A Unique Vaccine for Preventing Pregnancy Without Derangement of Ovulation and Menstrual Regularity is Back on Clinical Trial
Talwar GP1*, Jagdish C Gupta1, Rohini Sehgal2, Alka Kriplani2 and Indrani Ganguli3
1 Talwar Research Foundation, India
2 All India Institute of Medical Sciences, India
3 Sir Ganga Ram Hospital, India
Submission:May 29, 2018 ; Published: August 14, 2018
*Corresponding author: Talwar GP, Talwar Research Foundation, New Delhi, India, Email:gptalwar@gmail.com
How to cite this article: Talwar GP, Jagdish C G, Rohini S, Alka K, Indrani G. A Unique Vaccine for Preventing Pregnancy Without Derangement of Ovulation and Menstrual Regularity is Back on Clinical Trial J Gynecol Women’s Health. 2018: 11(2): 555807. DOI: 10.19080/JGWH.2018.11.555807
Keywords: Human chorionic gonadotropin; Bioactivity; Immunogenicity; Heterospecies dimer; PGIMER; Antibodies; Mycobacterium indicus pranii
Abbrevations:HCG: Human Chorionic Gonadotropin; HSD: Heterospecies Dimer; PGIMER: Postgraduate Institute of Medical Education and Research; MIP: Mycobacterium Indicus Pranii; DCGI: Drugs Controller General of India
Introduction
A vaccine was developed many years back which was capable of inducing antibodies against human chorionic gonadotropin (hCG) in women [1]. The strategy adopted was to link the beta subunit of hCG with a carrier, such as tetanus toxoid. The antibodies were competent to neutralize the bioactivity of hCG. Figure 1 shows that independent sets of antibodies were generated against both hCG and TT, the later protected the women from tetanus, which in those years used to take a heavy toll of life of rural non-TT immunized women giving birth to children in the field.
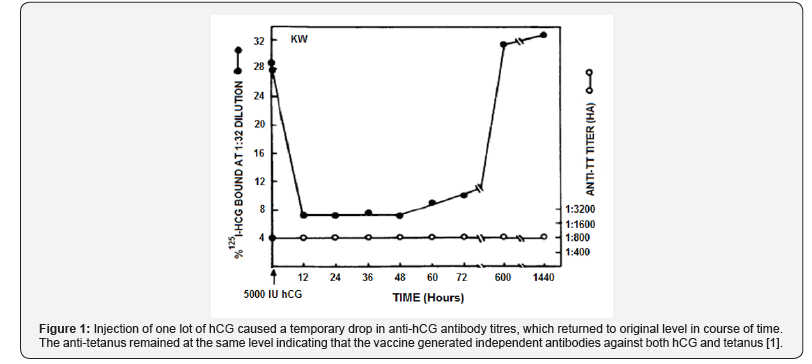
Thus, the vaccine was designed to achieve 2 purposes; to protect at least against tetanus, and if our premise of anti-hCG would have the competence of preventing pregnancy was correct, then vaccination would also achieve the purpose of preventing pregnancy. Over the years, the validity of this approach, the safety and reversibility of the vaccine and its ability to prevent pregnancy in sexually active women has been proved and will be briefly recapitulated in this article.
Enhancement of Immunogenicity of Hcgβ-Tt
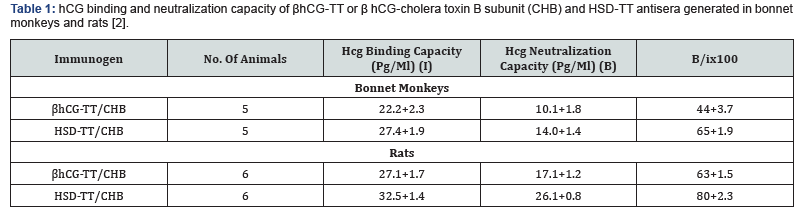
As hCG is made in fairly large amounts by women in early stages of pregnancy, the immunogenicity of βhCG-TT vaccine had to be improved. This was done by linking non-covalently βhCG with alpha subunit of ovine LH. Table 1 shows that the heterospecies dimer (HSD) thus created linked to TT generated higher titres of anti-hCG antibodies [2].
Safety and Efficacy Studies
After obtaining approval of the Institutional Ethics Committees and the Drugs Controller General of India (DCGI), Phase I safety and the historical Phase II efficacy trials were conducted at the All India Institute of Medical Sciences New Delhi, Postgraduate Institute of Medical Education and Research (PGIMER) Chandigarh and the Safdarjung Hospital New Delhi. 148 sexually active women of proven fertility with at least 2 children were enrolled after they had given written consent to participate in the study. They were given 3 injections of the HSDTT vaccine at 6 weeks interval as primary immunization. All of them generated both anti-hCG and anti-tetanus antibodies. In 110 of these women, the titres remained above 50ng/ml hCG neutralizing antibodies (the putative threshold fixed for testing) for 3 months or more. They were followed up for learning whether vaccination indeed prevented pregnancy [3]. Figure 2 represents the observations on 4 such women. It will be observed that the menstrual cycles of these women remained regular. The luteal phase progesterone was between 14-44nm, thus indicating that they continued to ovulate.
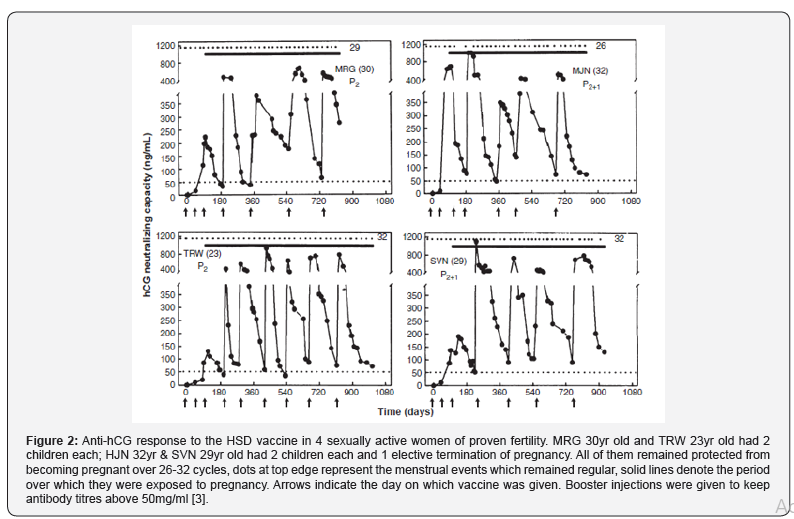
The vaccine was highly effective, only 1 pregnancy took place in 1224 cycles. All women kept ovulating normally and had regular menstrual cycles. Eight women were protected for 30 cycles without becoming pregnant, nine were protected over 24-29 cycles, 15 for 12-17 cycles and 21 for 6-11 cycles. Thus, the vaccine had high efficacy. More importantly, contraception was achieved without derangement of menstrual regularity and hormonal profiles.
Reversibility and Regain of Fertility
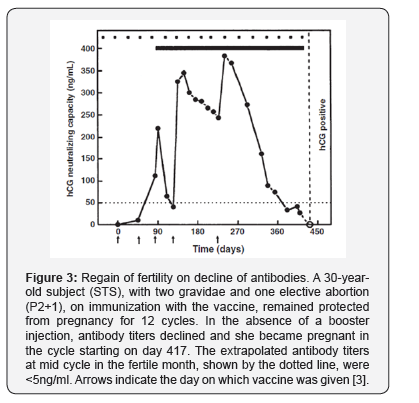
The vaccine was fully reversible. On decline of antibodies below 20ng/ml, STS Became pregnant and delivered a normal child (Figure 3). Four other women desirous of another child, did not take boosters of the vaccine, and became pregnant on decline of antibodies. They delivered normal babies, whose developmental landmarks and cognitive abilities were akin to their siblings [4]. Thus, the vaccine induced bio-effective antihCG antibodies preventing pregnancy. The antibodies declined to near zero level in all women in the absence of boosters, and the women regain fertility delivering normal babies.
Recombinant Anti-Hcg Vaccine
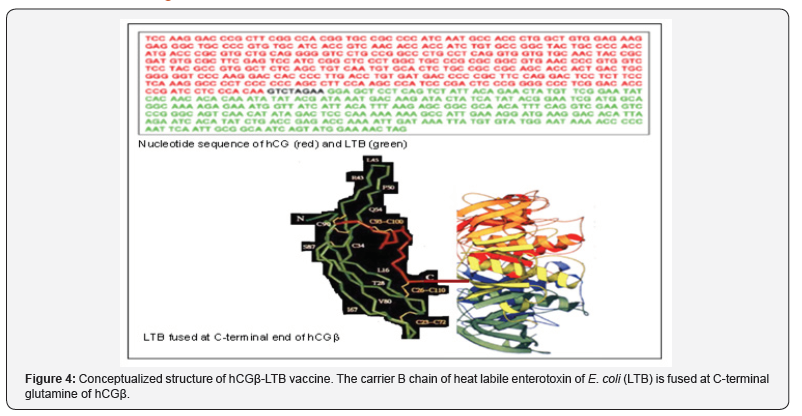
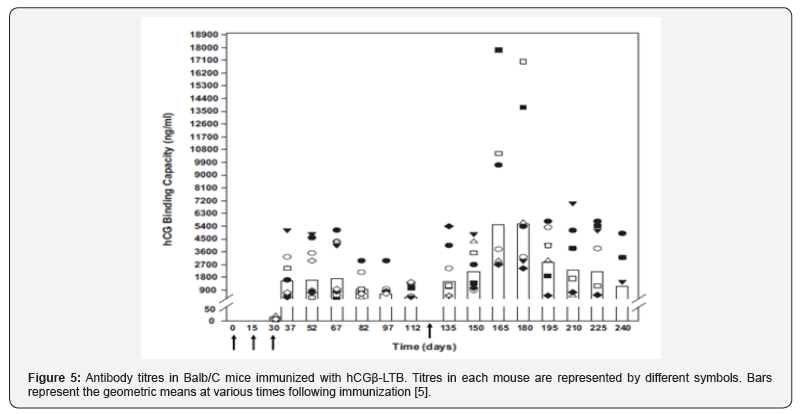
In order to render the vaccine amenable to industrial production, a genetically engineered vaccine was made. Figure 4 gives the nucleotide sequence of hCGβ-LTB vaccine. The vaccine adsorbed on alhydrogel given along with autoclaved Mycobacterium indicus pranii (MIP) as adjuvant, induced fairly high anti-hCG titres in Balb/C mice [5] as well as other strains of mice [6]. The complete safety of this vaccine was tested by a GLP compliant Company.
Their findings showed that the vaccine was non-sensitizing to the skin of guinea pigs with no clinical signs of toxicity, mortality and changes in body weight. The vaccine was non-mutagenic at the highest concentration tested by Bacterial Reverse Mutation and Mammalian Chromosome Aberration Tests. Similar observations on non-mutagenic property of the vaccines was made In-vivo by Mammalian Erythrocyte Micronucleus Test in Mice.
Single dose acute toxicity study was conducted in Sprague Dawley rats. No mortality, clinical signs of toxicity and treatment related changes in the body weight, were observed. No changes in gross pathology (external and internal) were observed at even the highest dose tested. Repeat doses of the vaccine were also administered to rats, which were followed up to 90 days post immunization. These studies showed no treatment related changes in physical, physiological, clinical, hematological parameters, as also in histopathology profiles of the organs. Segment II studies in rats showed that vaccine did not affect the embryo-foetal development. Body weight, food consumption, gross pathology remained normal, and no abnormal effect was observed in fetal sex ratio, fetal weight, external, visceral and skeletal norms of fetuses. The vaccine received approval of the RCGM (National Review Committee on Genetic manipulation). It also received the approval of the Drugs Controller General of India (DCGI) for going back to clinical trials.
The trials would be conducted under the Indian Council of Medical Research at the All India Institute of Medical Sciences, New Delhi and Sir Gangaram Hospital, New Delhi on a combined Phase I/II protocol. In Phase I, the immunogenicity and safety of the vaccine will be tested in 50 women of reproductive age and proven fertility, 10 women each at doses of 100μg, 200μg, 300μg, 400μg and 500μg per injection. The safety criteria include haematological parameters, lipid profile, liver function tests, kidney function tests, serum calcium and phosphorus, total protein, albumin, globulin, progesterone, TSH, prolactin, estrogen, auto antibodies, etc. Subjects will also be clinically examined for weight, blood pressure, and rogenism, pelvic TVS ultrasound, pyrexia, H/O joint pains, local reaction at the site of injection, persistent pain at the site of injection and swelling, if any.
After confirming the full safety of the vaccine and defining the optimum dose of the vaccine to generate high antibody titres against hCG, Phase II efficacy studies will begin in 70 women of proven fertility who are also sexually active.
Acknowledgement
The work received research grants from the Department of Biotechnology, Govt of India and the Indian Council of Medical Research, New Delhi.
References
- Talwar GP, Sharma NC, Dubey SK, Salahuddin M, Das C, et al (1976) Isoimmunization against human chorionic gonadotropin with conjugates of processed β-subunit of the hormone and tetanus toxoid. Proc Nac Acad Sci 73(1): 218-222.
- Talwar GP, Singh O, Rao LV (1988) An improved immunogen for antihuman chorionic gonadotropin vaccine elicited antibodies reactive with a conformation native to the hormone without cross-reaction with human follicle stimulating hormone and human thyroid stimulating hormone. Journal of Reprod Immunol 14(3): 203-212.
- Talwar GP, Singh O, Pal R, Chatterjee N, Sahai P, et al. (1994) A vaccine that prevents pregnancy in women. Proc Natl Acad Sci USA 91(18): 8532-8536.
- Singh M, Das SK, Suri S, Singh O, Talwar GP, et al. (1998) Regain of fertility and normality of progeny born at below protective threshold antibody titres in women immunized with HSD-TT vaccine. Am J Reprod Immunol 39(6): 395-398.
- Purswani S, Talwar GP (2011) Development of a highly immunogenic recombinant candidate vaccine against human chorionic gonadotropin. Vaccine 29(12): 2341-2348.
- Purswani S, Talwar GP, Vohra R, Pal R, Panda AK, et al. (2011) Mycobacterium indicus pranii is a potent immunomodulator for a recombinant vaccine against human chorionic gonadotropin. J Reprod Immunol 91(1-2): 24-30.






























