Long-Term Follow-Up in a Male Patient with Micro-TSH-Oma Diagnosed at 8-Yr-Old
Adriane A Cardoso-Demartini1*, Mauro A Czepielewski2, Suzana Nesi-França1, Rosana Marques-Pereira1, Nelson P Ferreira3 and Luiz De Lacerda1
1Department of Pediatrics, Universidade Federal do Paraná, Curitiba, Brazil
2Department of Internal Medicine, Universidade Federal do Rio Grande do Sul, Brazil
3Department of Neurosurgery, Santa Casa de Misericórdia de Porto Alegre, Brazil
Submission: July 01, 2019;Published: July 18, 2019
*Corresponding author: Adriane A Cardoso-Demartini, Pediatric Endocrinology Unit, Department of Pediatrics, Federal University of Paraná, Curitiba, Rua Padre Camargo, 250 – Curitiba, PR, Brazil Introduction
How to cite this article: Adriane A Cardoso-Demartini, Mauro A Czepielewski, Suzana Nesi-França, Rosana Marques-Pereira, Nelson P Ferreira, Luiz De Lacerda. Long-Term Follow-Up in a Male Patient with Micro-TSH-Oma Diagnosed at 8-Yr-Old. J Endocrinol Thyroid Res. 2019; 4(5): 555646. DOI:10.19080/JETR.2019.04.555646
Abstract
Background:TSH-secreting pituitary adenomas (TSH-omas) are very rare and an infrequent cause of thyrotoxicosis.
Case Report:A 7.9-yr-old boy was referred to our Pediatric Endocrinology Unit due to a goiter. On admission, patient was thyrotoxic with diffuse goiter. Laboratory evaluation suggested inappropriate TSH secretion as the cause of hyperthyroidism: high serum TSH in presence of elevated levels of TT4, TT3 and fT4, and TSH unresponsive to TRH stimulation and to T3 suppression. Initially, α-subunit (aSU) was in the upper limit of normalcy and pituitary MRI was normal. One year after, patient was still hyperthyroid, despite regular use of methimazole; TSH was 12.6 mU/mL, αSU was elevated and MRI detected a pituitary 8 mm width adenoma, establishing the diagnosis of TSH-oma. Peak GH (ng/dL) on ITT and TSH after TRH were 5.9 and 4.2, respectively. Cortisol and prolactin (PRL) responded normally to ITT and TRH tests. Transsphenoidal surgery was done and, postoperatively, transient diabetes insipidus and adrenal insufficiency ensued. Two and five months after surgery fT4, TT4 and TT3 were normal, albeit peak TSH after TRH was 1.54. PRL and GH were unresponsive to adequate stimuli. Fourteen months after surgery, TT4, TT3 and fT4 were low normal. He presented with low IGF-1, low GH peak on dinanic tests and hypogonadism and was treated with recombinant human growth hormone (rhGH) and testostenone. At 16 yr-old, we reached final height, above target height.
Conclusion: TSH-oma may be an etiology of thyrotoxicosis in children. To our knowledge, this is one of the youngest patients with TSH-oma yet reported.
Keywords: Pituitary tumor; TSH-oma; Hyperthyroidism; Thyrotoxicosis; Transsphenoidal surgery; Final height
Introduction
In most children with thyrotoxicosis the main cause is Graves’ disease. Other causes include toxic adenoma, thyroiditis, iodine-induced hyperthyroidism, McCune-Albright syndrome, syndrome of resistance to thyroid hormone (RTH) and thytroid-stimulating hormone (TSH) secreting pituitary adenoma (TSH-oma). TSH-oma comprises 0.5 to 3% of all pituitary tumors. Patients present with signs and symptoms related to thyroid hormone (TH) excess and/or to tumor size (headache, visual field disturbances, cranial nerve palsies). The presence of goiter is frequent [1,2]. Elevated TH levels in presence of non-suppressed TSH should occur in TSH-omas, as well as in other conditions such as early phase of destructive thyroiditis, irregular replacement of l-thyroxine, assay interference of heterophilic antibodies and RTH. The combination of high serum free TH, inappropriately normal or elevated TSH, high serum α-subunit (αSU) or increased αSU/TSH molar ratio and a pituitary tumor strongly suggests the diagnosis of a TSH-oma.
Triiodothyronine (T3) suppression test is generally reserved for patients with inconclusive results in above tests, because genetic tests for detection of mutations in thyroid receptor (TR)α and TRβ genes are expensive. Administering long-acting somatostatin analogs has been proposed for distinguishing between thyrotropinomas and RTH, since patients with thyrotropinomas would be likely to show a significant reduction in free thyroxine (fT4) and T3 levels. Approximately one third of patients with TSH-oma were misdiagnosed as having primary hyperthyroidism and mistakenly treated with thyroidectomy or radioiodine [2]. The majority of TSH-omas is monoclonal in origin, like other types of pituitary adenomas. Pituitary-specific transcription factor-1 (Pit-1) may play a role in adenomatous cell proliferation and its overexpression was detected in growth hormone- (GH), prolactin- (PRL) and TSH-secreting adenomas more frequently than in normal pituitary. Reduced expression of TR was demonstrated, and it could explain the abnormal negative feedback of TH on TSH production by tumor cells [2,3].
TSH-omas are more fibrotic than other pituitary tumors and it can worsen surgical outcome and somatostatin analog treatment should be considered as the first-line treatment in adults with macroinvasive TSH-omas [2,4,5]. Such an adenoma is infrequent in adults and has rarely been report in children, we describe an 8-yr-old boy with TSH-oma, and his follow-up until final height. The patient and his mother assigned consentient term.
Case Presentation
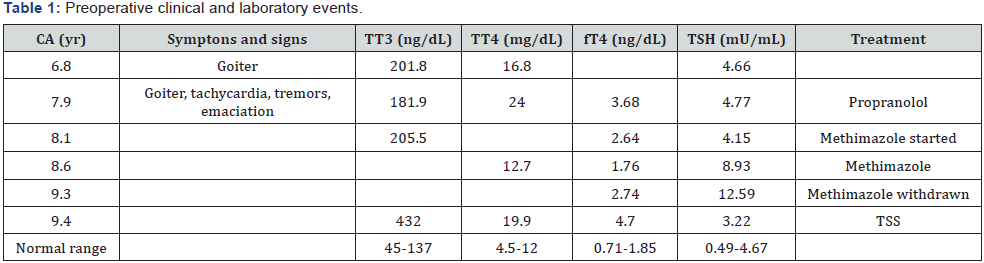
A 7.9-yr-old white pre-pubertal boy was referred to Pediatric Endocrinology Unit due to goiter. His mother noticed he was more irritable, and lost weight albeit an increased appetite. History was negative for insomnia, headache or visual disturbance. Physical examination disclosed a lean and hyperactive child with stare opened eyes, warm and moist hands, with fine tremors. Height was 138cm (1.78SDS; target height -0.96SDS), weight 27.2kg (0.40SDS), and BMI 14.28 (-1.14SDS). Pulse rate was regular (108bpm) and blood pressure 100/60mmHg. Thyroid was tender, diffusely enlarged (app.30g). Deep tendon reflexes were exacerbated. Laboratory work-up revealed a bone age (BA, Greulich & Pyle) of 9-yr, and the following thyroid function profile (normal values in brackets) was found: TT3 181.9 (45- 137ng/dL), TT4 24 (6-12μg/dL), fT4 3.68 (0.71-1.85ng/dL), TSH 4.77 (0.49-4.67μU/mL); basal and peak TSH on TRH test 4.6 and 6.2, respectively; pre and post T3 suppression test RAIU (24h) values were 42.1 and 30% respectively, while TSH did not change significantly (4.15) whereas fT4 exhibited some reduction (2.64). Anti-thyroid receptor (TRAb), anti-thyroglobulin (anti-TG) and anti-thyroid peroxidase (anti-TPO) antibodies were negative. Calcium and PTH levels were normal. αSU was 0.86 (≤0.8ng/mL), αSU/TSH molar ratio 2 (<1) and magnetic resonance imaging (MRI) of pituitary was normal.
Patient was managed with propranolol (1 mg/kg/day) and methimazole (MTZ, 0.5 mg/kg/day) and thereafter, with MTZ exclusively. Table 1 summarizes main preoperative clinical and laboratory events. While on MTZ, T4 and T3 did not normalize, TSH values ranged between 5.81 and 12.59 and goiter was slightly enlarged. MTZ was withdrawn and thyroid and pituitary functions were evaluated three weeks later. On combined insulin hypoglycemia (ITT)/TRH tests, prolactin (PRL) and cortisol rose properly, peak GH (ng/mL) was 5.9 and TSH was unresponsive (Table 2). Basal LH and FSH were normal and IGF-I was 434 (30- 289 ng/mL). RAIU was elevated (75.8%) and rose paradoxically (87%) after T3 suppression test. Sex hormone-binding globulin (SHBG) was 233 (13-71 nmol/L), and T4-binding globulin (TBG) was 16 (10-29 mg/dL). Repeated TRAb, anti-TG and anti-TPO were negative. At this time αSU was high to 0.949 (αSU/TSH 4.7) and pituitary MRI revealed the presence of an 8 mm width microadenoma (Figure 1).
CA = Chronological age
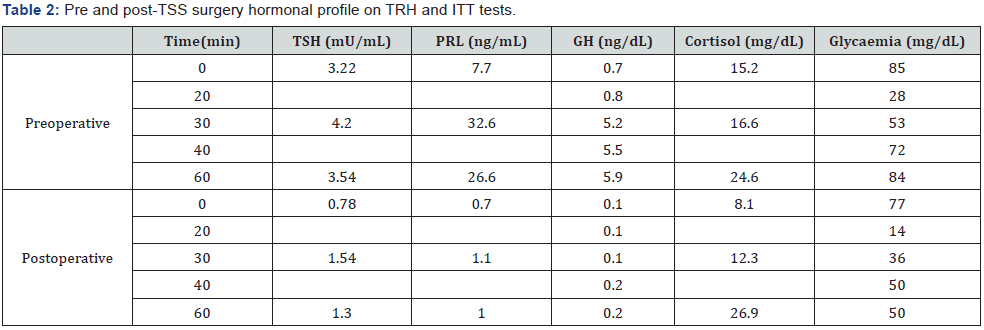
vTranssphenoidal surgery (TSS) was performed and a well-demarcated, fibrous and firm adenoma was excised. The pathologic specimen showed adenoma cells that were immunopositively only for TSH and chromogranin and negative for LH, FSH, PRL, ACTH, and GH. Eighteen hours after surgery, serum TSH and fT4 descended to 0.53 and 1.8, respectively and goiter and thyrotoxicosis signs diminished as well. On the 3rd day postoperatively, acute adrenal insufficiency and transient diabetes ensued. Hydrocortisone and DDAVP were given and maintained for 2 and 14 months, respectively. Two months after TSS, ACTH was 12 (10-50 pg/mL) and IGF-1 64 (74-388 ng/mL). Peak GH and cortisol (μg/dL) on ITT were 0.2 and 26.9 respectively. PRL and TSH responses to TRH were blunted; however, RAIU was normal (23.8%). One year after surgery, BA was 11.5, TT3, TT4 and fT4 were in the low-normal range for age, calorimetry was sub-normal and pituitary MRI showed no evidence of tumor.
He had gained weight, but growth velocity was <1 cm/yr despite adequate replacement dose of l-thyroxine (88 μg/day) He was put on rhGH (0,033 mg/kg/day) and growth velocity improved significantly (9.2 cm/yr). Three years after surgery, he is still pre-pubertal and growing normally (on both l-thyroxine and rGH). Last pituitary MRI was normal and aSU lower than 0.05 (aSU/TSH 0.61). His BA was 13.0 (chronological age 12.5) and peak LH and FSH after GnRH were 1.1 and 1.4 mU/mL, respectively. At that time, testosterone replacement was started and after 9 months, he was pubertal. Five years after surgery, rhGH was suspended, because he reached height above target height. Six months later, testosterone replacement was stopped. However, pubertal stage did not evolve and IGF-1 was 145 (226- 903 ng/mL), testosterone (250 mg/month) and rhGH (0.6 mg/ day) were re-started. At his last visit, at 16.3 yr.-old, height was 178.9 cm (0.59 SDS) and pituitary MRI was normal. Figure 2 shows his height and weight SDS during follow-up and table 3 summarizes main postoperative clinical and laboratory events.
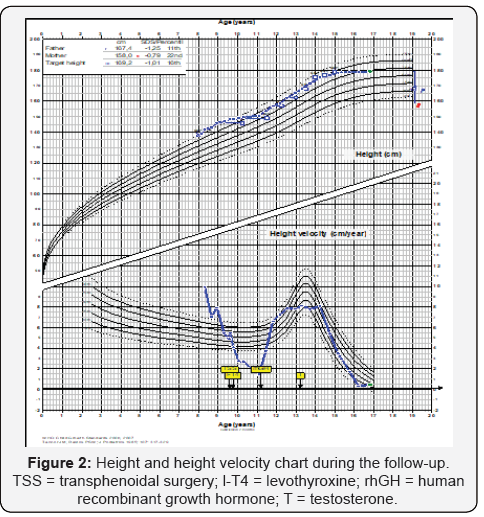
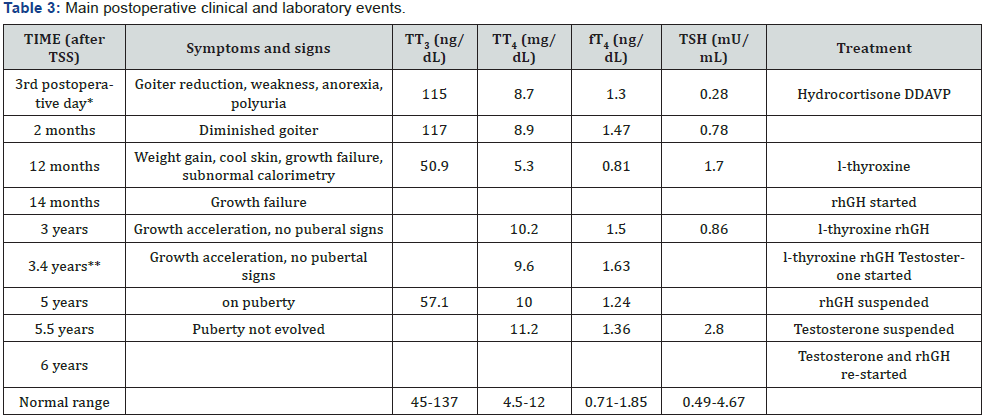
*Basal cortisol = 3.43 mg/dL (6-19); urine density = 1005; ** Total testosterone = 106 ng/dL (<100); peak LH and FSH after GnRH = 1.1 and 1.4 mU/mL; CA = Chronological age; BA = bone age; rhGH = ecombinant human growth hormone.
Discussion
Once inappropriate TSH secretion syndrome is identified, specific investigation to differentiate a TSH-oma of RTH is mandatory [2]. In our patient, TSH was not responsive to TRH stimulation test and both aSU and aSU/TSH molar ratio were high. In not previously treated subjects with RTH, the TSH response to TRH is preserved, and aSU/TSH is normal. Moreover, in RTH subjects a decreased secretion of TSH after supraphysiological doses of TH is usually accompanied by a reduction in RAIU [6], what was not observed in our patient. These findings suggest that RTH was not likely. TSH-omas are rare in adults and to our knowledge our patient is one of the youngest children with hyperthyroidism due to TSH-oma ever reported. Other 13 children or adolescents described were 8-yr. or older (8 to 16yr) and had macroadenoma except a 13 yr-old girl who had microadenoma and a 15 yr-old girl whose tumor size was not decribed [7-19].
In this case, pituitary MRI suggested microadenoma, although 88% of TSH-omas are usually large and invasive [2,20]. Patient underwent TSS because the primary goal of treatment of TSHomas is, whenever possible, the complete removal of the tumor [2]. TSH, TH and aSU levels reduced soon after surgery and one week after, patient was euthyroid. TSS was successful in regard the complete removal of the tumor, although in the follow-up central hypothyroidism, and GH, PRL, LH and FSH deficiencies succeeded. Panhypopituitarism and diabetes insipidus also have been reported [14].
The first case of a patient with TSH-oma and normal aSU was described in 1991 [21]. Valdes-Socin et al. observed normal aSU in more than 60% of the cases. High aSU is often associated with bad prognosis and was found more frequently in macro than in microadenomas [4]. The high percentage of patients with normal aSU could difficult differential diagnosis with RTH. Absence of TSH response to TRH may be suggestive of presence of a TSHoma. In difficult cases, genetic analysis looking for the presence of a mutation in TRβ gene may easily help to discriminate between the two disorders [2]. SHBG could also be a useful test yet its level was almost invariably normal in patients with RTH but often high in thyrotoxic patients with TSH-oma [2]. One challenging situation is those patients with an invisible adenoma on MRI and near-normal aSU, as initially occurred in our patient, whose diagnosis was done one year after inappropriate treatment with MTZ; possibly, that promoted tumor growth.
This case shows interesting aspects: the age of the patient at diagnosis; the finding of a normal MRI in contraposition to the elevated aSU/TSH molar ratio that was not adequately interpreted; growth of the tumor during MTZ, blunted TSH response to TRH in the post-operative phase in contradiction to diminished calorimetry, low-normal values of fT4, TT4 and TT3, and normal RAIU. We presented a comprehensive evaluation of a patient with TSH-oma followed for more than 8yr, who attained a final height, in accordance with the target height due to adequate therapeutic management.
Learning points
i. Once inappropriate TSH secretion syndrome is identified, specific investigation to differentiate a TSH-oma of RTH is mandatory, even in children.
ii. Patients who have TSH-oma could be misdiagnosed as having primary hyperthyroidism and, thus, mistakenly treated with antithyroid drugs or thyroid ablation.
iii. TSH-oma may be a microadenoma and be present even when aSU is near-normal or normal and it is a challenging situation.
iv. After surgery, follow-up should be prolonged and hormonal deficiencies should be diagnosed and treated.
References
- Beck-Peccoz P, Persani L, Mannavola D, Campi I (2009) Pituitary tumours: TSH-secreting adenomas. Best Pract Res Clin Endocrinol Metab 23(5): 597-606.
- Amlashi FG, Tritos NA (2016) Thyrotropin-secreting pituitary adenomas: epidemiology, diagnosis, and management. Endocrine 52(3): 427-440.
- Sanno N, Teramoto A, Osamura RY (2001) Thyrotropin-secreting pituitary adenomas. Clinical and biological heterogeneity and current treatment. J Neurooncol 54(2): 179-186.
- Valdes-Socin H, Chanson P, Delemer B, Tabarin A, Rohmer V, et al. (2003) The changing spectrum of TSH-secreting pituitary adenomas: diagnosis and management in 43 patients. Eur J Endocrinol 148(4): 433-442.
- Ness-Abramof R, Ishay A, Harel G, Sylvetzky N, Baron E, et al. (2007) TSH-secreting pituitary adenomas: follow-up of 11 cases and review of the literature. Pituitary 10(3): 307-310.
- Refetoff S, Weiss RE, Usala SJ (1993) The syndromes of resistance to thyroid hormone. Endocr Rev 14(3): 348-399.
- Rabbiosi S, Peroni E, Tronconi GM, Chiumello G, Losa M, et al. (2012) Asymptomatic thyrotropin-secreting pituitary macroadenoma in a 13-year-old girl: successful first-line treatment with somatostatin analogs. Thyroid 22(10): 1076-1079.
- Brucker-Davis F, Oldfield EH, Skarulis MC, Doppman JL, Weintraub BD (1999) Thyrotropin-secreting pituitary tumors: diagnostic criteria, thyroid hormone sensivity, and treatment outcome in 25 patients followed at the National Institutes of Health. J Clin Endocrinol Metab 84(2): 476-486
- Korn EA, Gaich G, Brines M, Carpenter TO (1994) Thyrotropin-secreting adenoma in an adolescent girl without increased serum thyrotropin-alpha. Horm Res 42: 120-123.
- Nyhan WL, Green M (1964) Hyperthyroidism in a patient with a pituitary adenoma. J Pediatr 65: 583-589.
- Suntornlohanakul S, Vasiknanont P, Mo-Suwan L, Phuenpathom N, Chongchitnant N (1900) TSH secreting pituitary adenoma in children: a case report. J Med Assoc Thai 73(3): 175-178.
- Polak M, Bertherat J, Li JY, Kujas M, Le Dafniet M, et al. (1991) A human TSH-secreting adenoma: endocrine, biochemical and morphological studies. Evidence of somatostatin receptors by using quantitative autoradiography. Clinical and biological improvement by SMS 201-995 treatment. Acta Endocrinol (Copenh) 124(4): 479-486.
- Avramides A, Karapiperis A, Triantafyllidou E, Vayas S, Moshidou A, et al. (1992) TSH-secreting pituitary macroadenoma in an 11-year-old girl. Acta Paediatr 81(12): 1058-1060.
- Phillip M, Hershkovitz E, Kornmehl P, Cohen A, Leiberman E (1995) Thyrotropin secreting pituitary adenoma associated with hypopituitarism and diabetes insipidus in an adolescent boy. J Pediatr Endocrinol Metab 8(1): 47-50.
- Gannage MH, Maacaron C, Okais N, Halaby G (1997) Adenome hypophysaire a TSH. A propos d'un cas. J Med Liban 45: 97-101.
- Safer JD, Colan SD, Fraser LM, Wondisford FE (2001) A pituitary tumor in a patient with thyroid hormone resistance: a diagnostic dilemma. Thyroid 11(3): 281-291.
- Kessler M, David R, Pawelczak M, Hanono A, Shah B (2010) Thyrotropin-secreting pituitary adenoma in an adolescent boy: challenges in management. Pediatrics 126(2): e474-478.
- Nakayama Y, Jinguji S, Kumakura S, Nagasaki K, Natsumeda M, et al. (2012) Thyroid-stimulating hormone (thyrotropin)-secretion pituitary adenoma in an 8-year-old boy: case report. Pituitary 15(1): 110-115.
- Mazerkina N, Trunin Y, Gorelyshev S, Golanov A, Kadashev B, et al. (2016) Thyrotropin-secreting pituitary adenoma in an 11-year-old boy with type 1 autoimmune polyglandular syndrome. J Pediatr Endocrinol Metab 29(2): 237-240.
- Sarlis NJ, Gourgiotis L, Koch CA, Skarulis MC, Brucker-Davis F, et al. (2003) MR imaging features of thyrotropin-secreting pituitary adenomas at initial presentation. AJR Am J Roentgenol 181(2): 577-582.
- Beckers A, Abs R, Mahler C, Vandalem JL, Pirens G, et al. (1991) Thyrotropin-secreting pituitary adenomas: report of seven cases. J Clin Endocrinol Metab 72(2): 477-483.






























