Real World Efficacy and Tolerability of Heme Iron Polypeptide in Non-Pregnant and Pregnant Women with Iron Deficiency Anemia
Varsha Narayanan1* and Amit Bhargava2
1 DGM Medical services, India
2 VP Medical Services, India
Submission: June 14, 2018 Published: August 20, 2018
*Corresponding author: Varsha Narayanan, DGM-Medical Services, Lupin Ltd, Mumbai, India, Email:varshanarayanan@lupin.com
How to cite this article: Varsha N, Amit B. Real World Efficacy and Tolerability of Heme Iron Polypeptide in Non-Pregnant and Pregnant Women with Iron Deficiency Anemia. J Gynecol Women’s Health. 2018: 11(2): 555810. DOI: 10.19080/JGWH.2018.11.555810
Abstract
Background: Iron deficiency anemia is a common condition affecting the quality of life, as well as maternal and fetal outcomes in non-pregnant and pregnant women respectively. Adequate and effective iron supplementation is the mainstay of therapy. Oral iron supplementation represents the most convenient and cost effective form of therapy. Conventional Non-heme iron oral supplements are known to be associated with poor gastrointestinal tolerability, and therefore reduced compliance and efficacy. Oral Heme iron supplementation represents a better tolerated and more bioavailable option for oral iron therapy. Though evidence of its efficacy and tolerability are available through some randomized clinical trials, real world data from its regular use in pregnant and non-pregnant patients is lacking.
Methodology: In this study, 378 non-pregnant and pregnant women visiting 56 Gynecology clinics across India with an in-clinic diagnosis of Iron deficiency anemia based on baseline laboratory evaluation of Hemoglobin (Hb), along with Ferritin, MCV and Hematocrit, were treated with daily tablets of Heme Iron polypeptide (HIP) for an average duration of 3 months. Laboratory parameters assessed at baseline were repeated at final follow up and the patients’ tolerability to therapy was also recorded.
Result: In non-pregnant women, the average rise in Hb was 1.17, 2.06 and 3.28 (g/dl), in groups with baseline mild, moderate and severe anemia, while in pregnant women, the average Hb rise was 2.70 and 3.53 (g/dl) in groups with baseline moderate and severe anemia. The rise in Hb was highly significant in both pregnant and non-pregnant women with baseline moderate and severe anemia. There was also a significant decline in the number of patients with moderate and severe anemia, as well as in percentage patients with low ferritin, MCV and hematocrit values. The tolerability of HIP was satisfactory with a treatment adverse event related discontinuation rate of less than 0.8%.
Conclusion: This real world study suggests that oral HIP therapy can be an effective treatment option in pregnant and non-pregnant women with Iron deficiency due to its better gastrointestinal tolerability and efficacy in improving hematological parameters.
SynopsisSkilled-birth attendants’ postpartum hemorrhage skill simulation pass rate was low. Vaginal delivery performance was greater one year compared to two years after training.
Keywords: Iron deficiency; Heme iron polypeptide; Hemoglobin; Ferritin; Pregnant; Hepcidin; GI tolerability
Abbrevations:GI: Gastrointestinal; HIP: Heme Iron Polypeptide; IDA: Iron Deficiency Anemia; AE: Adverse Events
Introduction
30% of the world’s population which is around 2 billion people, are affected by anemia, with 56% of pregnant women and 43% of non-pregnant women in developing countries being anemic [1,2]. The causes of anemia are manifold, but iron deficiency is by far the most important cause of nutritional anemia worldwide [3].
Prevalence of anemia in in developing countries like India is around 53.6% and can go up to 89.6%-100% in pregnant women [4-6]. Iron deficiency is considered the most common cause of anemia in India. Dietary deficiencies, poor bioavailability and tolerability of iron supplements, parasitic infections, closely spaced pregnancies and heavy menstrual blood losses are some of the common causes of anemia in India [7]. Anemia in pregnancy is dilutional in nature due to increasing total blood volume and red cell mass as well as increased demand from growing fetus [8].
Adequate and effective supplementation with iron is the mainstay of anemia therapy and also helps in reducing maternal and fetal morbidity and mortality [9]. The recommended iron intake in non-pregnant women is 12-18mg/day however over 90% women in India have a dietary intake below this recommendation at an average of only 9mg/day [10]. The recommended iron intake is further increased in pregnancy to around 35mg/day [11]. Therefore dietary iron alone is not sufficient for iron requirements in non-pregnant women with iron deficiency or in pregnant women, to maintain hemoglobin levels.
Dietary iron is contained in two forms namely non-heme and heme. While non-heme iron supplements have been conventionally prescribed, they are associated with poor gastrointestinal (GI) tolerability and constipation which is a major therapeutic and compliance limitation [12,13]. GI tolerability of conventional iron supplements is further reduced in pregnant women. Other requirements with conventional non-heme iron supplements like administration only on empty stomach, varying bioavailability of diverse salts, and drug and food interactions further reduce compliance and treatment adherence.
Heme iron, commercially available as heme iron polypeptide (HIP) for oral supplementation represents an approach for iron supplementation with higher bioavailability and gastrointestinal tolerability, and a lower dose requirement [14]. To our knowledge this is the first real world data from India on the effectiveness and tolerability of oral Heme Iron Polypeptide (HIP) supplementation in both non pregnant and pregnant women diagnosed with Iron deficiency anemia (IDA).
Methodology
378 female patients with an in-clinic diagnosis of iron deficiency anemia (IDA) were prescribed oral Iron in the form of Heme Iron Polypeptide (HIP), and the real world data for the same was recorded from 56 Gynecologists across India (15 in north, 16 in east zone, 14 in west zone and 11 in south zone). Baseline blood parameters to diagnose IDA recorded were: Hemoglobin, Serum Ferritin, MCV and Hematocrit. Patients were classified into mild, moderate and severe iron deficiency anemia based on the WHO cut offs for Hemoglobin in non-pregnant and pregnant women [15]. Patients were also classified to have low, or normal ferritin levels based on WHO cut off values [16]. Laboratory references were used for MCV and Hematocrit cut off values [17].
Thereafter the patients with IDA were given 1-2 tablets of 12mg Heme Iron polypeptide (HIP) daily with or without meals. Average duration of therapy was around 3 months. The blood parameters done at baseline were again repeated at final follow up. The patients were assessed at final follow up for mean rise in the blood parameters recorded at baseline. Primary end points were mean rise in Hemoglobin and tolerability with HIP. Secondary end points included overall reduction in severity of anemia, increase in serum ferritin, MCV and Hematocrit with HIP treatment. Ethical principles, patient consent and confidentiality of identity were adhered to while capturing the real world inclinic data.
Result
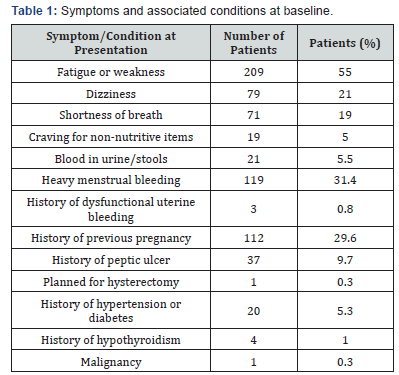
Out of the 378 female patients who were prescribed HIP daily, 83% patients were in the 20-40 years age group while 13% were in the 41-50 age group. 2% patients each were in the 10- 19 years and >50 years age group. 23 women were pregnant. Percentage patients presenting with Iron deficiency symptoms and associated conditions at baseline before start of HIP therapy is given in Table 1. One third of the patients were on some concomitant medication which included either a PPI, NSAID or a vitamin-mineral supplement. Almost 70% patients had history of previous non-compliance or gastro-intestinal intolerance to oral iron therapy with conventional non-heme iron supplements. The mean rise in hemoglobin was seen to be significant in nonpregnant and pregnant women in each of the categories classified as mild, moderate and severe anemia at baseline based on WHO cut offs (Figure 1&2).
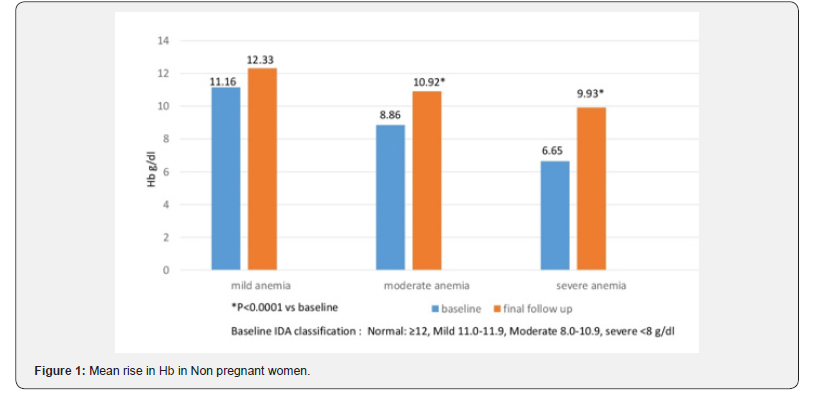
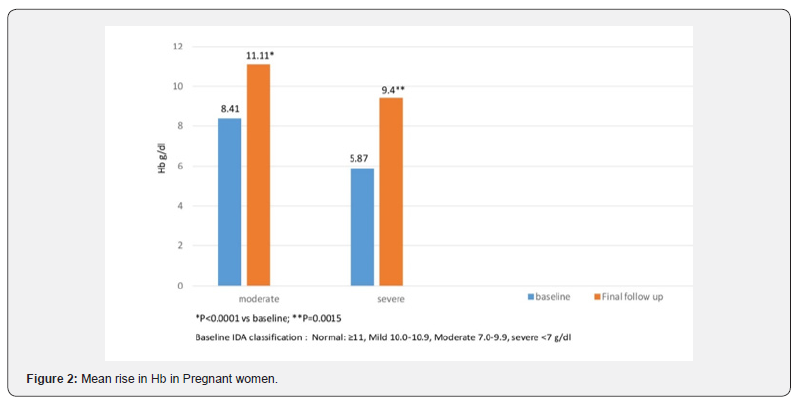
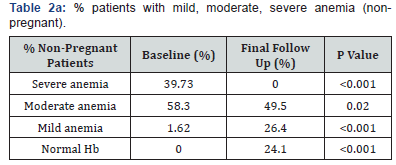
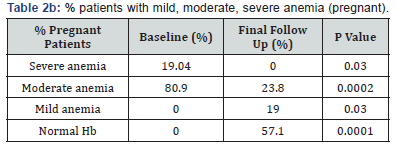
The percentage of patients who had severe anemia, decreased significantly and the percentage of patients with normal hemoglobin increased significantly in both non-pregnant and pregnant women at final follow up as compared to baseline Table 2(a&b)
Baseline classification based on WHO cut offs: Normal: ≥11, Mild 10.0-10.9, Moderate 7.0-9.9, severe < 7g/dl15 Average serum ferritin was seen to increase from 94.87ug/dl to 112.8ug/dl, and this absolute increase was not found to be significant. However the percentage of patients with low ferritin levels decreased from 20% and 37.5% to nil in non-pregnant (P< 0.0001) and pregnant women (not significant due to small sample size) respectively. (cut off for low ferritin/depleted iron stores: 15ug/dl) [16].
The percentage of non-pregnant patients showing low levels of MCV and Hematocrit decreased from 69.4% to 42.85% and 70.53% to 42.8% from baseline to final follow up (P< 0.0001). A similar drop was seen in percentage of pregnant women with low MCV and Hematocrit (66.6% to 58.3% and 57.14% to 28.57% from baseline to final follow up) but significance was not assessed due to small number of pregnant women. (Cut off for low MCV and Hematocrit: 80fl/red cell and 34.9% respectively) [17].
Adverse events (AE) occurred in 27 patients (7%). All AEs were mild, transient and were mainly gastrointestinal disturbance, which included nausea, abdominal cramps and constipation. 24 patients required symptomatic treatment, and symptoms subsided in 19 (79%) of them. Out of the 5 patients in whom symptoms persisted, 3 (0.79%) had to discontinue treatment after which symptoms subsided.
Discussion
HIP is a form of iron supplementation with higher bioavailability and gastrointestinal tolerability [18]. HIP is produced by hydrolysis of animal source hemoglobin with proteolytic enzymes which leaves the peptides from the hemoglobin subunits covalently bound to the heme ring [19]. This increases solubility at low pH and enhances its binding to heme receptor and transport into the cell, thereby increasing its bioavailability [20]. A 10 times higher bioavailability of heme iron compared to non-heme iron, have been reported in volunteer studies [21].
Non heme-iron is taken up by DMT1 (divalent metal transporter 1), which are non-selective and non-regulated iron channels on the enterocytes. Heme iron is taken up by heme transporter protein coupled folate transfer/heme carrier protein 1 (PCFT/HCP1) directly into the cytoplasm of enterocyte. The intact heme is then transported across basolateral membrane by the feline leukemia virus subgroup C receptor (FLVCR) protein and then binds to hemopexin in the circulation [19,22]. Also, some heme iron may be acted upon by heme oxygenase releasing non-heme iron and biliverdin.
Heme iron absorption is regulated by heme receptor and body iron levels, unlike non-heme iron salts. A study on HIP supplementation observed significant increase in serum iron levels in subjects with low initial serum iron levels (< 80μg/dl) compared to those with normal initial iron levels, suggesting that continual supplementation of heme-iron may not lead to iron overload [19].
Substances in food like tannins and phytates (present commonly in Indian diets) and chelators like desferrioxamine can reduce the availability of non heme iron but do not reduce heme iron bioavailability [23]. In a study where HIP, ferrous fumarate, and placebo was administered with breakfast to patients, HIP led to increased iron absorption even with meal as compared to ferrous fumarate and placebo (p < 0.03 and p < 0.02 respectively) [21]. No patients from heme-iron group had any side effects.
In two different studies, Young et al evaluated intrinsically labelled Fe-heme and non-heme Fe (ferrous sulphate) given to pregnant women and non-pregnant women [24,25]. In pregnant women in third trimester, maternally absorbed Fe tracer present in the neonates was significantly greater compared to the Fe tracer (p=0.02). Also, fetal transfer of Fe was inversely associated with maternal Hb and ferritin levels, whereas total body iron significantly correlated with heme iron transfer, suggesting favourable transport across placenta of heme-iron administered to the pregnant women [25]. In the other study in both pregnant and non-pregnant women, blood samples obtained 2-week post dosing to assess iron status indicators, serum hepcidin and iron utilization based on RBC incorporation of iron isotopes showed that heme-iron utilization was significantly higher relative to utilization of ferrous sulphate among both pregnant (p = 0.04) and non-pregnant women (p < 0.0001) [24]. While nonheme iron utilization is inversely related to serum hepcidin (as evidenced by women with undetectable levels of serum hepcidin having significantly greater non-heme iron utilization, and a 24% variation in non-heme iron utilization seen in healthy pregnant and non-pregnant participants due to hepcidin levels), heme iron utilization is not seen to be significantly influenced by iron stores or serum hepcidin [24].
A Korean volunteer double blind study investigated two HIP 12 mg preparations from Korea and Japan with 2 strengths of ferrous aminoacetate as well as placebo, and found increased serum iron levels with the Korean HIP suggesting a better absorption of HIP than iron-salts. In two different studies [19].
Suzuki et al evaluated the efficacy of Heme Iron Polypeptide 3mg in pregnancy and in females with bleeding disorder, and demonstrated gradual rise in Hb over a 14-week period with most patients reaching an Hb above 11gm/dl with twice daily regime [26]. None of the subjects experienced any unexpected adverse events and there were no drop outs due to the same. Authors have suggested that average dose of 6.5mg elemental iron as HIP or maximum dose of 9mg elemental iron as HIP is sufficient to improve anemia in pregnant women when administered in the third trimester.
Compliance to Heme Iron therapy due to low GI side effects has been a major reason for the efficacy results in clinical studies. Habib et al in their study showed that Hb levels improved significantly only amongst strictly compliant pregnant women and anemia was significantly associated with noncompliance with iron supplementation, the adjusted OR being 6.19 (95% CI 2.55 - 15.02, p < 0.0001) [27]. Eskeland et al in their study in pregnant and post-partum women noted a compliance of < 50% in 4% of the heme-iron group, 12% of the non-heme iron group and 5% of the placebo group. Women who failed to maintain an acceptable hematologic status were 10%, 20% and 45% in heme iron, non-heme iron and placebo group respectively [28].
A recent study comparing oral HIP and intravenous iron saccharate complex in pregnant women with iron deficiency anemia (Hb< 10gm/dL) showed Hb increase from 8.5 +/- 3.5 to 11.3 +/- 1.3g/dl in HIP treatment group, and an Hb increase from 8.7+/- 2.5 to 11.7 +- 0.9g/dl in IV iron treatment group accompanied by increased ferritin in both groups at end of three months (no significant differences between groups for change in Hb or ferritin levels) [29]. GI upset was reported in 1.6% of patients receiving oral HIP. This suggested oral HIP as an effective and tolerable treatment which can be considered as an alternative to intravenous iron saccharate complex for iron deficiency anemia of pregnancy.
Our real world findings corroborate the results of earlier studies with HIP. In non-pregnant women, the average rise in Hb seen in our study was 1.17, 2.06 and 3.28g/dl, in the baseline mild, moderate and severe anemia groups while in pregnant women, the average Hb rise was 2.70 and 3.53g/dl in the baseline moderate and severe anemia groups over an average of 3 month study period. The rise in Hb was highly significant in both pregnant and non-pregnant women with baseline moderate and severe anemia, and Hb rise was seen to be more in patients with lower baseline Hb, as has also been seen in other studies. There was also a decline in the number of patients with moderate and severe anemia, as well as in percentage patients with low ferritin, MCV and hematocrit values. The tolerability of HIP in our study was also similar to other published studies with a treatment adverse event rate of 7% and a discontinuation rate of less than 0.8% suggesting satisfactory patient compliance.
Conclusion
Oral HIP therapy can be an effective treatment and an option of choice for both pregnant and non- pregnant women with Iron deficiency anemia, due to better absorption, GI tolerability and efficacy in improving hematological parameters. More real world studies comparing various heme and non-heme oral iron supplements can further enhance effective clinical decision making in the management of iron deficiency in pregnant and non-pregnant women.
References
- Maeyer DEM, Adiels TM (1985) The prevalence of anaemia in the world. World Health Stat Q 38(3): 302-316.
- World Health Organization (WHO) (1992) The prevalence of anaemia in women: A tabulation of available information. Geneva, Switzerland.
- Maeyer DEM, Dallman P, Gurney JM, Hallberg L, Sood SK, et al. (1989) Preventing and controlling iron deficiency anaemia through primary health care. Geneva, Switzerland, p. 58.
- World bank (2011) Prevalence of Anemia among Pregnant Women (%).
- Bora R, Sable C, Wolfson J, Boro K, Rao R, et al. (2014) Prevalence of Anemia in Pregnant Women and Its Effect on Neonatal Outcomes in Northeast India. J Matern Fetal Neonatal Med 27(9): 887-891.
- Vemulapalli B, Rao KK (2014) Prevalence of Anemia among Pregnant Women of Rural community in Vizianagram North Costal Andhra Pradesh, India. AJMS 5(2): 21-25.
- Bharati P, Som S, Chakrabarty S, Bharati S, Pal M, et al. (2008) Prevalence of Anemia and Its Determinants among Nonpregnant and Pregnant Women in India. Asia Pac J Public Health 20(4): 347-359.
- Chandra S, Tripathi AK, Mishra S, Amzarul M, Vaish AK, et al. (2012) Physiological Changes in Hematological Parameters during Pregnancy. Indian J Hematol Blood Transfus 28(3): 144-146.
- (2008) EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS))- Scientific Opinion on the Safety of Iron (Blood Peptonates) for the Proposed Uses as a Source of Iron Added for Nutritional Purposes to Foods for the General Population, Including Food Supplements. EFSA Journal 8: 1585.
- Milman N (2006) Iron and Pregnancy-A Delicate Balance. Annals of Hematology 85(9): 559-565.
- (2011) Indian Council for Medical Research-Dietary Guidelines for Indians-A Manual. In: (2nd edn), National Institute of Nutrition, Hyderabad, India, p. 89.
- Pavord S, Myers B, Robinson S, Allard S, Strong J, et al. (2012) UK Guidelines on the Management of Iron Deficiency in Pregnancy. Br J Haematol 156(5): 588-600.
- Johnson-Wimbley TD, Graham DY (2011) Diagnosis and Management of Iron Deficiency Anemia in the 21st Century. Therap Adv Gastroenterol 4(3): 177-184.
- Hoppe M, Brun B, Larsson MP, Moraeus L, Hulthen L, et al. (2013) Heme Iron-Based Dietary Intervention for Improvement of Iron Status in Young Women. Nutrition 29(1): 89-95.
- WHO (2011) Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. p. 6.
- WHO (2011) Serum ferritin concentrations for the assessment of iron status and iron deficiency in populations. P. 5.
- https://www.mayoclinic.org/tests-procedures/complete-bloodcount/ about/pac-20384919
- Nagaraju SP, Cohn A, Akbari A, Davis JL, Zimmerman DL, et al. (2013) Iron Polypeptide for the Treatment of Iron Deficiency Anemia in Non- Dialysis Chronic Kidney Disease Patients: A Randomized Controlled Trial. BMC Nephrology 14: 64.
- Nam T, Shim JY, Kim B, Rah SY, Park K, et al. (2006) Clinical Study on the Iron Absorption from Heme-Iron Polypeptide and Nonheme-Iron. Nutritional Sciences 9(4): 295-300.
- Nissenson AR, Charytan C (2003) Controversies in Iron Management. Kidney Int Suppl 64: S64-S71.
- Seligman PA, Moore GM, Schleicher RB (2000) Clinical Studies of HIP: An Oral-Iron Product. Nutrition Research 20(9): 1279-1286.
- West AR, Oates PS (2008) Mechanisms of Iron Absorption: Current Questions and Controversies. World J Gastroenterol 14(26): 4101- 4110.
- Kalaivani K (2009) Prevalence & Consequences of Anemia in Pregnancy. Indian J Med Res 130: 627-633.
- Young MF, Griffin I, Pressman E, McIntyre AW, Cooper E, et al. (2010) Utilization of Iron from an Animal-Based Iron Source Is Greater than That of Ferrous Sulfate in Pregnant and Nonpregnant Women. The Journal of Nutrition 140(12): 2162-2166.
- Young MF, Griffin I, Pressman E, McIntyre AW, Cooper E, et al. (2012) Maternal Hepcidin Is Associated with Placental Transfer of Iron Derived from Dietary and Non-Sources. J Nutr 142(1): 33-39.
- Suzuki S, Ito H, Kiyokawa H (1990) The Effect of BioIron-150 on Pregnant Women and Women with Females Associated Bleeding Disorders. The World of Obstetrics and Gynecology 42: 33-42.
- Habib F, Habib ZAE, Alenazy M, Nooh R (2009) Compliance to Iron Supplementation during Pregnancy. J Obstet Gynaecol 29(6): 487-492.
- Eskeland B, Malterud K, Ulvik RJ, Hunskaar S (1997) Iron Supplementation in Pregnancy: Is Less Enough? A Randomized, Placebo Controlled Trial of Low Dose Iron Supplementation with and without Iron. Acta Obstet Gynecol Scand 76(9): 822-828.
- Abdelazimn IA, Abu-Faza M, Elbiaa AAM, Othman HS, Alsharif DA, et al. (2017) Heme Iron Polypeptide (Proferrin®-ES) versus Iron Saccharate Complex (Ferrosac) for Treatment of Iron Deficiency Anemia during Pregnancy. Acta Medica International 4(11): 56-61.






























