The Impact of Hematinics Supplementation during Pregnancy on Maternal Anemia and Perinatal Outcome among Parturients in Southern Nigeria - a Prospective Study
Ogu Rosemary N* and Ikimalo John
Department of Obstetrics and Gynaecology, University of Port Harcourt, Nigeria
Submission: February 13, 2018 ; Published: March 28, 2018
*Corresponding author: Ikimalo John I, Department of Obstetrics and Gynaecology, University of Port Harcourt, Nigeria, Email: rosemary.ogu@uniport.edu.ng
How to cite this article: Ogu Rosemary N, Ikimalo John I. The Impact of Hematinics Supplementation during Pregnancy on Maternal Anemia and Perinatal Outcome among Parturients in Southern Nigeria - a Prospective Study. J Gynecol Women's Health 2018; 9(2): 555758. DOI: 10.19080/JGWH.2018.09.555758
Abstract
Introduction: Anemia is the commonest pregnancy complication worldwide. Anemia in pregnancy is a leading cause of maternal and perinatal morbidity and mortality especially in developing countries. It is a medical condition that is usually preventable through public health interventions, which are often feasible and cost effective. In order to strengthen the understanding of the disease for improved patient outcomes, this study sought to ascertain the prevalence of anemia in pregnancy, the impact of hematinics supplementation on maternal anemia prevalence, and the effect of maternal anemia on fetal outcomes at the University of Port Harcourt Teaching Hospital, in South-South Nigeria.
Methodology: The study was a prospective study of 430 pregnant women attending the antenatal clinic at the University of Port Harcourt Teaching Hospital. Participants were consecutive patients with singleton pregnancies booking for antenatal care who gave consent to be recruited for the study. Participants were enrolled at their booking antenatal visit and monitored through pregnancy till delivery. Hemoglobin concentration was used to assess the level of anemia.
Results: The prevalence of maternal anemia at the University of Port Harcourt Teaching Hospital using the WHO definition of less than 11g/ dl is 73.5% at booking and 47.9% at delivery. Defining anemia as hemoglobin less than 10g/dl, 34.0% of the participants were anemic at booking and 10.9% at delivery. Participants who received routine hematinics and antenatal care had hemoglobin levels which showed severity of anemia at booking greatly reduced by the time of delivery such that only 10.9% of parturients had moderate anemia compared to 39.5% of clients at booking. The incidence of low birth weight babies and preterm deliveries in women with anemia in pregnancy was statistically different from that of parturients with normal hemoglobin levels (p<0.05).
Conclusion: This study confirms that anemia in pregnancy is prevalent in our environment. The study also showed that the incidence of low birth weight babies and preterm deliveries in women with anemia in pregnancy was statistically different from that of parturients with normal hemoglobin levels. The only stillbirths recorded in this study were in patients with moderate anemia.
Keywords: Maternal anemia; Hematinics supplementation; Pregnancy
Introduction
Anemia in pregnancy has assumed important public health status worldwide. It is the commonest complication in pregnancy in sub Saharan Africa, more so with the advent of the HIV/AIDS pandermic [1]. Anemia in pregnancy is a leading cause of maternal and perinatal morbidity and mortality especially in developing countries. It is a medical condition that is usually preventable through public health interventions, which are often feasible and cost effective.
The World Health Organization (WHO) defines anemia in pregnancy as a hemoglobin concentration less than 11g/ dl and estimates that more than half of pregnant women in the world have a hemoglobin level indicative of anemia [2]. In combination with obstetric haemorrhage, anemia is estimated to be responsible for 17% to 46% of causes of maternal deaths [3-5]. The prevalence of anemia in Nigeria using WHO criterion range from 76% in Abeokuta [6], 67.4% in Enugu [7], 56.1% in Bayelsa [8] to 51.4% in Ibadan [9]. Anemia is classified as mild, moderate, severe and very severe and the WHO describes the hemoglobin level for each of these classes of anemia in pregnancy as 10-10.9g/dl, 7-9.9g/dl, 4-6.9g/dl and less than 4g/ dl, respectively [10].
The red cell is the component of the blood which has the major responsibility of transporting oxygen from the lungs to body tissues. Traditionally, anemia is defined as the presence of few red blood cells in the circulation or low hemoglobin concentration in the red blood cells [11]. In order to perform its important function of carrying oxygen from the lungs to body tissues, red blood cells contain hemoglobin. Normal hemoglobin consists of four globin chains each holding a haem molecule that contains iron. Globin is a complex protein containing a precise sequence of amino acids that allows it to fold into a complex pattern to favour uptake or unloading of oxygen [12]. In pregnancy, there is a dramatic disproportionate increase in total blood volume (up to 50% increase) and red cell mass (up to 18 - 25% increase depending on iron status) results in a decrease in hemoglobin concentration [13]. This does not normally fall below 11g/dl at any time during pregnancy. However deficiency of essential hematinics arising from increased requirements and inadequate intake have far-reaching effects on mother, fetus and neonate [13]. Increased blood loss at delivery, preterm birth, low birth weight babies, anemia in the first year of life, behavioral abnormalities and poor performance are observed associated complications of maternal anemia [13].
Iron deficiency is the most common cause of anemia in pregnancy. Iron deficiency anemia (IDA), has been reported to account for 75-95% of cases of anemia in pregnancy [14]. Iron-deficiency anemia, the late manifestation of chronic iron deficiency, is thought to be the most common nutrient deficiency among pregnant women [15]. It has been reported that over 40 million pregnant women suffer from iron deficiency and its consequences in developing countries [16].The high physiological requirement for iron in pregnancy is difficult to meet with most diets; this is so especially in developing countries where food requirement is a problem. This dietary difficulty therefore underlines the need for iron supplementation during pregnancy. Although routinely implemented, the effectiveness of hematinics supplementation in sub-Saharan Africa still needs to be documented.
Furthermore, the extent to which maternal anemia affects maternal and neonatal health is still uncertain [17]. Various studies have demonstrated a strong association between low hemoglobin concentration before delivery and adverse outcomes in terms of low birth weight, preterm delivery [18-20], poor apgar scores and neonatal morbidity [21,22], while others have not [23].
We therefore sought to determine the prevalence of anemia among our parturients and to determine whether this was affected by iron supplementation, and to ascertain the impact of maternal anemia to pregnancy outcome in our practice.
Methods
Study population
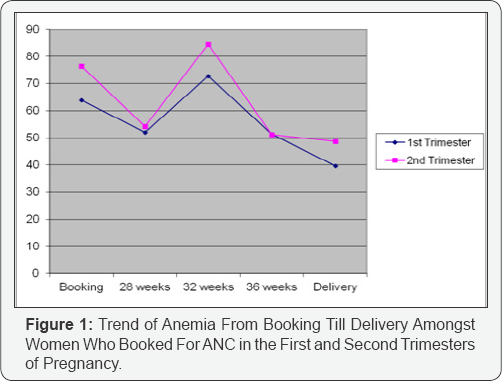

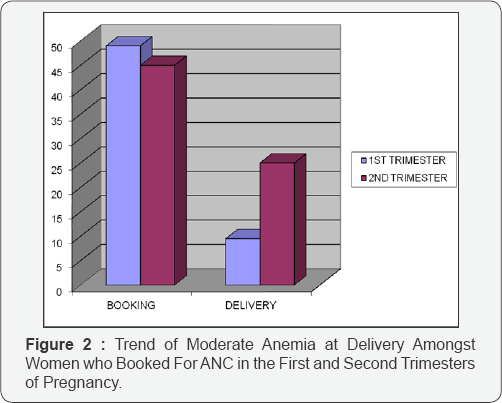
The study was a (prospective) cohort study of430 consecutive women with singleton pregnancies presenting for antenatal care at the University of Port Harcourt Teaching Hospital. The study was in 2 parts; in the first stage, a cross sectional survey was done to determine the prevalence and cases of anemia in pregnancy at booking. In the second stage, a cohort design was employed to measure the level of association between anemia and pregnancy outcome among the cohort of women. Women with anemia according to the WHO criteria of <11g/dl were identified as “cases”, while the “controls” were women seen at ante-natal clinic who had normal hemoglobin and were matched for age, parity and gestational length. Anemia is classified as mild, moderate, severe and very severe and the WHO describes the hemoglobin level for each of these classes of anemia in pregnancy as 10-10.9g/dl, 7-9.9g/dl, 4-6.9g/dl and less than 4g/dl, respectively [10]. In the University Of Port Harcourt Teaching Hospital, a study [14] on maternal anemia was carried out in 2004. This was however, a cross sectional study that used hemoglobin concentration of 10g/dl as definition of anemia.This value was also used as a benchmark in the evaluation of the prevalence of maternal anemia in this study (Figure 1 & 2).
The details of the study were thoroughly explained to all women booking for antenatal care at the booking clinic during the health talk session at the beginning of the clinic. Those who consented were then examined one after the other and the required information as well as phone numbers collected and entered onto the designed proforma. Blood samples were also collected and processed that same day. This cohort of women was followed up till delivery and phone calls were made to trace those missing any clinic appointments or passing their expected dates of delivery. Pregnancy outcomes were compared for both groups. The cohort of women was followed up till delivery. All participants were on routine hematinics of daily folic acid and ferrous sulphate as well as antimalarial prophylaxis with sulphadoxine pyrimethamine as usually given during the antenatal period. Any participant developing anemia at any point was treated as normally done in the antenatal period. Women used as controls who eventually became anemic were treated. All anemic patients, at recruitment or during the course of the study were treated. Analysis revealed the prevalence of anemia at booking, 28weeks, 32weeks and 36 weeks of gestation as well as the proportion of women developing anemia after booking, i.e. during the course of pregnancy.
The gestational age at booking and delivery, hemoglobin concentration at booking, 28weeks, 32weeks, 36 weeks and delivery as well as weight and Apgar score of the newborn were recorded. Designed proforma was used to record demographic information (age, educational status, marital status, parity, gestational age), clinical correlates (History of current pregnancy, Height, weight, blood group, genotype, hemoglobin concentration, blood film findings, urine analysis) and outcome measures (hemoglobin level at 28weeks, 32weeks, 36 weeks and delivery, gestational age at delivery, weight of newborn, apgar scores of newborn) for each participant. Written informed consent was obtained from participants and the ethical committee of the hospital gave approval for the study [24].
Sample size determination
Average prevalence of anemia in pregnancy is 50% [25]. The estimate from this study was desired to be within 5% of the actual prevalence with 95% confidence level (5% significance level). The sample size was thus calculated from the formula [26].
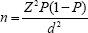
Where n is the sample size, Z is the proportion of normal distribution corresponding to the required significance level (5%) which is 1.96, P is the proportion of pregnant women with anemia which is 50% and d is the relative precision or tolerable error of the estimate from the study which is 5%.
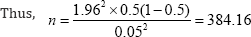
Deliberate over sampling for drop out and loss to follow up was 11.2%, the average attrition rate seen in similar studies done in Nigeria [9,24]. 11.2% of 384.16 are 42.26.
Therefore the minimum sample size for this study was 384 + 42 = 426.
For the cohort study, the minimum sample size for the study is determined as stated below using the formula for estimating sample size in comparison of two proportions [27]
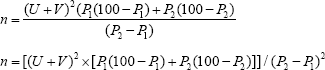
Where
n = minimum required sample size
P1 = estimated proportion of anemic pregnant women with adverse outcome
P2 = estimated proportion of non- anemic pregnant women with adverse outcome
U = the probability of finding a significant result = 0.84 which is the power of the study
V = percentage of the normal distribution corresponding to the two-sided significance level (at 5% significance level, V = 1.96)
However, 430 recruited for the cross sectional survey was used. This increased the internal validity of the study
Statistical analysis
All results were entered into the proforma and thence, into an excel worksheet in the computer and analysed using Epi- Info statistical software package. Descriptive statistics were computed for all relevant data. Association between anemia and pregnancy outcome was tested using ANOVA wherein the square root of the F value is equivalent to the Student's T- Test. All significance were reported at P<0.05.
Results
Maternal age and parity distribution
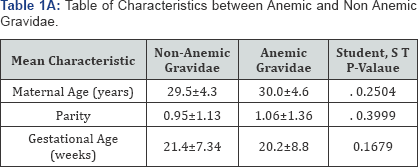
The maternal age of the subjects ranged between 14 and 43 years with a mean of 29.6±4.4 years. The mean age of the normal and anemic patients was 29.5±4.3 and 30.0±4.6 years respectively. This was not statistically significant with a p value >0.05 (Table 1A). Three hundred and sixteen (50.2%) of the subjects were aged between 25 and 34 years. The parity ranged between 0 and 5 with a mean of 0.98±1.2 for all subjects while it was 0.95±1.13 and 1.06±1.13 for the non anemic and anemic subjects respectively. This was not statistically significant with a p value >0.05. Most of the subjects 72.8% (303) were Para 0 or 1 while 1.4% (6) were grand multiparous.
*Statistically significant; P values less than 0.05.
Prevalence of anemia
Out of the 430 subjects recruited for the study, 316 had hemoglobin concentration less than 11g/dl at the time of booking, giving a prevalence rate of anemia at booking of 73.5%. Of the anemic subjects, 39.5% (170) and 33.5% (144) were mildly and moderately anemic respectively. Two (2) subjects i.e. 0.5% had severe anemia at booking (Table 1B). When a minimum hemoglobin concentration of less than 10g/dl was used to define anemia, 34.0% of the subjects were anemic at booking.
The prevalence of anemia at delivery after hematinics supplementation was 47.9% (158), with 37.0% (122) and 10.9% (36) having mild and moderate anemia respectively. No parturient presented with severe anemia. Using the minimum of hemoglobin concentration of less than 10g/dl to define anemia, only 10.9% (36) of the subjects were anemic at delivery.
The prevalence of anemia amongst primigravidae at booking was 74.1% while at delivery, it was 52.2%. At booking, mild, moderate and severe anemia were 35.5%, 38.1% and 0.5% respectively while at delivery, mild anemia was 39.1% while moderate anemia constituted 13.4%. Using the minimum of hemoglobin concentration of less than 10g/dl to define anemia, only 13.4% of primigravid parturients were anemic.
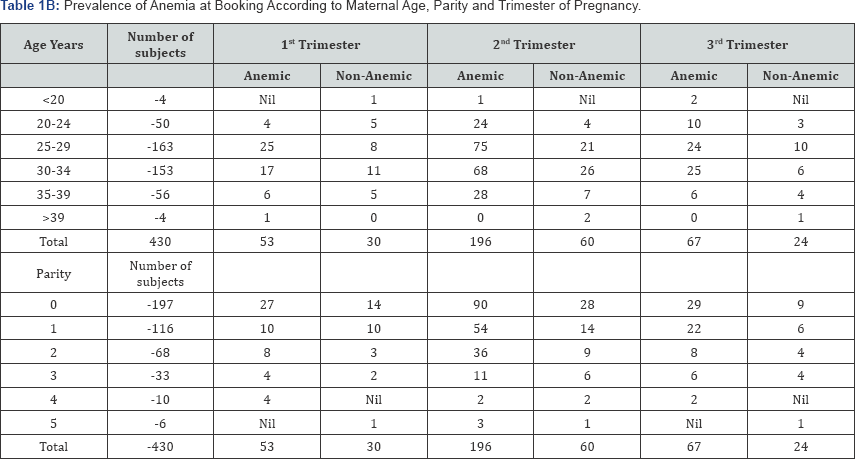
The prevalence of anemia amongst multiparae at booking was 73.0% while at delivery; it was 44.8%. At booking, mild and moderate anemia were 42.9% and 29.6% respectively while at delivery, mild anemia was 35.4% while moderate anemia constituted 9.4%. Using the minimum of hemoglobin concentration of less than 10g/dl to define anemia, only 9.4% of multiparous parturients were anemic.
Trend of anemia between time of booking & time of delivery; Effect of hematinics supplementation
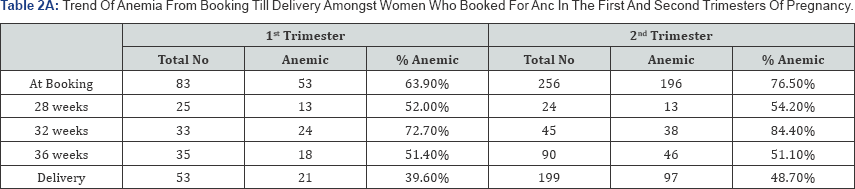
Eighty-three subjects were in their first trimester when they registered for antenatal care; of these, 63.9% (53) were anemic, by delivery, this had reduced to 39.6% (Table 2A). Amongst subjects registering for antenatal care in the first trimester, 49% (26) of those anemic had moderate anemia, by delivery, this had reduced to 9.5% (2) (Table 2B). This result revealed an improvement of maternal hemoglobin levels by 80.6%.
Of the two hundred and fifty six (256) subjects who registered for antenatal care in their second trimester, 76.5% (196) were anemic, by delivery; this had reduced to 48.7% (Table 2A). Amongst subjects registering for antenatal care in the second trimester, 45% (89) of those anemic had moderate anemia, by delivery, this had reduced to 25% (24) (Table 2B). This showed an improvement on maternal hemoglobin concentration by 44.4%.
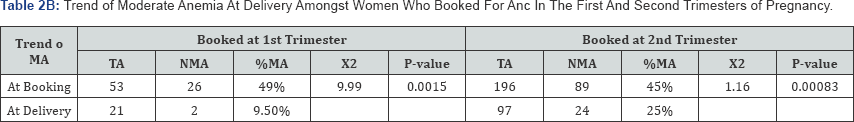
TA: Total Anemic, NMA: Number with Moderate Anemia, %MA: Percentage with Moderate Anemia,
*Statistically significant; P values less than 0.05.
Mean gestational age at booking and delivery
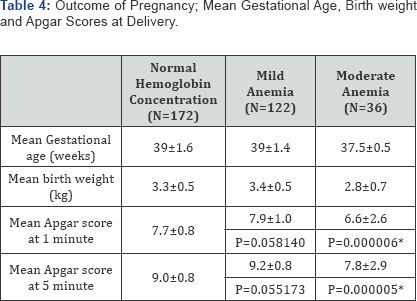
The total mean gestational age at booking was 21.1±7.7 weeks, range of 5 to 39 weeks while the median and modal age coincided at 20 weeks. The mean gestational length for the non anemic and anemic participants was 21.4±7.3 and 20.2±8.8 weeks respectively. This was not statistically significant with a p value > 0.05 (Table 1A). The total mean gestational age at delivery was 39.2±1.9 weeks. The range was 29 to 44 weeks while the modal age was 40 weeks and the median age 39 weeks. The mean gestational age at delivery was 39±1.6 weeks (range 29- 42), 39±1.4 weeks (range 37-44) and 37.5±0.5 weeks (range 30- 40) for the normal, mildly and moderately anemic clients respectively.
Mode of delivery (Table 3)
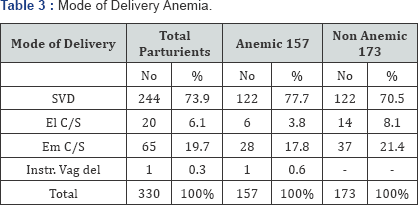

Spontaneous vertex delivery occurred in 244 (73.9%) patients. Most of the anemic patients (77.7%) achieved this as compared with 70.5% non anemic patients. Delivered by caesarean section occurred in 21.6% of the anemic patients as compared to 29.5% in the non-anemic patients. This was not statistically significant (p>0.05).
Fetal outcome, birth weight and apgar score (Table 4)
Three hundred and twenty six (98.8%) of the patients had live births, 100% of the non -anemic patients compared to 97.5% of the anemic patients. All the patients with mild anemia had live births. Still births occurred only in the moderately anemic patients.
*Statistically significant; P values less than 0.05.
The mean birth weight for the normal subjects was 3.3±0.5kg, range 1.1 to 3.6kg. For the mildly anemic subjects; it was 3.4±0.5kg, range 2.0 to 4.5kg. For the moderately anemic subjects; it was 2.8±0.7kg, range 0.7 to 4.6kg. This was statistically significant (p<0.05).
The mildly anemic and non anemic subjects had babies with better apgar scores compared with the moderately anemic patients. This was however not clinically significant (p>0.05) [28,29].
Discussion
The WHO defines anemia in pregnancy as a hemoglobin concentration less than 11 g/dl and estimates that more than half of pregnant women in the world have a hemoglobin level indicative of anemia.2The management and control of anemia in pregnancy is enhanced by the availability of local prevalence statistics for effective planning and implementation.
Table 1A shows the mean characteristics in terms of age, parity and gestational length between non- anemic and anemic participants and statistical analysis shows that the participants were well matched with no significant difference between the anemic and non-anemic participants.
The prevalence of anemia at booking using the WHO benchmark of 11g/dl recorded in this study (73.5%) is an indication that anemia during pregnancy is still a major problem in Nigeria. The high anemia prevalence is in agreement with data Ivoke et al28who reported a prevalence of 60% of maternal anemia in South-Eastern Nigeria. A study by Olatunbosun et al29in South-Southern Nigeria however reported a slightly lower prevalence of 54.5%. Data from the literature in developing countries have reported prevalence of anemia in pregnancy that ranged from 35.0 to 75.0% [30]. A prevalence of 73.5% in this study puts our finding in the upper limits of the range of maternal anemia in developing countries. The prevalence of anemia in pregnancy varies considerably because of differences in socio-economic conditions, life-styles and health-seeking behaviours across varying cultural settings. The prevalence of anemia at delivery was 47.9% with 37.0% and 10.9% having mild and moderate anemia respectively. No parturient presented with severe anemia.
When the minimum of hemoglobin concentration was set at less than 10g/dl to define anemia, only 10.9% of the subjects were anemic at delivery. These variations highlight the need to establish reference levels for local populations and for more confirmatory research. The severity of anemia at booking was greatly reduced by the time of delivery such that only 9.5% of parturients had moderate anemia compared to 49% of subjects booking at the first trimester (X2=9.99, p=0.0015). Similarly, there was also a statistically significant difference between the hemoglobin levels at booking in the second trimester and at delivery (X2=1.16, p=0.00083) (Table 2B) This finding is similar to that reported by Ouedraogo et al. [31] who also described a significant difference between hemoglobin levels at booking and at delivery following hematinics supplementation intrapartum. In developing countries, where diets are poor in iron but often rich in inhibitors of iron absorption [32], the issue of iron supplementation in pregnant women cannot be overemphasized. In tropical areas, many women start pregnancy with a lower store of iron [33] and undergo higher risks of anemia because of the increase in iron needs during gestation. As pregnancy progresses, it is known that iron requirements for fetal growth rise in proportion to the weight of the fetus, with most of the iron accumulating in the fetus during the third trimester. This therefore buttresses the importance of antenatal care and the efficacy of routine antenatal hematinics and anti- malarials.
From Table 2B, it can be seen that the measure of correction of anemia was greatest when subjects booked for antenatal care in the first trimester (80.6% versus 44.4%). Registering for antenatal care as early as possible should thus be advocated for
The total mean gestational age at delivery was 39.2±1.9 weeks. The mean gestational age at delivery was 39±1.6 weeks (range 29-42), 39±1.4 weeks (range 37-44) and 37.5±0.5 weeks (range 30-40) for the normal, mildly and moderately anemic subjects respectively. We found that 98.8% of the subjects had live births; 100% of the non-anemic subjects compared to 97.5% of the anemic subjects. Still births occurred only in the moderately anemic patients. This finding was in concert with that reported by Nair et al. [34] who found that perinatal mortality was more likely in women who were anemic during pregnancy Another study in Pakistan also found maternal anemia to be associated with increased risk of stillbirth [35]. The mean birth weight for the normal subjects was 3.3±0.5kg, range 1.1 to 3.6kg. For the mildly anemic subjects; it was 3.4±0.5kg, range 2.0 to 4.5kg while for the moderately anemic subjects; it was 2.8±0.7kg, range 0.7 to 4.6kg. This was statistically significant (p -value <0.05). The relation between maternal anemia and birth weight has been extensively reported by several investigators. In several studies, a U-shaped association was observed between maternal hemoglobin concentrations and birth weight [36,37]. Abnormally high hemoglobin concentrations usually indicate poor plasma volume expansion, which is also a risk for low birth weight [37]. Likewise, lower birth weights in anemic women have been reported in several studies [17,38]. We found that the mildly anemic and non anemic subjects had babies with better apgars cores compared with the moderately anemic patients although this was not clinically significant (Table 4). An association between maternal anemia and lower infant Apgar scores has been reported in some studies. In a Pakistani study, higher maternal hemoglobin concentrations were correlated with better Apgar scores and with a lower risk of birth asphyxia [39]. Also, when pregnant women were treated with iron or a placebo in Niger, Apgar scores were significantly higher in those infants whose mothers received iron [40].
Conclusion
This study confirms that anemia in pregnancy is prevalent in Nigeria. Moderate anemia is associated with poor fetal outcomes in terms of birth weight and gestational age at delivery. The situation is probably worse than reported because the population studied is a low risk group compared to those who do not receive antenatal care. Educating women on early ANC booking and compliance with the use of prescribed medications should be emphasized and is herein advocated for.
Limitation
We did not consider the effect of parasitic infections which can lead to anemia. We were therefore not able to determine their contribution to anemia in our study population.
References
- Kassebaum NJ, GBD 2013 Anemia Collaborators (2013) The global burden of anemia. Hematol Oncol Clin North Am 30(2): 247-308.
- WHO (2015) The global prevalence of anemia in 2011. World Health Organization, Geneva, Switzerland.
- Stevens GA, Finucane MM, De-Regil LM, Paciorek CJ, Flaxman SR, et al. (2013) Global, regional, and national trends in hemoglobin concentration and prevalence of total and severe anemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population-representative data. Lancet Glob Health 1(1): e16-e25.
- Harrison KA (1989) Maternal Mortality. Trans R Soc Trop Med Hyg 83(4): 449-453.
- Van den Broek N (2001) Anemia in Pregnancy in sub Saharan countries. Eur J Obstet Gynaecol Repro Biol 96(1): 4-6.
- Idowu DA, Mafiana CF, Sotiloye D (2005) Anemia in Pregnancy: A survey of pregnant women in Abeokuta, Nigeria. Afr H Sci 5(4): 295299.
- Iloabachie GC, Meniru GI (1990) The increasing frequency of anemia in pregnancy in Nigeria. Med Orient J Med 2(4): 194-197.
- Oboro VO, Tabowei TA, Jemikalajah J (2002) Prevalence and Risk Factors for Anemia in pregnancy in South Southern Nigeria. J Obstet Gynaecol 22(6): 610-613.
- Aimakhu CO, Olayemi O (2003) Maternal haematocrit and pregnancy outcome in Nigerian women. W Afr J Med 22(1): 18-21.
- WHO (2017) Nutritional Anemias: Tools for Effective Prevention and Control. World Health Organisation, Geneva, Switzerland.
- WHO (2011) Hemoglobin concentrations for the diagnosis of anemia and assessment of severity. Vitamin and Mineral Nutrition Information System. World Health Organization, Geneva, Switzerland.
- Forget BG, Bunn HF (2013) Classification of the Disorders of Hemoglobin. Cold Spring Harb Perspect Med 3(2): a011684.
- Soma-Pillay P, Nelson-Piercy C, Tolppanen H, Mebazaa A (2016) Physiological changes in pregnancy. Cardiovasc J Afr 27(2): 89-94.
- Halimi S, Halimi SMA, Shoaib M (2011) Oral versus parenteral iron therapy for correction of iron deficiency anemia in preganancy. Gomal Journal of Medical Sciences 9(1): 3-5.
- WHO (1992) The Prevalence of Anemia in Women: A Tabulation of Available Information, World Health Organization, Geneva, Switzerland.
- Development Initiatives (2017) Global Nutrition Report 2017: Nourishing the SDGs. Development Initiatives, Bristol, UK.
- Ahmad MO, Kalsoom U, Sughra U, Hadi U, Imran M (2011) Effect of maternal anemia on birth weight. J Ayub Med Coll Abbottabad 23(1): 77-79.
- Lone FW, Qureshi RN, Emanuel F (2004) Maternal anemia and its impact on perinatal outcome. Trop Med Int Health 9(4): 486-490.
- Steer P, Alam MA, Wadsworth J, Welch A (1995) Relationship between maternal hemoglobin concentration and birth weight in different ethnic groups. BMJ 310(6978): 489-491.
- Khan MM (2001) Effect of maternal anemia on fetal parameters. J Ayub Med Coll Abbottabad 13(2): 38-41.
- Ahmad MO, Kalsoom U (2015) Effect of maternal anemia on APGAR Score of newborn. Journal of Rawalpindi Medical College (JRMC) 19(3): 239-242.
- Abdel-Raoufabdel-Aziz R, Ali DK, Talkhan HM (2013) Pregnancy outcome and the effect of maternal nutritional status. J Egypt Soc Parasitol 43(1): 125-132.
- Masukume G, Khashan AS, Kenny LC, Baker PN, Nelson G (2015) Scope Consortium. Risk Factors and Birth Outcomes of Anemia in Early Pregnancy in a Nulliparous Cohort. PLoS One 10(4): e0122729.
- Fiebai PO (2003) The Prevalence of anemia in pregnancy at the University of Port Harcourt Teaching Hospital at booking and 36 week of gestation. Long commentary in obstetrics in partial fulfillment of the Part 2 book of the West Africa College of Surgeons.
- Bashiri A, Burstein E, Sheiner E, Mazor M (2003) Aneamia during pregnancy and treatment with intravenous Iron. Review of the literature. Eur J Obstet Gynecol Reprod Biol 110(1): 2-7.
- Mahajan BK (1997) Methods in Biostatistics for Medical Students and Research workers. In: Mahajan BK (Ed.), (6th edn), Jaypee Brothers Medical Publishers Ltd, New Delhi, India, pp. 88-102.
- Kirkwood B (1988) Essentials of Medical Statistics. Blackwell Scientific Publications, Oxford, UK, pp. 213-216.
- Ivoke N, Eyo JE, Ivoke ON, Nwani CD, Odii ES, et al. (2013) Anemia prevalence and associated factors among women attending antenatal clinics in South- Western Ebonyi State, Nigeria. International Journal of Medicine and Medical Sciences 46(4): 1354-1359.
- Olatunbosun O, Abasiattai AM, Bassey EA, James RS, Ibanga G, et al. (2014) Prevalence of anemia among pregnant women at booking in the University of Uyo Teaching Hospital, Uyo, Nigeria. BioMed Research International Volume 2014: Article ID 849080.
- Omigbodun AO (2004) Recent trends in the management of anemia in pregnancy. Tropical Journal of Obstetrics and Gynaecology 21(1): 1-3.
- Ouedraogo S, Koura GK, Accrombessi K, Bodeau-Livinec F, Massougbodji A, et al. (2012) Maternal Anemia at First Antenatal Visit: Prevalence and Risk Factors in a Malaria-Endemic Area in Benin. Am J Trop Med Hyg 87(3): 418-424.
- Baltussen R, Knai C, Sharan M (2004) Iron fortification and iron supplementation are cost effective interventions to reduce iron deficiency in four sub-regions of the world. J Nutr 134(10): 2678-2684.
- Scholl T (2011) Maternal iron status: Relation to fetal growth, length of gestation and the neonate's iron endowment. Nutr Rev 69(Suppl1): S23-S29.
- Nair M, Choudhury MK, Choudhury SS, Kakoty SD, Sarma UC, et al. (2016) Association between maternal anemia and pregnancy outcomes: a cohort study in Assam, India. BMJ Global Health 1: e000026.
- Lone FW, Qureshi RN, Emmanuel F (2004) Maternal anemia and its impact on perinatal outcome in a tertiary care hospital in Pakistan. East Mediterr Health J 10(6): 801-807.
- Dewey KG, Oaks BM (2007) U-shaped curve for risk associated with maternal hemoglobin, iron status, or iron supplementation. Am J Clin Nutr 106(Suppl 6): 1694S-1702S.
- Moghaddam TF, Barjasteh S (2015) Maternal Hemoglobin Levels during Pregnancy and their Association with Birth Weight of Neonates. Iranian Journal of Pediatric Hematology Oncology (5)4: 211-217.
- Rahmati S, Delpishe A, Azami M, Ahmadi MRH, Sayehmiri K (2017) Maternal Anemia during pregnancy and infant low birth weight: A systematic review and Meta-analysis. Int J Reprod Bio Med 15(3): 125134.
- Ahmad MO, Kalsoom U (2015) Effect of Maternal Anemia on Apgar score of Newborn. Journal of Rawalpindi Medical College 19(3): 239242.
- Preziosi P, Prual A, Galan P, Daouda H, Hercberg S (1997) Effect of iron supplementation on the iron status of pregnant women: Consequences for newborns. Am J Clin Nutr 66(5): 1178-1182.






























