Administration of Tocolytic Agents is not Only un useful but also Culprit to Prevent Preterm Labor
Masamitsu Nakamura* and Akihiko Sekizawa
Department of Obstetrics and Gynecology, Showa University School of Medicine, Japan
Submission: March 01, 2018; Published: March 13, 2018
*Corresponding author: Masamitsu Nakamura, Department of Obstetrics and Gynecology Showa University School of Medicine, 1-5-8 Hatanodai, Shinagawa-ku, Tokyo 142-8666, Japan, Tel: +81-3-3784-8551; Fax: +81-3-3784-8355; Email: mashamitsul975@gmail.com
How to cite this article: Masamitsu N, Akihiko S. Administration of Tocolytic Agents is not Only un useful but also Culprit to Prevent Preterm Labor. J Gynecol Women�s Health 2018; 8(5): 555750.DOI:10.19080/JGWH.2018.08.555750
Abstract
In Japan, long term tocolytic therapy for threatened preterm labor is still performed at many perinatal centers despite of less evidence for prevention against preterm delivery. There are some reports which tocolytic agents can prolong preterm delivery during antenatal corticosteroid therapy for maturation of fetal lung, or for maternal transport to a tertiary perinatal care center and which the administration of tocolytic agents to patients with threatened premature delivery was restricted due to side effects such as maternal lung edema or rhabdomyolysis. In addition to these side effects, long term administration for mother with intrauterine exposure for fetus in beta-2 receptor agonist including ritodrine hydrochloride is risk factor of childhood asthma. It was reviewed two reports about tocolytic agents including beta-2 receptor agonist to reveal that there is no evidence performing long term tocolytic therapy.
Keywords: Tocolytic therapy; Ritodrine hydrochloride; Asthma; Side effect
Needless of Long Term Tocolytic Therapy
Short-term tocolysis using ritodrine and/or magnesium sulfate for threatened preterm labor is not common in Japan. Long term tocolytic management strategy to prevent preterm birth may contribute to the epidemiological data that the Japanese preterm delivery rate was less than 6% in 2007, with the lowest perinatal mortality in the world [1]. However, there is few reports which long-term tocolysis was useful to prolong the pregnancy period [2].
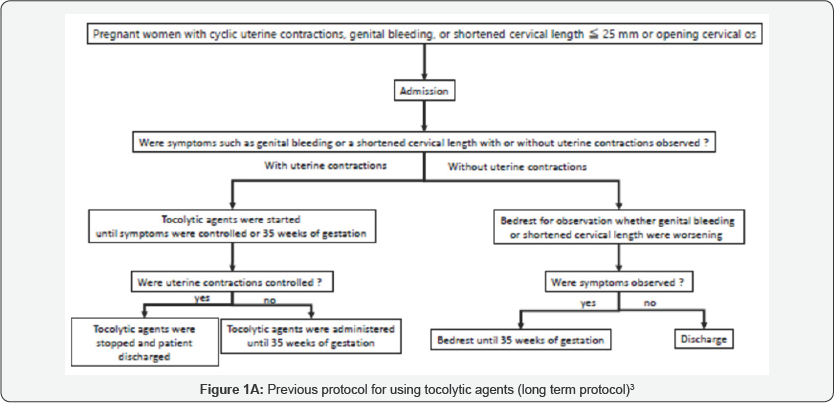
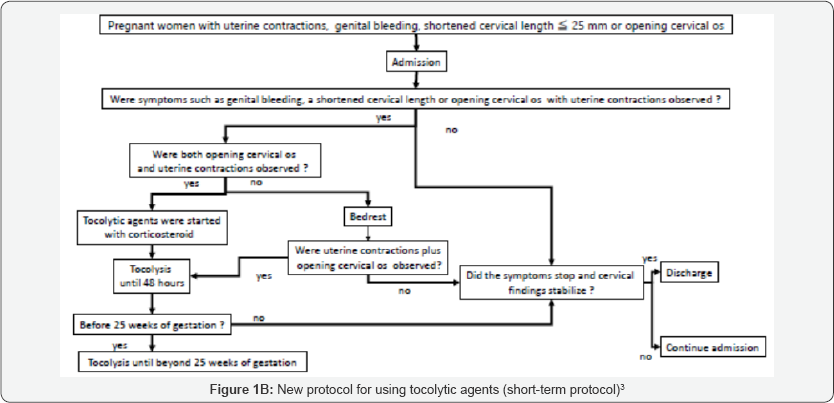
Natural preterm delivery due to onset of labor is thought to be caused by some inflammations in uterine cave as a maternal protective reaction. Such a onset of labor which would be occurred term or preterm cannot be controlled using tocolytic agents including ritodrine hydrochloride and magnesium sulfate. Nakamura et al. changed the management protocol for threatened preterm birth in January 2014 from long-term tocolysis to short-term tocolysis3 ( Figure 1). There were 1548 and 1444 deliveries before after 2014 during the study period. There was no significant change Gestational age (week) at delivery, neonatal body weight, and Apgar score (1/5 minutes) between the two groups, respectively. The outcomes of the subjects in both groups are demonstrated in Table 1. No significant differences were observed between the two groups for any of the parameters in the study. Japan currently has the lowest perinatal mortality worldwide [4]. Japanese obstetricians believe that long-term tocolysis has a definite contribution to the low preterm delivery and perinatal mortality rates. Although the rates of singleton preterm delivery in Estonia, Finland, Ireland, Lithuania and Sweden were less than 5% among European countries [5,6].
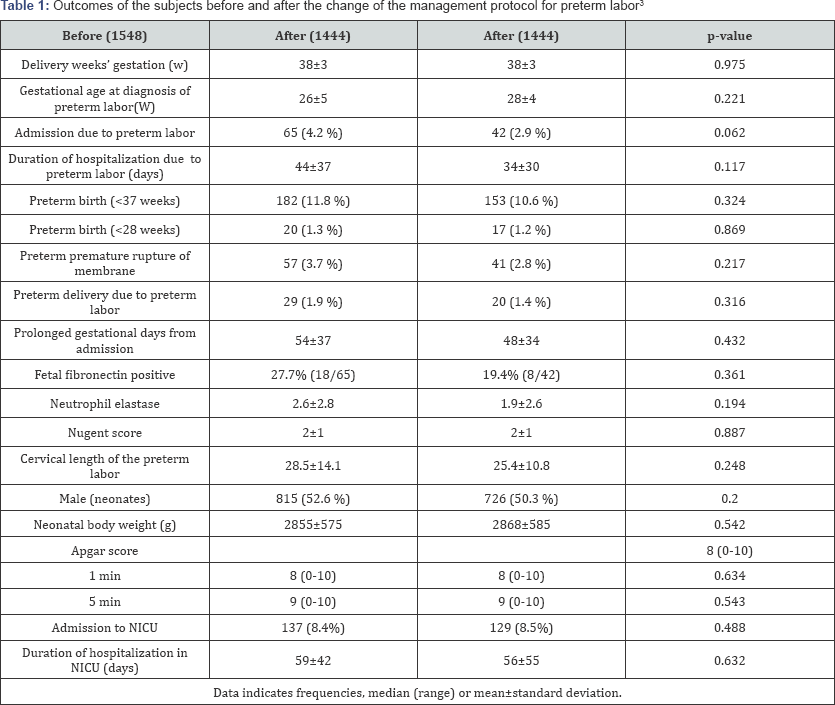
Because there was no significant change of perinatal outcomes before and after 2014, it was no need at least long term tocolytic therapy to extend a delivery period in cases with threatened preterm labor.
Maternal and Childhood Risks of Long Term Tocolytic Therapy
It has been reported that the frequencies of side effects of tocolytic agents were lower than those of the FDA recommendation6. Although it was reported that there are some 18.7% of side effects with ritodrine hydrochloride such as elevated liver enzyme, rhabdomyolysis, edema of lung, granulocytopenia, palpitation tachycardia, parotid bubo and hyeramylasemia in Japan7, the frequencies of side effects might be lower than in Western countries due to a lower prescribed dosage. Because long term tocolytic therapy could not improve the outcomes in cases with threatened preterm labor as mentioned previously, tocolytic agents might give a risk of side effect to such cases.
Ogawa K. et al. [8] have reported that beta-2 receptor agonist (B2RA) exposure to fetus in utero performed intravenous administration is a risk factor for childhood asthma until 5 years old. 1158 children were analyzed in this study. Children exposed to B2RA in utero with current asthma at 5 years old were increased to adjusted odds ratio 2.04 (95% confidence interval 1.02-4.05) compared to them non-exposed group. The risk for current asthma at 5 years old was total dose dependent during pregnancy according to the point which B2RA administration with low-dose was not significant compared to non-exposed group. In addition, Ogawa K et al. [9] reported that as dosage of oral administration was much smaller than that of intravenous, oral B2RA administration during pregnancy would not be a risk for current asthma at 5 years old. European Medicine Agency had decided to restrict the period of usage in Ritodrine hydrochloride and to revoke insurance approbation for oral administration of Ritodrine.
Discussion
It would be needed to be reconsidered the management using tocolytic agents for threatened preterm labor to the aspects of the side effects, cost and the bed for pregnant women who would need the perinatal management without threatened preterm labor.
Conclusion
In conclusion, merit of tocolysis for threatened preterm labor not only was small but also have some problems for maternal, fetal and childhood periods.
References
- Beck S, Wojdyla D, Say L, Pilar Betran A, Merialdi M, et al. (2009) The worldwide incidence of preterm birth: a systematic review of maternal mortality and morbidity. Bull World Health Organ 88: 31-38.
- Takagi K, Satoh T (2009) Multicentre Premature Labour Study G. Is long-term tocolysis effective for threatened premature labour? J Int Med Res 37: 227-239.
- Nakamura M, Hasegawa J, Arakaki T, Hamada S, Takita H et al. (2016) Comparison of perinatal outcomes between long-term and short-term use of tocolytic agent: a historical cohort study in a single perinatal hospital. J Obstet Gynaecol Res 42(12): 1680-1685.
- World Health Organization (2012) Neonatal and Perinatal Mortality: Country, Regional and Global Estimates. WHO, Geneva, Switzerland.
- Zeitlin J, Szamotulska K, Drewniak N, Mohangoo AD, Chalmers J, et al. (2013) Preterm birth time trends in Europe: a study of 19 countries. BJOG 120(11): 1356-1365.
- (2012) Statistics and Infomation Department, Minister's Secretariat, Mnistry of Health, Labour and Welfare: Vital Statistics of Japan 2010, Vol 1. Health, Labour and Welfare Statistics Association, Tokyo, Japan.
- Otsuki K (2016) The research report of Japan Perinatal Medicine Symposium working Committee, The Research report in Survey of Usage and Side effects in ritodrine hydrochloride. Perinatal Symposium 34: 15-18.
- Ogawa K, Tanaka S, Limin Y, Arata N, Sago H, et al. (2017) Beta-2 receptor agonist exposure in the uterus associated with subsequent risk of childhood asthma. Pediatr Allergy Immunol 28(8): 746-753.
- European Medicine Agency (2013) Restrictions on use of short- acting beta-agonists in obstetric indications, CMDh endorses PRAC recommendations. EMA, London, UK.






























