Prognostic impact of serum markers and BRCA mutation on histological types of ovarian cancer
Bizoń Magdalena*, Zielińska Aleksandra and Cendrowski Krzysztof
Chair and Department of Obstetrics, Gynecology and Gynecological Oncology, Medical University of Warsaw, Poland
Submission: October 28, 2021; Published: November 12, 2021
*Corresponding author: Bizoń Magdalena, Chair and Department of Obstetrics, Gynecology and Gynecological Oncology, Medical University of Warsaw, Poland
How to cite this article: Bizoń M, Zielińska A, Cendrowski K. Prognostic impact of serum markers and BRCA mutation on histological types of ovarian cancer. J Gynecol Women’s Health 2021: 22(4): 556091. DOI: 10.19080/JGWH.2021.22.556091
Abstract
Morbidity of ovarian cancer has been increasing for years. The lack of specific and sensitive methods of diagnosis causes delays in diagnosis. The most frequently used markers in the diagnostic process are CA125, HE4 and the Risk of Ovarian Malignancy Algorithm (ROMA). The aim of the study was to evaluate the correlation between the value of markers used in primary diagnosis of ovarian cancer and histological type of cancer and genetic status of patients with ovarian cancer.
Materials and Methods: The analysis was based on 55 patients from the clinic with a diagnosis of ovarian cancer with mean age 60.78±12.99 (median: 63.25 years). Each patient underwent surgical treatment. In every case tissue of ovarian tumor was investigated for BRCA mutation using NGS (next generation sequencing) methods.
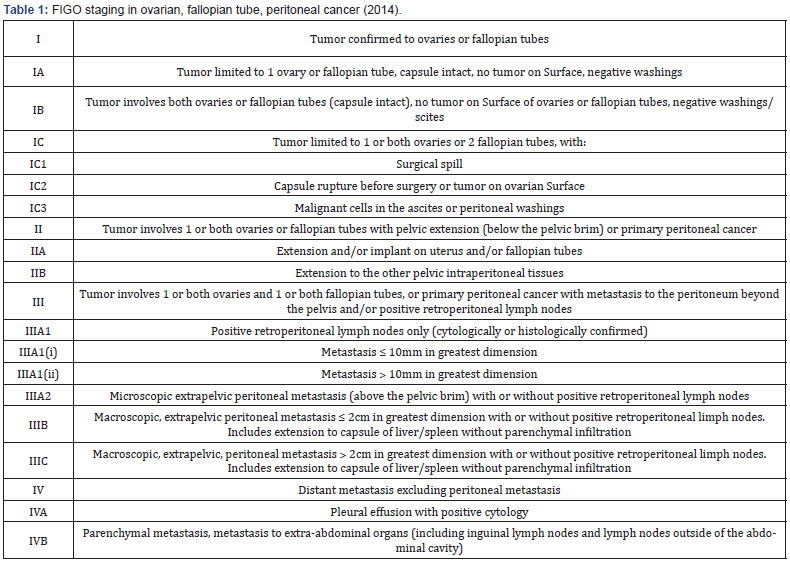
Results: Level of CA125 at the time of diagnosis was statistically significant for serous (p=0.006) and endometrioid (p=0.01) ovarian cancer. A significant level of HE4 was observed in serous ovarian cancer (p = 0.021). There was a significant correlation between the calculated value of the ROMA algorithm and serous type of ovarian cancer (p=0.02).
Conclusion: CA125, which is correlated with histological type of tumor, has a significant role in monitoring the effectiveness of chemotherapy. Presence of BRCA mutation is a risk factor of morbidity of ovarian cancer and is not restricted to only one histological type. This is a preliminary report, which will be continued.
Keywords: Ovarian cancer; BRCA mutation; CA125 in ovarian cancer; HE4 in ovarian cancer
Introduction
Ovarian cancer is the fourth most common neoplastic disease in women. Morbidity has been increasing for years. In Poland over 3500 new diagnoses are made every year. In most cases ovarian cancer is diagnosed in an advanced stage due to lack of symptoms in the early stage of disease. The lack of specific and sensitive diagnostic methods causes delays in diagnosis. The most frequently used laboratory test is cancer antigen 125 (CA125), which is a protein used to assess the risk of ovarian cancer diagnosis and follow up the treatment [1]. CA125 was first described in the 1980s as carbohydrate antigen 125. The upper limit of CA125 in reproductive and postmenopausal age was 35 U/ml. Nevertheless, sensitivity of CA125 in the early stage of ovarian cancer was lower. In stage I disease CA125 was elevated by 23-50% [2].
Unfortunately, CA125 is not specific to ovarian cancer and can also be elevated in endometriosis, fibroids or pregnancy, whereas in cirrhosis of the liver it occurs together with ascites. Especially, it is elevated in about 80% of epithelial ovarian cancer cases in advanced stages. The normal range of CA125 was observed in 50% of early-stage cases of ovarian cancer [3]. For better assessment, glycoprotein HE4 (human epididymis protein 4) was introduced [4]. Preoperative assessment of a pelvic mass tumor is highly important to choose the best way of treatment. Zhang et al. reported that the highest level of identification of the tumor is through using a combination of transvaginal ultrasound, color Doppler and serum markers. The sensitivity was calculated as 90.63%, specificity 97.14%. Positive prediction was assessed as 93.94%, negative as 98.55% [5].
Zheng et al. confirmed sensitivity and specificity of CA125 as 64.29% and 53.57% in stage 1 and 2 and 91.43% and 88.57% in stage 3 and 4 of ovarian cancer, respectively. Sensitivity and specificity of HE4 were assessed as 46.4% and 43.3% in stage 1 and 2 and 88.6% and 49.2% in stage 3 and 4 of disease [6]. For differential diagnosis also CA19-9, CEA and estradiol are used. However, there is no specific marker for diagnosis of ovarian cancer.
Progression and treatment of ovarian cancer depend on many factors. Firstly, histological type of disease plays a significant role. The most frequent is the serous one, which in most cases is also correlated with BRCA mutation. The second one is endometrioid type of ovarian cancer and then clear cell type. Both types - endometrioid and clear cell - can be affected by long-term endometriosis. Wentzensen et al. [7] observed in their study a correlation between risk factors and different histological types of ovarian cancer [7]. In every case of ovarian cancer the genetic background should be evaluated. Especially BRCA mutation is often observed. Genetic predisposition for morbidity influences progression of the disease and early onset. New methods of treatment, e.g. PARP inhibitors or immunotherapy, can be used in these cases so it is necessary to know the genetic status of ovarian tumor. The aim of the study was to determine the correlation between the value of markers used in primary diagnosis of ovarian cancer and histological type of cancer and genetic status of patients with ovarian cancer.
Materials and Methods
The study is a preliminary report, where further investigation is needed. Analysis was based on 55 patients from the clinic with diagnosis of ovarian cancer with mean age 60.78±12.99 (median: 63.25 years). Each patient underwent surgical treatment. After the surgical procedure patients were classified according to the FIGO classification from stage IC to IVA [Table 1]. Histological type of ovarian cancer was confirmed after the hematoxylin and eosin staining protocol and immunohistochemistry procedures. Patients were divided into 2 groups. Group A consisted of 36 women with serous type of ovarian cancer with mean age 62.76±12.09 (median: 64.58 years). Group B consisted of 19 women with endometrioid type with mean age 58.13±14.95 (median: 53.8 years). In every case tissue from the ovarian tumor was investigated for BRCA mutation using NGS (next generation sequencing) methods. In all cases adjuvant chemotherapy based on carboplatin and paclitaxel was initiated after surgical treatment.
Laboratory Tests
Before the operation laboratory tests were performed. Value of CA125 (normal range: 0-35 U/ml), HE4 (with normal range below 70pmol/L for premenopausal women and 140pmol/L after menopause) were monitored. In every case the ROMA algorithm was used for calculation of the risk of ovarian cancer according to reproductive or postmenopausal age. Higher risk of epithelial ovarian cancer related to ROMA is calculated as more than 25.3% for postmenopausal women and more than 7.4% in reproductive age. Other tests performed in differential diagnosis were: CA19- 9 (normal range: 0-37 U/ml), CEA (0-5ng/ml) and estradiol (normal range in correlation to menstrual cycle). After 6 courses of chemotherapy the value of CA125 was assessed to monitor the effectiveness of treatment.
Statistical Methods
To test significant differences between groups, Student’s t-test and the Mann-Whitney test were used. P<0.05 was considered to indicate a statistically significant difference. Also the chi-square test was used due to compare two features.
Results
Value of CA125
The level of CA125 was compared before the surgical procedure and after 6 courses of chemotherapy based on paclitaxel and carboplatin. In group A the mean value of CA125 was 1705.94±2468.67 IU/ml (median 380.5), and HE4 was 532.08±815.74 (median 127). After calculation the range of ROMA algorithm was calculated as 70.03%±31.66 (median 82.59%). The mean value of CA125 after chemotherapy decreased to 159.22±336.42 IU/ml (median 19.5). In group B mean value of CA125 was 364±699.18 (median 46), HE4 183.74±326.88 (median 56) .ROMA score was 42.16%±30.78 (median 25.6%). Mean value of CA125 after chemotherapy was 12.28±6.88 (median 10).
The level of CA125 at the time of diagnosis was statistically significant for serous (p=0.006) and endometrioid (p=0.01) type of ovarian cancer. Statistical significance of level of HE4 was observed in serous ovarian cancer (p = 0.021) [Figure 1].
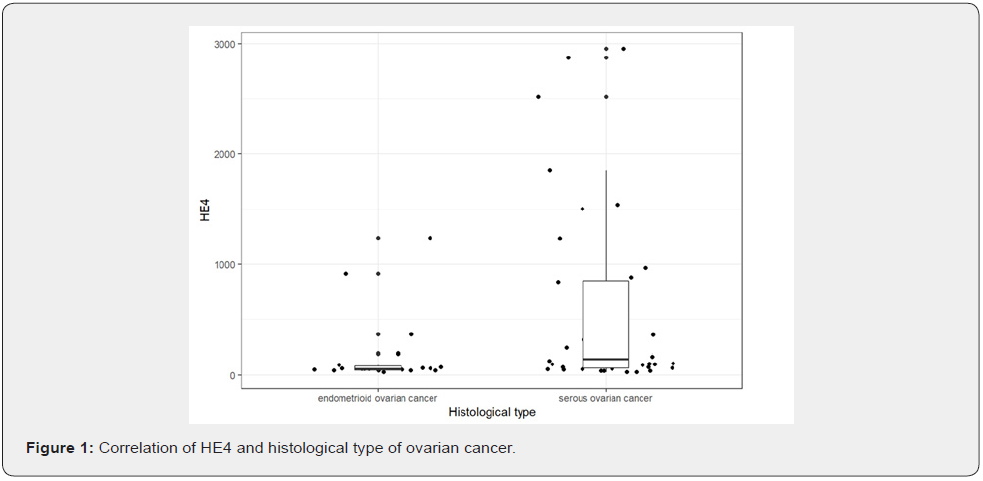
There was a significant correlation between the calculated value of the ROMA algorithm and serous type of ovarian cancer (p=0.02) [Figure 2].
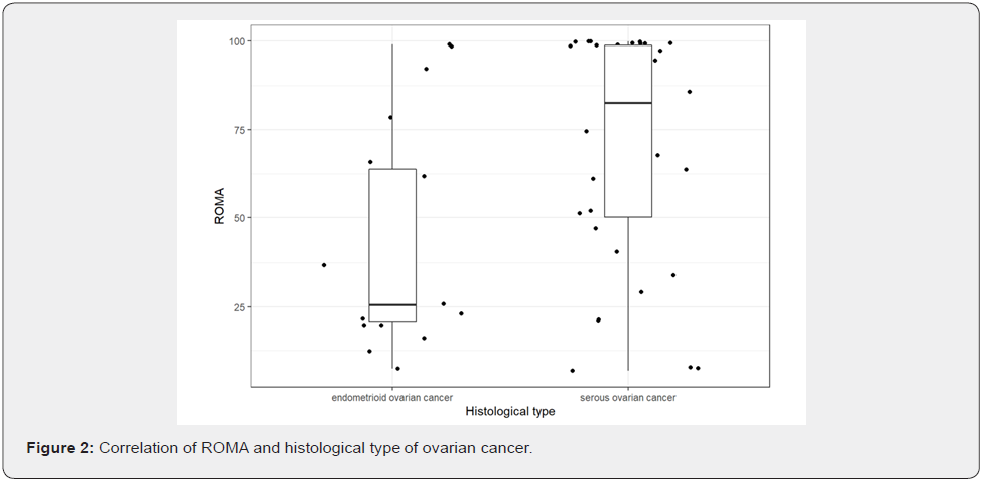
Statistical analysis revealed a higher value of CA125 after chemotherapy in serous type of ovarian cancer in comparison with endometrioid ovarian cancer. A decreasing level of CA125 was correlated with histological type (p<0.05) [Figure 3].
Value of Other Markers
Ca19-9 was in the normal range in 91.7% of group A and 89.5% of group B. In group A in 5.5% the level of CEA was increased. Otherwise, all women from group B were in the normal range of CEA. The correct value of estradiol was confirmed in all patients. There was no statistical correlation with histological type of ovarian cancer.
Genetic Tests
Presence of BRCA mutation in ovarian tumor was confirmed in 8.3% of group A and 15.8% of group B. Mean age of patients with serous type of ovarian cancer and BRCA mutation was 62.26±10.52 (median 64.31 years). Mean age of women with endometrioid type and BRCA positive was 46.79±3.35 (median 48.19 years).
There was no correlation of presence of BRCA mutation and histological type of ovarian cancer. No significant correlation was observed between presence of BRCA mutation and value of CA125 before the surgical procedure and after chemotherapy. The lack of statistical significance may be due to the small population size.
Discussion
Early diagnosis of ovarian cancer allows for quicker implementation of treatment. Extensive analysis performed in the United States of America revealed a correlation between histological types and effects on treatment and longer survival. Higher mortality is observed in carcinosarcoma. The most common ovarian cancers are high-grade serous type and endometrioid. As they are so frequent, they have been the target of many investigations [8].
In retrospective analysis in China sensitivity and specificity in suspicion of ovarian cancer were assessed as 93.2% and 87.5% [9]. Probability of malignancy is significant in preoperative assessment and qualification for treatment. Ferraro et al. assessed specificity of CA125 as 78% [10]. Grandi et al. [11] revealed CA125 in 1st stage ovarian cancer in 54.4%, which increased in the second stage to 78%. Higher specificity is observed in endometrioid type of ovarian cancer.
May et al. observed prognostic value of CA125 in high-grade serous ovarian cancer. The cut off value was defined as 174U/ml. Comparison of the preoperative and postoperative value of CA125 revealed a decreasing concentration of this marker [12]. Lee et al. [13] reported a correlation between lower value of CA125 after chemotherapy and longer overall survival in advanced ovarian cancer. In our analysis concentration of CA125 also decreased after the surgical procedure and then after 6 courses of chemotherapy. Moreover, in our research, a higher level of CA125 was observed in the serous type of ovarian cancer than in the endometrioid one.
The prognostic role of CA125 before and after treatment was investigated by Fadera et al. [14] In the first stage of ovarian cancer CA125 before treatment was not prognostic and had no influence on overall survival. However, during treatment the value of CA125 is normalized. The value of CA125 before the second line chemotherapy has an influence on decreasing risk of death because of ovarian cancer.
Shen et al. published a study with superior sensitivity of HE4 than CA125 in predicting the surgical outcome of primary debulking surgery and interval debulking surgery. They claimed that the change of HE4 during neoadjuvant chemotherapy could predict the outcome of interval debulking surgery [15]. Similarly, Steffensen et al. [16] revealed HE4 as a strong indicator of epithelial ovarian cancer. They observed a correlation between higher concentration of HE4 and worse prognosis of disease. According to our investigation, HE4 is statistically correlated with serous type of ovarian cancer. Further studies are necessary for better usage of this indicator.
The ROMA algorithm was created for better preoperative prediction of ovarian cancer according to presence of ovarian tumor. Calculation is correlated with reproductive or postmenopausal age. Sensitivity and specificity of ROMA for reproductive age were reported as 0.714 and 0.972, for postmenopausal age as 0.929 and 0.8. Nevertheless, no correlation was found according to histological type of ovarian cancer and stage of disease [17]. Simmons et al. [18] observed a positive role of the markers CA125, HE4, MMP-7 and CA 72-4 in early detection of ovarian cancer. We detected in our investigation a statistical significant correlation between the value of CA125 and serous and endometrial type of ovarian cancer.
The marker CA19-9 and its role in diagnosis of ovarian cancer were described by Zhang et al. [19] The aim of that study was to determine contrast enhanced ultrasound with CA19-9/CA125 to differentiate ovarian serous carcinoma from ovarian malignant epithelial cancer. Concentrations of CA19-9 and CA125 were significantly higher in ovarian malignant epithelial cancer. In our research no statistical significance of use of CA19-9 in ovarian cancer diagnosis was found.
Koshiyama et al. [20] described differential diagnosis of histological types of ovarian cancer according to classification of type I (endometrioid, clear cell, mucinous, low-grade serous) and II (high-grade serous). Presence of BRCA mutation was higher in II type ovarian cancer. In our analysis due to the smaller population estimation of gene mutation was not statistically significant.
Frequency of BRCA mutation was described by Zhang et al. [21] in analysis based on 1342 women. They confirmed coexistence of serous type of ovarian cancer and BRCA positive mutation in 18% of cases. No mutation was seen in mucinous carcinoma. Due to the smaller population, the percentage of positive BRCA mutation was lower. Kim et al. in their investigation found a correlation with positive BRCA mutation and longer overall survival in comparison to patients with no genetic background [22].
On the other hand, Zheng et al. [23] conducted a study on familial risk of ovarian cancer. Early morbidity connected with familial background was observed especially in endometrioid and serous types of ovarian cancer. Raspollini et al. [24] reported correlations of CA125 and COX-2 with survival and clinical responsiveness to chemotherapy in ovarian cancer. Level of CA125 before onset of treatment was significant. Further investigations are necessary to better understanding the role of COX-2 in the pathomechanism of ovarian cancer.
Conclusion
Laboratory tests used in clinical practice for diagnosis of ovarian cancer are not sufficiently specific to differentiate histological types of disease. However, CA125 has a significant role in monitoring effectiveness after chemotherapy, which is correlated with histological type of tumor. Presence of BRCA mutation is a risk factor of morbidity of ovarian cancer and is not linked to only one histological type. Further investigations on a larger population are needed. Our investigation is an introduction to a long-term project. This preliminary report exposes the problem of lack of specific and sensitive methods to differentiate ovarian cancer and its histological types. In the future we are planning to publish a longer report based on our observations.
References
- Froyman W, Timmerman D (2019) Methods of assesing ovarian masses: International Ovarian Tumor Analysis Approach. Obstet Gynecol Clin North Am 46(4): 625-641.
- Hellstroem I, Raycraft J, Hayden-Ledbetter M (2005) The HE4 (WFDC2) protein is a biomarker for ovarian carcinoma. Cancer Res 56: 2162-2169.
- Dochez V, Caillon H, Vaucel E, Dimet J, Winer N, et al. (2019) Biomarkers and algorithms for diagnosis of ovarian cancer:Ca125, HE4, RMI and ROMA, a review. J Ovarian Res 12(1): 28.
- Ortiz-Munoz B, Aznar-Oroval E, Garcia AG, Peris AC, Ballestero PP, et al. (2014) HE4, Ca125 and ROMA algorithm for differentia diagnosis between benign gynaecological diseases and ovarian cancer. Tumour Biol 35(7): 7249-7258.
- Zhang F, Zhang ZL (2015) The Diagnostic Value of Transvaginal Sonograph (TVS), Color Doppler, and Serum Tumor Marker CA125, CEA, and AFP in Ovarian Cancer. Cell Biochem Biophys 72(2): 353-357.
- Zheng X, Chen S, Li L, Liu X, Liu X, et al. (2018) Evaluation of HE4 and TTR for diagnosis of ovarian cancer: comparison with Ca-125. J Gynecol Obstet Hum Reprod 47(6): 227-230.
- Wentzensen N, Poole EM, Trabert B, White E, Arslan AA, et al. (2016) Ovarian Cancer Risk Factors by Histologic Subtype: An Analysis From the Ovarian Cancer Cohort Consortium. J Clin Oncol 34(24): 2888-2898.
- Peres LC, Cushing-Haugen KL, Kobel M, Harris HR, Berchuck A, et al. (2019) Invasive epithelial ovarian cancer survival by histotype and disease stage. JNCI J Natl Cancer Inst 111(1): 1.
- Chen L, Wang X, Shu J, Xu S, Wu Q, et al. (2019) Diagnostic value of serum D-dimer, Ca125 and neutrophil-to-lymphocyte ratio in differentiating ovarian cancer and endometriosis. Int J Gynaecol Obstet 147(2): 212-218.
- Ferraro S, Braga F, Lanzoni M, Boracchi P, Biganzoli EM, et al. (2013) Serum human epididymis protein 4 vs carbohydrate antigen 125 for ovarian cancer diagnosis: a systematic review. J Clin Pathol 66(4): 273-281.
- Grandi G, Perrone A, Toss A, Vitagliano A, Friso S, et al. (2020) The generally low sensitivity of Ca125 for FIGO stage I ovarian cancer diagnosis increases for endometrioid histotype. Minerva Med 111(2):133-140.
- May T, Stewart JM, Bernardini MQ, Ferguson SE, Laframboise S, et al. (2018) The prognostic value if perioperative, presystemic therapy CA125 levels in patients with high-grade serous ovarian cancer. Int J Gynaecol Obstet 140(2): 247-252.
- Lee M, Chang MY, Yoo H, Lee KE, Chay DB, et al. (2016) Clinical Significance of CA125 Level after the First Cycle of Chemotherapy on Survival of Patients with Advanced Ovarian Cancer. Yonsei Med J 57(3): 580-587.
- Fadera AN, Javab J, Krivakc TC, Bristowd RE, Tergasa AI, et al. (2014) The prognostic significance of pre- and post-treatment CA-125 in grade 1 serous ovarian carcinoma: A Gynecologic Oncology Group. Gynecol Oncol 132(3): 560-565.
- Shen Y, Li L (2016) Serum HE4 superior to CA125 in predicting poorer surgical outcome of epithelial ovarian cancer. Tumour Biol 37(11): 14765-14772.
- Steffensen KD, Waldstrom M, Brandslund I, Jakobsen A (2011) Prognostic impact of prechemotherapy serum levels of HER2, CA125 and HE4 in ovarian cancer patients. Int J Gynecol Cancer 21(6): 1040-1047.
- Kim B, Park Y, Kim B, Ahn HJ, Lee K-A, et al. (2019) Diagnostic performance of CA125, HE4, adn risk of ovarian malignancy algorithm for ovarian cancer. J Clin Lab Anal 33(1): e22624.
- Simmons AR, Fourkala EO, Gentry-Maharaj A, Ryan A, Sutton MN, et al. (2019) Complementary longitudinal serum biomarkers to CA125 for early detection of ovarian cancer. Cancer Prev Res (Phila) 12(6): 391-400.
- Zhang W, Wang L, Xin Z (2018) Combination of serum CA19-9 and CA125 levels and contrast-enhanced ultrasound parametric data facilitates to differentiate ovarian serous carcinoma from ovarian malignant epithelial cancer. Medicine (Baltimore) 97(16): e0358.
- Koshiyama M, Matsumura N, Konishi I (2017) Subtypes of Ovarian Cancer and Ovarian Cancer Screening.
Diagnostics (Basel) 7(1): 12. - Zhang S, Royer R, Li S, McLaughlin JR, Rosen B, et al. (2011) Frequencies of BRCA1 and BRCA2 mutations among 1 342 unselected patients with invasive ovarian cancer. Gynecol Oncol 121(2): 353-357.
- Kim SI, Lee M, Kim HS, Chung HH, Kim JW, et al. (2019) Effect of BRCA mutational status on survival outcome in advanced-stage high-grade serous ovarian cancer. J Ovarian Res 12(1): 40.
- Zheng G, YuID H, Kanerva A, Forsti A, Sundquist K, et al. (2018) Familial risks of ovarian cancer by age at diagnosis, proband type and histology. PLoS ONE 13(10): e0205000.
- Raspollini MR, Amunii G, Villanucci A, Boddi V, Taddei GL, et al. (2006) COX-2 and preoperative CA-125 level are strongly correlated with survival and clinical responsiveness to chemotherapy in ovarian cancer. Acta Obstet Gynecol Scand 85(4): 493-498.






























