Determination of Micronutrient Concentrations and Physico Chemical Characterization of Alcohol Industries Effluents
Tesfalem Belay Woldeamanuale*
Department of Environmental pollution, Ethiopia Environment and Forest Research Institute, Ethiopia
Submission: November 20, 2017; Published: November 30, 2017
*Corresponding author: Tesfalem Belay Woldeamanuale, Department of Environmental pollution, Ethiopia Environment and Forest Research Institute, Central Ethiopia Environment and Forest Research Center, Ethiopia, Email: tesbel23@gmail.com
How to cite this article: Tesfalem B W. Determination of Micronutrient Concentrations and Physico Chemical Characterization of Alcohol Industries Effluents. J Forensic Sci & Criminal Invest. 2017; 6(3): 555691. DOI: 10.19080/JFSCI.2017.06.555691
Abstract
Physico-chemical parameters and Concentration of some Micronutrients were determined in waste water samples collected from four selected industries in Addis Ababa, Ethiopia using standard analytical procedure. Sampling was carried out August, 2017. The results of the physico-chemical analysis were obtained in the following range; pH (4.98-8.7), temperature (27.5-34.20C), Conductivity (1264.57-1787.14 μs/ cm), chloride (91.73-919.04mg/l,) sulphate (25-3700mg/l), TDS (2180.2-2951.mg/l),alkalinity(212.5-537.5 mg/L), SS (1006.2-1189.21mg/ l),B0D(2146.02-3256.3mg/ l) NO3-N (437.5-1000.23 mg/l), and sulphied (10.63-4900mg/l).The concentration of some heavy metals (mg/l) in the samples analyzed were found to be in the following range; Cd (BDL), Cr (0.39- 0.53), Pb (0.23-0.53). The results of Cr and Pb were above the permissible limits set by Federal Environmental Protection Agency (FEPA) and World Health Organization (WHO) most especially, effluent from Ethanol and textile Industries but Cd was below detection limit in all samples. It can be concluded that all the effluents required further treatment before releasing them into water body or land in other to prevent pollution. Proper monitoring of industrial effluent by regulatory bodies and adequate measures should be put in place in other to curb indiscriminate release of industrial effluents to the environment.
Keywords: Waste-Water; Pollution; Heavy Metals; Physiochemical Analysis
Abbreviations: FEPA: Federal Environmental Protection Agency; WHO: World Health Organization; Pb: Lead; Cd: Cadmium; Cr: Chromium; TS: Total Solids; COD: Chemical Oxygen Demand; EC: Electrical Conductivity; TSS: Total Suspended Solids; TDS: Total Dissolved Solids; BOD: biological oxygen demand; US-EPA: United States- Environmental Pollution Agency
Introduction
Industrialization plays a vital role in growth and development of any country but, it has direct and indirect adverse effect on our environment. The continuous increase in industries has become sources of pollution. These industries include battery manufacturing, iron and steel, plastics, chemicals, fertilizer, textile, food and beverages, breweries, pharmaceuticals, soap, petroleum and petrochemical, automobile, tannery, paper mill and cosmetics, tobacco and paint industries [1]. The up surge in urban population and the establishment of industries involved in the manufacture of various agrochemicals. Industrial Groundwater is threatened with pollution from sources like; domestic wastes, industrial wastes, runoff from urban areas, suspended and dissolved soils, organics and pathogens. Wastewater treatment is not given the necessary priority it deserves and therefore, industrial waste and domestic sewage are discharged into receiving water bodies without treatment. The consequence of this is increased river pollution, loss of aquatic life and uptake of polluted water by plants and animals, which eventually gets into human body resulting in health related problems.
In the third world, many surface waters are known locally to be very contaminated but these cases are rarely documented, due to lack of basic tools such as established monitoring network as well as logistical and analytical capacities [2]. The quality of drinking water in Ethiopia cities has been deteriorated in the recent years mainly due to growth of population and improper disposal of waste water from industries [3]. Approximately 1.2 billion people in developing nations lack clean water because most household and industrial wastes are dumped into rivers and lakes without treatment contributing to many waterborne diseases in humans [4]. High levels of pollutants mainly organic matter in river water because an increase in biological oxygen demand, chemical oxygen demand, and total dissolved solids and total suspended solids. They make water unsuitable for drinking, irrigation [5] or any other use. Mismanagement of these wastes which are dumped in soils and unsanitary landfills are subjected to weathering and leaching processes by rain and other atmospheric influences resulting in the release of hazardous substances such as cyanides, minerals, heavy metals and organic acids which get to underground water systems and inland water bodies untreated.
Their effects render underground and surface waters unsafe for human, recreational and agricultural use. Biotic life is destroyed and natural ecosystems are poisoned. Human life is threatened and the principle of sustainable development is compromised [6]. Some of the heavy metals commonly found in industrial waste water includes: lead (Pb), cadmium (Cd), Chromium (Cr) etc. The pH of the wastewater may affect the quality of the water by increasing the toxicity of the metal as the pH decreases. Industrial effluents not only contain toxic materials but also have other nutrients that enhance the growth of crop plants. Therefore, it is essential that the implications of the use of industrial effluents in the crop field and their effect on soil characteristics should be assessed before they are recommended for use in irrigation. The aim of this study was to determine concentration of micronutrients and physicochemical characterization of effluents discharge from four industries which are; Three Ethanol industries (A, B, C) and textile industry (D). These levels were compared with the maximum permissible levels stipulated by FEPA [7] and WHO [8].
Materials and Methods
Sample collection
Samples were collected from four different industries in duplicate. Sampling was carried out using well cleaned amber bottles (2L) that are airtight from the effluent reservoir tank of each company. Sampling bottles were pre-cleaned by soaking them in detergent for 24 hours, followed by rinsing with tap water and then rinsed with 5% nitric acid and lastly with distilled water [9] the water samples were preserved with 1ml conc. HNO3 and stored at 4oC until use. Samples were kept in a portable cooler containing ice, to maintain an inert temperature condition for the effluent and were transported to the laboratory for analysis. The physico-chemical analysis done includes: The pH, conductivity, alkalinity and total solids. All field meters and equipment were checked and calibrated according to the manufacturer's specifications. The pH meter was calibrated using [10] buffers of pH 4.0, 7.0 and 10.0. The pH was measured using pH meter, a conductivity meter measured the conductivity and the alkalinity was determined by titration method while the total dissolved solids were determined using gravimetric analysis.
Physical and Nutrient load analysis
The following physical parameters were assessed using standard methods for examination of water and wastewater [11] pH, total solids (TS), chemical oxygen demand (COD) and biological oxygen demand. Nutrient Load was assessed using the following parameters: sulphate, nitrate and total nitrogen. Water samples were filtered using what man filter paper No 4 before analysis in order to removes suspended particles. All chemicals used were of Analytical grade. Each sample was digested using a mixture of 10ml each of 40% HF and 70% HClO4 in a fume chamber. The resulting clear colorless solution was allowed to cool down, filtered and made to 100ml with deionized water and finally stored in a polythene container in readiness for instrumental analysis. The heavy metal analysis was also done using the Atomic Absorption Spectrophotometer.
Results and Discussion
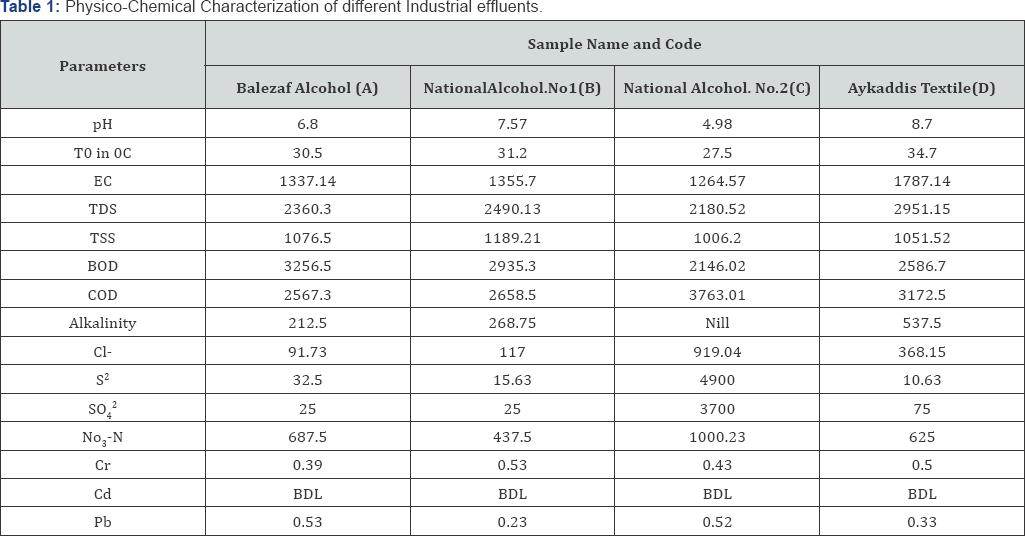
Industrial effluents are the main source of surface and ground water pollution. To evaluate the pollution content of industrial effluent, four samples in duplicate the four industries were analyzed for various physical and chemical parameters such as temperature, pH, electrical conductivity (EC), total suspended solids (TSS), total dissolved solids (TDS), biological oxygen demand (BOD) and Concentration of Micronutrients. The results were compared with the standard values of Federal Environmental Protection Agency (FEPA) for industrial effluents, United States- Environmental Pollution Agency (US-EPA) and World Health Organization (WHO) for drinking water given in:
Temperature
Temperature is basically important for its effect on other properties of wastewater. Temperature values for various samples are presented in Table 1, ranging from 27.5-34.20C. The highest value was found in the sample D followed by, B and A, while sample C has the lowest temperature. The temperature values of all the samples analyzed were within the permissible limits of FEPA and WHO. Release of high temperature waste water into water bodies may speed up some reactions in the water body. It will also reduce solubility of oxygen and amplified odor due to anaerobic reaction (less oxygen).
Determination of pH
The pH of the samples ranged from 4.98-8.7 so presented in Table 1. Sample C has the lowest pH (4.98), while sample D has the highest pH value of 8.7. All the pH values were within the permissible limits for industrial effluents set by FEPA except sample C. The pH of samples A, B and D are within the permissible standard for drinking water set by USEPA and W.H.O while the pH of sample C was found to be below the standard and it has acidic nature.
Electrical Conductivity EC
Conductivity of waste water which is a useful indicator of its salinity or total salt electrical conductivity is a use full indicator of mineralization and salinity or total salt in a water sample. EC of the samples shown in (Table 1) ranged from 1264.57 to 1787.14 μs/cm. Sample D has the highest EC of 1787.14μs/cm,while sample C has the lowest EC of 1264.57 μs/cm. The mean guideline values of 1000 μ s/cm. All samples of waste water are conductivity values for all the samples were above the WHO not discharge through channel into stream.
*All parameters are in mg/l except PH*;
BLD: Blow detection limit Total Suspended Solids TSS
The TSS values (Table 1) of the samples ranged from 1006.2 to 1189.21 mg L-1. FEPA standard for TSS is 30mg L-1. So it is clear that all the samples have very high TSS values which may be due to the fact that the waste contains many suspended particles. The maximum value of TSS (1189.21 mg L-1) was recorded in the effluent of sample B. Effluents of such high TSS may cause handling problem, if directly applied to agricultural field, or if this effluent is discharged to river or stream, it will make it unsuitable for aquatic life. Literature classified wastewater TSS as follows: TSS < 100mg/l as weak, TSS > 100mg/las but < 220mg/l as medium and TSS > 220mg/l as strong wastewater. Therefore, waste water from all samples are strong and cannot be discharged into stream.
Total Dissolved Solids TDS
The TDS values of the samples ranged from 2180.52 -2951.15 mg L-1 (Table 1). The lowest values were found in sample C while the highest value was found in sample D. The FEPA standard for TDS is 2000 mg L-1. All the samples have values above the permissible limit. High TDS is caused by high solids loading.
Biological and Chemical Oxygen Demand (BOD &COD)
An indication of organic oxygen demand content of wastewater can be obtained by measuring the amount of oxygen required for its stabilization either as BOD and COD. Biological Oxygen demand (BOD) is the measure of the oxygen required by microorganisms whilst breaking down organic matter. While Chemical Oxygen Demand (COD) is the measure of amount of oxygen required by both potassium dichromate and concentrated sulphuric acid to breakdown both organic and inorganic matters. The BOD values ranged from 2146.23256.5 mg L-1as presented in (Table 1). The COD values ranged from 2567.3-3763.01mg/L. Sample A has the highest BOD and lowest COD while sample C has the lowest BOD and highest COD. These effluents on entering fresh water (rivers, stream etc.) make the O2 depleted, causing suffocation of fish and other aquatic fauna and flora resulting in the death of aquatic life. The concentrations of BOD and COD in all the samples were higher than the WHO and FEPA values of 50mg/l and 1000mg/l for the discharged of waste water into stream. High COD and BOD concentration observed in the wastewater might be due to the use of chemicals, which are organic or inorganic that are oxygen demand in nature.
Anionic Analysis
The chloride level of the samples measured fall within this range 91.73- 919.04mg/L. Chloride levels in all samples were found to be below the permissible level (600mg/L) except sample C. The concentrations of nitrogen in nitrate, alkalinity, sulphate and sulphied in all the samples analyzed varied between 437.5-1000.25mg/l for nitrogen in Nitrate; 221.5 to 537.5mg/l for alkalinity; 25- 3700mg/l for sulphate and 10.63 to 4900mg/l for sulphied respectively (Table 1). Supplied levels in all samples were found to be above FEPA permissible level. Hydrogen sulphied is formed under conditions of deficient oxygen in the presence of organic materials and sulphate [12]. This could be a possible reason for the high sulphied measured in the effluents analyzed. The levels of nitrogen in nitrate above the WHO limits of 45mg/l for nitrate in waste water, the levels of nitrogen may give rise to methaemoglobinemia, also the levels of nitrogen reported in all sample can cause eutrophication and may pose a problem for other uses.
Heavy Metals
Investigated metals in the industrial effluents of various industries are presented in (Table 1). The composition of metals in the waste water samples ranged from 0.39 to 0.53mg/l for Cr, 0.23 to 0.53mg/l for Pb, Blow detection limit for Cd. the concentrations of metals in the samples analyzed are in the following order Cr > Pb > Cd. Highest amount of all the heavy metals analyzed were found in sample C while, the lowest amount heavy metals were found in sample B. The concentration of heavy metals (Pb, and Cr) in all the samples analyzed was above permissible level recommended by FEPA and WHO but The concentration of heavy metals (Cd) in all the samples analyzed were below detection limit.
Conclusion
Based on the result of these analysis the order of the level of contaminants in the samples analyzed collected from the four chemical producing industries may be assumed to follow this sequence C > D > A > B. The concentration of contaminants in all the effluents studied ware mostly above the set limits by the Federal Environmental Protection Agency (FEPA) and World Health Organization (WHO). Though, the effluents were found to be rich in plant nutrients required for plant growth but due to the presence of high levels of toxic heavy metals (Pb and Cr), these waste water may not be good for irrigation in other to avoid accumulation of these metals in soils, and if the effluent are released into the environment without proper treatment, it may affect underground water and aquatic life if released into water bodies.
References
- Rahman A, Lee HK, Khan MA (1997) Domestic water contamination in rapidly growing megacities of Asia: Case of Karachi, Pakistan. Environ Monitor Assessm 44(1-3): 339- 360.
- (1998) Federal Environmental Protection Agency FEPA. Guidelines and Standards for environmental Pollution control in Nigeria. Decree 58 of 1988, pp. 238.
- (1998) DWAF, Quality of Domestic Water Supplies. AssessmentGuide.1 (2nd edn) Department of Water Affairs and Forestry, Department of Health and Water Research Commission.
- Hari O, Nepal S, Aryo M, Singh N (1994) Combined effect of waste of distillery and sugar mill on seed germination, seeding growth and biomass of okra (Abelmoschusesculentus l0 Moench). J of Environ Bio 15(3): 171-175.
- APHA, AWWA and WEF 1998. Standard methods for the examination of water and wastewater, (20th edn) Clesceri LS, Greenberg AE, Eaton AD (Eds.), American Public Health Association, American Water Work Association, Water Environment Federation, Washington DC, USA.
- (1992) American Public Health Association (APH), Standard methods for the examination of water and wastewater. (18th edn), Greenberg AE, Clesceri LS and Eaton AD (Eds.), Apha, WEF and AWWA, Washington DC, USA, pp. 1134.
- Brown JS, Rattry EAS, Paton GI, Reid G, Caffoor I, et al. (1996) Comparative assessment of the toxicity of a paper mill effluent by respiratory and luminescence based bacteria assay. Chemosphere 32: 1553-1556.n
- (1997) HACH, Water Analysis Handbook, (3" Love land, Colorado, USA.
- Clement TP (2011) Bio-remediation of contaminated ground water systems, Ground water Quantity and Quality Management. American Society of Civil Engineers (ASCE), MM Aral, S Taylor (Eds.) pp. 522-559.
- Venkata subramani R, Meenambal T (2007) Study of sub-surface water quality in Mattupalayam Taluk of Coimbatore district Tamil Nadu. Nat Environ Poll Tech 6(2): 307-310.
- Branco AM, Suassuna J, Vainsencher SA (2005) Improving Access to water resources through rain water harvesting as a minigation measure: the case of the Brazilian semi-arid region. Mitigation and Adaptation Strategies for Global Change 10(3): 393-409.
- WHO. 2000, WHO Air Quality Guidelines, (2nd edn), World Health Organization, Europe Regional Office, Copenhagen, Denmark.






























