Syrenomelia: A Reported Intrauteral Case Secondary to Embryotoxicity with Phenylephrine
Ramon Miguel Vargas Vera1*, Martha Verónica Placencia Ibadango1, Kalid Stefano Vargas-Silva2, Daniela Zalamea Holguin1, Saul Alexander Alen Castro Placencia3 and Martin Chiriguaya-Castro4
1University of Guayaquil, USA
2Cepegen, USA
3Catholic University Santiago de Guayaquil, USA
4Hospital Matilde Hidalgo de Procel, USA
Submission: August 19, 2021; Published: August 24, 2021
*Corresponding author: Ramon Miguel Vargas Vera, University of Guayaquil, USA
How to cite this article: Ramon M V V, Martha V P I, Kalid S V-S, Daniela Z H, Saul A A C P, et al. Syrenomelia: A Reported Intrauteral Case Secondary to Embryotoxicity with Phenylephrine. J Gynecol Women’s Health. 2021: 22(1): 556080. DOI: 10.19080/JGWH.2021.22.556080
Keywords: Mermaid syndrome; Sirenomelia; Single lower limb; Single umbilical artery
Mermaid syndrome (sirenomelia) is an extremely rare anomaly, an incidence of 1 in 100,000 births, in which a newborn with joined legs has a mermaid appearance (head and trunk like humans and tail like fish), and in the majority they die at birth. Gastrointestinal, urogenital, and single umbilical artery abnormalities are a clinical result of this syndrome. There are two important hypotheses for the pathogenesis of mermaid syndrome: the hypothesis of the subtraction of the umbilical artery by the vitelline artery and the hypothesis of defective blastogenesis. The cause of mermaid syndrome is unknown, but there are some possible factors such as age younger than 20 years and older than 40 years in the mother and exposure of the fetus to teratogens. We present the case of a 28-year-old woman with a fetus with mermaid syndrome secondary to exposure to phenylephrine. The mother did not present a genealogical or environmental history as a risk factor, the product was diagnosed by ultrasound at 10 weeks of gestation, it died at 10.3 weeks.
Introduction
Mermaid syndrome (Sirenomelia) is a very rare congenital anomaly secondary to a developmental defect in the caudal region with varying degrees of leg adherence, causing the complete absence of the lower limb. The first reported case of the siren syndrome dates back to the 16th century [1,2]. The prevalence of this malformation is 1 in 100,000 births. Currently 350 cases are reported around the world of this anomaly. The incidence between men and women is [3]: 1.3, and the frequency in homozygous twins is 150 to 200 times; it is frequent to see this cases on diabetic mothers with a 15% [4,5]. The objective of this presentation is to report the case of a 28-year-old patient who was diagnosed at 10 weeks of gestation with a product with sirenomelia secondary to the administration of phenylephrine.
Reported Case
28-year-old pregnant woman, primiparous, Hispanic, accountant; With no significant genealogical and personal history, at 3-4 weeks of embryonic life or 5-6 weeks of gestation, she ingested 9 phenylephrine tablets to combat allergic rhinitis, ignoring her pregnancy status, no alterations were evidenced in the first control ultrasound (7th weeks of gestation), at prenatal control at 10 weeks, fetal death was evidenced with a congenital malformation compatible with sirenomelia. (Figure 1) The 10-week embryo weighed 32g a crown-coccyx length of 6.5cm. Standing length 0.7cm. Head circumference of 7.5cm, thorax 6.5cm and abdomen 6cm.with ambiguous genitalia and fused lower limbs (Figure 2); No other phenotypic alterations were evidenced on physical examination; It was not possible to perform a cytogenetic or pathological study because it was not authorized by the parents.
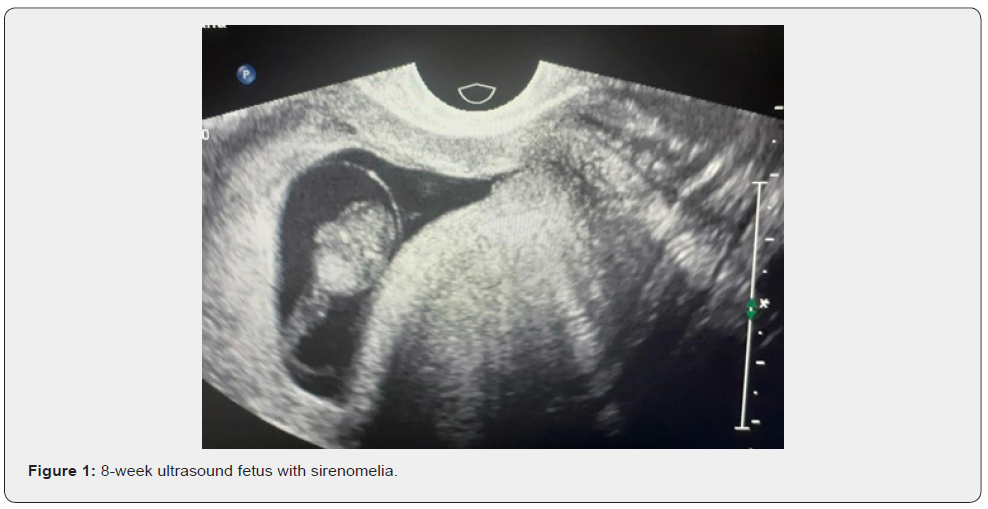
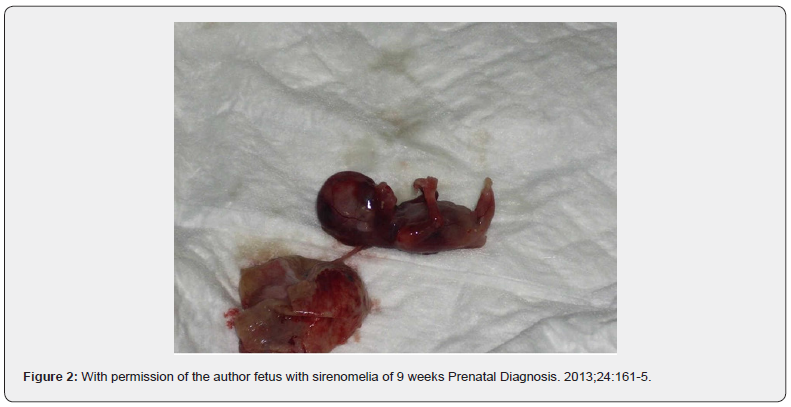
Discussion
Sirenomelia is an extremely rare malformation first reported in 1542 by Rocheus et al. Palfyn et al. in 1543. The mermaid means that the trunk looks human and the back looks like a fish [5,6]. In 1961, Duhamel classified the mermaid syndrome as caudal regression syndrome type 5 (CRS) because of the similarity with the anomalies of the CRS [7]. We now know that it is a separate syndrome from caudal regression. The diagnostic key is the presence of a single umbilical artery and renal agenesis, which is a characteristic clinical syndrome to the siren, whereas, in CRS, there is dysfunction and no fatal renal abnormality. Neonates born with mermaid syndrome often have a normal karyotype [8]. In 1987, Stocker and Heifetz introduced the theory of vitelline artery sequestration and reported that all patients with sirenomelia had a large umbilical artery, separated from the superior abdominal aorta below the celiac artery, the branches of the other aorta have not evolved. The lack of blood flow and poor nutrition, causes the lower part of the body to be blocked leading to sacral agenesis and fusion of the limbs, imperforate anus, rectal agenesis, absence of external and internal genitalia and renal agenesis [9]. The main cause of mermaid syndrome is unknown, there are two important hypotheses for the etiology of mermaid syndrome: hypothesis of sequestration of the yolk artery and defective hypothesis of blastogenesis. Under the hypothesis of the vitelline artery sequestration theory, due to discoloration of the vitelline artery, the celiac artery separates from the abdominal aorta, and the rest of the aortic artery is absent or hypoplastic. The vitelline artery reduces blood flow and nutrition to the caudal portion of the embryo by diverting blood flow to the placenta. This occurs in the third and fourth weeks of embryonic life causing disturbances in the caudal region of the fetus [10]. The theory of defective blastogenesis, in which the organs of the caudal region have inadequate angiogenesis leading to incomplete growth and development in the caudal region [11]. Although genetic defects in humans are unknown, two defective genes have been studied in mice, Cyp26a1 and BMP7, leading to mermaid syndrome. The Cyp26a1 gene is responsible for encoding the enzyme that breaks down retinoic acid (the metabolite of vitamin A). Retinoic acid temporarily helps the vasculature in the caudal region of the embryo. The Cyp26a1 gene mutation leads to incomplete development of the caudal region of the embryo, giving rise to mermaid syndrome in mice. Bone morphogenic protein 7 is a protein that plays an important role in angiogenesis in vitro. They stimulate endothelial cells in the caudal region, producing tissue and vascular growth that leads to normal growth of the lower extremities in the fetus [12-14]. According to Orioli et al. [15] in 249 cases of mermaid syndrome, there was no recurrence in the family. Gestational diabetes mellitus is the only known maternal disease associated with mermaid syndrome. Mothers under 20 and over 40 are vulnerable [15-19]. Hyperthermia and amniotic band sequence are underlying causes of mermaid syndrome. Exposure to teratogenic factors, such as air pollution, the mother's contact with drugs such as cocaine, tobacco, alcohol, and radiation, as well as fetal exposure to cadmium, lithium, phenytoin, sodium valproate, carbamazepine, warfarin, methylergonovine, diethylpropion, trimethoprim, and ochratoxin (a type of fungus) have been associated with mermaid syndrome [20,21]. Several experimental studies conducted regarding xanthines and adrenergic drugs regarding teratogenic alterations in the vascular system. Hyperthermia and amniotic band sequence are underlying causes of mermaid syndrome. Exposure to teratogenic factors, such as air pollution, the mother's contact with drugs such as cocaine, tobacco, alcohol, and radiation, as well as fetal exposure to cadmium, lithium, phenytoin, sodium valproate, carbamazepine, warfarin, methylergonovine, diethylpropion, trimethoprim, and ochratoxin (a type of fungus) have been associated with mermaid syndrome [20,21]. Several experimental studies conducted regarding xanthines and adrenergic drugs regarding teratogenic alterations in the vascular system. Hyperthermia and amniotic band sequence are underlying causes of mermaid syndrome. Exposure to teratogenic factors, such as air pollution, the mother's contact with drugs such as cocaine, tobacco, alcohol, and radiation, as well as fetal exposure to cadmium, lithium, phenytoin, sodium valproate, carbamazepine, warfarin, methylergonovine, diethylpropion, trimethoprim, and ochratoxin (a type of fungus) have been associated with mermaid syndrome [20,21]. Several experimental studies conducted regarding xanthines and adrenergic drugs regarding teratogenic alterations in the vascular system. as well as fetal exposure to cadmium, lithium, phenytoin, sodium valproate, carbamazepine, warfarin, methylergonovine, diethylpropion, trimethoprim and ochratoxin (a type of fungus) has been associated with mermaid syndrome [20,21]. Several experimental studies conducted regarding xanthines and adrenergic drugs regarding teratogenic alterations in the vascular system. as well as fetal exposure to cadmium, lithium, phenytoin, sodium valproate, carbamazepine, warfarin, methylergonovine, diethylpropion, trimethoprim and ochratoxin (a type of fungus) has been associated with mermaid syndrome [20,21]. Several experimental studies conducted regarding xanthines and adrenergic drugs regarding teratogenic alterations in the vascular system. Ugen et al. [22] demonstrated limb defects such as ectrodactyly in rat embryos when acetazolamide was administered a carbonic anhydrase inhibitor with phenylephrine. Ugen et al. [23] previously demonstrated that phenylephrine, a selective postsynaptic alpha-1-adrenergic agonist As well as acetazolamide, it significantly reinforces the incidence of alteration of the right forelimb such as ectrodactyly. As reported, phenylephrine decreases maternal uterine blood flow compared to the control group. Which would enhance the teratogenicity of phenylephrine or acetazolamide Hodach et al. [24] showed that the activation of the beta-adrenergic sympathomimetic receptors isoproterenol, epinephrine, norepinephrine and phenylephrine are directly related to cardiovascular abnormalities produced in chicken embryos. Osuala et al. [25] They observed the role of the development of the heart with Adrenergic hormones, which influence the metabolism of AR by inactivating the dopamine β-hydroxylase (Dbh) gene, which can lead to heart disease. Fujinaga M et al. [26] Reported that rat embryos cultured from the presomite stage in medium containing the alpha-1 adrenergic agonist, phenylephrine, have a high incidence of situs inversus. Bruyere and Col demonstrated that the exposure of methylxanthones and adrenergic agents in chicken embryos increases intracellular cyclic AMP, which may explain the hypoplasia and loss of structures as a result of inhibition of mitosis and / or necrosis of the embryonic tissue of the limbs [27,28]. Sander and Col demonstrated in a review of 171 articles that the exposure of some drugs such as acetazolamide, adenine, 1.7 dimethylxanthine, xanthine derived aminophylline, retinoic acid, acetoxy-methyl-methyl nitrosamine, aspirin ,This alteration can be diagnosed prenatally with ultrasound in the first trimester of pregnancy with the following signs: nuchal translucency, fused lower limb, single lower limb, renal agenesis, single umbilical artery, and oligo hydramnio [29,30] as occurred in our case.
Conclusion
According to the findings of the mermaid syndrome, caudal hemorrhage from the area has been identified as an important cause and other factors including gestational diabetes mellitus and fetal exposure to teratogenic substances have been reported. Experimental and case-control studies are recommended regarding phenylephrine as a potential teratogenic agent for this vascular anomaly.
Acknowledgment
We thank Inter hospital and the Center for Genetic and Perinatal Studies (CEPEGEN) for their logistical and financial support to carry out this publication.
References
- Duesterhoeft SM, Ernst LM, Siebert JR, Kapur RP (2007) Five cases of caudal regression with an aberrant abdominal umbilical artery: further support for a caudal regression sirenomelia spectrum. Am J Med Genet A 143A (24): 3175-3184.
- Garrido-Allepuz C, Haro E, González-Lamuño D, Martínez-Frías ML, Bertocchini F, et al. (2011) A clinical and experimental overview of sirenomelia: insight into the mechanisms of congenital limb malformations. Dis Model Mech 4(3): 289-299.
- Taee N, Tarhani F, Goodarzi MF, Safdari M, Bajelan A, et al. (2018) Mermaid Syndrome: A Case Report of a Rare Congenital Anomaly in Full-Term Neonate with Thumb Deformity. AJP Rep 8(4): e328-e331.
- Taori KB, Mitra K, Ghonga NP, Gandhi RO, Mammen T, et al. (2002) Sirenomelia sequence (mermaid): report of three cases. Indian J Radiol Imaging 12(3): 399.
- Sikandar R, Munim S (2009) Sirenomelia, the mermaid syndrome: case report and a brief review of literature. J Pak Med Assoc 59(10): 721-723.
- Schiesser M, Holzgreve W, Lapaire O, Willi N, Hans L, et al. (2003) Sirenomelia, the mermaid syndrome - detection in the first trimester. Prenat Diagn 23(6): 493-495.
- Duhamel B (1961) From the mermaid to anal imperforation: the syndrome of caudal regression. Arch Dis Child 36(186): 152-155.
- Szumera E, Jasiewicz B, Potaczek T (2018) Atypical caudal regression syndrome with agenesis of lumbar spine and presence of sacrum - case report and literature review. J Spinal Cord Med 41(4): 496-500.
- Dayasiri K, Thadchanamoorthy V, Thudugala K, Ranaweera A, Parthipan N, et al. (2020) Clinical and Radiological Characterization of an Infant with Caudal Regression Syndrome Type III. Case Reports in Neurological Medicine.
- Kavunga E, Kambale G, Mumbere M, Kasereka C (2019) Sirenomelia associated with an anterior abdominal wall defect: a case report. Journal of Medical Case Reports 13(1): 213.
- Opitz JM, Zanni G, Reynolds JF Jr, Gilbert-Barness E (2002) Defects of blastogenesis. Am J Med Genet 115(4): 269-286.
- Zakin L, Reversade B, Kuroda H, Lyons KM, Robertis DEM, et al. (2005) Sirenomelia in Bmp7 and Tsg compound mutant mice: requirement for Bmp signaling in the development of ventral posterior mesoderm. Development 132(10): 2489-2499.
- Ribes V, Fraulob V, Petkovich M, Dollé P (2007) The oxidizing enzyme CYP26a1 tightly regulates the availability of retinoic acid in the gastrulating mouse embryo to ensure proper head development and vasculogenesis. Dev Dyn 236(3): 644-653.
- Sheng N, Xie Z, Wang C, Bai G, Kejing Z, et al. (2010) Retinoic acid regulates bone morphogenic protein signal duration by promoting the degradation of phosphorylated Smad1. Proc Natl Acad Sci USA 107(44): 18886-18891.
- Orioli IM, Amar E, Arteaga-Vazquez J, Bakker MK, Bianca S, et al. (2011) Sirenomelia: an epidemiologic study in a large dataset from the International Clearinghouse of Birth Defects Surveillance and Research, and literature review. Am J Med Genet C Semin Med Genet 157(4): 358-373.
- Al-Haggar M, Yahia S, Abdel-Hadi D, Grill F, Al Kaissi A, et al. (2010) Sirenomelia (symelia apus) with Potter's syndrome in connection with gestational diabetes mellitus: a case report and literature review. Afr Health Sci 10(4): 395-399.
- Chan BW, Chan KS, Koide T, Sau-Man Y, Leung MBW, et al. (2002) Maternal diabetes increases the risk of caudal regression caused by retinoic acid. Diabetes 51(9): 2811-2816.
- Taee N, Tarhani F, Safdary A (2005) A case of sirenomelia in newborn of a diabetic mother. Int J Diabetes Metab 5(1): 83-87.
- Dharmraj M, Gaur S (2012) Sirenomelia: a rare case of fetal congenital anomaly. J Clin Neonatol 1(4): 221-223.
- Tica OS, Tica AA, Brailoiu CG, Cernea N, Tica VI, et al. (2013) Sirenomelia after phenobarbital and carbamazepine therapy in pregnancy. Birth Defects Res A Clin Mol Teratol 97(6): 425-428.
- Barański B (1984) Effect of exposure of pregnant rats to cadmium on prenatal and postnatal development of the young. J Hyg Epidemiol Microbiol Immunol 29(3): 253-262.
- Ugen KE, Scott WJ (1986) Acetazolamide teratogenesis in Wistar rats: potentiation and antagonism by adrenergic agents. Teratology 34(2): 195-200.
- Ugen KE, Scott WJ (1987) Reduction of uterine blood flow by phenylephrine, an alpha adrenergic agonist, in the day 11 pregnant rat: relationship to potentiation of acetazolamide teratogenesis. Teratology 36(1): 133-41.
- Hodach RJ, Hodach AE, Fallon JF, Folts JD, Bruyere HJ, et al. (1975) The role of beta adrenergic activity in the production of cardiac and aortic arch anomalies in chick embryos. Teratology 12(1): 33-45.
- Osuala K, Baker CN, Nguyen HL, Martinez C, Weinshenker D, et al. (2012) Physiological and genomic consequences of adrenergic deficiency during embryonic / fetal development in mice: impact on retinoic acid metabolism. Physiol Genomics 44(19): 934-947.
- Fujinaga M, Baden JM (1991) Critical period of rat development when sidedness of asymmetric body structures is determined. Teratology 44(4): 453-462.
- Bruyere HJ, Fallon JF, Gilbert EF (1983) External malformations in chick embryos following concomitant administration of methylxanthines and beta-adrenomimetic agents: 1. Gross pathologic features. Teratology 28(2): 257-269.
- Sanders DD, Stephens TD (1991) Review of drug-induced limb defects in mammals. Teratology 44(3): 335-354.
- Akbayir O, Gungorduk K, Sudolmus S, Gulkilik A, Ark C, et al. (2008) First trimester diagnosis of sirenomelia: a case report and review of the literature. Arch Gynecol Obstet 278(6): 589-592.
- López-García A (2013) Prenatal diagnosis of 2 cases of Sirenomelia in the first trimester. Clinical case diagnprenat 4: 161-165.






























