Dysmenorrhea: Pathophysiology and Treatment with Physiological Modulators
Hernández Santos JR1, Cornelli U2* and Recchia M3
1Centro Medico Nacional de 20 Noviembre, Av Féliz Cuevas, Mexico
2Loyola University Medical School – Chicago, USA
3Medstat, Via A Salaino 7, Milan Italy
Submission: February 04, 2021; Published: February 14, 2022
*Corresponding author: Umberto Cornelli, Loyola University Medical School – Chicago, USA
How to cite this article: Wendy K. The Donor Sibling Registry: Connecting Donor-Conceived People with Their Half-Siblings and Donors. J Gynecol Women’s Health 2022: 23(1): 556103. DOI: 10.19080/JGWH.2022.23.556103
Abstract
Background: Dysmenorrhea is a condition which affects 45% of menstruating women on average.
Objective: Assessment of the clinical activity of a combination of physiological modulators (A28).
Material and methods: Ninety-eight women were treated for two months, after a two-month period of stable dysmenorrhea as determined according to sixteen behavioral and sixteen somatic symptoms each measured on a scale ranging from 0 to 5. As soon as symptoms began, the women were treated with a combination of physiological modulators (A28) at a dose of six capsules (2 x 3) on the first day, followed by four capsules (2 x 2) on the second day. The treatment was repeated for two subsequent months. At the end of each monthly period, the women were asked to assess the efficacy of the treatment (satisfaction) according to a score from 0 to 4. The statistical calculations were based on the chi-squared method and Markovian algorithm.
Result: The two baseline measurements provided very similar data (p>0.05), and the same was true after two months of treatment (p>0.05). The activity of A28 was evident in over 70% of the women without any side effects. The relief was particularly evident for somatic symptoms such as colic, headache and back pain, and for behavioral symptoms such as anger, irritability, mood swing, and crying easily. The overall assessment of the therapy was at least satisfactory in more than 85% of cases.
Conclusion: Physiological modulators are effective in the treatment of dysmenorrhea.
Keywords: Dysmenorrhea; Physiological modulators; Behavioral symptoms; Somatic symptoms
Abbreviations: ECD: Extra Cellular Dominion; VAS: Visual Analogue Scale; BBB: Blood Brain Barrier; NSAIDs: Nonsteroidal Anti-Inflammatory Drugs
Introduction
Dysmenorrhea affects about half of menstruating women during their menses [1], causing an extensive personal and public health problem consisting of daily discomfort, absenteeism, and severe economic loss. The symptoms usually start the day before the onset of menses and persist for 2 to 3 days. There are various symptoms, both somatic (e.g., colic pain, hot flashes, etc.) and behavioral (e.g., sadness, aggression, etc.). Although the disease can sometimes be caused by pelvic abnormalities, in the majority of cases it is primary and cannot be explained by structural gynecological disorders. The pain is thought to result from uterine contraction and ischemia, probably mediated by prostaglandins or other inflammatory mediators produced by the secretory endometrium. We should also consider the involvement of GABAA receptors, widespread in the CNS and GI tract [2,3], and CaSRs (calcium sensing receptors), which are more frequently found in the stomach, colon, bones, kidneys, and parathyroid gland [4,5].
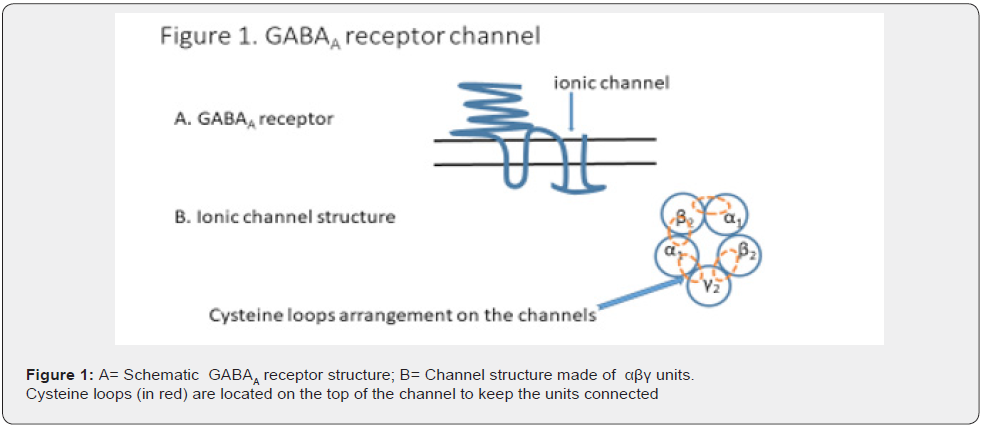
GABAA receptors have an inhibitory action and have estrogens and progestogens as ligands. The receptor contains an ionic channel which is composed of three different subunits α1 ,β2, and γ2, bound through cysteine residuals. Hormone levels decline just before and for a few days after the menses and the inhibitory activity of the receptors is highly reduced. This event gives rise to an uncontrolled release of neurotransmitters and inflammatory mediators thus causing the common peripheral and central symptoms found in dysmenorrhea.
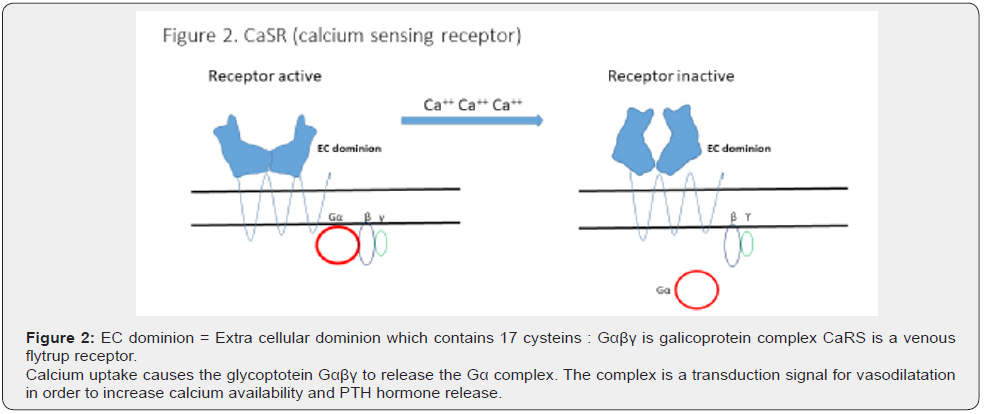
CaSRs may be indirectly involved, mainly due to the calcium shortage induced by the fall in hormone levels. CaSR modulation is considered important in the GI tract to prevent the dysregulation of contractions [6].
The two kinds of receptor, GABAA and CaSRs, have common characteristics since their activity may also be reduced by oxidative stress (OS). This is dependent on the integrity of the double bonds of cysteines found in the ionic channels of GABAA receptors, and in the ECD (extra cellular dominion) of CaSRs (Figure 1&2). OS affects the cysteines which are key to maintaining the quaternary structure of the structural glycoproteins found in both receptors (Figures 1&2). Both events, hormone shortage and OS, are responsible for many dysmenorrhea symptoms. In theory, an increase in antioxidant capacity in the gut and CNS should improve the activity of GABAA receptors and CaSRs by reducing the transfer of OS from one compartment to the other.
This transfer may be caused by circulating lymphocytes, which are activated and can reach every tissue. Although lymphocytes have a large set of antioxidant enzymes [7], OS can overcome their antioxidant capacity turning them into vehicles of oxidation. This can be controlled by increasing their antioxidant capacity. Furthermore, the increase in calcium availability may keep CaSRs inactive and prevent the alteration of GI blood flow and stimulation of PTH secretion. This investigation aims to analyze the activity of some physiological modulators (PMs) [8,9] in controlling the symptoms caused by dysmenorrhea.
Material and Methods
The Product
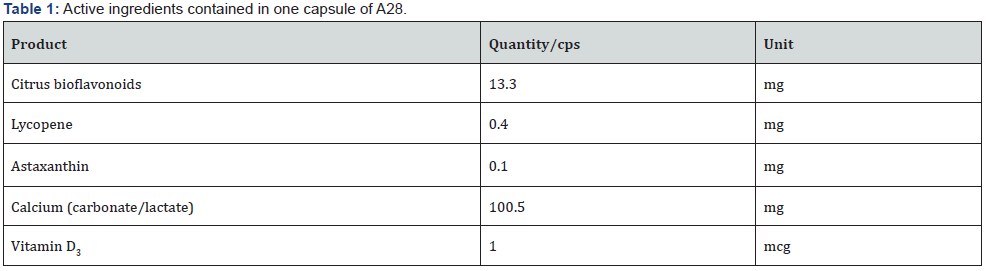
Table 1 shows the formula of the physiological modulators (A28). The product was chosen on the basis of the pharmacological characteristics of each of its components. Citrus bioflavonoids: to reduce the OS in the GI tract [10] and protect the ionotropic GABAA receptors [11,12]. Lycopene: to protect lymphocytes from oxidative damage [13] and limit the transfer of oxidation into the circulation and tissues. Astaxanthin: to protect neurons from oxidative stress [11,15]. Calcium lactate and calcium carbonate: to make calcium available to the CaSRs in the GI tract. Vitamin D3: to improve calcium absorption. The dosage of the antioxidant components (citrus bioflavonoids, lycopene and astaxanthin) was determined in order to provide an antioxidant capacity [16,17] of at least 500 U.Cor. for each compound. The total combined antioxidant capacity of the citrus bioflavonoids, lycopene and astaxanthin contained in A28 was 1500 U.Cor. In terms of ascorbic acid, a value of 1500 U.Cor. can be achieved with about 80 mg.
Subjects
Women of between 20 and 35 years of age whose menarche occurred at 12-16 years old were admitted.
Symptoms and Scores
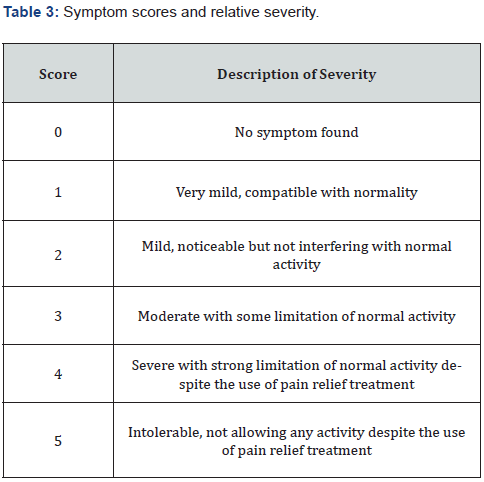
The severity of dysmenorrhea was measured according to 32 symptoms, some somatic and others behavioral as shown in Table 2. Each symptom was given a score of between 0 and 5 using a 10 cm VAS (visual analogue scale) as shown in Table 3.

Admission Criteria
The evaluation was carried out over three days each time during two immediately subsequent menses. Only women with at least three symptoms with scores of over 3 were admitted, provided the scores were constant or that the difference was no greater than one point between the two assessments.
Exclusion Criteria
Subjects suffering from any form of cancer, who had undergone bariatric surgery, or with chronic gastrointestinal disease (Crohn’s disease, irritable bowel syndrome, etc.), allergy, intolerance, or with a BWI of less than 19 or greater than 35. Women with Alzheimer’s disease, depression, anorexia, and paranoia were also excluded. Other diseases, such as hypertension and dyslipidemia, were not considered exclusion factors if the therapy prescribed was effective, safe, and ongoing for at least three months. Women taking an oral contraceptive were not excluded provided the contraceptive and dosages had been the same for at least six months.
Treatment
Two capsules of product were administered the moment the symptoms began, followed by two more capsules every six hours (total of six capsules in the first day). During the following day, two capsules of product were administered every twelve hours (total of four capsules). Two boxes containing ten capsules each were given to each patient. Pain relief was allowed only if the product was not effective (no reduction or worsening of the symptoms).
Assessment of the Activity

The assessment was carried out in the first and second months of the baseline and treatment periods. The scores were calculated on the third day of menses. Each score consisted of the severity of the symptoms over the three days. At the end of each month of treatment the subjects were asked to complete a questionnaire with a score of between 0 and 4 (satisfaction score) to assess the activity of the product (Table 4).
Compliance
Compliance was measured by counting the capsules left in the boxes given to the women.
Statistical Analysis
A previous preventive study with the same product and the same score assessment system [18] suggested that a total of twenty cases was sufficient to achieve a discrimination power (1- β) of 0.8 with α = 0.05. In this study, a total of 60 cases was sufficient to increase the discrimination power (1-β) to 0.9 with α = 0.05. A non-parametric exact Fisher test (chi squared test) was used together with Markovian chains to measure the changes in symptom scores [19]. JMP 14 pro (Sas Institute corporation) was used for all the calculations.
Result
All 98 women completed the study. Compliance was 100% and no side effects were reported. The first evaluation consisted of comparing the scores recorded at baseline and the same scores after two months of treatment. The two baseline scores were similar (chi-squared p>0.05), as were those after two months of treatment (chi-squared p>0.05). The difference between the two periods was statistically significant (p<0.001) suggesting that the treatment was very effectively reducing almost all the symptoms. Tables 5 & 6 show the disappearance or improvement in symptoms (Markovian algorithm) (Table 7).
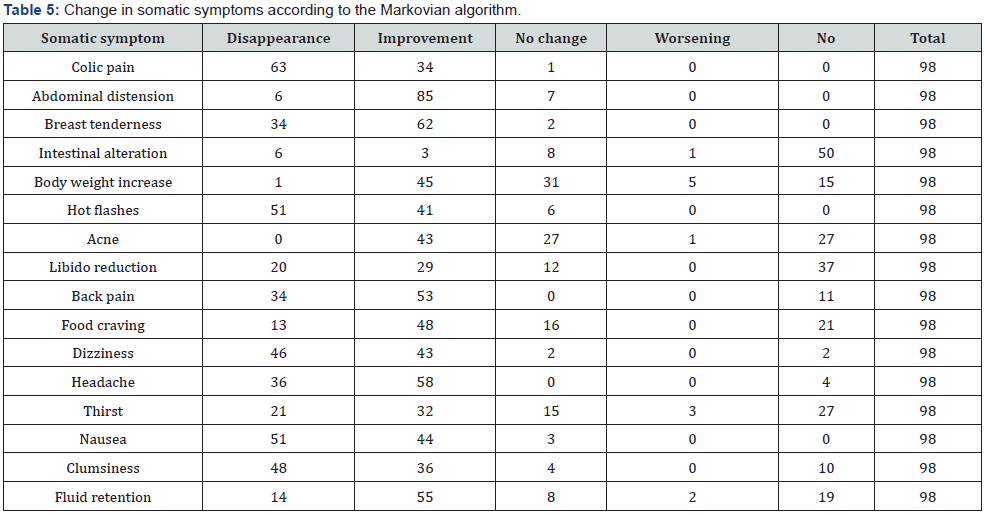
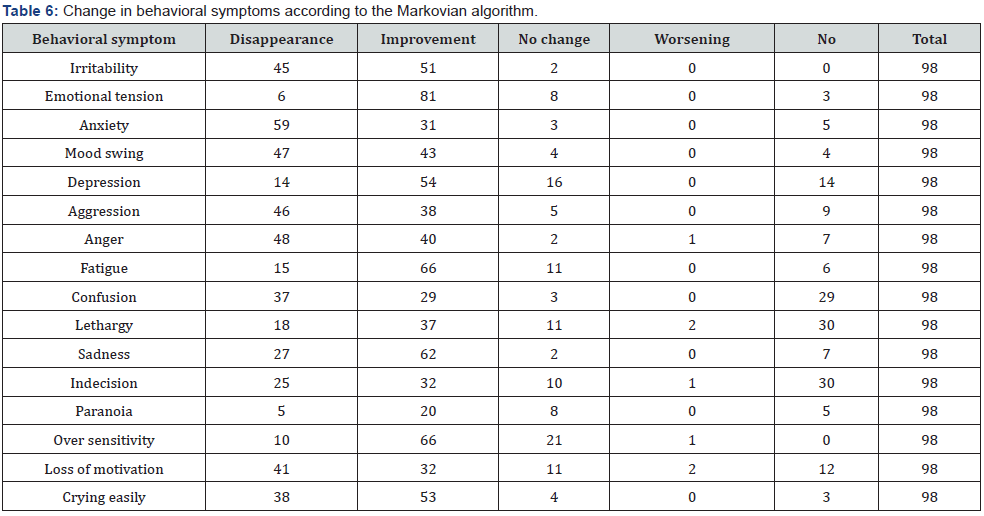
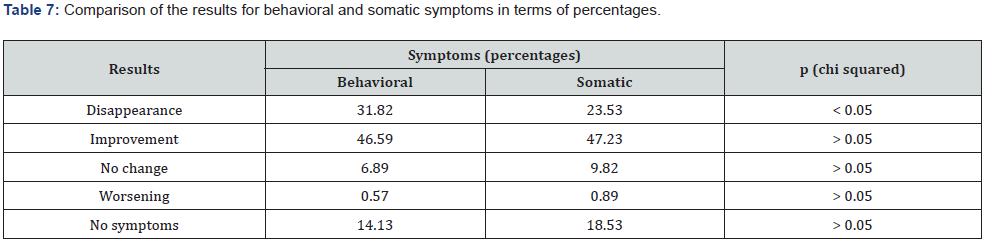
A more positive effect was found for the disappearance of behavioral symptoms (p<0.05). In terms of improvement, the activity is similar for both kinds of symptom. The same is true for worsening, which was limited to less than 1% of cases. Pain relief treatment was needed in 8 cases only (8.4%). The best improvements among the somatic symptoms were seen for colic pain, headache, back pain, breast tenderness, nausea, hot flashes, and dizziness (Table 5). The best results among the behavioral symptoms were found for irritability, mood swing, anger, sadness and crying easily.
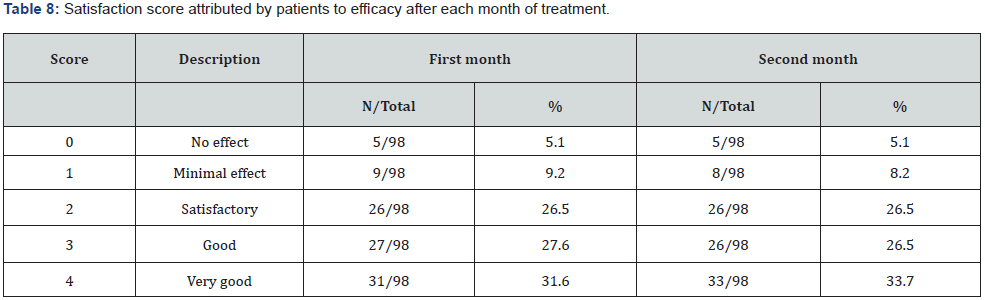
Table 8 shows the patients’ assessment. The patients’ assessment is almost identical in both months (chi-squared p> 0.8) and the efficacy was considered at least satisfactory in more than 85% of cases.
Discussion
Our study confirms previous clinical pharmacolgy findings on dysmenorrhea based on the assessment of daily discomfort using similar protocols [19]. Considering the complete picture in terms of improvement and disappearance, the behavioral symptoms seem to be more positively affected (78.4% and 70.8% respectively), but the difference is not statistically significant (chi-squared p>0.05). Among the somatic symptoms, pain in all its forms and nausea improved the most. As regards behavioral symptoms, irritability, mood swing, anger, sadness, and crying easily were more effectively reduced. The therapy was considered by the patients to be at least satisfactory in more than 85% of cases.
The efficacy of the product in controlling pain, nausea, breast tenderness and hot flashes is consistent with a synergistic effect on the behavioral symptoms. In other words, the improvement in somatic symptoms may lead to a decrease in the behavioral ones, which apparently seem more affected. It is more likely that a reduction in pain causes the disappearance of irritability, mood swing and anger than the other way around. Our data show that the use of PMs is a good approach to the treatment of dysmenorrhea, particularly since the results are achieved with natural compounds, with very low dosages of each component and without any report of side effects. All the components could be taken just following a diet with the difference that most of the time it is hard for women to follow a diet during an episode of dysmenorrhea.
Considering that the dosage on the first day was six capsules, it is hard to take the same total quantities (79.8 mg of bioflavonoids, 2.4 mg of lycopene, 0.6 mg of astaxanthin, 602 mg of calcium and 6 mcg of Vitamin D3) by simply eating, particularly in the case of food repulsion. Many dysmenorrhea treatments have been investigated, including changes to lifestyle (rest, sleep, diet, etc.) and - if necessary - drug support.
The use of isoflavones in the diet has given conflicting results [20,21], which may be attributed to ethnic differences or bias due to total energy intake. Hormonal treatment using an oral contraceptive, with progestins (e.g., levorgestrel, medroxyprogesterone, etc.), or gonadotropin-releasing hormone agonist, is frequently used together with the addition of NSAIDs (nonsteroidal anti-inflammatory drugs) to control the pain due to prostaglandin inhibition. Hypnosis and other non-drug therapies have also been proposed such as acupuncture, chiropractic therapy, and transcutaneous electrical nerve stimulation. Our approach was different, based on the control of GABAA receptors and CaSRs to avoid OS damage. The membranes of all receptors are particularly sensitive to oxidation, which may alter the structure of the rafts (phospholipids and proteins) that support their location and function.
Furthermore, when there are several cysteines in the protein complex, such as in the ionic channels and in the ECD, the double bonds are easily opened, causing the quaternary structure to alter. These effects compromise the neuronal activity of GABAA, and CaSR calcium intake.
OS tends to spread out from the GI tract and reach the circulation, and only efficient quenching of the reactive substances (e.g., O2•, OH•, etc.) can counteract its spread. Similarly, if the OS is generated in the brain, it may transfer to the rest of the body. The antioxidant systems found in CNS and GI cells may not be sufficient to provide protection, despite the presence of circulating antioxidants (e.g., albumin, uric acid, ascorbic acid, etc.) and the activity of the internal antioxidant system (e.g., GSH).
The intake of antioxidants with food is usually not sufficient since most of them (e.g., bioflavonoids) are only active locally in the GI tract and do not cross the blood brain barrier (BBB). This is what the astaxanthin in the formula we used is for; it is known to cross the BBB and reach the neuronal membranes. For peripheral oxidation control, we focused our attention on lycopene, which is one of the essential antioxidants that protect lymphocytes from oxidation.
Dosages are fundamental for any antioxidant, since it is known that every antioxidant could become a prooxidant if the quantity administered is excessive. For example, high dosages of ascorbic acid can carbonylate proteins making them inefficient and prooxidant [22], and vitamin E at high dosages can antagonize the activity of vitamin K [23].
For this reason, low dosages have been used in A28. Are they sufficient for the activity required? The astaxanthin dosage of 0.6mg corresponds to 1017 molecules; even if we assume only 0.1% availability in the brain, this comes to somewhere in the range of 1014 molecules of astaxanthin, which means that each brain cell (about 1 x 1011) may have about 1000 molecules of astaxanthin available to it. Since these molecules are susceptible to metabolism and excretion, their levels must be renewed with subsequent administrations.
The same applies for all the other antioxidant components.
Lycopene administration aims at supporting the antioxidant capacity of lymphocytes, which account for 20-30% of all leucocytes. They number between 4 x 106 and 1 x 107 in the blood. Lycopene, together with β-carotene and lutein, are the most abundant carotenoids found in human serum and tissues. Epidemiological data suggest that lycopene may be associated with an array of chronic conditions [24]. At least 34% is absorbed [25], which means that no less than 1 x 1017 molecules will be available for circulating cells at a dose of 2.4 mg. This quantity is more than sufficient to bind all the circulating cells, lipoproteins, and platelets.
In physiological terms, it seems that the real problem of any PM is not the quantity available but its specific antioxidant activity, which derives from the characteristics of its components. In other words, OS cannot be reduced by simply taking vitamin C or vitamin E, particularly when these compounds are taken at high doses.
In the end, each chronic disease must be tackled with appropriate antioxidants given at appropriate doses.
Those active in the case of OS during menses [26] are different from those needed in the case of hypothyroidism treated with levothyroxine [27-29], or used together with oral contraceptives [30], or used in the case of anemia due to menstrual blood loss [31,32], or needed for the therapy of Alzheimer’s disease [33] or used to improve the antioxidant capacity of saliva [34].
The oxidative quenching is different if it is caused by mitochondrial dysfunction than if it is caused by amyloid induced damage to the brain (e.g., Alzheimer’s disease). Specific PMs with antioxidant activity must be used for each disease. It is essential from a clinical point of view to validate any PM composition in patients. This study shows that the pathophysiology of dysmenorrhea symptoms is susceptible to the clinical effect of an appropriate combination of PMs.
The Problem of A28 Dosage
In a previous clinical investigation [18], two capsules before menses began and two to eight capsules in the following two days were shown to reduce daily discomfort by 79%. The average number of capsules taken was seven (ranging from four to ten). This means that different subjects may have different sensitivity to the treatment. It cannot be ruled out that the product was not effective in some cases owing to the many unknown variables that cause dysmenorrhea. These needs analyzing in further studies.
In this study, we chose to fix the dosage at ten capsules in two days (six capsules on the first day and four capsules the following day) and to start the treatment when menses began. This dosage may overcome the problem of sensitivity to the treatment as far as is possible. We were able to do this because, even at high dosages, the amounts of the components of A28 are still in the range commonly found in the diet. According to the feedback from the patients, a dosage of two capsules was in most cases sufficient to reduce symptoms for about six hours. However, larger clinical trials with dose finding protocols are necessary to define this aspect more clearly.
Conclusion
The use of a combination of physiological modulators was shown to be effective in treating dysmenorrhea. A regular intake of four/six capsules from the onset of symptoms was found to reduce both behavioral and somatic symptoms in at least 70% of cases. The patients’ assessment was favorable in more than 85% of cases.
References
- Iacovides S, Avidon I, Baker FC 2015 What we know about primary dysmenorrhea today: a clinical review. Hum Rep Update 21(6): 762-778.
- Foresti W. GABA receptors. Biotrend Rev 20111 no 7/1.
- Sigel E, Michael ES (2012) Structure, function, and modulation of GABAA J Biol Chem 287(48): 40224-40231.
- Alfadda TI, Saleh AMA, Houillier P, Gelbel JP (2014) Calcium-sensing receptors 20 years later. Am J Cell Physiol 307(3): C221-C231.
- Hanna FM, Kallay E, Chang W, Maria LB, Rajesh VT, et al. (2018) Calcium-sensing receptor in physiology and calcitropic and non-calcitropic diseases. Nat Rev Endocrinol 15(1): 33-51.
- Thakker RV (2012) Calcium-sensing receptor: role in heath and disease. Ind J Endocrinol Metab 16(suppl 2): S213-S216.
- Fisher G, Schwartz DD, Quindry J, Barberio MD, Foster EB, et al. (2011) Lymphocyte enzyme antioxidant response to oxidative stress following high-intensity interval exercise. J Appl Physiol 110(3): 730-737.
- Olson JA (1996) Benefit and liabilities of Vitamin A and carotenoids. J Nutr 126 (suppl 4): 1208S-1212S.
- Cornelli U. I Modulatori Fisiologici 2010 MediService Cava Manara PV-Italy.
- Mahmoud AM, Hernández BTJ, Saudun MA, Huissein OE (2019) Beneficial effect of Citrus Bioflavonoids on cardiovascular and metabolic health. Ox Med Cell Longevity.
- Hanrahan JR, Chebib M, Johnston GA (2015) Interaction of flavonoids with ionotropic GABA receptors. Adv Pharmacol 72: 189-200.
- Johnston GA (2015) Flavonoids nutraceuticals and ionotropic receptors for the inhibitory neurotransmitter GABA. Neurochen Int 89: 120-125.
- Porrini M, Riso P (2000) Lymphocyte lycopene and DNA protection from oxidative damage in women after a short period of tomato consumption. J Nutr 130(2): 189-192.
- Galasso C, Orefice I, Pallone P, Paola C, Roberta M, et al. (2018) On neuroprotective role of astaxanthin: new perspective? Mar Drugs 16(8): 247-251.
- Fakri S, Aneva IY Farzaei MF, Sorbaro Sánchez (2019) The neuroprotective effect of astaxanthin: therapeutic targets and clinical perspective. Molecules 24(14): 2640-2658.
- Cornelli U, Belcaro G, Nardi GM, Cesarone MR, Dugall M, et al. (2010) Action of an antioxidant complex on the antioxidant power of saliva. Pan Med 52(2 Suppl 1): 69-73.
- Serena B, Mariangela P, Annarosa F, Franco C, Umberto C, et al. (2014) Validation of a patented method to determine the antioxidant capacity of human saliva based on the reduction of iron: the SAT test. Clin Lab 60(3): 475-482.
- Belcaro G, Cornelli U, Hernández Santos JR (2020) Treatmennt of dysmenorrhea with Physiological modulators (A28): a registry study. J Preg Women’s Health Care 1(1): 1-4.
- Caracciolo di Forino A (1968) String processing languages and generalized Markov algorithms.In: DG Bobrow (Ed.), In Symbol manipulation languages and techniques, North-Holland Publ Co, Netherlands, pp. 191-206.
- Kim HW, Kwon MK, Kin NS, Reame NE (2006) Intake of soy isoflavones in relation to perimenstrual symptoms of Korean women living in the USA. Nursing Health Sci 8(2): 108-113.
- Nagata C, Hirokawa K, Shimizu N, Shimizy H (2005) Association of menstrual pain with intakes of soy, fat and dietary fiber in Japanese women. Eur J Clin Nutr 59(1): 88-92.
- Arballo J, Amengual J, Erdman JW (2021) Lycopene: A Critical Review of Digestion, Absorption, Metabolism, and Excretion. Antioxidants 10(3): 342-360.
- Anderesen M, Regueira T, Bruhn A, Druso P, Pablo S, et al. (2008) Lipoperoxidation and protein oxidative damage exhibit different kinetics during septic shock. Mediators of inflammation.
- Booth SL, Golly I, Sacheck JM, Ronenn R, Gerard ED, et al. (2004) Evvect of vitamin E supplementation on vitamin K in adults with normal coagulation status. Am J Clin Nutr 80: 143-148.
- Diwadkar NV, Novotny JA, Gusttin DM, Jeffery AS, Keith AR, et al. (2003) A physiological pharmacokinetic model describing the disposition of lycopene in healthy men. J Lipid Research 44: 1927-1939.
- Cornelli U, Belcaro G, Cesarone MR, Finco A (2013) Analysis of oxidative stress during menstrual cycle. Repr Biol Endocrinol 11: 74.
- Cornelli U, Belcaro G, Ledda A, Feregalli B (2011) Activity of some physiological modulators in reducing the side effects of levothyroxine in patients suffering from hypothyroidism. Pan Med 53 (3suppl 1): 99-103.
- Cornelli U, Belcaro G, Ledda A, Feregalli B (2011) Oxidative stress following administration of levothyroxine in subjects suffering from primary hypothyroidism. Pan Med 53 (3 suppl 1): 95-98.
- Cornelli U, Ledda A, Belcaro G, Finco A (2015) Activity of a combination of physiological modulators in limiting side effects in patients suffering from primary hypothyroidism during levothyroxine treatment. Austin J Nutr Metab 2(2): 1018.
- Finco A, Belcaro G, Cesarone MR (2011) Evaluation of oxidative stress after treatment with low estrogen contraceptive either alone or associated with specific antioxidant therapy. Contraception 85(5): 503-508.
- Cornelli U, Belcaro G, Fareed J (2016) Use of physiological modulators in the management of anemia due to menstrual blood loss. Jacobs J Hemat 2: 025.
- Cornelli U, Belcaro G (2015) Treatment of anemia owing to increased menstrual blood loss: activity of Physiological Modulators. J Hematology 4(2): 164-170.
- Cornelli U (2010) Treatment of Alzheimer’s disease with a cholinesterase inhibitor combined with antioxidants. Neurodegener Dis 7(1-3): 193-202.
- Cornelli U, Belcaro G, Cesarone MR, GM Nardi, M Dugall, et al. (2010) Action of an antioxidant complex on the antioxidant power of saliva. Pan Med 52(2 suppl 1): 69-73.






























