MTHFR Testing in ART Should be Compulsory for both Male and Female Patients Including Oocyte Donation Programs
Laetitia Jacquesson-Fournols1, Edouard J Servy2, Brian Dale3,4, Arthur Clement5 and Yves JR Menezo*4,5
1Boulevard de Courcelles, France
2Servy-Massey Fertility Institute, USA
3Centro Fecondazione Assistita, Italy
4London fertility associates, UK
5Laboratoire Clement, avenue d'Eylau, France
Submission: September 14, 2017 ; Published: October 04, 2017
*Corresponding author: Yves JR Menezo, Laboratoire Clement, avenue d'Eylau, France, Email: yves.menezo@gmail.com
How to cite this article: Laetitia J F, Edouard J S, Brian D, Arthur C, Yves JR M. MTHFR Testing in ART Should be Compulsory for both Male and Female Patients Including Oocyte Donation Programs. J Gynecol Women�s Health 2017; 7(2): 555706 DOI:10.19080/JGWH.2017.07.555706
Summary
In a short cohort of patients with repeat miscarriages and ART failures, we observed a strong impact of the C677T MTHFR isoform. Both partners seem to be responsible for the failure, not only the woman. We observed that oocyte donation failures could be attributed in part to the male partner. It is imperative to address a question regarding oocyte donors, since MTHFR testing is not usually performed by oocyte banks. Also, testing should be mandatory in couples with repeat miscarriages and ART failures. Testing the donor, the recipient and the male partner should be considered as proper medical practice Testing donors in sperm cryobanks should not be overlooked.
Introduction
Methylation is a fundamental biochemical process where methyl groups are added covalently to lipids, proteins and DNA. In the reproduction field, the most important events are DNA and histone methylation which are involved in the process of epigenesis and imprinting. Most of the DNA methyl tags are erased during early embryo development and are restored during prenatal life in males, and during post-natal follicle development in females. Germ-cell-specific methylation and genomic imprints are established via de novo DNA methylation during oogenesis and spermatogenesis, through DNA methyltransferase(s) activity. The universal cofactor for methylation is S-Adenosyl methionine (SAM). Normally, the oocyte and early embryo have a total capacity to synthesize SAM [1] and all the in the methylation process: all the step-enzymes involved are expressed but this capacity is decreasing with maternal age [2]. Once the target molecule has been methylated, S-Adenosyl homocysteine (SAH) is formed (Figure 1) and homocysteine (Hcy) is released after hydrolysis. Hcy is a toxic compound that must be recycled in Methionine via the One Carbon Cycle (1-CC); but the efficacy of the 1-CC relies strongly on the folic acid cycle (Figure 1). To be metabolically active, folic acid (and folates) must be converted to Methyl Tetrahydrofolate (MTHF) by the Dihydrofolate reductase (DHFR). Then THF can be transformed to the biologically active l-methyl folate by the enzyme methylenetetrahydrofolate reductase (MTHFR) and enter the 1-CC, to allow the conversion of Homocysteine to Methionine. MTHFR activity is a critical step. Even if some data in literature are confusing [3] there are now strong evidences indicating that MTHFR isoforms, especially T677T are noxious for fertility in women [4] and men [5]. T677T isoform severely alters preimplantation development and increases chromosomal abnormalities. As this isoform is generally not tested in couples, we have studied the genetic background of several patients with recurrent severe infertility, especially in oocyte donation failures. We then tested in these isoform carriers whether treatment with preparations containing 5-MTHF (Impryl R, Parthenogen Switzerland and Tetrafolic R, NuriliaR France), could help in solving some of the problems. For the patients, we have prescribed an oral daily dose of 500 micrograms of Methylfolate preparation containing also a B complex vitamin. It has to be administrated for a period of 4 months preceding ART or unprotected intercourse In this brief report we present our preliminary observations.
Case Reports
Patients with repeat miscarriages
Five patients with a total of 23 miscarriages, all the women had a normal Karyotypes. After control, four women are MTHFR C677T heterozygote (HTZ) and their partners are also C677T carriers. After 5-MTHF treatment of the 2 members of the couples, (One with metafolin, two with Impryl one with tetrafolic), two women have already delivered (two girls), one is 12 week pregnant and the other one has also an ongoing pregnancy at 25 weeks. For another couple, the female patient is Wild type (WT C677C) and her husband is homozygote (HMZ T677T); they are currently under treatment. Two of these patients had been previously counselled for oocyte donation.
Repeat Assisted Reproduction technology (ART) failures
Patient DC/Y: 3 IVF-ICSI, 27 oocytes collected, 2 embryo transfers, no pregnancy. Both patients are HMZ T677T, currently under treatment.
Patient LFF/B: 2 miscarriages post spontaneous pregnancies, 2 IVF-ICSI failures for 23 oocytes retrieved, no blastocyst formation observed; Woman HTZ, man HMZ T677T isoform. Currently under treatment.
Patient DDL/B: 4 IVF-ICSI failures for 25 oocytes retrieved. Post control Woman HMZ T677T, Man HTZ. Patients have given up.
Patients with oocyte donation failures
Patient LFJ/T: Two oocyte donation failures. Woman HTZ, Man HMZ for MTHFR T677T. Currently under treatment.
Patient LJF/ S: one miscarriage post spontaneous pregnancy 3 oocyte donation failures. Woman WT, Man HMZ for MTHFR T677T, his level of circulating homocysteine is dangerously high 55micromoles/L.The patients are currently under treatment.
Patient LFJ/D: Several ART failures. 2 oocyte donation failures. Post control both partners are C677T.
Discussion and Conclusion
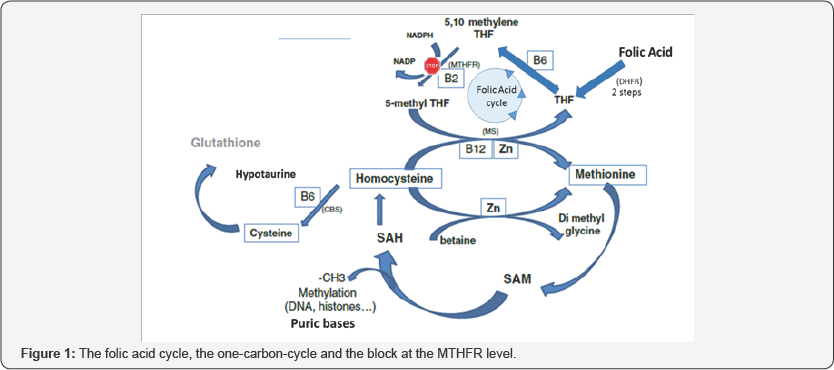
Our data on a small cohort of patients confirm some recent observations in the scientific literature [4] First, MTHFR isoform has a real negative impact on oocytes and subsequently on early embryonic development, directly driven by the maternal stores/reserves accumulated during oocyte growth. But what is now clear is the negative impact on the male gamete, confirming some of our recent data [5]. Of great interest is the positive effect of supplementation with Methylfolate. This observation is important since folate-receptor-1 and folate- transporter-member-1 are two of the most expressed mRNAs in the oocyte [1]. A functional Methylfolate supply is necessary for the oocyte DNA repair capacity [6]: this is why chromosomal anomalies are observed at a high rate in the embryos of those patients [4]. Methylfolate is also i effective in the prevention of neural tube defects (NTDs, 7). High doses of folic acid (FA) have no positive impact in those patients carrying the MTHFR isoforms: Folic acid does not really enter the 1-CC as it cannot be transformed to 5-MTHF (Figure 1). Moreover excess of FA can negatively compete at the level of absorption and binding on the receptor and transporter, with the natural Methylfolate ingested found in food; therefore it may amplify the negative impact [7]. Excess of FA will lead to the UMFA (Unmetabolised folic acid) syndrome. This syndrome is still a matter of debate [8] in term of health. Although there is no evidence that an excess of UMFA would directly cause any oncological problems, there is a significant risk in accelerating growth and metastases of already existing tumors. In the mouse high doses of folic acid alter lipid metabolism in the liver [9].
The negative impact of MTHFR C677T isoform on sperm quality is confirmed [5]. The paternal effect on embryo development should not be overlooked as trophoblast development and differentiation are strongly under paternal control and request a high methylation activity. The paternal effect appears later in the embryo development than the maternal effect.
Oocyte donation is not an inevitable strategy for patients with recurrent miscarriages and/or ART failures. Oocyte donation has become increasingly popular worldwide; it has an obvious commercial gain for both the ART clinics who offer the technique, and young donors. It is too often considered as "the easy way out”. Incorrect testing of donors is poor medical practice, but as observed here, owing to the stringent requirements of the embryo for methyl donors, the recipients should also be tested. In particular, we should not overlook testing the male partner Finally, we would like to raise the question of the potential cancer risks for the young donors [10]. We are not suggesting that oocyte donation should not be practiced, but only to use this technique in ART when it is absolutely necessary, and where there are mutual benefits for the recipients and the donors.
References
- Ménézo Y, Lichtblau I, Elder K (2013) New insights into human preimplantation metabolism in vivo and in vitro. Assist Reprod Genet 30(3): 293-303.
- Richardson B (2003) Impact of aging on DNA methylation. Ageing Res Rev 2(3): 245-261.
- D'Elia PQ, dos Santos AA, Bianco B, Barbosa CP, Christofolini DM, et al. (2014) MTHFR polymorphisms C677T and A1298C and associations with IVF outcomes in Brazilian women. Reprod Biomed Online 28(6): 733-738.
- Enciso M, Sarasa J, Xanthopoulou L, Bristow S, Bowles M, et al. (2016) Polymorphisms in the MTHFR gene influence embryo viability and the incidence of aneuploidy. Hum Genet 135(5): 555-568.
- Cornet D, Cohen M, Clement A, Amar E, Fournols L, et al. (2017) Association between the MTHFR-C677T isoform and structure of sperm DNA. J Assist Reprod Genet 34.
- Ménézo Y, Dale B, Cohen M (2010) DNA damage and repair in human oocytes and embryos: a review. Zygote 18(4): 357-365.
- Obeid R, Holzgreve W, Pietrzik K (2013) Is 5-methyltetrahydrofolate an alternative to folic acid for the prevention of neural tube defects? J Perinat Med 41(5): 469-483.
- Ulrich CM, Potter JD (2006) Folate supplementation: too much of a good thing? Cancer Epidemiol Biomarkers Prev 15(2): 189-193.
- Christensen KE, Mikael LG, Leung KY, Levesque N, Deng L, et al. (2015) High folic acid consumption leads to pseudo-MTHFR deficiency, altered lipid metabolism, and liver injury in mice. Am J Clin Nutr 101(3): 646658.
- Schneider J, Lahl J, Kramer W (2017) Long term breasr cancer risk following ovarian stimulation in young egg donors: a call for follow up, research and informed consent Reprod. Bio Med Online 34(5): 480485.






























