Hepatitis C in Pregnancy - Universal Screening in the Era of Newer Antivirals
Vedhapriya Srinivasan*1, Mark G Martens1 and Kathleen K Casey2
1Department of OBGYN, Jersey Shore Medical University Hospital, USA
2Department of Infectious Diseases, Jersey Shore Medical University Hospital, USA
Submission: August 30, 2017 ; Published: October 04, 2017
*Corresponding author: Vedhapriya Srinivasan, Department of OBGYN, Jersey Shore Medical University Hospital, Neptune, New Jersey, USA, Tel: 917-794-7380; Email: vedhavimal612@gmail.com
How to cite this article: Vedhapriya S, Mark G M, Kathleen K C. Hepatitis C in Pregnancy - Universal Screening in the Era of Newer Antivirals. J Gynecol Women�s Health 2017; 7(2): 555708.DOI:10.19080/JGWH.2017.07.555708
Abstract
Chronic hepatitis due to Hepatitis C virus is a public health problem of significant magnitude in the USA, affecting around 2.9-3.5 million people. In recent years, increased incidence of HCV infections have been noted among young people aged between 20-29 years and 3039 years. During the period between 2010-2014, it has been reported that HCV antibody detection rates have increased 22% in women of childbearing age in parallel with a 14% increase in detection of HCV RNA or antibodies in children <2 years in the United States. Intravenous drug abuse has been identified as the most common associated factor with this increased incidence in younger age groups. Studies have reported that self-reporting of risks factors like drug use especially past use may not be reliable for screening of Hepatitis C virus infections. Subsequently, the current risk factor based screening recommendations may under report the prevalence of HCV infections. Since 2011, newer direct acting antiviral drugs against HCV infections have been very effective in attaining sustained virological response with short treatment duration and minimal side effects. Expansion of screening in younger people irrespective of the risk factors helps in reducing the burden of the disease significantly. Universal screening for HCV infections in women of reproductive age group helps in diagnoses and treatment of affected women either pre -conceptionally or in postnatal period thereby decreasing disease burden and also eliminating vertical transmission of HCV, the most common cause of pediatric infections in developed countries.
Keywords: Hepatitis C; Direct acting antivirals; Injection drug abuse; Universal screening
Abbrevations: DAA: Direct Acting Antivirals; SVR: Sustained Virological Response; AASLD: American Association for the Study of Liver Diseases
Introduction
Hepatitis C virus (HCV) is a single stranded RNA virus causing mild transient illness that clears spontaneously in 2025% of cases. In 50-80% of infected persons, it may persist as chronic hepatitis C that may then progress to liver cirrhosis. Approximately 10-20% of people developing cirrhosis go on to develop complications such as decompensated liver disease, portal hypertension and hepatocellular carcinoma [1,2]. The Centers for Disease Control and Prevention [CDC] estimates that roughly 2.7-3.9 million people in the United States have chronic hepatitis C. Untreated chronic hepatitis C related mortality became the most common cause of infectious diseases related mortality in the USA in 2013, surpassing combined mortality due to 60 other reportable infectious diseases [3]. Though prevalence of the disease is highest among people born between 19451975, during the period between 2010-2014 the largest increase in incidence of acute hepatitis C infection was among people aged 20-29 years and 30-39 years [4]. Also during the period between 2010-2014, it has been reported that HCV antibody detection rates have increased 22% in women of childbearing age in parallel with a 14% increase in detection of HCV RNA or antibodies in children <2 years in the United States. This recent spike in incidence among younger age groups is associated especially with a history of injection drug abuse [5]. Recently approved Direct Acting Antivirals [DAA] have demonstrated a post treatment sustained virological response (SVR) of> 95% after a short treatment duration of only 8-12 weeks. Universal screening in previously unscreened women for HCV antibodies during gynecology office visits and during pregnancy may prevent underreporting of HCV infections that can occur with risk based screening strategy and give women effective treatment options. This should in turn, eliminate pediatric infections due to vertical transmission, the most common cause of chronic hepatitis C in children.
Hepatitis C in Pregnancy
In pregnant women, Hepatitis C infection is rarely diagnosed, as most of the infections are asymptomatic and screening is not universal. In 20-25% of these cases, the infection resolves but in the remainder it persists as chronic hepatitis C infection [6]. Chronic hepatitis C infection has not been previously found to have any impact on the course and outcome of pregnancy [7], but there are recent studies showing poor pregnancy and neonatal outcomes in women with HCV infection including increased risk of pre-term deliveries, pre labor rupture of membranes, gestational diabetes in those with excess weight gain, intrauterine growth retardation, low birth weight and NICU admission [8,9]. One of the most significant concerns of HCV infection during pregnancy is the vertical transmission of HCV. Vertical transmission of HCV occurs in about 3-5% mothers infected with chronic hepatitis C, with increased rates of transmission in mothers with HIV co infection (19%), intravenous drug use and high maternal HCV load. Transmission of the Hepatitis C virus can occur during intrauterine and perinatal period but postnatal transmission is very rare [10,11]. Factors associated with increased risk of maternal to child transmission are high viral load, prolonged rupture of membranes >6 hours, invasive intrapartum monitoring during labor such as scalp electrodes and exposure to maternal blood such as from second and third degree perineal lacerations. Mode of delivery does not seem to influence rates of transmission and there is no evidence supporting use of cesarean delivery to prevent Hepatitis C maternal to child transmission [12,13].
Pediatric Burden of Hepatitis C
Worldwide prevalence of Hepatitis C in children is 0.05% to 0.4% in developed countries and 2 -5% in resource limited settings, with vertical transmission as the most common cause of pediatric hepatitis C infection in developed countries [14]. The diagnosis of vertically transmitted hepatitis C infection is based on detecting HCV RNA in the infants between 2-5 months of age and again between 18-24 months or by persistence of HCV antibodies beyond 18 months as passively transferred maternal antibodies to the fetus may take 15 months to clear [15]. The course of HCV infection in children is generally milder than in adults but in a few it progresses to cirrhosis. Perinatal infections are asymptomatic and ALT levels don't correlate with severity of the disease. In a study done by the European Pediatric Hepatitis C Infection Network, 266 children infected by vertical infection were identified and were followed up until a median time of 4 years, 20-25% cleared their infections, 50% had chronic asymptomatic infection, 30% had chronic active infection. Most of the children were asymptomatic and only 10% had hepatomegaly. Growth was not affected in these children and there was no reduction in quality of life [16]. There were 67 pediatric patients who underwent liver transplantation for HCV infection in USA between 1988 and 2005. According to the recent NHANES survey, prevalence of chronic hepatitis C in children is between 26,000-46, 000 in the United States with projected health care cost between 199-336 million dollars [17]. Since perinatal infections are mostly asymptomatic, without pre-conceptional or antenatal screening of women for hepatitis C infection, most of the vertically infected infants can go undiagnosed. The FDA has recently approved the Sofosbuvir/ Ledipasvir combination for treatment of children aged between 12-17 years with chronic hepatitis C due to HCV genotypes 1,4,5, and 6 [18].
Risk Factor Based Vs Universal Screening
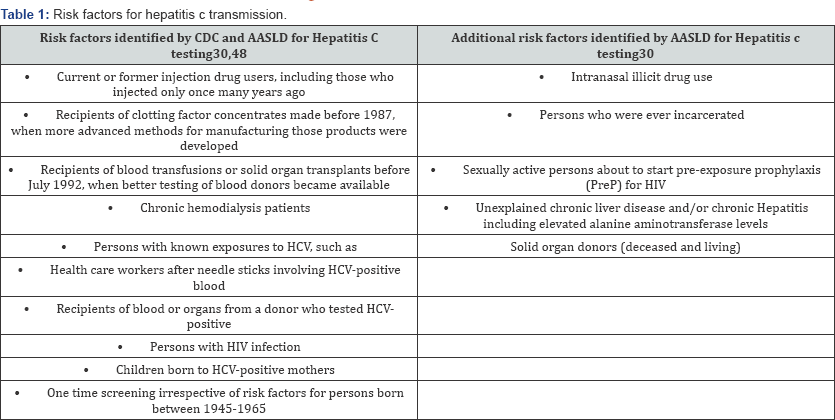
The CDC and American Association for the Study of Liver Diseases (AASLD) do not recommend universal screening for HCV among persons with no risk factors (Table 1). The emphasis on one time screening of people born between 1945-1965 has improved detection rates of chronic hepatitis C in baby boomers, but in recent years there is increased incidence of HCV in younger people with intravenous drug abuse emerging as the principal risk factor. Statewide surveillance for HCV infection between 2006-2012 showed a trend of increase in HCV infections in persons aged under 40 years and in around 73% of them injection drug use was identified as the principal risk factor [19]. Other studies have reported intranasal cocaine/heroin use to be a risk factor for HCV infection independent of intravenous drug use as HCV is found in nasal secretions of intra nasal cocaine users. Transmission of HCV among intranasal illicit drug users may be due to sharing of straws and other snorting devices and due to epistaxis experienced by intranasal illicit drug users [20-22]. A 2016 survey by National Institute on Drug Abuse found that 14.3% of 12 graders interviewed admitted using illicitdrugs, 1.3% of 10 th graders interviewed had history of cocaine use and 3.8% of 8th graders with inhalant use in the USA [23]. Selfreporting of cocaine and injectable drug use especially regarding past use may be unreliable and severely underreported especially in adolescents when used as the sole screening methodology and in close to 70% of cases no risk factor were identified on routine questionnaire [24,25]. Expansion of screening and treatment of HCV infections can reduce the burden of the disease significantly A study by Durham and colleagues, reported projected reduction in case burden of chronic hepatitis C by 80% as early as 2025, if screening is expanded and treatment rates are increased among persons with history of injection drug abuse. The same study reported that universal screening in people with no apparent risk factors could reduce the prevalence of HCV even further [26]. Studies have also reported that people prefer universal screening for infectious diseases [27] and women are most receptive to health interventions and screenings during preconception period and during pregnancy [28].
Direct Acting Anti Virals
Hepatitis C virus is made of core, E1, E2, p7structural proteins and non-structural (NS) proteins NS2, NS3, NS4A, NS4B, NS5A, and NS5B. The non-structural proteins NS3, NS4A, NS4B, NS5A, and NS5B are essential for virus replication and assembly; NS5B is a RNA dependent RNA polymerase. The newer direct acting antivirals target these proteins, disrupting viral replication and assembly [29,30]. Before the approval of DAA's, the standard therapy against HCV was combination of Interferon and Ribavirin, both of them contraindicated in pregnancy. Ribavarin is a known teratogenic drug and when used in reproductive age women, pregnancy should be ruled out before starting the drug and conception avoided until 6 months after treatment ends. In addition, the safety profile, efficacy in terms of sustained virological response is low and treatment duration longer comparedto the newer direct acting anti-virals. The newer DAA'S have shorter treatment duration [8-12 weeks], sustained virological responses of 95% and the regimens are ribavirin and interferon free. A brief overview of the newer DAA is given in Table 2.
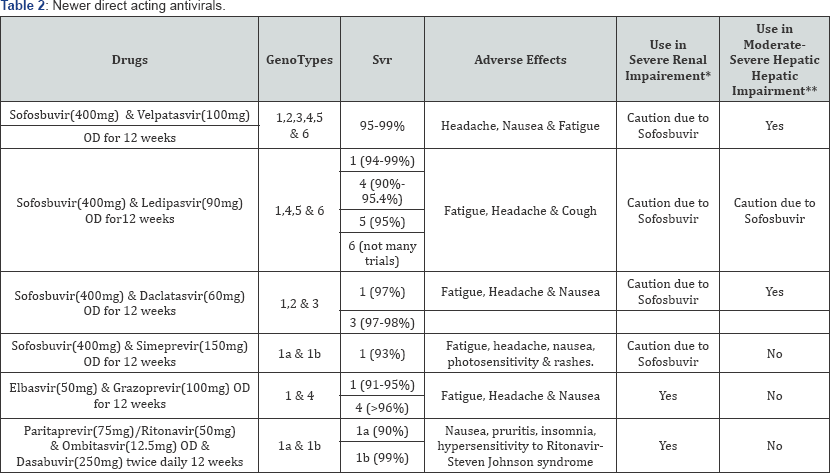
*Creatinine clearance is less than 30 ml/min,
**CTP B OR C with one or more of ascites, jaundice, hepatic encephalopathy, variceal hemorrhage.
Sofosbuvir is a nucleotide analogue inhibitor of HCV NS5B RNA dependent RNA polymerase and Velpatasvir is a new NS5A protein inhibitor with activity against all genotypes of HCV and a high barrier to resistance [31,32]. Sofosbuvir 400mg plus Velpatasvir 100mg in a fixed dose once daily combination has 99% sustained viral response rates against all genotypes and also in patients with cirrhosis and prior treatment failure [33]. Ledipasvir plus Sofosbuvir for 12 weeks has 94-99% rates of achieving sustained virological response against Hepatitis C virus genotype 1, 90-95% against genotype 4 and 95% against genotype 5 [34]. Daclatasvir, a HCV NS5A inhibitor has been approved for treatment of HCV genotypes 1 and 3 as single dose [60mg] once daily combination with Sofosbuvir 400mg with treatment duration of 12 weeks [35]. Though FDA has not approved this drug for treatment of chronic hepatitis C due to genotype 2, the AASLD recommends this drug combination for treatment of HCV genotype 2 [30].
Simeprevir, a competitive, reversible NS3/4A serine protease inhibitor, has been approved for treatment of HCV genotypes 1a and 1b in combination with sofosbuvir once daily for 12 weeks [SVR- 93%] [36]. Elbasvir 50mg and Grazoprevir 100mg taken orally once daily for 12 weeks has SVR of 91-95% against genotype 1 and SVR of >96% against genotype 4. This drug has been approved for treatment of chronic hepatitis C and compensated cirrhosis due to HCV genotypes 1 & 4 [30,37]. Ombitasvir is an inhibitor of NS5A, the phosphoprotein involved in HCV replication. Paritaprevir is an inhibitor of NS3/NS4A protease needed for viral replication. The plasma concentration of Paritaprevir is increased significantly when Ritonavir, a CYP3A4 inhibitor with no antiviral activity is added to regime and this also prolongs the elimination half-life of Paritaprevir [38]. Combination of Ombitasvir (12.5mg) /Paritaprevir (75mg)/Ritonavir (50mg) two tablets once daily and Dasabuvir (250mg) (PROD) a NS5B RNA polymerase enzyme twice daily has been approved for treatment of chronic Hepatitis C caused by HCV genotypes 1a and 1 b [30].
These drugs have good safety profile with fatigue, nausea and headache as their most common side effects. In patients with moderate or severe hepatic impairment, simeprevir, elbasvir-grazoprevir and ombitasvir/paritaprevir/ritonavir are contraindicated as they can worsen liver function further [38-40]. Sofosbuvir is predominantly renally excreted and safety of using the drug in patients with end stage renal disease and on dialysis has not been established [32,33].
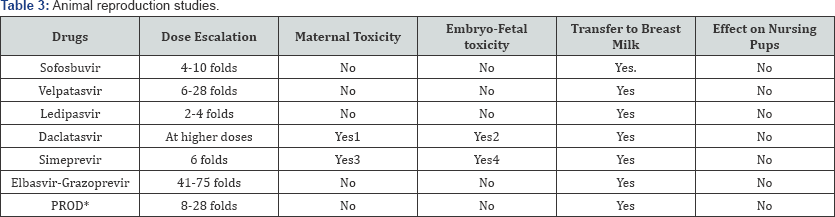
*Ombitasvir-Paritaprevir-Ritonavir-Dasabuvir- Ombitasvir
1. Reduced body weight & reduced food consumption
2. Malformations of Brain, skull, eyes, ears, lips, palate and limbs & reduced birth weight and growth
3. Maternal death
4. Early and late IU fetal losses & reduced fetal weight & skeletal variations.
None of the newer DAAs are approved to be used in pregnancy and during breast-feeding, as there are no human trials documenting the safety of these drugs in pregnant women. In animal reproductive studies done in pregnant rats and rabbits, Daclatasvir and Simeprevir use has been associated with maternal toxicity such as reduced food consumption, reduced body weight and embryo-fetal toxicity with early and late fetal losses, anomalies of skull, eyes, ears, palate, lips and skeletal system [41,42]. All DAA's are secreted into breast milk when given to lactating animals at varying concentrations, with Velpatasvir present in highest concentration in breast milk but none of them appear to have any negative effect on the nursing pups (Table 3) [43-46]. Though other combinations have shown no embryotoxicity in animal studies pregnant women are susceptible to drug induced hepatitis and these animal studies are not reflective of humans. So until drug registries for DAA'S have been established and human trials have reported the safety of these drugs during pregnancy its best to treat HCV infections pre conceptionally or in the post natal period.
Conclusion
Hepatitis C infection is a serious public health problem affecting approximately 2.9 -3.7 million people in the United States. The recent CDC statistics on surveillance for HCV infections for the year 2015 shows as pattern of increased incidence among young people with injection drug abuse as the most common associated factor [47]. The current screening recommendation for Hepatitis C is based on presence of risk factors. Multiple studies have reported that this risk factor based recommendation for screening of Hepatitis C may underreport the prevalence of chronic hepatitis Cin young people with history of past or current intravenous drug abuse. Expansion of screening for chronic hepatitis Cirrespective of risk factors will help in improving detection rates of chronichepatitis C and increase the linkage to care.
Universal screening for HCV antibody in women on their routine office visits or during preconception visits may help in diagnosis and treatment before conception. This in turn will help in reducing the disease burden significantly and in eliminating mother to child transmission of Hepatitis C virus. Women who have not been screened during preconception visits can be screened during antenatal visit. This helps in testing and follows up of children who are vertically infected and treatment of the infected mothers postnatal before subsequent conception.
In many places, obstetricians and gynecologists are the only doctors women seek for their health care. So it is incumbent upon us to create awareness of Hepatitis C among young women and to expand screening for HCV irrespective of the risk factors. Improved detection, treatment of affected women and elimination of perinatal infections in children ultimately help in achieving the goal of eradicating liver diseases due to chronic hepatitis C infection.
References
- Ponziani FR, Mangiola F, Binda C, Zocco MA, Siciliano M, et al. (2017) Future of liver disease in the era of direct acting antivirals for the treatment of Hepatitis C. World J Hepatol 9(7): 352-367.
- Hoshida Y, Fuchs BC, Bardeesy N, Baumert TF, Chung RT (2014) Pathogenesis and prevention of Hepatitis C virus-induced hepatocellular carcinoma. J Hepatol 61(1 Suppl): S79-S90.
- Ly KN, Hughes EM, Ruth B Jiles, Scott D Holmberg Division of Viral Hepatitis, Centers for Disease Control and Prevention, Atlanta, Georgia- Rising Mortality Associated With Hepatitis C Virus in the United States, 2003-2013- Clinical Infectious Diseases.
- Centers for Disease control and Prevention-Surveillance for viral Hepatitis 2014.
- Koneru A, Nelson N, Hariri S, Canary L, Sanders KJ, et al. (2016) Increased Hepatitis C Virus (HCV) Detection in Women of Childbearing Age and Potential Risk for Vertical Transmission- United States and Kentucky, 2011-2014. MMWR Morb Mortal Wkly Rep 65: 705-710.
- Prasad MR, Honegger JR (2013) Hepatitis C Virus in Pregnancy. Am J Perinatol 30(2): 149-159.
- Floreani A, Paternoster D, Zappala F, Cusinato R, Bombi G, et al. (1996) Hepatitis C virus infection in pregnancy. Br J Obstet Gynaecol 103(4): 325-329.
- Huang Q, Hang L, Zhong M, Gao Y, Luo M, et al. (2016) Maternal HCV infection is associated with intrauterine fetal growth disturbance: A meta-analysis of observational studies. Medicine (Baltimore) 95(35): e4777.
- Pergam SA, Wang CC, Gardella CM, Sandison TG, Phipps WT, et al. (2008) Pregnancy Complications Associated with Hepatitis C: Data from a 2003-2005 Washington State Birth Cohort. Am J Obstet Gynecol 199(1): 38.e1-38.e9.
- Floreani A (2013) Hepatitis C and pregnancy. World Journal of Gastroenterology : WJG 19(40): 6714-6720.
- Yeung CY, Lee HC, Chan WT, Jiang CB, Chang SW, et al. (2014) Vertical transmission of Hepatitis C virus: Current knowledge and perspectives. World Journal of Hepatology 6(9): 643-651.
- Tovo PA, Calitri C, Scolfaro C, Gabiano C, Garazzino S (2016) Vertically acquired Hepatitis C virus infection: Correlates oftransmission and disease progression. World J Gastroenterol 22(4): 1382-1392.
- European Paediatric Hepatitis C Virus Network (2001) Effects of mode of delivery and infant feeding on the risk of mother-to-child transmission of Hepatitis C virus. BJOG 108(4): 371-377.
- Khaderi S, Shepherd R, Goss JA, Leung DH (2014) Hepatitis C in the pediatric population: Transmission, natural history, treatment and liver transplantation. World J Gastroenterol 20(32): 11281-11286.
- El-Guindi MA (2016) Hepatitis C Viral Infection in Children: Updated Review. Pediatr Gastroenterol Hepatol Nutr 19(2): 83-95.
- The European Paediatric Hepatitis C Virus Network (2005) Three Broad Modalities in the Natural History of Vertically Acquired Hepatitis C Virus Infection. Clin Infect Dis 41(1): 45-51.
- Jhaveri R, Grant W, Kauf TL, McHutchison J (2006) The burden of Hepatitis C virus infection in children: estimated direct medical costs over a 10-year period. J Pediatr 148(3): 353-358.
- Infectious Disease Society of America (2017) FDA approval of treatment for HCV in children and adolescents.
- Zibbell JE, Iqbal K, Patel RC, Suryaprasad A, Sanders KJ, et al. (2015) Increases in Hepatitis C Virus Infection Related to Injection Drug Use Among Persons Aged ≤30 Years- Kentucky, Tennessee, Virginia, and West Virginia, 2006-2012- Morbidity and Mortality Weekly report.
- Allison RD, Conry-Cantilena C, Koziol D, Schechterly C, Ness P, et al. (2012) A 25-Year Study of the Clinical and Histologic Outcomes of Hepatitis C Virus Infection and Its Modes of Transmission in a Cohort of Initially Asymptomatic Blood Donors. J Infect Dis 206(5): 654-661.
- Keen L, Khan M, Clifford L, Harrell PT, Latimer WW (2014) Injection and Non-Injection Drug Use and Infectious Disease in Baltimore City: Differences by Race. Addict Behav 39(9): 1325-1328.
- McMahon JM, Simm M, Milano D, Clatts M (2004) Detection of Hepatitis C virus in the nasal secretions of an intranasal drug-user. Annals of Clinical Microbiology and Antimicrobial 3: 6.
- NIDA (2016) Teen substance use shows promising decline. National Institute on Drug Abuse website.
- Delaney-Black V, Chiodo LM, Hannigan JH, Greenwald MK, Janisse J, et al. (2010) Just Say "I Don't”: Lack of Concordance between Teen Report and Biological Measures of Drug Use. Pediatrics 126(5): 887-893.
- Garg M, Garrison L, Leeman L, Hamidovic A, Borrego M, et al. (2016) Validity Of Self-Reported Drug Use Information Among Pregnant Women. Maternal and Child Health Journal 20(1): 41-47.
- Durham DP, Skrip LA, Bruce RD, et al. The Impact of Enhanced Screening and Treatment on Hepatitis C in the United States. Clin Infect Dis 62(3): 298-304.
- Coffin PO, Stevens AM, Scott JD, Stekler JD, Golden MR (2011) Patient acceptance of universal screening for Hepatitis C virus infection. BMC Infectious Diseases 11: 160.
- Ward C, Tudor-Williams G, Cotzias T, Hargreaves S, Regan L, et al. (2000) Prevalence of Hepatitis C among pregnant women attending an inner London obstetric department: uptake and acceptability of named antenatal testing. Gut 47(2): 277-280.
- Aligeti M, Roder A, Horner SM (2015) Cooperation between the Hepatitis C Virus p7 and NS5B Proteins Enhances Virion Infectivity. Journal of Virology 89(22): 11523-11533.
- Recommendations for Testing, Managing, and Treating Hepatitis C -American Association for the Study of Liver Diseases.
- Puoti M, Panzeri C, Rossotti R, Baiguera C (2014) Efficacy of Sofosbuvir- based therapies in HIV/HCV infected patients and persons who inject drugs. Digestive and Liver Disease 46(Suppl 5): S206-S211.
- Smolders EJ, de Kanter CTMM, van Hoek B, Arends JE, Drenth JPH, et al. (2016) Pharmacokinetics, Efficacy, and Safety of Hepatitis C Virus Drugs in Patients with Liver and/or Renal Impairment. Drug Safety 39(7): 589-611.
- Bonaventura A, Montecu cco F (2016) Sofosbuvir/velpatasvir: A promising combination. World J Hepatol 8(19): 785-789.
- Nkuize M, Serste T, Buset M, Mulkay JP (2016) Combinational edipasvir- Sofosbuvir for the treatment of chronic Hepatitis C virus infection: a review and clinical perspective. Ther Clin Risk Manag 12: 861-872.
- Young J, Weis N, Hofer H, Irving W, Weiland O, et al. (2017) The effectiveness of daclatasvir based therapy in European patients with chronic Hepatitis C and advanced liver disease. BMC Infectious Diseases 17(1): 45.
- Izquierdo L, Helle F, Francois C, Castelain S, Duverlie G, et al. (2014) Simeprevir for the treatment of Hepatitis C virus infection. Pharmgenomics Pers Med 7: 241-249.
- Yao Y, Yue M, Wang J, Chen H, Liu M et al. (2017) Grazoprevir and Elbasvir in Patients with Genotype 1 Hepatitis C Virus Infection: A Comprehensive Efficacy and Safety Analysis. Can J Gastroenterol Hepatol 8186275.
- Smith MA, Lim A (2015) Profile of paritaprevir/ritonavir/ombitasvir plus dasabuvir in the treatment of chronic Hepatitis C virus genotype 1 infection. Drug Design, Development and Therapy 9: 6083-6094.
- Gaetano JN (2014) Benefit-risk assessment of new and emerging treatments for Hepatitis C: focus on simeprevir and Sofosbuvir. Drug, Healthcare and Patient Safety 6: 37-45.
- Bell AM, Wagner JL, Barber KE, Stover KR (2016) Elbasvir/ Grazoprevir: A Review of the Latest Agent in the Fight against Hepatitis C. International Journal of Hepatologyn 2016: 3852126.
- Center for Drug Evaluation and Research- US food and drug administration-daklinza (daclatasvir) tablets, for oral use-highlights of prescribing information.
- Center for Drug Evaluation and Research- U.S. food and drug administration- olysio (simeprevir) capsules, for oral use.
- Center for Drug Evaluation and Research- U.S. Food And Drug Administration-Epclusa® (Sofosbuvir and velpatasvir) tablets, for oral use Initial U.S.
- Center for Drug Evaluation and Research- U.S. Food And Drug Administration.
- Center for Drug Evaluation and Research- U.S. Food And Drug Administration- Zepatier™ (Elbasvir And Grazoprevir) Highlights Of Prescribing Information.
- Center for Drug Evaluation and Research- U.S. Food And Drug Administration -Viekira Pak (Ombitasvir, Paritaprevir, And Ritonavir Tablets; Dasabuvir Tablets), Co-Packaged For Oral Use.
- Centers for Disease Control and Preventio -Summary Viral Hepatitis Surveillance summary - United States.
- Centers for Disease Control and Prevention-Viral Hepatitis-Testing Recommendations for Hepatitis C infection.






























