Covid Vaccine as a Trigger for Grave’s Hyperthyroidism: Cases and Literature Review
Sarah Ghandour1 Nisrine AlGhazal2
1Department of Endocrinology, Mediclinic Middle East, Mohamad Bin Rashed University of Medicine and Health Sciences, UAE
2Department of Endocrinology, Mediclinic Middle East, UAE
Submission: November 09, 2021; Published: January 20, 2022
*Corresponding author: Nisrine Al Ghazal, Department of Endocrinology, Mediclinic Middle East, UAE
How to cite this article: Sara El G, Nisrine Al G. Covid Vaccine as a Trigger for Grave’s Hyperthyroidism: Cases and Literature Review. J Endocrinol Thyroid Res. 2022; 6(4): 555693. DOI:10.19080/JETR.2022.06.555693
Abstract
Objective: Grave’s Disease is a systemic autoimmune disease directly caused by circulating auto-antibodies that bind to the thyrotropin receptor, subsequently inducing the production and release of thyroid hormone, proliferation of thyrocytes, and enlargement of the thyroid gland. Clinical practice has shown possible uncovering of Grave’s Disease following SARS-Cov-2 vaccine. The aim of this report is to shed the light on Grave’s Disease cases post SARS-Cov-2 vaccine, while reviewing the link between the vaccine and subsequent trigger of thyroid autoimmune hyperactivity.
Cases: We present the case of a middle-aged woman with Grave’s disease following Hashimoto hypothyroidism manifesting 1 month post vaccine.
A 36-year-old man presents with asymptomatic Grave’s disease 1 week following the vaccine.
A 33-year-old woman presents with full blown Grave’s disease 9 days following vaccination.
A 33-year-old man develops symptomatic Grave’s disease 3 weeks after the vaccine.
Review: Cases presented above reflect different presentations of Grave’s disease. Moreso, the literature harbors similar cases of Grave’s disease recurrence, initial manifestation and exacerbation following SARS-Cov-2 vaccine. Grave’s disease etiology is autoimmune but environmental, genetic, and immunological factors contribute to its onset. The vaccine can potentially trigger autoimmune hyper-reactivity through the hyper-inflammatory response to the virus itself but also through the adjuvant found in the vaccine (Shoenfeld’s Syndrome)
Conclusion: Cases of Grave’s disease manifestation or exacerbation post SARS-Cov-2 vaccines are reported. Theories are like SARS- Cov-2 -induced Grave’s Disease but also other mechanisms are uniquely mediated by vaccines. That vaccination benefit outweighs the potential risks but one should have a low threshold to test and follow patients at risk for auto-immune thyroid diseases after receiving SARS-CovV-2 vaccine.
Introduction
Grave’s Disease (GD) is a systemic autoimmune disease directly caused by circulating autoantibodies (Abs) that bind to the thyrotropin receptor (TSH-R), subsequently inducing the production and release of thyroid hormone, proliferation of thyrocytes, and enlargement of the thyroid gland [1]. Hence, the stimulatory TSH-R-Ab (TRAb) is the causative agent in GD. The prevalence of GD approximates 1% to 1.5% in the population as a whole: the incidence is 20 to 30 new cases/100 000/year. An increased incidence is observed among African Americans. Both genetic and environment factors, for example, familial clustering, negative life experiences, high iodine intake, and smoking, predispose to GD [1]. Vaccines in general have been shown to trigger immune responses that leads to a multiple autoimmune disease.
Recent clinical practice has shown a potential link between GD and Covid vaccine, but also a possible flare of GD related to the vaccine. There are currently four types of COVID-19 vaccines. These include mRNA vaccines (BNT162b2, Pfizer-BioNTech; mRNA-1273, Moderna), protein subunit vaccines (NVX-CoV2373, Novavax), vector vaccines (Ad26.COV2, Janssen Johnson & Johnson; ChAdOx1 nCoV-19/ AZD1222, Oxford- AstraZeneca), and whole virus vaccines (PiCoVacc, Sinovac19; BBIBP-CorV, Sinopharm) [2]. In an attempt to provide more insight on the topic, we hereby present four cases of GD occurring within 4 weeks of Covid vaccine. Cases vary in severity, presentation, and course. A literature review detailing reported cases, pathogenesis of GD and the effect of the vaccine on its development is presented as well.
Cases
Case 1
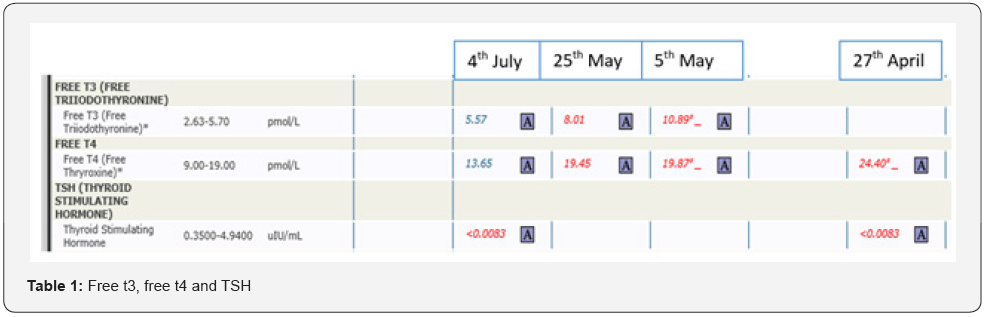
45 years old lady known to have hypothyroidism for 8 years on levothyroxine presented (27/4/2021) for symptoms of hyperthyroidism over the past few weeks. She received her Covid Pfizer vaccination 1 month prior to presentation. On exam, she was tachycardic (pulse rate of 108 BPM) and had significant hand tremor. Laboratory studies confirmed hyperthyroidism (Table 1), that persisted two weeks after stopping levothyroxine; Furthermore, TRAb was 3.46 IUI/L (<1.75 IUI/L). On 25th of May, she was started on anti-thyroid medications (carbimazole), subsequently, her symptoms improved and her free t3 and free t4 normalized.
Case 2
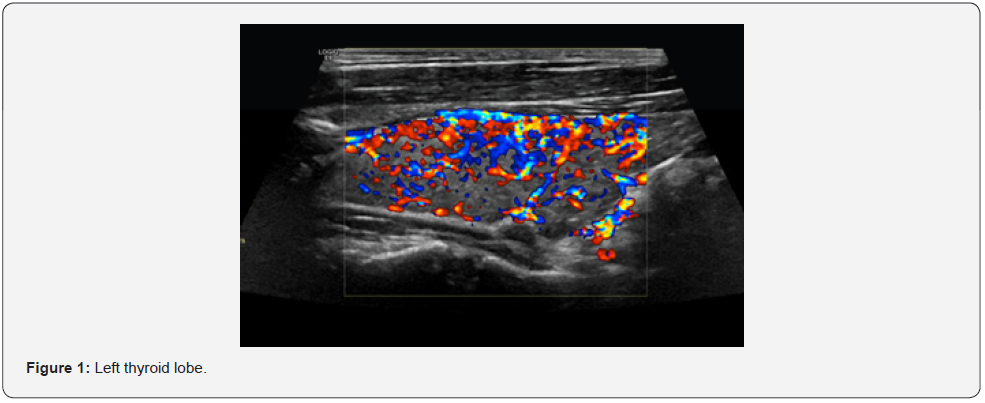
36-year-old man found to have abnormal thyroid function tests incidentally upon testing. He received Covid vaccine (Pfizer) 1 week prior to testing. On physical exam, he was clinically euthyroid and had an enlarged goiter. His laboratory investigations showed biochemical hyperthyroidism and elevated TRAb. Technetium scan was compatible with GD as there was an increased Global Tc-99m uptake (6.25%) and ultrasound showed a heterogeneous thyroid gland with increased vascularity, picture compatible with thyroiditis (Figure 1). Treatment options offered to the patient, and he was started on anti-thyroid medications.
Case 3
33-year-old woman was evaluated 9 days post first dose of Pfizer vaccine for palpitations and excessive sweating. She has no family history of thyroid diseases. She was found to have an undetectably low TSH, free T4 more than 3 times upper normal and free T3 more than 6 times upper normal. Liver function tests were elevated as well and TRAB was positive. Ultrasound showed hyper vascular heterogenous thyroid. Physical exam was significant for goiter, hyper-reflexia and tachycardia. She was started on Carbimazole and her thyroid function tests subsequently improved significantly 4 weeks later.
Case 4
33-year-old man was evaluated 3 weeks post first dose of Pfizer vaccine for palpitations, fatigue, insomnia, weight loss, blurred vision, and excessive sweating. He was found to have an undetectably low TSH, free T4 and free T3 between 1.5-2 times upper normal. Inflammatory markers were negative and TRAB was positive. Both his father and sister have Grave’s disease. Physical exam was significant for a small goiter and resting tremor. He was started on Carbimazole and his thyroid function tests normalized 2 months later.
Review
Cases presented above reflect different presentations of GD. Presentations occurred 1-4 weeks post vaccine and ranged from being asymptomatic to manifesting severe hyperthyroidism. No ocular involvement was seen in all cases on presentation. One case manifested as Hashi-toxicosis with GD manifesting post years of Hashimoto hypothyroidism.
Literature Review
Onset of GD in two subjects was reported a few days after the first dose of BNT162b2 [3]. Both received a dose of the BNT162b2 vaccine and reported symptoms 2–3 days later [3]. Also, no description of ocular symptoms was included in the study [3]. The study found the subjects’ presentation to fit the diagnostic criteria for autoimmune/ inflammatory syndrome induced by adjuvants (ASIA), also known as Shoenfeld’s Syndrome. Later, G Zettnig reported two further cases: a late recurrence after 17 years (5 weeks after the second dose of the vaccine) and an initial manifestation (2 weeks after the vaccine) [4]. The authors postulated that ASIA as well as the vaccine itself could be the trigger of auto-immune thyroid disease. Chutintorn Sriphrapradang reported worsening of Graves’ hyperthyroidism 4 days after a booster vaccine with a replication-deficient chimpanzee adenovirus-vectored vaccine ChAdOx1 nCoV-19 (AZD1222, Oxford-AstraZeneca) [5]. The booster is a strategy to cover the variants. It also improves humoral and cellular responses in mice. This study also postulated ASIA to be the reason behind the flare.
GD Pathogenesis
GD is an autoimmune disorder that affects thyroid and is the foremost common explanation for hyperthyroidism. It has a female predominance and peaks in middle age [6]. It is also more commonly seen in smokers. GD is characterized by diffuse goiter and hypersecretion of thyroid hormones and symptoms typically develop gradually (weight loss, heat intolerance, muscle weakness, insomnia, tachycardia, sweating, irritability, restlessness, anxiousness, and fatigue, frequent bowel movements, irregular menstrual cycles in females, and erectile dysfunction in males among others) [6]. Exophtalmous could accompany GD and rarely, patients manifest thickened, reddish skin lesions called pretibial myxedema. GD’s etiology is autoimmune but environmental, genetic, and immunological factors contribute to its onset. GD is frequently associated with high titers of TRAb.
SARS-CoV-2 and GD
SARS-CoV-2 exploits ACE2 like a receptor for its initial entry. ACE2 expression was demonstrated to be higher in thyroid gland among others. It is likely therefore, that the thyroid gets affected by the virus as thyroid cells ubiquitously express the ACE2 protein [6]. By the same logic, COVID-19 could correlate with a higher risk of thyrotoxicosis (GD or subacute thyroiditis). Cases of SARSCoV- 2 infection-induced Graves’ disease have been reported [7-9].
As stress has been implicated in the relapses of GD, many viral infections have been considered environmental factors. Also, SARS-CoV-2 triggers a hyper-inflammatory condition that can set off the immunological response associated with GD (for instance IL6 is high in GD and in COVID) [6]. Moreso, Th2-mediated immune activity is likely to activate antigen-specific B lymphocytes to make TRAb. In addition, the naïve CD4+ T cells could differentiate into Th1, Th2, Th17, and Tregs depending on the cytokine milieu. For instance, IL-6 and TGF-β cytokines were shown to be detected prior to the elevated Th17 levels and Th17 itself plays a major role in development of GD. Consistently, a study evidenced that SARSCoV- 2 infection-associated hyper inflammation could induce Th17 [10].
ASIA
An adjuvant is an immunological or pharmacological substance or group of substances that can be added to a given agent to enhance its effect in terms of efficacy, effectiveness, and potency [11]. Adjuvants are commonly found in vaccines, immunization products, mineral oils, cosmetics, silicone breast implants and other therapeutic/medical devices, being usually safe and effective. However, in a fraction of genetically susceptible subjects, the administration of adjuvants may lead to serious sideeffects [11]. Indeed, ASIA is linked to genetic predisposition (for instance HLA-DRB1*01 or HLA-DRB4) and results from exposure to external or endogenous factors triggering autoimmunity [12]. AISA was attributed to molecular mimicry between microbial and human antigens that transformed a defensive immune-response into auto-immune response [13]. In 2017, a review of ASIA and auto-immune thyroid diseases shed the light over the fact that very few reports and animal models’ studies were published to show the relationship between endocrinopathies and adjuvants; However, the minority of cases was attributed to lack of awareness rather than rarity [13].
Conclusion
Cases of Grave’s disease manifestation or exacerbation post SARS-Cov-2 vaccines are reported. Theories are like SARS- Cov- 2 -induced GD but also other mechanisms are uniquely mediated by vaccines. That vaccination benefit outweighs the potential risks but one should have a low threshold to test and follow patients at risk for auto-immune thyroid diseases after receiving SARSCovV- 2 vaccine.
References
- Kahaly G (2020) Management of Graves Thyroidal and Extrathyroidal Disease: An Update. The Journal of Clinical Endocrinology & Metabolism 105(12): 3704-3720.
- Ng X, Betzler B, Testi I, Ho S, Tien M, et al. (2021) Ocular Adverse Events After COVID-19 Vaccination. Ocul Immunol Inflamm 29(6): 1216-1224.
- Vera-Lastra O, Ordinola Navarro A, Cruz Domiguez M, Medina G, Sánchez Valadez T, et al. (2021) Two Cases of Graves' Disease Following SARS-CoV-2 Vaccination: An Autoimmune/Inflammatory Syndrome Induced by Adjuvants. Thyroid 31(9): 1436-1439.
- Zettinig G, Krebs M (2021) Two further cases of Graves’ disease following SARS-Cov-2 vaccination. Journal of Endocrinological Investigation.
- Sriphrapradang C (2021) Aggravation of hyperthyroidism after heterologous prime-boost immunization with inactivated and adenovirus-vectored SARS-CoV-2 vaccine in a patient with Graves’ disease. Endocrine 74(2): 226-227.
- Murugan A, Alzahrani A (2021) SARS-CoV-2 plays a pivotal role in inducing hyperthyroidism of Graves’ disease. Endocrine 73(2): 243-254.
- Mateu-Salat M, Urgell E, Chico A (2020) SARS-COV-2 as a trigger for autoimmune disease: report of two cases of Graves’ disease after COVID-19. J Endocrinol Invest 43(10): 1527-1528.
- Jiménez-Blanco S, Pla-Peris B, Marazuela M (2020) COVID-19: a cause of recurrent Graves’ hyperthyroidism?. J Endocrinol Invest 44(2): 387-388.
- Harris A, Al Mushref M (2021) Graves’ Thyrotoxicosis Following SARS-CoV-2 Infection. AACE Clin Case Rep 7(1): 14-16.
- Costela-Ruiz V, Illescas-Montes R, Puerta-Puerta J, Ruiz C, Melguizo-Rodríguez L (2020) SARS-CoV-2 infection: The role of cytokines in COVID-19 disease. Cytokine & Growth Factor Rev 54: 62-75.
- Bragazzi N, Hejly A, Watad A, Adawi M, Amital H, et al. (2020) ASIA syndrome and endocrine autoimmune disorders. Best Pract Res Clin Endocrinol Metab 34(1): 101412.
- Watad A, Quaresma M, Brown S, Cohen Tervaert J, Rodríguez-Pint I, et al. (2017) Autoimmune/inflammatory syndrome induced by adjuvants (Shoenfeld’s syndrome) – An update. Lupus 26(7): 675-681.
- Watad A, David P, Brown S, Shoenfeld Y (2017) Autoimmune/Inflammatory Syndrome Induced by Adjuvants and Thyroid Autoimmunity. Front Endocrinol (Lausanne) 7: 150.






























