Consequences of Maternal, Neonatal, And Prognostic Factors for Severity of Covid-19 in Pregnant Women
Mitra Darbandi1, Kamyar Mansori2, Badieh Pakdin1, Farid Najafi1, Ebrahim Shakiba1 and Fatemeh Khosravi Shadmani1*
1Research Center for Environmental Determinants of Health (RCEDH), Kermanshah University of Medical Sciences, Iran
2Department of Biostatistics and Epidemiology, Zanjan University of Medical Sciences, Iran
Submission: June 06, 2022;Published: June 14, 2022
*Corresponding author: Fatemeh Khosravi Shadmani, Research Center for Environmental Determinants of Health (RCEDH), Health Institute, Kermanshah University of Medical Sciences, Kermanshah, Iran
How to cite this article: Mitra Darbandi, Kamyar Mansori, Badieh Pakdin, Farid Najafi, Ebrahim Shakiba, Fatemeh Khosravi Shadmani. Consequences of Maternal, Neonatal, And Prognostic Factors for Severity of Covid-19 in Pregnant Women. J Gynecol Women’s Health 2022: 23(4): 556117. DOI: 10.19080/JGWH.2022.23.556117
Abstract
Background: The aim of this study was to evaluate the consequences of pregnancy, neonatal, and prognostic factors of the severity of pregnant women with COVID-19 in Kermanshah province, west of Iran.
Methods: This historical cohort study was conducted on all pregnant women with COVID-19 in Kermanshah province from April 2020 to April 202. The data collection tool was a checklist including demographic, epidemiological, clinical, laboratory, radiological, and therapeutic variables that were extracted through interviews with patients and their medical records. Univariate and Multivariable Logistic Regression model were used to data analysis by STATA 14 software.
Results: In this study, 375 pregnant women were evaluated with COVID-19 infection, severity of the disease was mild in 54.4% (204 women) and severe in 45.6% (171 women). A total of 21 pregnant women were hospitalized that 5 of whom were admitted to the ICU. Also, the most common neonatal events including low birth weight (4.8%) and spontaneous abortion (4.0%) was the most common consequence of pregnancy complications. Hospitalization (7.1 (1.6-32.0)) and distress (8.5 (4.0-17.8)) were the most important risk factors of the severity.
Conclusion: COVID-19 infection in pregnant women can increase preterm delivery, low birth weight, vertical transmission, cesarean delivery and spontaneous abortion. In addition, the clinical manifestations of pregnant women with COVID-19 were similar to those of general COVID-19 patients. Is necessary to increase the care of pregnant women during the COVID-19 epidemic and to receive specialized care in case of infection covid-19.
Keywords: COVID 19; Pregnant woman; Neonatal; Complication; Spontaneous abortion
Abbreviations: SD: Standard Deviation; OR: Odds Ratio; CI: Confidence Interval; LBW: Low Birth Weight; SARS: Severe Acute Respiratory Syndrome; MGP: Multiple Gestation Pregnancy
Introduction
The novel coronavirus epidemic called COVID-19 started in late 2019 in China and quickly spread to other parts of the world and turned to a pandemic [1-3]. This highly contagious virus infects all population strata. According to the WHO, on November 16, 2021, the new case and deaths were 254631818 and 5123367 in the world and 6045212 and 128272 in Iran, respectively [4] One of the most sensitive groups at risk during the COVID-19 pandemic is pregnant women and their fetuses [5-8]. In recent decades, epidemics of infectious diseases have incurred harmful effects of the viruses on pregnant women and their fetuses [9,10]. The H1N1 flu and the Z1KA epidemics are examples with severe complications in pregnant women and their fetuses [10-14].
Studies have shown pregnancy does not increase the risk of developing COVID-19, but the complications can be more severe and COVID-19 can take longer as compared with non-pregnant individuals [15,16]. It is not clear whether the trend of natural immunological changes in women during pregnancy can increase the incidence and exacerbation of COVID-19 signs and symptoms. Studies have reported that developing COVID-19 in pregnant women increases the risk of ICU admission, especially in mothers who develop kidney failure and sepsis in the third trimester [15,17,18]. Other complications reported in pregnant women with COVID-19 include increased rates of preterm childbirth and cesarean section. Fever and hypoxemia may increase the risk of preterm childbirth, premature rupture of membranes, and abnormal fetal heart patterns. Preterm childbirth also occurs in cases without severe respiratory disease [6,19].
The spread of the coronavirus has raised concerns about its intrauterine transmission from mother to fetus in pregnant women [1,20,21]. However, a number of studies rule out this possibility [22,23]. Viral pneumonia caused by any infectious agent is one of the leading causes of death during pregnancy worldwide and is the most common infection (excluding obstetric infections) in pregnant women [24]. Currently, the following questions are raised regarding COVID-19 in pregnant women: what are the signs and symptoms of COVID-19 and how severe are they in pregnant women? What are the odds of vertical transmission? And what is the status of the pregnant mother and her fetus after 40 days? Accordingly, the present study was conducted to evaluating the consequences of maternal, neonatal, and prognostic factors for severity of COVID-19 in pregnant women Kermanshah Province, west of Iran.
Materials and Methods
Study design and Subjects
TThe present research was a cohort study which designed and performed to determine the outcomes of pregnancy and neonatal in pregnant women with COVID-19 and the factors affecting its severity. The study population was all pregnant women with COVID-19 who were confirmed by RT-PCR test or CT scan test in Kermanshah province from April 2020 to April 2021.Considering all pregnant women with COVID-19 were examined during the above-mentioned period, the sampling method was census and there was no need to calculate the sample size. The main criteria for inclusion in the study included a pregnant woman with COVID-19 approved by RT-PCR test or CT scan test. To conduct the study, pregnant women with COVID-19 were included in the study from the time of diagnosis to three months after delivery and then examined monthly. It should be noted that the study was open cohort and pregnant women with COVID-19 could enter the study during the study.
Data collection
A researcher-made checklist was used to collect data from pregnant mothers. The checklist variables included demographic, epidemiological, clinical, laboratory, radiological, and therapeutic data which was preceded by relevant studies and consultation with experts. Demographic and clinical data were collected through interviews with the patient or her companion and the rest of the information was extracted from the patient’s medical record. Data were collected from the time of diagnosis to three months after delivery.
Statistical analysis
Data were analyzed using Stata software version 14.0 (Stata Corp, College Station, TX, USA). For descriptive analyses, the mean, standard deviation (SD), and number (%) were used. Then, univariate and multivariate logistic regression model was used to determine the predictors of the severity of COVID-19 in pregnant women and finally crude and adjusted odds ratio (OR) with 95% confidence interval (CI) were estimated. P-value <0.05 was considered as a significant level.
Ethics considerations
This study was performed according to the principles expressed in the Declaration of Helsinki and was approved by the Deputy of Research and Ethics Committee of Kermanshah University of Medical Sciences (Iran) (ID: IR.KUMS. REC.1399.718).
Result

In this research, carried out in the Iranian province of Kermanshah, 375 pregnant women with COVID-19 infection were studied. The subjects were diagnosed using RT-PCR test. Also, the consequences of maternal, neonatal, and prognostic factors for severity of COVID-19 in pregnant women were investigated. It worth mentioning that the pregnant women were studied at all gestational ages. (Table 1) has shown the characteristics of the pregnant women. The total number of 204 (54.4%) and 171 (45.6%) women in the study were diagnosed with mild and severe cases of covid-19 infection respectively. Plus, the mean age of pregnant women was 36.6±7.2. Although women with milder illnesses were younger, there was no statistically significant difference between the two groups of patients in terms of age (p=0.147).

A total of 21 pregnant women were hospitalized, out of whom 4 were diagnosed with mild cases of Covid 19 which revealed a significant difference between the groups (0.001) in terms of hospitalization. Of the hospitalized patients, 5 had severe COVID-19 infections and were admitted to the ICU. The median of ICU stay for these patients was calculated to be 3 (0- 5) days. Also, 72.5% of the patients had a history of exposure to the patient with Covid-19 which showed a significant difference between severe and mild case groups (p<0.001). Plus, in 33 (8.8%) patients, lung involvement was observed via CT scan; of these cases, 25 had severe disease and were significantly different from patients with mild cases (p <0.001). The most common presenting symptoms were loss of smell (59.7%), muscular pain (44.8%), and loss of taste (41.6%). These symptoms were significantly different between the severe and mild groups (Table 1). (Table 2) has indicated the results of the study; showing that 121 (32.3%) women had a cesarean delivery.
Also, neonatal related complications including low birth weight (LBW) (4.8%), preterm delivery (0.5%) and vertical transmission (0.5%) were significantly higher in women with severe cases compared to those with mild COVID-19 cases. In addition, spontaneous abortion (4.0%) was the most common pregnancy complications in the two groups. (Table 3) has indicated the prognostic factors for severity in patients infected with Covid 19. Hospitalization (7.1 (1.6-32.0)), loss of smell (2.3 (1.1-4.7)), muscular pain (3.6 (2.1-6.2)), fever (3.2 (1.8-5.7),headache (3.3 (1.8-6.1)), distress (8.5 (4.0-17.8)), and cough(2.8 (1.5-5.3)) were counted as the risk factors. According to the finding, for instance, women with muscular pain are 3.6 times more likely to be diagnosed with a severe case of COVID-19 (Table 3).
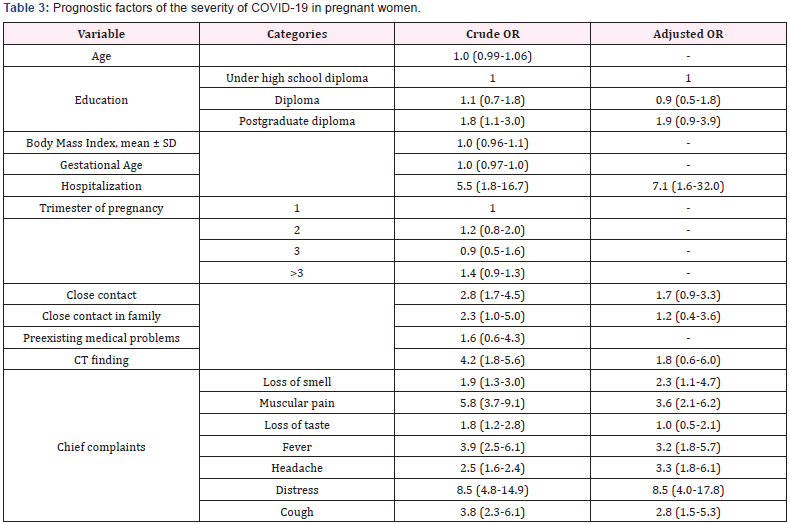
Discussion
The aim of this study was to determine the consequences of pregnancy, neonatal and prognostic factors of the severity of pregnant women with COVID-19 in Kermanshah province, west of Iran. In this study, 375 pregnant women were evaluated with COVID-19 infection, severity of the disease was mild in 54.4% (204 women) and severe in 45.6% (171 women). In addition, the results of this study demonstrated that neonatal events including LBW, preterm delivery and vertical transmission of COVID-19 were significantly higher in women with sever disease compared to the mild type. In addition, severe cases were significantly higher in participant who were in close contact with patient Covid-19. Previous studies have shown that pregnant women with COVID-19 infection are associated with increased preterm delivery and LBW.
A study in Wuhan-China has shown that compared to pregnant women without COVID-19, pregnant women with a confirmed COVID-19 diagnosis had an increased risk of preterm birth (OR 3.34, 95% CI 1.60-7.00) [18]. A study by Wong SF et al. have been showed that %80 pregnant women with severe acute respiratory syndrome (SARS) had preterm delivery [25]. It was reported in the study by Lee et al. that the prevalence of preterm delivery increased in women with COVID-19 infection [6]. In a systematic review, preterm delivery was calculated about 25% [26]. As well as the LBW in infants born to pregnant mothers with COVID-19 has been reported in the Lassi et al. study 16.6% and in the Abedzadeh-Kalahroudi et al. study 34.6% [27,28]. Therefore, preterm delivery and LBW are adverse outcomes in pregnant women with COVID-19 infection and these two consequences can cause other side effects for the infant.
In this study, vertical transmission was observed in two pregnant women confirmed COVID-19 diagnosis. The vertical transfer rate in the study of Chi J et al. was reported to be 3.91% by SARS-CoV-2 nucleic acid tests [26]. Although, some studies show that there is no evidence for intrauterine infection caused by vertical transmission in women who develop COVID-19 pneumonia in late pregnancy [29,30]. However, the possibility of vertical transmission cannot be ruled out. In a study, the laboratory findings displaying inflammation and liver damage in a neonate indirectly support this possibility of vertical transmission [30]. Studies that have examined this association have often been cross-sectional, case-report or based on medical records. Therefore, the results should be reported with caution and further investigation is needed.
In this study, the percent of caesarean section was higher than vaginal delivery (25.3% vs. 32.3%).
Although, the mode of delivery was not significantly different in patients with severe and mild of COVID-19 infection. Studies on pregnant women with Covid-19 infection have shown that cesarean section is more common than vaginal delivery [26,31]. In addition, a population-based cohort study in Wuhan on 1,1078 participants, show that compared to pregnant women without COVID-19, pregnant women with a confirmed COVID-19 diagnosis had an increased risk of cesarean section (OR 3.63, 95% CI 1.95-6.76) [18]. However, caesarean section is not a recommended method of childbirth in pregnant women with COVID-19 infection. A systematic review study by Cai J et. al. has reported the rate of neonatal COVID-19 infection, neonatal deaths, and maternal deaths is no greater when the mother gave birth through vaginal delivery [32]. Our findings show that 15 women pregnant (4%) with COVID-19 infection had spontaneous abortion, this outcome 6.1% was reported in Russia [33].
A study in India showed spontaneous abortion in singleton pregnancy 1.7% and in multiple gestation pregnancy (MGP) 10% [34]. Therefore, it is necessary to increase the care of pregnant women during the COVID-19 epidemic and to receive specialized care in case of infection covid-19. The findings of this study showed that the clinical manifestations of pregnant women with COVID-19 were similar to those of general COVID-19 patients [35,36]. In pregnant women with sever Covid-19 infection, symptoms of the disease including cough, fever, loss of smell, muscular pain, headache and distress were more than patients with mild severity. According to the importance of vertical transfer, the lack of sufficient information on vertical transfer to infants was a limitation in this study. The absence of a control group was another limitation of the present study. Large sample size and number of significant variables were the strengths of the study.
Conclusion
The findings of this study demonstrated that COVID-19 infection in pregnant women can increase preterm delivery, LBW, vertical transmission, cesarean section and spontaneous abortion. In addition, severe cases were significantly higher in participant who were in close contact with patient COVID-19. The clinical manifestations of pregnant women with COVID-19 were similar to those of general COVID-19 patients.
Acknowledgements
We thank the colleagues of the Deputy Minister of Health and the Deputy Minister of Treatment at Kermanshah University of Medical Sciences.
References
- Berghella V (2020) Coronavirus disease 2019 (COVID-19): Pregnancy issues.
- Carlson A, Thung SF, Norwitz ER (2009) H1N1 influenza in pregnancy: what all obstetric care providers ought to know. Rev Obstet Gynecol 2(3): 139-145.
- Keshavarz AT, Vahdat K, Mohsen (2020) Novel coronavirus disease 2019 (COVID-19): an emerging infectious disease in the 21st century. Iranian South Medical Journal 22(6): 432-450.
- (2021) Worldometer. Coronavirus.
- Di Mascio D, Khalil A, Saccone G, Rizzo G, Buca D, et al. (2020) Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol MFM 2(2): 100107.
- Liu H, Wang L-L, Zhao S-J, Kwak-Kim J, Mor G, et al. (2020) Why are pregnant women susceptible to COVID-19? An immunological viewpoint. J Reprod immunol 139: 103122.
- Zaigham M, Andersson O (2020) Maternal and perinatal outcomes with COVID‐19: a systematic review of 108 pregnancies. Acta Obstet Gynecol Scand 99(7): 823-829.
- Schwartz DA (2020) The Effects of Pregnancy on Women With COVID-19: Maternal and Infant Outcomes. Clin Infect Dis 71(16): 2042-2044.
- Alfaraj SH, Al-Tawfiq JA, Memish ZA (2019) Middle East Respiratory Syndrome Coronavirus (MERS-CoV) infection during pregnancy: Report of two cases & review of the literature. J Microbiol Immunol Infect 52(3): 501-503.
- Omer SB, Beigi RH (2016) Pregnancy in the time of Zika: addressing barriers for developing vaccines and other measures for pregnant women. Jama 315(12): 1227-1228.
- Ticconi C, Pietropolli A, Rezza G (2016) Zika virus infection and pregnancy: what we do and do not know. Pathog Glob Health 110(7-8): 262-268.
- Schwartz DA (2019) Maternal and infant death and the rVSV-ZEBOV vaccine through three recent Ebola virus epidemics-West Africa, DRC Equateur and DRC Kivu: 4 years of excluding pregnant and lactating women and their infants from immunization. Current Tropical Medicine Reports 6(4): 213-222.
- Fuentes-Zacarias P, Murrieta-Coxca JM, Gutierrez-Samudio RN, Schmidt A, Schmidt A, et al. (2021) Pregnancy and pandemics: Interaction of viral surface proteins and placenta cells. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1867(11): 166218.
- Marban-Castro E, Gonce A, Fumado V, Romero-Acevedo L, Bardaji A (2021) Zika virus infection in pregnant women and their children: A review. Eur J Obstet Gynecol Reprod Biol 265: 162-168.
- Kourtis AP, Read JS, Jamieson DJ (2014) Pregnancy and infection. N Engl J Med 370(23): 2211-2218.
- Panagiotakopoulos L, Myers TR, Gee J, Lipkind HS, Kharbanda EO, et al. (2020) SARS-CoV-2 infection among hospitalized pregnant women: reasons for admission and pregnancy characteristics-eight US health care centers, March 1–May 30, 2020. 2020. MMWR Morb Mortal Wkly Rep 69(38): 1355-1359.
- Rasmussen SA, Smulian JC, Lednicky JA, Wen TS, Jamieson DJ (2020) Coronavirus Disease 2019 (COVID-19) and Pregnancy: What obstetricians need to know. A J Obstet Gynecol 222(5): 415-426.
- Yang R, Mei H, Zheng T, Fu Q, Zhang Y, et al. (2020) Pregnant women with COVID-19 and risk of adverse birth outcomes and maternal-fetal vertical transmission: a population-based cohort study in Wuhan, China. BMC Medicine 18(1): 1-7.
- Banaei M, Ghasemi V, Naz MSG, Kiani Z, Rashidi-Fakari F, et al. (2020) Obstetrics and neonatal outcomes in pregnant women with COVID-19: a systematic review. Iran J Public Health 49(Suppl 1): 38-47.
- Wang X, Zhou Z, Zhang J, Zhu F, Tang Y, et al. (2020) A case of 2019 Novel Coronavirus in a pregnant woman with preterm delivery. Clin Infect Dis 71(15): 844-846.
- Shaoshuai W, Lili G, Ling C, Weiyong L, Yong C, et al. (2020) A case report of neonatal COVID-19 infection in China. Clin Infect Dis 71(15): 853-857.
- Mounesan L, Eybpoosh S, Haghdoost A, Moradi G, Mostafavi E (2020) Is reporting many cases of COVID-19 in Iran due to strength or weakness of Iran’s health system? Iran J Microbiol 12(2): 73-6.
- Zhu H, Wang L, Fang C, Peng S, Zhang L, et al. (2020) Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Transl Pediatr 9(1): 51-60.
- (2020) Novel coronavirus situation report-2. Organization WH.
- Wong SF, Chow KM, Leung TN, Ng WF, Ng TK, et al. (2004) Pregnancy and perinatal outcomes of women with severe acute respiratory syndrome. A J Obstet Gynecol 191(1): 292-297.
- Chi J, Gong W, Gao Q (2021) Clinical characteristics and outcomes of pregnant women with COVID-19 and the risk of vertical transmission: a systematic review. Arch Gynecol Obstet 303(2): 337-45.
- Abedzadeh-Kalahroudi M, Sehat M, Vahedpour Z, Talebian P, Haghighi A (2021) Clinical and obstetric characteristics of pregnant women with Covid-19: A case series study on 26 patients. Taiwan J Obstet Gynecol 60(3): 458-62.
- Lassi ZS, Ana A, Das JK, Salam RA, Padhani ZA, et al. (2021) A systematic review and meta-analysis of data on pregnant women with confirmed COVID-19: Clinical presentation, and pregnancy and perinatal outcomes based on COVID-19 severity. J Glob Health 11-05018.
- Chen H, Guo J, Wang C, Luo F, Yu X, et al. (2020) Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. The lancet 395(10226): 809-15.
- Dong L, Tian J, He S, Zhu C, Wang J, et al. (2020) Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn. Jama 323(18): 1846-1848.
- Giaxi P, Maniatelli E, Vivilaki VG (2020) Evaluation of mode of delivery in pregnant women infected with COVID-19. Eur J Midwifery 4: 28.
- Cai J, Tang M, Gao Y, Zhang H, Yang Y, et al. (2021) Cesarean section or vaginal delivery to prevent possible vertical transmission from a pregnant mother confirmed with COVID-19 to a neonate: a systematic review. Front Med 8: 634949.
- Shmakov RG, Prikhodko A, Polushkina E, Shmakova E, Pyregov A, et al. (2020) Clinical course of novel COVID-19 infection in pregnant women. J Matern Fetal Neonatal Med Pp: 1-7.
- Mahajan NN, Ansari M, Gaikwad C, Jadhav P, Tirkey D, et al. (2021) Impact of SARS‐CoV‐2 on multiple gestation pregnancy. International Journal of Gynecology & Obstetrics 152(2): 220-5.
- Ge H, Wang X, Yuan X, Xiao G, Wang C, et al. (2020) The epidemiology and clinical information about COVID-19. Eur J Clin Microbiol Infect Dis 39(6): 1011-1019.
- Guan W-j, Ni Z-y, Hu Y, Liang W-h, Ou C-q, et al. (2020) Clinical characteristics of coronavirus disease 2019 in China. New England Journal of Medicine 382(18): 1708-1720.






























