discussion
VVCs are among the most common vaginal infections that directly affect patients’ quality of life. Acute or recurrent vaginal infections are indeed associated with psychological changes often leading to depression and anxiety [10,34]. Various risk factors such as stress, hospitalization, other pathologies, or drug-dependent immunosuppression can contribute to VVC. In addition, VVC can be exacerbated by long-term stress, a situation that is very common in the population today, especially in light of the current global SARS Cov-2 pandemic [35,36]. Finding effective and safe co-adjuvant treatments to help eradicate infections and reduce the likelihood of recurrence could be of significant benefit. The frequent occurrence of these infections in the population requires the development of alternative treatments to effectively eliminate Candida species and, at the same time, prevent recurrences without inducing unwanted side effects.
The novelty of this study should be underlined as current investigations of the beneficial effects of pea protein and grape seed extract on vaginal mucosa and inflammation are limited to preclinical studies. Indeed, the authors of this study investigated for the first time the efficacy and safety of a topical PGL formulation compared with topical lactic acid alone, in combination with clotrimazole vaginal tablets in a cohort of patients affected by VVC. The main findings of the study are that PGL is a safe treatment, since no systemic or local side effects were reported, and that, in agreement with pre-clinical data obtained in a murine model of VVC [27], co-adjuvant treatment with PGL resulted in a significant reduction of the mycological count, suggesting that addition of pea protein and grape seed extract reinforces the activity of lactic acid in controlling vaginal infections.
Patients affected by C. albicans infections experience a high degree of discomfort due to itching, vaginal discharge and soreness, symptoms caused by both inflammation and damage to the vaginal mucosa [6]. Accordingly, these symptoms - both severe and moderate - were represented in the patients participating in the study. One of the most bothersome complaints reported by patients suffering from VVC is itching. Notably, the topical application of PGL resolved this symptom within 2 days of treatment; PGL was more effective than lactic acid in relieving severe itching and vaginal discharge [25,26].
Complaints of VVC peak when patients report dysuria and dyspareunia. By 5 and 6 days after treatment, all cases of moderate dysuria and dyspareunia had disappeared in the group treated with PGL compared to those treated with lactic acid alone. The positive effects induced by PGL are clinically important, as they may contribute to improve women’s psychological condition by reducing depression and anxiety associated with VVC. Maintenance of proper vaginal pH is critical for neutralizing potentially infectious microorganisms [1,37]. Importantly, only PGL was able to restore vaginal pH to an optimal level.
The beneficial effects of the formulation investigated in the present study could be attributed to the restoration of barrier activity of the vaginal mucosa due to the presence of mucomimetic substances. Indeed, grape seed extract has been shown to contribute to the repair of a damaged epithelium by restoring tight junctions [35], while pea proteins are known for their mucoadhesive and film-forming properties [25] which help to maintain the integrity of the vaginal mucosa, essential to prevent local inflammation, pathogen invasion, and subsequent recurrence [27-29].
Due to the complexity of the study design (i.e., patients enrollment, follow-up, etc.), this study has evident limitations: the small number of patients and the lack of a control group treated only with the antifungal drug. Nevertheless, due to the statistically significant difference between the PGL and lactic acid groups, the results are solid enough to support the potential benefits of PGL as an adjunct to clotrimazole in the treatment of VVC symptoms. Overall, these results suggest that the use of the PGL formulation could be very helpful as adjunctive treatment to support standard pharmacological therapies in treating VVC and preventing its recurrence; for this reason, the results seem to confirm the evidence of PGL safety and efficacy for the management of patients with VVC. It may also be interesting to explore PGL as a therapeutic option for other types of vaginal infections, particularly those thought to be due to an imbalance in the vaginal microbiota. Future studies should examine the efficacy of PGL in a larger cohort of patients with RVVC to confirm these findings. In addition, it would be useful to study the efficacy of PGL without concurrent antifungal therapy to possibly reduce the use of azoles and thus avoid further selection of drug-resistant species.
Disclosure
The authors have no competing interests with Devintec SAGL and were independent clinicians selected by CEBIS International SRL. All Investigators declares that they received investigator honoraria for this study.
Acknowledgement
We acknowledge the activity of the Contract Research Organization (CRO) for the study performance, monitoring and statistical analysis (CEBIS International SRL; 47 Theodor Pallady, Helios Business Centre | Entrance B, 3rd Floor, 032275 Bucharest, Romania).
References
- Gonçalves B, Ferreira C, Alves CT, Henriques M, Azeredo J, et al. (2016) Vulvovaginal candidiasis: Epidemiology, microbiology and risk factors. Crit Rev Microbiol 42(6): 905-927.
- Tachedjian G, Aldunate M, Bradshaw CS, Cone RA (2017) The role of lactic acid production by probiotic Lactobacillus species in vaginal health. Res Microbiol 168(9-10): 782-792.
- Smith SB, Ravel J (2017) The vaginal microbiota, host defence and reproductive physiology. J Physiol 595(2): 451-463.
- Giraldo PC, Polpeta NC, Juliato CR, Yoshida LP, do Amaral RL, et al. (2012) Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med 9(3): 805-811.
- Bilardi JE, Walker S, Temple-Smith M, McNair R, Mooney-Somers J, et al. (2013) The burden of bacterial vaginosis: women's experience of the physical, emotional, sexual and social impact of living with recurrent bacterial vaginosis. PLoS One 8(9): e74378.
- Ehrström S, Kornfeld D, Rylander E (2007) Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosom Obstet Gynaecol 28(3): 169-176.
- Yano J, Sobel JD, Nyirjesy P, Ryan S, Williams VL, et al. (2019) Current patient perspectives of vulvovaginal candidiasis: incidence, symptoms, management and post-treatment outcomes. BMC Womens Health 19(1): 48.
- Leusink P, Kaptheijns A, Laan E, van Boven K, Lagro-Janssen A, et al. (2016) Comorbidities Among Women With Vulvovaginal Complaints in Family Practice. J Sex Med 13(2): 220-225.
- Fukazawa EI, Witkin SS, Robial R, Vinagre JG, Baracat EC, et al. (2019) Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet 300(3): 647-650.
- Aballéa S, Guelfucci F, Wagner J, Amine K, Jean-Paul D, et al. (2013) Subjective health status and health-related quality of life among women with Recurrent Vulvovaginal Candidosis (RVVC) in Europe and the USA. Health Qual Life Outcomes 11: 169.
- Anderson MR, Klink K, Cohrssen A (2004) Evaluation of vaginal complaints. JAMA 291(11): 1368-1379.
- Mtibaa L, Fakhfakh N, Kallel A, Belhadj S, Belhaj SN,et al. (2017) Vulvovaginal candidiasis: Etiology, symptomatology and risk factors. J Mycol Med 27(2): 153-158.
- Mayer FL, Wilson D, Hube B (2013) Candida albicans pathogenicity mechanisms. Virulence 4(2): 119-128.
- Sundstrom P, Balish E, Allen CM (2002) Essential role of the Candida albicans transglutaminase substrate, hyphal wall protein 1, in lethal oroesophageal candidiasis in immunodeficient mice. J Infect Dis 185(4): 521-530.
- Martin H, Kavanagh K, Velasco-Torrijos T (2021) Targeting adhesion in fungal pathogen Candida albicans. Future Med Chem 13(3): 313-334.
- Hoyer LL, Cota E (2016) Candida albicans Agglutinin-Like Sequence (Als) Family Vignettes: A Review of Als Protein Structure and Function. Front Microbiol 7: 280.
- Núñez-Beltrán A, López-Romero E, Cuéllar-Cruz M (2017) Identification of proteins involved in the adhesion of Candida species to different medical devices. Microb Pathog 107: 293-303.
- Nobile CJ, Johnson AD (2015) Candida albicans Biofilms and Human Disease. Annu Rev Microbiol 69: 71-92.
- Rodríguez-Cerdeira C, Martínez-Herrera E, Carnero-Gregorio M, López-BarcenasA, Gabriella F, et al. (2020) Pathogenesis and Clinical Relevance of Candida Biofilms in Vulvovaginal Candidiasis. Front Microbiol 11: 544480.
- Richardson JP, Willems HME, Moyes DL, Saeed S, Barker KS, et al. (2018) Candidalysin Drives Epithelial Signaling, Neutrophil Recruitment, and Immunopathology at the Vaginal Mucosa. Infect Immun 86(2): e00645-17.
- Willems HME, Lowes DJ, Barker KS, Palmer GE, Peters BM, et al. (2018) Comparative Analysis of the Capacity of the Candida Species to Elicit Vaginal Immunopathology. Infect Immun 86(12): e00527-18.
- Fisher JF, Sobel JD, Kauffman CA, Newman CA (2011) Candida urinary tract infections--treatment. Clin Infect Dis 52(Suppl 6): S457-S466.
- Benitez LL, Carver PL (2019) Adverse Effects Associated with Long-Term Administration of Azole Antifungal Agents. Drugs 79(8): 833-853.
- Arendrup MC, Patterson TF (2017) Multidrug-Resistant Candida: Epidemiology, Molecular Mechanisms, and Treatment. J Infect Dis 216(suppl_3): S445-S451.
- Utrilla MP, Peinado MJ, Ruiz R, Rodriguez-Nogales A, Francesca A, et al. (2015) Pea (Pisum sativum L.) seed albumin extracts show anti-inflammatory effect in the DSS model of mouse colitis. Mol Nutr Food Res 59: 807-819.
- Gupta M, Dey S, Marbaniang D, Pal P, Ray S, et al. (2020) Grape seed extract: having a potential health benefits. J Food Sci Technol 57: 1205-1215.
- Esposito E, Campolo M, Casili G, Marika L, Alessia F, et al. (2018) Effect of pea protein plus grape seed dry extract on a murine model of Candida albicans induced vaginitis. Future Microbiol 13: 1375-1382.
- Campolo M, Lanza M, Filippone A, Irene P, Giovanna C, et al. (2020) Evaluation of a Product Containing Xyloglucan and Pea Protein on Skin Barrier Permeability. Skin Pharmacol Physiol 33(4): 231-236.
- Han Y (2007) Synergic effect of grape seed extract with amphotericin B against disseminated candidiasis due to Candida albicans. Phytomedicine 14(11): 733-738.
- O'Hanlon DE, Moench TR, Cone RA (2011) In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infect Dis 11: 200.
- Edwards VL, Smith SB, McComb EJ, Tamarelle J, Bing M, et al. (2019) The Cervicovaginal Microbiota-Host Interaction Modulates Chlamydia trachomatis Infection mBio 10: e01548-19.
- Hearps AC, Tyssen D, Srbinovski D, BayiggaL, Diaz DJD, et al. (2017) Vaginal lactic acid elicits an anti-inflammatory response from human cervicovaginal epithelial cells and inhibits production of pro-inflammatory mediators associated with HIV acquisition. Mucosal Immunol 10(6): 1480-1490.
- Liu W, Zhang X, Liu Z, Luo X (2011) Impact of pH on the antifungal susceptibility of vaginal Candida albicans. Int J Gynaecol Obstet 114(3): 278-280.
- Nyirjesy P, Peyton C, Weitz MV, Mathew L, Culhane JF, et al. (2006) Causes of chronic vaginitis: analysis of a prospective database of affected women. Obstet Gynecol 108(5): 1185-1191.
- Moshfeghy Z, Tahari S, Janghorban R, Najib FS, Mani A, et al. (2019) Association of sexual function and psychological symptoms including depression, anxiety and stress in women with recurrent vulvovaginal candidiasis. J Turk Ger Gynecol Assoc 21(2): 90-96.
- Akimoto-Gunther L, Bonfim-Mendonça Pde S, Takahachi G, Mary MT, et al. (2016) Highlights Regarding Host Predisposing Factors to Recurrent Vulvovaginal Candidiasis: Chronic Stress and Reduced Antioxidant Capacity. PLoS One 11(7): e0158870.
- Nallathambi R, Poulev A, Zuk JB, Raskin I (2020) Proanthocyanidin-Rich Grape Seed Extract Reduces Inflammation and Oxidative Stress and Restores Tight Junction Barrier Function in Caco-2 Colon Cells. Nutrients 12(6): 1623.






























