Thyroid Lymphoma Presentıng with Compressıve Symptoms and Early Transformatıon into Diffuse Large B-Cell Lymphoma
Akdeniz Y1, Kucuk A I2*, Yilmaz F3, Capoglu R1 and Kocer B1
1 Department of Surgery, Sakarya University, Turkey
2 Department of Oncologic Surgery, Balıkesir Atatürk City Hospital, Turkey
3 Department of Patology, Sakarya University, Turkey
Submission: December 12, 2019; Published: January 07, 2020
*Corresponding author:Ali Imran Küçük, Department of Surgical Oncology, Balıkesir Atatürk City Hospital Altıeylül / Balıkesir, Turkey
How to cite this article:Akdeniz Y, Kucuk A I, Yilmaz F, Capoglu R, Kocer B. Thyroid Lymphoma Presentıng with Compressıve Symptoms and Early Transformatıon into Diffuse Large B-Cell Lymphoma. J Endocrinol Thyroid Res. 2020; 5(2): 555660. DOI: 10.19080/JETR.2020.05.555660
Abstract
Primary thyroid lymphoma (PTL) is a rare thyroid malignancy. Low-grade B-cell lymphomas can be seldomly transformed into diffuse large B-cell lymphoma (DLBCL). The rate of histological transformation in marginal zone lymphoma is 5% for every 5-10 years and 10% for 12 years. In some studies, histological transformation time was reported as 48 months (4-139 months). In a 63-year-old patient who presented to our outpatient clinic with a sudden swelling on her neck, a transformation occurred to DLBCL within 3 months after the operation, leading to compressive symptoms which resulted in respiratory distress in the patient. In the literature, no other thyroid low-grade B-cell lymphomas cases have been found to have relapsed and caused compressive symptoms such a short time and have undergone a subtype transformation after the second operation. In our article we will review the literature accompanied by our case.
Keywords: Thyroid lymphoma; Histological transformation; Surgery
Introduction
Primary thyroid lymphoma (PTL) is a rare thyroid malignancy. The incidence in all thyroid malignancies is approximately 1-5%. It constitutes approximately 1-2% of extranodal lymphomas [1,2]. Low-grade B-cell lymphomas (follicular lymphoma, MALT lymphoma and small lymphocytic lymphoma) can be transformed into diffuse large B-cell lymphoma (DLBCL). The transformation rate in follicular lymphoma is 3% per year and in small lymphocytic lymphoma this rate is 16% in 10 years [3,4]. It also varies between 3-12% in MALT lymphoma [5]. In a study, the rate of histological transformation in marginal zone lymphoma was 5% for every 5-10 years, whereas this rate was found to be 10% for 12 years [6]. In a large-scale study, 467 patients with MALT lymphoma were evaluated and 37 patients of them had DLBCL transformations. There were 16 cases of PTL in the histologically transformed patient group, but none showed any transformation to DLBCL [7]. We present a case of extranodal marginal zone B-cell lymphoma in thyroid transformed into DLBCL within 3 (three) months after surgery. In the literature, no case of PTL was observed with histological transformation in such a short time.
PTL is about 3-5 times more common in women than men. The most common age group is between the 5th and 8th decades. The incidence before the age of 40 is very low [8,9]. Various causes of PTL pathogenesis have been proposed. The most important risk factor is the presence of Hashimato Thyroiditis (HT) in patient. Chronic antigenic stimulation and various autoimmune diseases are also blamed. The overall rate of PTL development in the entire HT patient population is approximately 0.5-0.6%. The risk in this patient group increased by 40-80 times compared to the normal population. 80% of PTL patients have a history of HT. PTL develops approximately 20-30 years after the diagnosis of HT [10-12].
Approximately 80% of patients present with a rapidly growing mass in the neck region for the last 1-3 months. Depending on the rapidly growing mass; compressive symptoms such as dyspnea, dysphagia, stridor and hoarseness can be seen in patients. Rarely, cases of Horner syndrome related to the Superior Vena Cava compression of the mass have been reported. In addition to clinical findings, cervical lymphadenopathy is rarely seen in patients [13,14]. Weight loss, fever and nocturnal sweats, called B-type symptoms, can be seen in approximately 20% of patients [15]. In our study; we will analyze our case of PTL, which can be diagnosed postoperatively and have undergone subtype transformation with early recurrence during treatment planning.
Case Report
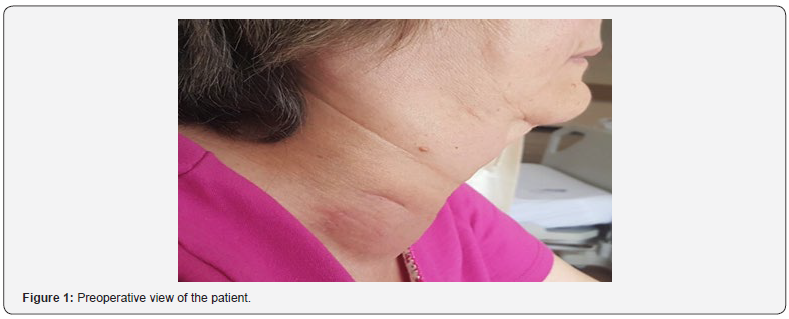
A 63-year-old female patient was admitted to our outpatient clinic with sudden onset of neck swelling. On physical examination, the thyroid gland was bilateral palpable. In the right lobe at isthmus junction, there was a palpable nodule of approximately 6 cm, with a soft consistency and with no clearly selected margins (Figure 1). Our patient had no nocturnal sweats, weight loss and fever symptoms. In the thyroid ultrasound (USG) examination of the patient, the right lobe was 50X52X72 mm, isthmus 20 mm and the left lobe was 32X24X63 mm. 35 mm hypoechoic areas, the largest of which were located in isthmus and which could not differentiate between nodule and pseudo nodule, detected in thyroid. Cervical or supraclavicular lymph nodes were not present. The thyroid function tests of patient were normal. Fine needle aspiration biopsy (FNAB) was performed and the result was compatible with benign cystic colloidal nodule. After 3 months, the largest hypoechoic area in the control USG was 40 mm, and again the FNAB was requested. FNAB results were reported as thyroiditis. Because of increased thyroid and nodule size on physical examination and presence of compressive symptoms and respiratory distress, operation was decided.

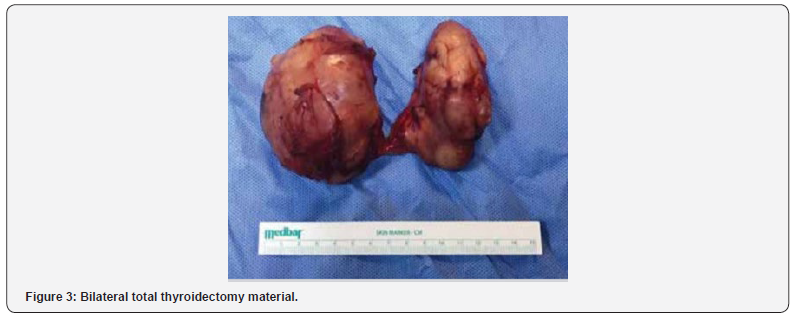
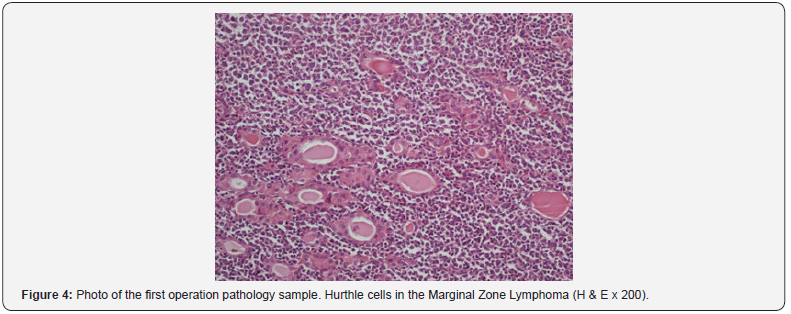
The preoperative MRI showed a multinodular thyroid gland which pushed the surrounding tissue with no invasion (Figure 2). The patient underwent bilateral total thyroidectomy (BTT) (Figure 3). Postoperative pathology result was extranodal marginal zone B-cell lymphoma (Figure 4). Immunohistochemical staining with CD 20, CD79a, Bcl-2, MUM-1 were positive, but staining with CD3, CD10 and Bcl-6 were negative. Ki-67 index was evaluated as 10- 15%. The patient was then referred to Hematology outpatient clinic for treatment planning. A bone marrow biopsy was performed, and the result was normal. Within three-month post operative treatment period, the patient had complaints of swelling and dyspnea. In her physical examination, she had a soft mass at operation margin. The 18F-FDG positron emission tomography (PET) of the patient revealed a mass that deviated the trachea to the left and filled the supra and the retroclavicular area and which was thought to be invasive to the esophagus. In the USG performed to the patient; there was a 55x42 mm sized mass in the supraclavicular area in the right half of the cervical region. The boundary between the mass and right CCA was lost and the internal jugular vein was dilated due to compression. The patient was operated again because of increased respiratory distress and compressive symptoms.
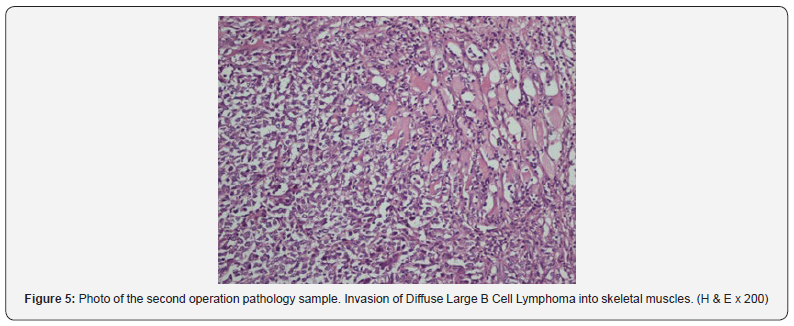
Thyroid was too tightly adhered surrounding soft tissues, esophagus and invaded to trachea, so dissection and resection of the thyroid were impossible, so only incisional biopsy was taken. Postoperative pathology result was infiltration of diffuse large B-cell lymphoma; because of the presence of small atypical lymphocytes and infiltration in the surrounding muscle tissue, it was thought that extranodal marginal zone lymphoma was transformed into large cell lymphoma (Figure 5). Cross sections showed diffuse tumor infiltration in the surrounding soft tissue and striated muscle tissue. The tumor consisted of atypical small centrocytic lymphocytes as well as atypical large centroblastic cells including atypical mitosis and hyperchromatic one or more nuclei adjacent to the nuclear membrane and some vesicular nucleus. Tumor cells were stained with CD20, CD79a and bcl-6; bcl-2 was positive in small lymphocytes, CD10, CD23, CD3, CD5, cyclin D1 was negative in immunohistochemical staining. Ki-67 proliferation index was evaluated as approximately 40%. Three cycles of cyclophosphamide, doxorubicin, vincristine, prednisolone and rituximab (R-CHOP) chemotherapy was administered to the patient. Then, 60 Gy RT was applied to the patient. 18F-FDG PET did not show any recurrence in 18 months postoperatively and follow-up is ongoing.
Discussion
PTL should be suspected in a woman with a long history of Hashimato thyroiditis and a rapidly growing mass in the neck. The first procedure to be performed for diagnosis after physical examination is thyroid ultrasonography (USG). In USG, PTL can be seen in three ways; they are nodular, diffuse or mixed type [16]. USG shows a hypoechoic, homogeneous solid lesion with internal echoes and unilateral hypervascularity in noduler type. In diffuse type; bilateral enlarged thyroid gland with hypoechoic structure is observed [17,18]. Internal and posterior echoes can be seen in all three types. Especially, an increase in posterior echogenic appearance is important in differentiating PTL from other fast-growing thyroid lesions [16]. In their study comparing USG images with pathological diagnosis; Ota et al. found that the positive predictive value of the nodular and mixed type was higher than the diffuse type (64.9%/63.2%/33.7%)[19]. FNAB is the most important diagnostic tool for thyroid lesions. False negative rate of FNAB are high in PTL. Due to the small number of patients, there are small scale studies in the literature. In the study of Matsuzuka et al.[13] in which maximum number of patients, the rate of successful diagnosis with FNAB was 78%. In 2010, in a retrospective study of 70 patients by Morgen et al. FNAB success rate was 65% [20]. In the study of Dustin et al. all 15 patients were diagnosed with FNAB [21]. FNAB was combined with immunohistochemical staining and flow cytometry methods to reduce false negativity [22]. Histopathological, it is important to differentiate MALT lymphoma with DLBCL or to differentiate autoimmune thyroiditis with MALT lymphoma. In immunohistochemical staining, DLBCL is usually stained with CD19, CD20, bcl-6 and CD45 while in MALT lymphoma there is no staining with CD5, CD10 and CD23 [23]. Due to characteristic cells, diagnosis with FNAB in DLBCL is easier than lymphomas caused by MALT lymphomas or Hashimato thyroiditis. Because of this insufficiency of FNAB in the diagnosis of PTL, tru-cut biopsy or open surgical biopsy remains important in suspected cases [12,18].
In addition, the higher amount of tissue obtained by tru-cut biyopsy and open surgery, allows the histological examination of the tumor, immunohistochemical staining and gene research. It is necessary to perform staging in patients with PTL before treatment. Various imaging methods such as USG, CT, MRI and PET / CT can be used for staging. There are also several studies suggesting that PET/CT can be used at the time of initial diagnosis and for evaluating the response of the disease to treatment [24- 26]. Ann-Arbor classification is used for pre-treatment staging [27]. When the stages of the studies are examined; 56% of patients were stage 1E, 32% were stage 2E, 2% were 3E and 11% were stage 4E. Five-year disease-free survival rates were 86% in stage 1E, 81% in stage 2E, and 64% in stage 3-4. When the five-year disease-free survival rates according to histological subtypes are examined; MALT lymphoma is observed to be 96%, DLBCL is 75%, Follicular lymphoma is 87% and other lymphoma subtypes are approximately 83% [28,29].
Poor prognostic factors affecting survival can be evaluated as advanced age, presence of B-type symptoms, presence of lymph node metastases, large tumor size and rapid clinical growth [30,31]. In general, the clinical course of patients with MALT lymphoma is slow. Five-year survival rates in all MALT lymphomas are approximately 89% [32]. It is observed that this ratio decreased to 57% after the histological transformation [6]. MALT lymphomas can also be immunophenotypical CD20 +, CD79a +, CD5 - / +, CD10-, CD23, CD43 - / +, BCL6- and MUM1 - / +. DLBCL developed from MALT lymphoma after histological transformation has no immunophenotypical distinctive properties. However, CD10, BCL6 and MUM1 expression increased in these cases. When we look at all MALT lymphomas, the mean histological transformation rate is 8% [33]. This rate is 7% in early stage disease and 15% in advanced stage disease. In the study on 441 patients with MALT lymphoma, the risk of histological transformation at 5, 10 and 15 years is examined; 3%, 3% and 5% respectively
These rates were 2%, 2% and 4% in 385 patients in the early stage; In 56 patients in the advanced stage, the rates were 7%, 7% and 16%, respectively. In this study, the mean time of histological transformation was 48 months (4-139 months) [7]. In our case, a transformation occurred to DLBCL within 3 months after the operation and caused enough compression symptoms to cause respiratory distress in the patient. In the literature, no other cases have been found which transformed to DLBCL with subtype transformation in the pathology report after the second operation which causes recurrent compression symptoms and which relapses in such a short time. The treatment in PTL may vary depending on the stage and subtype of the disease. PTL is sensitive to chemotherapy and radiotherapy such as other lymphomas. The role of surgery in the treatment of PTL is controversial. In a study of 62 patients with stage 1E and 2E at the Mayo clinic in 1992, no advantage of debulking surgery on open biopsy could be demonstrated. Biopsy + RT treatment was compared with Surgery + RT treatment in this study and Biopsy + RT treatment was found to be more successful (88% & 85%) [34].
Surgery can only be performed in patients who could not be diagnosed preoperatively as in our case. There is no need for adjuvant therapy in patients with no preoperative diagnosis and with postoperative pathology report resulting in stage 1E MALT lymphoma. In the study which was published by Derringer et al. in 2000, 16 patients with MALT lymphoma were treated only with surgery and no recurrence developed in 7 years of follow-up [8]. In our case, the postoperative first pathology results were extranodal marginal zone B-cell lymphoma in the MALT type. There was no dissemination of lymphoma outside the thyroid gland during the preoperative examinations and during the operation. Except MALT lymphoma, PTL subtypes are treated by combination of chemotherapy and radiotherapy. The standard regimen is CHOP chemotherapy. PTL responds quickly to this treatment regimen. Chemotherapy treatment combined with radiotherapy further reduces local recurrence rates. In general, radiotherapy is added to the treatment plan after 3 cycles of CHOP chemotherapy [35].
In the study of Doria et al. [35] local recurrence rate after chemoradiotherapy treatment was 7.7%, this rate was 37.1% after radiotherapy treatment alone. In this study, the rate of local recurrence was 43% in chemotherapy alone [36]. In 2011, in the retrospective study of Onal et al.,[14] overall survival was investigated. Overall survival was 91% in combined therapy, 69% in radiotherapy, and 57% in chemotherapy alone. However, in the study of Skacel et al. [36], there was no difference between treatment modalities [37]. In the last 10 years, Rituximab therapy has been added to CHOP chemotherapy in B cell mediated PTL. Rituximab is a monoclonal antibody developed against CD20 which is a B cell surface antigen. The addition of Rituximab to chemotherapy treatment in DLBCL increases overall survival while decreasing local recurrence rates [38]. However, some studies have showed that tumors with BCL-2 and BCL-6 overexpression responds less to rituximab [39].
Conclusion
PTL is a rare disease. In patients who have been followed for a long time due to Hashimato thyroiditis; should be evaluated in differential diagnosis of rapidly growing thyroid gland masses. In patients with recurrent PTL in the early postoperative period, caution should be taken in terms of aggressive subtype transformation as in our case.
References
- Ansell SM, Grant CS, Habermann TM (1999) Primary thyroid lymphoma. Semin Oncol 26: 316-323.
- Green LD, Mack L, Pasieka JL (2006) Anaplastic thyroid cancer and primary thyroid lymphoma: A review of these rare thyroid malignancies. J Surg Oncol 94: 725-736.
- Montoto S, Davies AJ, Mathews J, Calaminici M, Norton AJ, et al. (2007) Risk and clinical implications of transformation of folicular lymphoma to diffuse large B-cell lymphoma. J Clin Oncol 25: 2426-2433.
- Rossi D, Cerri M, Capello D, Deambrogi C, Rossi FM, et al. (2008) Biological and clinical risk factors of chronic lymphocytic leukaemia transformation to Richter syndrome. Br J Haematol142: 202-215.
- Zucca E, Conconi A, Laszlo D, Bouabdallah R, Coiffier B, et al. (2013) Addition of rituximab to chlorambucil produces superior event-free survival in the treatment of patients with extranodal marginal zone B-cell lymphoma: 5-year analysis of the IELSG-19 Randomized study. J Clin Oncol 31: 565-572.
- Conconi A, Franceschetti S, Aprile von Hohenstaufen K, Margiotta-Casaluci G, Stathis A, et al. (2015) Histologic transformation in marginal zone lymphomas. Ann Oncol 26: 2329-2335.
- Maeshima AM, Tanuguchi H, Toyoda K, Toyoda K, Yamauchi N, et al. (2016) Clinicopathological features of histological transformation from extranodal marginal zone B- cell lymphoma of mucosa-associaed lymphoid tissue to diffuse large B- cell lymphoma: an analysis of 467 patients. Br J Haematol 174: 923-931.
- Derringer GA, Thompson LD, Frommelt RA, Bijwaard KE, Heffes CS, et al. (2000) Malignant lymphoma of the thyroid gland: a clinicopathologic study of 108 cases. Am J Surg Pathol 24: 623-639.
- Sarinah B, Hisham AN (2010) Primary lymphoma of the thyroid: diagnostic and therapeutic considerations. Asian J Surg 33: 20-24.
- Pedersen RK, Pedersen NT (1996) Primary non-Hodgkin’s lymphoma of the thyroid gland: A population based study. Histopathology 28: 25-32.
- Holm LE, Blomgren H, Lowhagen T (1985) Cancerrisks in patients with chronic lymphocytic thyroiditis. N Engl J Med 312(10): 601-604.
- Pasieka JL (2000) Hashimato’s disease and thyroid lymphoma: Role of the surgeon. World J Surg 24: 966-970.
- Matsuzuka F, Miyauchi A, Katayama S, Narabayashi I, İkeda H, et al. (1993) Clinacal aspects of primary thyroid lymphoma: diagnosis and treatment based on our experience of 119 cases. Thyroid 3: 93-99.
- Onal C, Li YX, Miller RC, Poortmans P, Constantinou N, et al. (2011) Treatment results and prognostic factors in primary thyroid lymphoma patients: a rare cancer network study. Ann Oncol 22: 156-164.
- Katna R,Shet T, Sengar M, Menon H, Laskar S, et al. (2013) Clinicopathologic study and outcome analysis of thyroid lymphomas: Experience from a tertiary cancer center. Head Neck 35: 165-171.
- Stein SA, Wartofsky L (2013) Primary thyroid lymphoma: a clinical rewiew. J Clin Endocrinol Metab 98: 3131-3138.
- Sharma A, Jasim S, Reading CC, Ristow KM, Villasboas Bisneto JC, et al. (2016) Clinical Presentation and diagnostic challenges of thyroid lymphoma: a cohort study. Thyroid 26: 1061-1067.
- Hu G, Zhu X (2016) Ultrasonographic features of agresive primary thyroid diffuse B-cell lymphoma: a report of two cases. Oncol Lett 11: 2487-2490.
- Ota H, Ito Y, Matsuzuka F, Kuma S, Fukata S, et al. (2006) Usefulness of ultrasonography for diagnosis of malignant lymphoma of the thyroid. Thyroid 16: 983-987.
- Morgen EK, Geddie W, Boerner S, Bailey D, Santos Gda C (2010) The role of fine – needle aspiration in the diagnosis of thyroid lymphoma: A retrospective study of nine cases and review of published series. J Clin Pathol 63: 129-133.
- Dustin SM, Jo VY, Hanley KZ, Stelow EB (2012) High sensitivity and positive predictive value of fine – needle aspiration for uncommon thyroid malignancies. Diagn Cytopathol 40: 416-421.
- Sangalli g, Serio G, Zampatti C, Lomuscio G, Colombo L (2001) Fine – needle aspiration cytology of primary lymphoma of the thyroid: A report of 17 cases. Cytopathology 12: 257-263.
- Dralle H, Musholt TJ, Schabram J, Steinmüller T, Frilling A, et al. (2013) Surgeons practice guideline fort he surgical management of malignant thyroid tumors. Langenbecks Arch Surg 398: 347-375.
- Basu S, Li G, Bural G, Alavi A (2009) Fluorodeoxyglucose positron emission tomography (FDG-PET) and PET computed tomography imaging characteristics of thyroid lymphoma and their potantial clinical utility. Acta Radiol 50: 201-201.
- Arabi M, Dvorak R, Smith LB, Kujawski L, Groos MD (2011) Fluorodeoxyglucose positron emission tomography in primary thyroid lymphoma with coexisting lymphocytic thyroiditis. Thyroid 21:1153-1156.
- Lin EC (2007) FDG PET/CT for assessing therapy response in primary thyroid lymphoma. Clin Nucl Med 32: 152-153.
- Carbone PP, Kaplan HS, Musshoff K, Smithers DW, Tubiana M (1971) Report of the committee on Hodgkin’s disease staging classification. Cancer Res 31: 1860-1861.
- Graff-Baker A, Roman SA (2009) Prognosis of primary thyroid lymphoma: demographic, clinical and pathologic predictors of survival in 1408 cases. Surgery 146(6): 1105-1115.
- Freeman C, Berg JW (1972) Occurence and prognosis of extranodal lymphomas. Cancer 29(1): 252-260.
- Alizadeh AA, Eisen MB,Davis RE, Ma C, Lossos IS, et al. (2000) Distinct types of diffuse large B- cell lymphoma identified by gene expression profiling. Nature 403(6769): 503-511.
- Rosenwald A,Wright G, Chan WG, Connors JM, Campo E, et al. (2002) The use of molecular profiling to predict survival after chemotherapy for diffuse large B- cell lymphoma. N Engl J Med 346(25): 1937-1947.
- Olszewski AJ, Castillo JJ (2013) Survival of patients with marginal zone lymphoma: analysis of the surveillance, epidemiology and end results database. Cancer 119: 629-638.
- Pyke CM, Grant CS, Haberman TM, Kurtin PJ, van Heerden JA, et al. (1992) Non Hodgkin’s lymphoma of the thyroid: İs more than biopsy necessary? World J Surg 16: 604-610.
- Miller TP, Dahlberg S, Cssady JR, Adelstein DJ, Spier CM, et al. (1998) Chemotherapy alone compared with chemotherapy plus radiotherapy for localized intermediate and high grade non Hodgkin’s lymphoma. N Eng J Med 339: 21-26.
- Doria R, Jekel JF, Cooper DL (1994) Thyroid lymphoma. The case for combined modality therapy. Cancer 73: 200-206.
- Skacel M, Ross CW, Hsi ED (2000) A reassessment of primary thyroid lymphoma: High-grade MALT-type lymphoma as a distinct subtype of diffuse large B cell lymphoma. Histopathology 37: 10-18.
- Hiddemann W, Kneba M, Dreyling M, Schmitz N, Lengfelder E, et al. (2005) Frontline therapy with rituximab added to the combination of cyclophosphamide, doxorubicin, vincristine and prednisone (CHOP) significantly improves the outcome for patients with advanced-stage follicular lymphoma compared with therapy with CHOP alone: Results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood 106: 3725-3732.
- Visco C, Tzankov A, Xu-Monette ZY, Miranda RN, Tai YC, et al. (2013) Patients with diffuse large B cell lymphoma of germinal center origin with BCL-2 translocations have poor outcome, irrespective of MYC status: A report from an international DLBCL rituximab-CHOP Consortium Program Study. Heamatologica 98: 255-263.
- Winter JN, Weller EA, Horning SJ, Krajewska M, Variakojis D, et al. (2006) Prognostic significance of Bcl-6 protein expression in DLBCL treated with CHOP or R- CHOP: A prospective correlative study. Blood 107: 4207-4213.






























