Sperm Morphological Detailed Aspects Impact on ICSI Reproductive Outcomes
Naglaa Hussien1* and Mohamed Elzayat2
1Department of Obstetrics and Gynecology, Tanta University, Egypt
2Department of Genetic Engineering and Biotechnology, Head of Royal Fertility Center, Egypt
Submission: September 13, 2019;Published: October 11, 2019
*Corresponding author: Naglaa Hussien, Department of Obstetrics and Gynecology, Tanta University, Egypt
How to cite this article: Naglaa Hussien, Mohamed Elzayat. Sperm Morphological Detailed Aspects Impact on ICSI Reproductive Outcomes. 2019: 17(1): 555951. DOI: 10.19080/JGWH.2019.17.555951
Abstract
Background: Reduced fertilization and implantation rates have been recognized by various research efforts to be a critical issue in cases of teratospermia, globospermia, and megalospermia affecting the IVF-ICSI management cycles rates of success. A real‐time and high‐magnification technique for selection of sperms together using a micromanipulation technique named intracytoplasmic morphologically selected sperm injection was observed by prior research efforts to have higher effectiveness than the traditional intracytoplasmic sperm injection
Aim:
to investigate the detailed sperm morphological features affect the outcome of intracytoplasmic sperm injection (ICSI).Methodology: The research study cohort was composed of the male partners of 140 random couples referred for ICSI treatment Inclusive research criteria involved females younger than 37 years old and more than 3 retrieved ovum per management cycle.
ICSI Procedure: Indications for ICSI have been as follows poor (less than 20%) or no (0%) fertilization in the first IVF cycle, immotile sperms, and low percentage of normally formed sperms (less than 4%) the motile sperm fractions have been prepared for ICSI management cycle from sperm ejaculated using the swim-up method cases of oligospermia (sperm concentration less than 15 *106 sperms /mL the density-gradient technique was used .Sperm selection for microinjection was conducted at a magnification of 200*400 the embryologist have excluded sperms from microinjection into the ovum those having severe head-shape abnormalities
Result: Receiver operating characteristic curve (ROC) for MSOME (%) as a predictors of (A) normal fertilization as regards normal spermatozoa at cut off point >2, AUC =0.833,statistical sensitivity =84.7%, specficty =67.6%, PPV=73.5%, NPV= 80.7% ,nucleus at cut off point > 27.8, AUC =0.711,statistical sensitivity=70.8%,specficty =66.2%,PPV=68.9%,NPV= 68.2 % and (B) successful pregnancy as regards normal spermatozoa at cut off point > 4,AUC = 0.785 ,statistical sensitivity =57.14 %,specificity =97.96 %,PPV=92.3 %,NPV= 84.2 % ,nucleus at cut off point > 27.1, AUC =0.87, statistical sensitivity =97.6%, specificity =57.1 %,PPV=49.4%, NPV= 98.2%.
Conclusion: Morphological assessment of the sperms at high magnifications and examining the organelle integrity is one of the cornerstone factors that could aid in elevating the success rates of ICSI at fertilization and pregnancy rates .However future research efforts are recommended to integrate the investigation of sperm DNA fragmentation tests in conjunction to detailed morphological sperm assessment.
Keywords: Fertilization; Teratospermia; Globospermia; Megalospermia; DNA fragmentation; Morphological sperm; Swim-up method; ICSI treatment; Head-shape abnormalities; Embryologist; Sperm motility
Introduction
Sperm morphology have been adopted by various embryologist all over the globe as one of the best predictability tools for the rates of fertilization, implantation and besides have been a predictability tool for the quality of the developing embryo [1-3]. Reduced fertilization and implantation rates have been recognized by various research efforts to be a critical issue in cases of teratospermia, globospermia , and megalospermia affecting the IVF-ICSI management cycles rates of success . at cellular and molecular levels researchers have denoted various explanations for this issue in which it was revealed and displayed by microscopic examination that morphologically normal sperm sare needed for successful passage via the female reproductive tract barriers and zona pellucida membrane, an abnormal head shape could denote an abnormal genetic makeup and reduced sperm motility and fertilization capabilities sperm abnormalities of the organelle system , have been correlated and linked to reduced favorable ICSI clinical outcomes [4-6].
A real‐time and high‐magnification technique for selection of sperms together using a micromanipulation technique named intracytoplasmic morphologically selected sperm injection was observed by prior research efforts to have higher effectiveness than the traditional intracytoplasmic sperm injection [7-9].
The suitable spermatozoa are selected by a well-trained embryologist performing the ICSI, according to morphological and motility criteria research evidence have revealed and displayed that there is a higher rate of miscarriage and congenital abnormalities detected, in cases of DNA fragmentation within the selected spermatozoa [10-12].
Physiological ICSI an enhanced sperm selection prortocol implemented in conjunction to ICSI technique in which the protocol uses the molecular knowledge that the mature sperm head have a surface cell membrane receptor having the capability to bind to hyaluronic acid. IMSI involves the usage of variance interference contrast microscopic technology at high magnifications having a range of ×6000 to ×13 000 of unstained sperms the technique in addition relies on based on motile sperm organelle morphological examination (MSOME), which discriminates the state of various sub‐cellular organelles such as the acrosome, post‐acrosomal lamina, neck, mitochondria, tail and nucleus of the spermatozoa. The observation and existence of large vacuoles have been correlated and linked in a statistically significant fashion in prior research studies to high rates of DNA fragmentation and aneuploidy [13,14].
Aim of The Work
to investigate the detailed sperm morphological features, affect the outcome of intracytoplasmic sperm injection (ICSI).
Methodology
The research study cohort conducted in the ART unit of gynecology department in Tanta university teaching hospital in the period from January 2019 to June 2019 that was composed of the male partners of 140 random couples referred for ICSI treatment Inclusive research criteria involved females younger than 37 years old and more than 3 retrieved ovum per management cycle. ICSI Procedure Indications for ICSI have been as follows poor (less than 20%) or no (0%) fertilization in the first IVF cycle, immotile sperms, and low percentage of normally formed sperms (less than 4% ) the motile sperm fractions have been prepared for ICSI management cycle from sperm ejaculated using the swim-up method cases of oligospermia (sperm concentration less than 15 *106 sperms /mL the density-gradient technique was used .Sperm selection for microinjection was conducted at a magnification of 200*400 the embryologist have excluded sperms from microinjection into the ovum those having severe head-shape abnormalities, e.g. pin, amorphous, tapered, round, or multinucleated head. Beginning on day 21 of the prior cycle or on day 1 to 3 of the current cycle, cases have been administered gonadotropin-releasing hormone agonist depot or daily leuprorelin 3.75 mg depot or triptorelin 0.1mg. as pituitary desensitization Have been accomplished, ovarian stimulation has been started using human follicle-stimulating hormone. Final oocyte maturation was induced with 10 000 IU human chorionic gonadotropin (hCG) when at least 2 follicles with a mean diameter of more than 17mm was observed. Oocyte retrieval was performed by transvaginal sonographic needle- aspiration at 34 hours after trigger by hCG injection. Fertilization have been documented if 2 pronuclei are observed after around 12 to 16 hours and, when embryonic cleavage occurred. Embryo transfer have been conducted on day 3; Pregnancy have been verified at 6 to 7 weeks by sonographic examination.
Sperm Preparation for Morphological Observation
The fraction of unused spermatozoa usually suspended in IVF medium or sperm medium was mixed. An aliquot of 1-2 _L of the sperm suspension containing a few thousand spermatozoa was transferred to a microdroplet of sperm medium containing 0%–8% polyvinyl pyrrolidone solution and placed in a sterile glass-bottomed dish under sterile paraffin oil. The temperature of the observed sample and the polyvinyl pyrrolidone concentration have been adjusted according to the intensity of the sperm motility. In cases of poor sperm motility, the temperature was elevated to 37 degree Celsius, no polyvinyl pyrrolidone was added to the sperm medium In cases of high sperm motility levels , the temperature have been lowered to around 20 degree Celsius and the polyvinyl pyrrolidone concentration levels have been kept elevated ( around 8%). Morphological evaluation of the sperm cells in motion was made possible by the creation of small bays extruding from the rim of the droplets, which captured the heads of the motile spermatozoa.
Sperm Observation Sperms were assessed under immersion oil by an inverted microscope the morphological assessment has been conducted on the monitor screen reaching a real magnification of 6300*.
Statistical Analysis
Data were collected, revised, coded and entered to the Statistical Package for Social Science (IBM SPSS) version 23. The quantitative data were presented as mean, standard deviations and ranges while qualitative data were presented as number and percentages. The comparison between groups with qualitative data was done by using Chi-square test. The comparison between two independent groups with quantitative data and parametric distribution was done by using Independent t-test. Receiver operating characteristic curve (ROC) was used to assess the best cut off point with its sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and area under curve (AUC). The confidence interval was set to 95% and the margin of error accepted was set to 5%. So, the p-value was considered significant at the level of < 0.05.
Result
Table 1 reveals and displays the basic characteristics of the investigated cases Mean ± SD, Women’s age (years),BMI (kg/ m2), Men’s age (years), Duration of infertility =33.55 ± 3.63, 28.9 ± 3.5, 35.32 ± 2.85 , 3.61 ± 1.65 consecutively ,as regards MSOME (%)Mean ± SD of Normal spermatozoa, Normal nucleus ,Follicles ≥ 17 =3.45 ± 1.59, 28.3 ± 2.1, 1.3 ± 0.3 consecutively, as regards Etiology, no. (%) Idiopathic ,Male mild, Cervical ,Ovulatory ,Endometriosis, Male mild=70 (50.0%), 34 (24.29%), 21 (15.00%), 8 (5.71%),5 (3.57%), 2 (1.43%) furthermore the total oocytes retrieved, number mean +/-SD Oocytes count per patient =11.3 ± 2.3, Mature oocytes, Fertilization occurrence, number (%)=1257 / 1582 (79.46%), 795 / 1257 (63.25%) consecutively, Mean ± SD of Fertilization rate per patient, Total sperm count (x106/ml) = 64.93 ± 5.67, 82.79 ± 34.6 consecutively as regards the Clinical pregnancy, no. (%) success =42 (30.0%), failed =98 (70.0%).

Table 2 Comparative statistical analysis between cases with successful pregnancy and those with failed pregnancy as regards the investigated parameters in which there was no statistical significance as regards Women’s age (years), BMI (kg/ m2), Men’s age (years), Duration of infertility, Follicles ≥ 17mm, Etiology, no. (%),DNA fragmentation (%), Oocytes count per patient, Mature oocytes injected, Fertilization occurrence, Fertilization rate per patient (%),Total sperm count (x106/ml), (p values =0.504, 0.490, 0.092, 0.065, 0.073, 0.073, 0.339, 0.172, 0.833,0.472, 0.924 consecutively ) however there was statistical significant difference as regards Normal spermatozoa ,nucleus( p values<0.001) as shown (Figure 1).
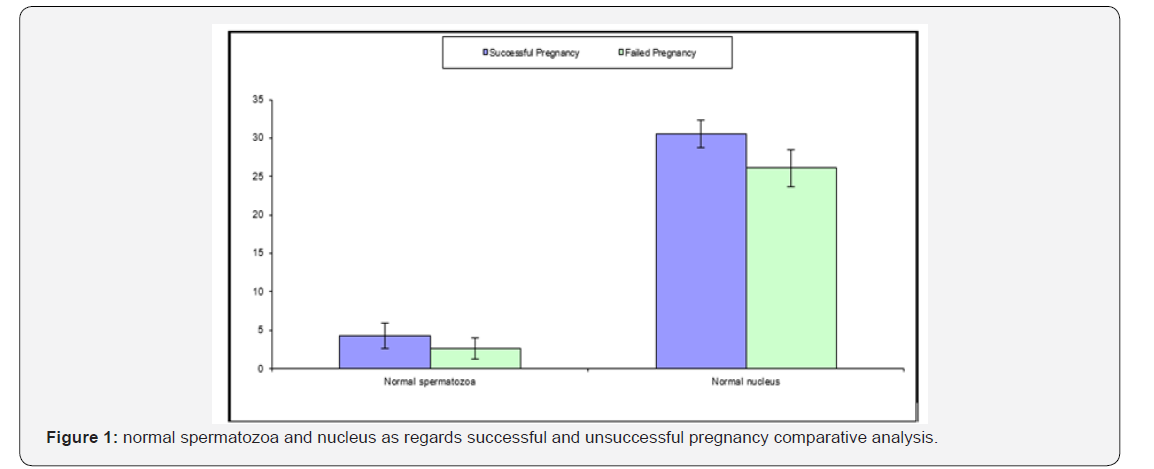

Table 3 statistical comparative analysis between cases as regards normal fertilization and those with decreased fertilization as regards the investigated parameters in which there was no statistical significance as regards Women’s age (years), BMI (kg/m2), Men’s age (years), Duration of infertility, Follicles ≥17mm, Etiology, no. (%), DNA fragmentation (%), Oocytes count per patient, Mature oocytes injected, Fertilization occurrence, Fertilization rate per patient (%),Total sperm count (x106/ml), (p values= 0.053, 0.113, 0.295, 0.488, 0.260, 0.559, 0.974, 0.132, 0.172,0.780, 0.619 consecutively) however there was statistical significant difference as regards Normal spermatozoa ,nucleus( p values<0.001) as shown (Figure 2).



Table 4 Receiver operating characteristic curve (ROC) for MSOME (%) as a predictors of (A) normal fertilization as regards normal spermatozoa at cut off point >2, AUC =0.833, statistical sensitivity =84.7%, specificity =67.6%, PPV=73.5%, NPV= 80.7%, nucleus at cut off point > 27.8, AUC =0.711, statistical sensitivity =70.8%, Specficty =66.2%,PPV=68.9%, NPV= 68.2 % and (B) successful pregnancy as regards normal spermatozoa at cut off point > 4, AUC = 0.785, statistical sensitivity =57.14 %, specificity =97.96 %, PPV=92.3 %, NPV= 84.2 %, nucleus at cut off point > 27.1, AUC =0.87, statistical sensitivity =97.6%, specificity =57.1 %, PPV=49.4%, NPV= 98.2%.
Discussion
The evaluation of the correlation between male fertility capacity and sperm morphological aspects is very complex, since human sperms have normally a high structural variability, and embryologists couldn’t be sure that the sperm cells randomly selected for evaluation represent those that are able to fertilize the ova normally. The morphological status of the sperm organelles is an enhanced embryological diagnostic approach particularly when integrated to sperm DNA fragmentation tests morphological analysis at the subcellular levels of single motile sperm cell enhances the capability to select the best functioning sperm [15-17].
A prospective prior research effort similar to the current study in approach and methodology aimed to investigate If sperm morphological characteristics influence The clinical outcome of intracytoplasmic sperm injection they revealed in manner similar to the current research that the morphological Normal appearance of whole sperm have been correlated To higher fertilization rates in ICSI management cycles those research findings reveal the fact that ICSI- rates of pregnancy are affected even by trivial morphological abnormalities of the sperm [1,3,5,7].
A prior research meta‐analysis revealed a statistically a significantly higher number of live births among the morphologically selected sperm research group t in comparison to the traditionally performed ICSI research group with a statistically significantly lower rate of miscarriage among the morphologically selected sperm research group in comparison to the traditionally conducted ICSI research group .those research findings support the current study observations and could be justified by the fact that the normal sperms morphologically have the highest probability of having a normal DNA content that permits the normal physiological developmental process of fertilization and embryonic development due to normal genetic mechanisms critical in development [2,4,8,11].
Furthermore previous embryological research efforts have revealed that sperms free of any particular morphological malformations had the highest positive statistical correlation with successful ICSI fertilization. prior research efforts have focused on the sperm nuclear abnormalities have revealed that the more the sperm nucleus had normal appearing structure particular ly with low DNA fragmentation rates nearly all ICSI outcomes are expected to be favorable particularly the fertilization and impanation rates [6,9,13].
The current research study findings are supported by prior research efforts revealing that there was low rates of pregnancy and implantation in clinical scenarios of teratospermia and low rates of fertilization in cases of globospermia and megalospermia [2,7,15].
Conclusions and Recommendations for Future Research
Morphological assessment of the sperms at high magnifications and examining the organelle integrity is one of the cornerstone factors that could aid in elevating the success rates of ICSI at fertilization and pregnancy rates .However future research efforts are recommended to integrate the investigation of sperm DNA fragmentation tests in conjunction to detailed morphological sperm assessment in a multicentric fashion that could aid in enhancement of the reproductive outcomes of ICSI practice.
References
- Teixeira DM, Barbosa MA, Ferriani RA (2013) Regular (ICSI) versus ultra‐high magnification (IMSI) sperm selection for assisted reproduction. Cochrane Database Syst Rev (7): CD010167.
- Avalos‐Duran G, Canedo‐Del AAME, Rivero‐Murillo J, Zambrano‐Guerrero JE, Carballo‐Mondragon E, et al. (2018) Physiological ICSI (PICSI) vs. conventional ICSI in couples with male factor: a systematic review. JBRA Assist Reprod 22(2): 139‐
- Khattabi EL, Dupont C, Sermondade N, Hugues JN, Poncelet C, et al. (2013) Is intracytoplasmic morphologically selected sperm injection effective in patients with infertility related to teratozoospermia or repeated implantation failure? Fertil Steril 100(1): 62‐
- Boitrelle F, Guthauser B, Alter L, Bailly M, Bergere M, et al. (2014) High‐magnification selection of spermatozoa prior to oocyte injection: confirmed and potential indications. Reprod Biomed Online 28(1): 6‐
- Delaroche L, Yazbeck C, Gout C, Kahn V, Oger P, et al. (2013) Intracytoplasmic morphologically selected sperm injection (IMSI) after repeated IVF or ICSI failures: a prospective comparative Eur J Obstet Gynecol Reprod Biol 167(1): 76‐80.
- Leandri RD, Gachet A, Pfeffer J, Celebi C, Rives N, et al. (2013) Is intracytoplasmic morphologically selected sperm injection (IMSI) beneficial in the first ART cycle? A multicentric randomized controlled trial. Andrology 1(5): 692‐
- Setti AS, Braga DP, Figueira RC, Iaconelli AJr, Borges E, et al. (2014) Intracytoplasmic morphologically selected sperm injection results in improved clinical outcomes in couples with previous ICSI failures or male factor infertility: A meta‐ Eur J Obstet Gynecol Reprod Biol 183: 96‐103.
- Sala LGB, Nicoli A, Fornaciari E, Falbo A, Rondini I, et al. (2015) Intracytoplasmic morphologically selected sperm injection versus conventional intracytoplasmic sperm injection: a randomized controlled trial. Reprod Biol Endocrinol 13: 97.
- Zegers‐Hochschild F, Adamson GD, Dyer S, Racowsky C, Mouzon DJ, et al. (2017) The International Glossary on Infertility and Fertility Care, 2017. Fertil Steril 108(3): 393-
- Orief YI, Elabd MM, Said THM, Ahmed NAAJ (2016) Comparative study between intracytoplasmic morphologically selected sperm injection versus intracytoplasmic sperm injection in patients with severe male factor infertility and repeated intra cytoplasmic sperm injection failure. Int J Reprod Contracep Obstet Gynecol 5: 1102‐
- Greco E, Scarselli F, Fabozzi G, Colasante A, Zavaglia D, et al. (2013) Sperm vacuoles negatively affect outcomes in intracytoplasmic morphologically selected sperm injection in terms of pregnancy, implantation, and live‐birth rates. Fertil Steril 100(2): 379‐
- Monte LG, Murisier F, Piva I, Germond M, Marci R, et al. (2013) Focus on intracytoplasmic morphologically selected sperm injection (IMSI): a mini‐ Asian J Androl 15(5): 608‐615.
- Vingris L, Setti AS, Ferreira Braga DADP, Figueira DCSR, Iaconelli A, et al. (2015) Sperm morphological normality under high magnification predicts laboratory and clinical outcomes in couples undergoing ICSI. Hum Fertil (Camb) 18(2): 81‐
- Kim HJ, Yoon HJ, Jang JM, Oh HS, Lee YJ, et al. (2014) Comparison between intracytoplasmic sperm injection and intracytoplasmic morphologically selected sperm injection in oligo‐astheno‐teratozoospermia patients. Clini Exp Reprod Med 41(1): 9‐
- Bradley CK, McArthur SJ, Gee AJ, Weiss KA, Schmidt U, et al. (2016) Intervention improves assisted conception intracytoplasmic sperm injection outcomes for patients with high levels of sperm DNA fragmentation: a retrospective analysis. Andrology 4(5): 903‐
- Gatimel N, Parinaud J, Leandri RD (2016) Intracytoplasmic morphologically selected sperm injection (IMSI) does not improve outcome in patients with two successive IVF‐ICSI failures. J Assist Reprod Genet 33(3): 349‐
- Witt KD, Beresford L, Bhattacharya S, Brian K, Coomarasamy A, et al. (2016) Hyaluronic Acid Binding Sperm Selection for assisted reproduction treatment (HAB Select): study protocol for a multicentre randomised controlled trial. BMJ Open 6(10): e012609.






























