Ectopic Pregnancy following in vitro Fertilization: A Case Report
Abayomi B Ajayi1, Victor D Ajayi, Ifeoluwa Oyetunji1, Oluwafunmilola Biobaku1 and Bamgboye M Afolabi2*
1Nordica Fertility and Endoscopy Centre, Nigeria
2Health, Environment and Development Foundation, Nigeria
Submission: June 12, 2017 ; Published: July 21, 2017
*Corresponding author: Bamgboye M Afolabi, Health, Environment and Development Foundation, 34 Montgomery Road, Yaba, Lagos Nigeria, Email: bmafolabi@gmail.com
How to cite this article: Ajayi AB, Ajayi VD, Oyetunji O, Biobaku O and Afolabi BM. Ectopic Pregnancy following in vitro Fertilization: A Case Report. J Gynecol Women's Health 2017; 6(1): 555679. DOI: 10.19080/JGWH.2017.06.555679.
Abstract
We present an uncommon case of ectopic pregnancy following In-vitro fertilization in a 32 year old infertile Banker who, prior to IVF treatment, was diagnosed with stage 4 endometriosis by laparoscopy. She presented after 4 years of being unable to conceive with associated severely painful menstrual periods and deep dyspareunia. She had In-vitro fertilization and embryo transfer of two Day 3 embryos but this resulted in a left tubal ectopic pregnancy. This was treated surgically and she had a successful repeat IVF treatment which resulted in a viable on-going singleton intrauterine pregnancy.
Keywords: Ectopic pregnancy; IVF; Endometriosis
Case Report
Mrs K.A was a 32 year old banker who had never conceived after 4 years of trying. She presented also with a history of severely painful menstrual periods, painful intercourse with deep penetration. She however had regular periods with moderate flow. Several ultrasound scans done previously showed recurrent cysts and HSG revealed bilateral tubal blockage. In 2014 she had surgery to remove gall stones and she had cyst drainage in 2015. She was married to a 34 year old banker who had no complaints.
Clinical evaluation revealed a stable patient with a healed midline supraumbilical surgical scar. A transvaginal ultrasound scan revealed a homogenous ovarian mass that measured 55.7 x 58.5mm. The right ovary and adnexae were normal.
She had a laparoscopy done and the findings were those of a normal sized uterus tethered to the POD and pelvic side walls by extensive endometriotic adhesions involving the sigmoid colon, appendix, rectum, both adnexae and anterior right iliac fossa. The right ovary and tube were matted anteriorly to round ligament and right half of utero-vesical pouch with appendix adherent to right uterine cervix.
A 5cm left endometrioma was buried within a tubo-ovarian mass in the left ovarian fossa, involving the sigmoid colon and the left utero-sacral ligament. Thicked uterosacral ligaments with rectum hitched up slightly. Eighty percent (80%) of the Pouch of Douglas was obliterated and swollen with tortuous tubes bilaterally, both with normal fimbriae. Careful adhesiolysis and left ovarian cystectomy was done. She also had hysteroscopy which was essentially normal but there was evidence of adenomyosis.
The clinical diagnosis at laparoscopy was Severe Endometriosis Grade 4. Specimen of the cyst wall was taken for histology and this returned with a diagnosis of endometriosis..
She was to have GnRH agonist for 3 months before proceeding to IVF.
The couple's evaluation for IVF revealed normal FSH level of 3.82 (1-10miu/ml) and a sperm count of 45x106 spermatozoa per ml, 70% progressive motility with mean progressive motility graded as GOOD (rapidly forward, fast progressive movement) and 50% normal forms.
She had a dummy transfer prior to commencement of her IVF cycle and endometrial catheterization was easy requiring canula only with an endometrial cavity length of 8.0cm.
She commenced the antagonist protocol in July 2016 with a starting dose of 375IU FSH which was increased to 450IU on day 6 of COH on account of poor response having only 2 follicles seen on scan. Antagonist was added on day 7. The 2 follicles which were slow in growth were triggered with 10,000IU of HCG on Day 15 of stimulation and 34 hours later 2 metaphase II eggs were retrieved and fertilized with her spouse's sperms. Two Day 3 embryos were smoothly transferred under abdominal ultrasound guidance into her uterine cavity dropping the embryos 2cm away from the fundus. A pregnancy test done two weeks later was positive. She had her first pregnancy scan a further two weeks later but the uterine cavity was empty with an endometrial thickness of 8.3mm. No intrauterine or extrauterine gestational sac was seen at this time and the right adnexae contained a mixed echo irregular cyst suspected to be an endometrioma while the left adnexae appeared normal. The vaginal ultrasound scan probe cover was stained with dark brown discharge. She had reported scanty vaginal bleeding spotting without any pains which lasted a few days and stopped.
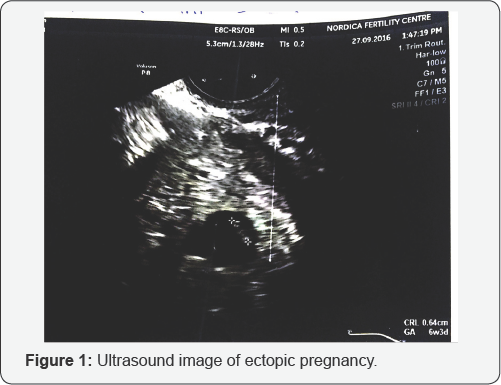
Serial quantitative BHCG was ordered. The first result was 9709Miu/ml and a repeat 48 hours later was 12,102.Miu/ml. a subsequent (3HCG was 15,549.0Miu/ml. The figures were increasing but not doubling. Spotting par vaginam persisted on and off and a repeat scan showed a left adnexial mass consistent with a gestational sac but with non active fetal pole seen. fetal pole with a CRL of 0.64cm, equivalent to 6 weeks and 3 days gestation (Figure 1). A diagnosis of suspected ectopic gestation was made and she was referred for an emergency surgery at the Lagos University Teaching Hospital where history, laboratory tests (positive serum pregnancy test) and ultrasound findings (left adnexal gestational sac with fetal pole, minimal fluid in pouch of Douglas and empty uterus) further confirmed an ectopic pregnancy.
The findings at exploratory laparotomy, having abandoned laparoscopy because of excessive pelvic adhesions, were as follows:
a. Haemoperitoneum 200mls
b. Left adnexal mass 6cm x 6cm
c. Moderate adhesion between uterus and left adnexal
d. Left fallopian tube not visualized
e. Grossly normal (Rt) tube and ovary
The left adnexa mass was excised and sent for histology. Histology report of surgical specimen confirmed ectopic gestation. She did well post-opera tively and was discharged home.
She was reviewed and opted to have another IVF cycle. An endometrial scratching was recommended and this was done on Day 21 of the cycle just before the repeat IVF treatment cycle.
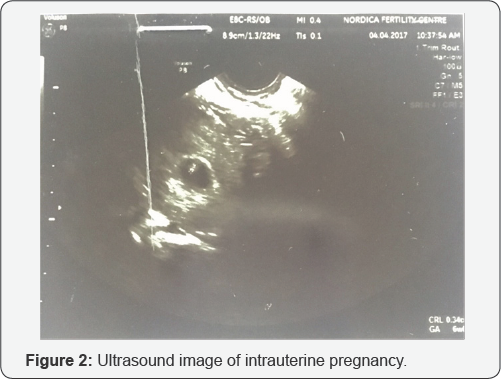
She had a similar antagonist protocol but this time the starting dose of FSH was 450IU. She produced 6 follicles from which 6 eggs were retrieved however only 3 were metaphase II eggs. Two of this fertilized following ICSI and two Day 3 embryos were smoothly transferred under abdominal ultrasound guidance into her uterine cavity placing them about 2cm short of the uterine fundus. She had a pregnancy test after an uneventful 2 week wait which was positive. An ultrasound scan done 2 weeks after revealed a singleton intrauterine gestational sac with a viable fetus with CRL=0.34cm compatible with 6 weeks gestation (Figure 2). There was no identifiable extrauterine gestational sac or pregnancy.
She had commenced her antenatal care and pregnancy is ongoing with no problems.
Discussion
Ectopic pregnancy (EP) is an unwanted and potentially dangerous complication of pregnancy. It is a major event in a woman's reproductive life and may complicate infertility treatment [1]. EP can be fatal especially in developing countries where it has been reported to account for up to 10% [2] of maternal death and this may be due to late presentation, detection and intervention. Ruptured tubal ectopic pregnancy is the leading cause of maternal mortality in the first trimester [3]. Case fatalities of 27.9 per 1000 and 30 per 1000 have been reported in Accra Ghana and Lagos Nigeria [4]. A Nigerian study reported an incidence of 1.5%, majority in the ampulla region of the fallopian tube (59.7%) and already ruptured at presentation in 70% of the cases [5]. Ectopic pregnancy can occur after IVF, though this is uncommon. The incidence of ectopic pregnancy with Assisted Reproductive Technology (ART) is in the range of 1.5-2.5% and majorities are tubal [6]. Some other reports estimate it be between 2.1-8.6% and up to 11% in women with a history of tubal factor [7].
Apart from the usual factors that have been generally linked to the occurrence of ectopic gestation, the risk factors for ectopic pregnancy following IVF include tubal infertility, previous ectopic pregnancy, previous tubal surgery, PID, endometriosis and its treatment, technique of embryo transfer [8-11]. In the case reported in this article, the patient had stage 4 endometriosis with tubal affectation. This could have increased her risk for ectopic pregnancy following embryo transfer
The exact mechanism responsible for an ectopic pregnancy following IVF is unknown. One of the mechanisms suspected is the migration of the transferred embryos up into the fallopian tubes due to local cellular activity or on account of fluid mechanics present inside the uterus. A dilated opening of the uterine end of the fallopian tube due to disease may make it easier for transferred embryos to enter the tubes and cause ectopic gestation [2]. Factors like altered normal mileu of the uterus due to controlled ovarian stimulation, day and stage of embryo transfer, multiple embryo transfer, volume of media used, and position of tip of catheter have been implicated in the causation of ectopic pregnancy. Ovarian stimulation has been thought to alter the endocrine mileu and decrease the endometrial receptivity. Failure of normal biological interaction between endometrium, fallopian tube (impaired tubal function) and the embryo can lead to ectopic gestation [8,11,12]. Like in this patient tubal issues as well as Controlled Ovarian Hyperstimulation (COH) for IVF could have synergistically acted as a double whammy to increase her chances of ectopic gestation [11, 12]. Endometrial and subendometrial blood flow on day of HCG trigger is significantly lower in IVF patients with miscarriage and ectopic pregnancy than those with viable intrauterine pregnancy [13].
High estradiol levels cause reverse migratory process of embryos [14] explain. Ectopic pregnancy has also been observed more in fresh than in frozen embryo transfer cycles [15-19]. This patient had a fresh transfer.
Some authors have speculated that assisted hatching increases the risk of ectopic pregnancy [8]. However randomized controlled trials do not support this [20]. The case reported did not have assisted hatching.
Blastocyst transfer decreases the risk of ectopic pregnancy as, according to some researchers, it is believed that by day 5 there is less uterine contractility [21,22]. Other studies did not find such difference between Day 3 and Day 5 transfers [22-24].
The quality of the embryos transferred has also been linked to ectopic pregnancy with poor egg and embryo quality more likely to lead to ectopic pregnancy [25]. IVF culture media leading to abnormal expression of adhesion molecules has also been linked to ectopic pregnancy following IVF [26,27].
The position of the tip of the embryo transfer catheter and distance from fundal endometrium could also influence the occurrence of ectopic pregnancy. Randomised controlled trials showed pregnancy and ectopic pregnancy rates of 12.4% and 1.5% and 14.2% and 0.4% for deep fundal transfer and mid fundal transfer respectively [3].
In this case report, the embryo transfer was done under ultrasound guidance, placing the embryos 2 cm away from the fundus as is our standard protocol. Ultrasound guided transfer, where embryos are carefully placed inside the uterine cavity, 1.52 cm away from the fundus and cornua, is said to reduce the risk of ectopic pregnancy. However, it may not completely remove the risk of ectopic pregnancy as seen in this case report [28].
Due to extensive adhesions on account of severe endometriosis laparoscopic approach could not be used for treatment in this case so a laparotomy had to be carried out.
'Migration' of embryos into the fallopian tubes from the cavity before implantation can lead to ectopic pregnancy. Mrs K.A had endometrial scratching before the repeat cycle. Could the possible improved chances of implantation following endometrial scratching have reduced the chances of this 'migration' resulting in a viable intrauterine pregnancy in this woman who remains at risk of another ectopic pregnancy? Clearly this single case report cannot claim this but should lead to a discussion and research on the possibility in the future.
Early identification and diagnosis is key to its management. Early transvaginal ultrasound scan and serial quantitative serum B HCG are the most reliable tools [3] and important steps in this direction and these were done in the reported case aiding early diagnosis and management. Treatment can be conservative using medical means like methotrexate or by laparoscopy Where there is heterotropic pregnancy or hydrosalpinx with EP salpingectomy is advised. In this case because of extensive adhesions attempts at laparoscopy had to be abandoned and a laparotomy and salpingectomy done. Conservative surgery was not possible in this case. Treatment should be tailored to clinical condition [3].
There is no apparent negative impact of the different approaches to treatment on subsequent IVF-ET except risk of recurrence [3]. It is good for the patient and her partner who were both devastated by the occurrence of the ectopic pregnancy and also good for the practitioners too to know that there is still a chance of a successful intrauterine IVF pregnancy after a prior ectopic pregnancy resulting from IVF.
Conclusion
Though the occurrence of ectopic pregnancy following IVF is uncommon in our practice, this case is reported to draw attention to its possible occurrence following IVF and ultrasound guided embryo transfer. Its possibility has to be considered by practitioners in women at risk undergoing IVF such as in this case of severe endometriosis with tubal affectation. Subsequent successful intrauterine pregnancy following repeat IVF is also still possible as seen in the reported case.
References
- Fernandez H, Gervaise A (2004) Ectopic pregnancies after infertility treatment: modern diagnosis and therapeutic strategy. Human Reproduction Update 10(6): 503-513.
- Khan KS, Wojdyla D, Say L, Gulmezoglu AM, Van Look PF (2006) WHO analysis of causes of maternal death: a systematic review. Lancet 367(9516): 1066-1074.
- Refaat B, Dalton E, Ledger WL (2015) Ectopic pregnancy Secondary to In Vitro Fertilization-Embryo Trnasfer: Pathogenic Mechanisms and Management Strategies. Reprod Biol Endocrinol 13: 30.
- Igwegbe A, Eleje G, Okpala B (2013) An appraisal of the management of ectopic pregnancy in a Nigerian tertiary hospital. Ann Med Health Sci Res 3(2): 166-170.
- Panti A, Ikechukwu NE, lukman OO, Yakubu A, Egondu SC, et al. (2012) Ectopic pregnancy at Usmanu Danfodiyo University Teaching Hospital Sokoto: A ten year review. Ann Nigerian Med 6(2): 87-91.
- Yoder N, Tal R, Martin JR (2016) Abdominal Ectopic Pregnancy After In Vitro Fertilization and Single Embryo Transfer: A Case report and Systematic Review. Reprod Biol Endocrinol 14(1): 69.
- Clayton HB, Schieve LA, Peterson HB, Jamieson DJ, Reynolds MA, et al. (2006) Ectopic pregnancy risk with assisted reproductive technology procedures. Obstet Gynecol 107(3): 595-604.
- Chang HJ, Suh CS (2010) Ectopic pregnancy after assisted reproductive technology: what are the risk factors? Curr Opin Obstet Gynecol 22: 202-207.
- Li C, Meng CX, Zhao WH, Lu HQ, Shi W, et al. (2014) Risk factors for ectopic pregnancy in women with planned pregnancy: a case-control study. Eur J Obstet Gynecol Reprod Biol 181: 176-182.
- Hjordt Hansen MV, Dalsgaard T, Hartwell D, Skovlund CW, Lidegaard O (2014) Reproductive prognosis in endometriosis. A national cohort study. Acta Obstet Gynecol Scand 93(5): 483-489.
- Shao R, Nutu M, Weijdegard B, Egecioglu E, Fernandez-Rodriguez J, et al. (2009) Clomiphene citrate causes aberrant tubal apoptosis and estrogen receptor activation in rat fallopian tube: implications for tubal ectopic pregnancy. Biol Reprod 80(6): 1262-1271.
- Jia-Rong Z, Shuang-Di L, Xiao-Ping W (2009) Eutopic or ectopic pregnancy: a competition between signals derived from the endometrium and the fallopian tube for blastocyst implantation. Placenta 30(10): 835-839.
- Wang L, Qiao J, Li R, Zhen X, Liu Z (2010) Role of endometrial blood flow assessment with color Doppler energy in predicting pregnancy outcome of IVF-ET cycles. Reprod Biol Endocrinol 8: 122.
- Shao R, Feng Y, Zou S, Weijdegard B, Wu G, et al. (2012) The role of estrogen in the pathophysiology of tubal ectopic pregnancy. Am J Transl Res 4: 269-278.
- Wang J, Wei Y, Diao F, Cui Y, Mao Y, Wang W, et al. (2013) The association between polycystic ovary syndrome and ectopic pregnancy after in vitro fertilization and embryo transfer. Am J Obstet Gynecol 209(2): 139.
- Zhang YL, Sun J, Su YC, Guo YH, Sun YP (2012) Study on the incidence and influences on ectopic pregnancy from embryo transfer of fresh cycles and frozen-thawed cycles. Zhonghua Fu Chan Ke Za Zhi 47(9): 655-658.
- Shen C, Shu D, Zhao X, Gao Y (2014) Comparison of clinical outcomes between fresh embryo transfers and frozen-thawed embryo transfers. Iran J Reprod Med 12(6): 409-414.
- Ishihara O, Kuwahara A, Saitoh H (2011) Frozen-thawed blastocyst transfer reduces ectopic pregnancy risk: an analysis of single embryo transfer cycles in Japan. Fertil Steril 95(6): 1966-1969.
- Huang B, Hu D, Qian K, Ai J, Li Y, et al. (2014) Is frozen embryo transfer cycle associated with a significantly lower incidence of ectopic pregnancy? An analysis of more than 30,000 cycles. Fertil Steril 102(5): 1345-1349.
- Hagemann AR, Lanzendorf SE, Jungheim ES, Chang AS, Ratts VS, et al. (2010) A prospective, randomized, double-blinded study of assisted hatching in women younger than 38 years undergoing in vitro fertilization. Fertil Steril 93(2): 586-591.
- Li RR, Dong YZ, Guo YH, Sun YP, Su YC, et al. (2013) Comparative study of pregnancy outcomes between day 3 embryo transfer and day 5 blastocyst transfer in patients with progesterone elevation. J Int Med Res 41(4): 1318-1325.
- Milki AA, Jun SH (2003) Ectopic pregnancy rates with day 3 versus day 5 embryo transfer: a retrospective analysis. BMC Pregnancy Childbirth 3(1): 7.
- Wang SS, Sun HX (2014) Blastocyst transfer ameliorates live birth rate compared with cleavage-stage embryos transfer in fresh in vitro fertilization or intracytoplasmic sperm injection cycles: reviews and meta-analysis. Yonsei Med J 55(3): 815-825.
- Smith LP, Oskowitz SP, Dodge LE, Hacker MR (2013) Risk of ectopic pregnancy following day-5 embryo transfer compared with day-3 transfer. Reprod Biomed Online 27(4): 407-413.
- Job-Spira N, Coste J, Bouyer J, Tharaux-Deneux C, Fernandez H (1996) Chromosomal abnormalities and ectopic pregnancy? New directions for aetiological research. Hum Reprod 11(2): 239-242.
- Wang LL, Chen X, Ye DS, Liu YD, He YX, et al. (2014) Misdiagnosis and delayed diagnosis for ectopic and heterotopic pregnancies after in vitro fertilization and embryo transfer. J Huazhong Univ Sci Technolog Med Sci 34(1): 103-107.
- Coroleu B, Barri PN, Carreras O, Martinez F, Parriego M, et al. (2002) The influence of the depth of embryo replacement into the uterine cavity on implantation rates after IVF: a controlled, ultrasound-guided study. Hum Reprod 17(2): 341-346.






























