The Spindle of Oocytes Observed by Polarized Light Microscope Can Predict Embryo Quality
Tung Nguyen Thanh and Hang Doan Thi*
Centre for Human Reproduction and Embryology, Millitary Medical University, Hanoi, Vietnam
Submission: March 06, 2018; Published: May 08, 2018
*Corresponding author: Hang Doan Thi, Centre for Human Reproduction and Embryology, Millitary Medical University, Hanoi, Vietnam, Email: hangdt166@gmail.com
How to cite this article: Tung N T, Hang D T. The Spindle of Oocytes Observed by Polarized Light Microscope Can Predict Embryo Quality. J Gynecol Women's Health 2018; 9(4): 555766. DOI: 10.19080/JGWH.2018.09.555766
Summary
Objective: to evaluate spindle position of mataphase II oocyte and the development of embryos originated from oocytes with spindle and without spindle.
Methods: 250 MII oocytes were analysed with polarized microscope in IVF centre of Viet Nam Military Medical University.
Results: Spindles were detected in 170 (77.98%) of 218 metaphase II oocytes, 115 spindles (67.65%) of MII oocytes is beneath or adjacent to the first polar body, 55 oocytes had the spindle located between 300 and 1800 away from the first polar body. Fertilization rate and the rate of good quality embryos in oocytes with a visible spindle (77.98% and 61.02%) were higher than those in oocytes without a visible spindle (22.02% and 36.84%), the difference was statistically significant with p <0.001 and p <0.05.
Conclusion: the spindle position of mataphase II oocytes is not always beneath or adjacent to the first polar body. Fertilization rate and the rate of good quality embryos in oocytes with a visible spindle were higher than those in oocytes without a visible spindle.
Keywords: ICSI (intracytoplasmic sperm injection); Metaphase II oocyte; Polscope; Spindle
Introduction
Ovarian stimulation to make multiple of good oocytes and embryos is one of the most important steps to increase the succeed rate of In-vitro Fertilization. The abnormal genetic oocytes are made by ovarian stimulation with impossibility in passing the physical limitation that can cause failure of embryos development inside uterus. Oocyte selection with best quality without genetic abnormalities by non-invasive technique is essential. Embryologists give many morphology criteria to assess and select good oocytes and put the embryos into uterus. Cytoplasm, zona pellucida, and polar body can be observed by conventional Microscope while some other parts of cell is invisible. Polarized Microscope allows embryologist to obverse morphology of the spindle. The appearance of spindle helps us recognize either oocyte maturation or the quality of laboratory.
There are 2 goals in this research:
a. Evaluation of spindle location of mature oocytes
b. Evaluation of embryos development by oocytes with spindle and oocytes with not-observed spindle
Methods
Clinical samples
During the period covering January to March 2018, mature oocytes of IVF patient at IVF Center of Viet Nam Military Medical University. Selection criteria: patients under 35 years old, 3ng/ ml < AMH < 10ng/ml, the number of secondary follicle of 5-15 on the second or third day of menses cycle; the absence of polycystic ovaries, low response to ovary stimulation, low quantity and quality sperm.
Equipments and materials
POLARIZED LIGHT MICROSCOPE Ti-U Nikon TE2000 with Module polar AIDE - Octax laser system, Digital Camera. Cultural media of Vitrolife (Sweden). Bench top incubators (Cook).
Methods
Cross-sectional analysis Research.
Oocyte preparation
The controlled ovarian stimulation, following the GnRH antagonist protocol. Ovary was under ultrasound daily until having at least 3 follicles bigger than 17mm. Ovulation was triggered by hCG 5000IU injection. Retrieve Oocytes after injection hCG 35-36h. After retrieving, put oocytes in incubator at 37 °C, 5% CO2 for 2 hours. Then denude Oocytes by enzyme hyaluronidase, put fresh Oocytes in incubator for 1 hour. Evaluate maturation level of oocytes and spindle before ICSI.
Cultures
After ICSI 16-18h, put zygote into new medium (G1 plus). Evaluate Embryos in the second day, and then put them into G2 plus in Incubator Cook. Classify Embryos in the third day basing on criteria of Alpha of ESHRE 2011 [1].
Results
Following Table 1: in 218 matured oocytes, there are 170 visible spindle oocytes (77.98%) and 48 not observed spindle oocytes (22.02%).

The result of Table 2 showed that of 170 visible spindle matured oocytes, there were 115 oocytes (67.65%) with spindle directly beneath and adjacent to the first polar body, 40 oocytes (23.53%) with spindle located between 30 and 60 degrees away from the first polar body, 10 oocytes (5.88%) with spindle located between 60 and 120 degrees angle, 5 oocytes (2.94%) with spindle located between 120 and 180 degrees angle (Figure 1).
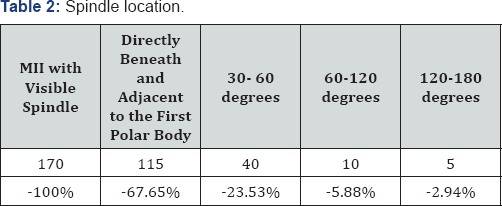
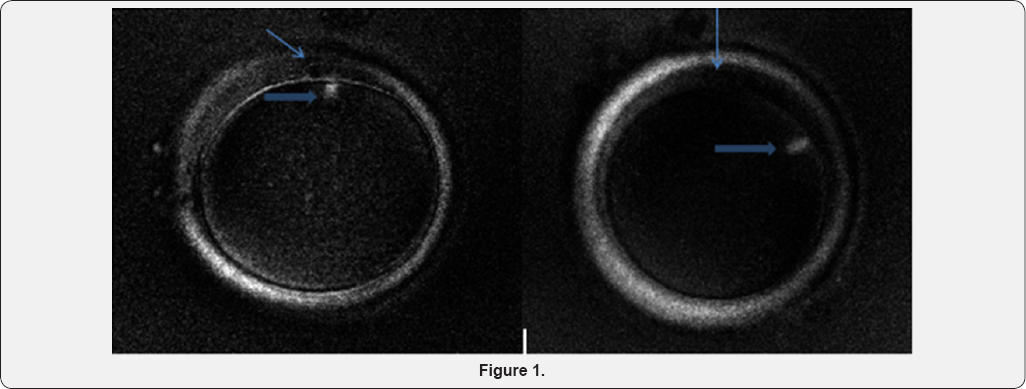
Image MII was observed by polarized microscope, with magnification 200X (the thin arrow: the first polar body. The bold arrow: meiosis spindle, beneath the first polar body, the right image shows that the spindle located in 70 degrees way from the first polar body.
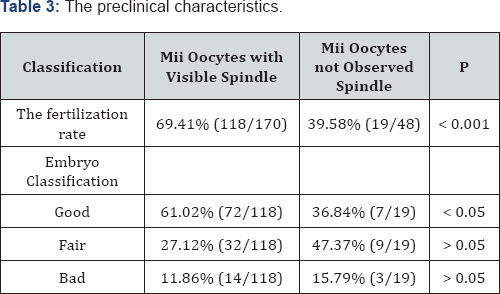
The fertilization rate of Mature oocytes with visible spindle was 69.41%, whereas this of mature oocytes with not-observed spindle was 39.58%. The difference was statistically significant with p <0.001. The rate of good embryos amonh MII oocytes with visible spindle was higher than that among MII oocytes with not-observed spindle (61.02% and 36.84%). The difference was statistically significant with p<0.05 (Table 3).
Discussion
In this research, oocytes were observed at 39 hours after hCG administration, which is the time that Cohen et. al. [2] had observed the spindle of MII oocytes with the highest rate (81.5%).
In this research, in spite of 48 oocytes with the first polar body, the rate of invisible spindle oocytes was 22.02%. To explain the absence spindle in MII oocytes, Montag et al. [3] using time- lapse to observe the oocyte maturation in the in-vitro showed that when MI oocytes extruded the first polar body, a spindle had established the bridge between the polar body and the oolemma for 75 -90 minutes. These imagine was recorded at Telophase 1 by polarized microscope. After extruding the first polar body, it took 115- 150 minutes to establish the spindle beneath the first polar body. Therefore, the spindle absence was in the oocyte meosis.
In ICSI, the spindle location of mature oocyte was involved to the first polar body. The ICSI micropipette had to avoid the spindle in the injection. However, several researches reported that the first polar body did not exactly predict the spindle location [4]. Thus the spindle could be injured when located far from the first polar body The use of polarized microscope could reduce spindle injured in ICSI. In this research, there were 8 oocytes (8.7%) with spindle located between 600 and 1200 degrees away the first polar body, this location was for injecting by the ICSI micropipette though out the 3-hour position in the oocyte. To explain the change of spindle location away from the first polar body, in the oocyte denudation by physical processing, the first polar body was moved. Another reason was that the fluid surround the spindle forced its movement [5].
Oocyte assessment by using polarization microscope improved the prediction of oocyte quality. In the Woodward's research 2008, the fertilization rate and embryo quality in day 3 and day 5 of the oocyte MII with visible spindle were higher and better than those of the oocyte MII with not-observed spindle [6]. In our research, fertilization rates between two groups were different significantly statistically (p <0.001). This result is similar to the result of Cohen et. al. 2004, Heindryckx 2011 [2,7].
There are many reasons for the IVF failure, in which the major reason is from chromosome abnormalities and 80% of aneuploidy embryos are from the oocyte meiosis 1 [8]. Chromosome dividing errors often happen in meiosis 1. The main mechanism is the error in homologous chromosome pairing in Prophase 1, recombination and early dividing of chromosome. Spindle controls the chromosome movement though many periods in meiosis and has a crucial role of meiosis [2]. Accordingn to Moon et. al. [6] implemented in 626 MII oocyte, the rate of good embryos among MII oocytes with visible spindle 64.2% was higher than that among MII oocytes with not-observed spindle 35,9% (p< 0.01), and the rate of multi-pronuclear in MII oocytes with visible spindle (3PN, 4PN) was reduced, compared to MU oocytes with not-observed spindle (5.9% and 10,7%) [9,10].
Conclusion
The spindle location may be far away from the first polar body. The fertilization and good embryo rates of MII oocytes with visible spindle were higher than those of MII oocytes with not- observed spindle.
References
- Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryo (2011) The Istabul consensus workshop on embryo assessment proceedings of an expert meeting. Human Reproduction 26(6): 1270-1283.
- Cohen Y, Malcov M, Schwartz T, Mey-Raz N, Carmon A, et al. (2004) Spindle iamging: a new marker for optimal timing of ICSI? Hum Reprod 19(3): 649-654.
- Montag M, Schimming T, van der Ven H (2006) Spindle imaging in human oocyte the impact of the meiotic cell cycle. Reprod Biomed Online 12(4): 442-446.
- Silva C S, Kapura K, Oldenboug R and Keefe D L (1999) The first polar body doesn't not predict accurately the location of the metaphase II meiotic spindle in mammalian oocytes. Fertil Reprod 13: 719-721.
- Moon JH, Hyun Ch S, Lee S W, Son W Y, Yoon S H, et al. (2003) Visualization of the metaphase II meiotic spindle in living human oocytes using the Polscope enables the prediction of embryonic developmental competence after ICSI. Human Reproduction 18(4): 817-820.
- Woodward B, Montgomery S, Hartshome G, Campbell K, Kennedy R (2008) Spindle position assessment prior to ICSI does not benefit fertilization or early embryo quality. Repro Biomed Online 16(2): 232238.
- Heindryckx B, Gheselle D S, Lierman S, Gerris J, Sutter DP (2011) Efficiency of palarized microscopy as a predictive tool for human oocyte quality. Human Reproduction 26(3): 535-544.
- Hassold T, Hunt P (2001) To err (meiotically; is human the genesis of human aneuploidy. Not Rev Genet 2(4): 280-291.
- Kathy L, Sharpe T, Randall L, Zimmer (2000) Oocyte and Pre-Embryo Classification. Hand book of the assisted reproduction laboratory, pp. 179-196.
- Palermo G, Joris H, Devroey P, Van Steirteghem AC (1992) Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 340(8810): 17-18.






























