Diagnostic Value of High Resolution Neckultrasongraghy, Fine Needle Aspiration Cytology and BRAFV600E Mutation in Diagnosis of Malignant Thyroid Nodules
Marwa Sayed1*, Marwa EL Sharkawy2, Hussein Okasha1, Husam Hussien3, Randa F Salam1, Waled Al Nabaoy4, Marianne Samir Makboul2, Usama AA Sharaf El Din1 and Mona Mansour Salem1
1Internal medicine Department, Cairo University, Egypt
2Clinical and Chemical Pathology Department, Cairo University, Egypt
3Pathology Department, Cairo University, Egypt
4Internal medicine department, Beny Sweef University, Egypt
Submission: April 24, 2017; Published: May 30, 2017
*Corresponding author: Marwa Sayed, MD, Department of internal medicine, Faculty of medicine, Cairo University, Egypt, Tel: 00201117313353; Email: meramar_s@hotmail.com
How to cite this article: Marwa S, Marwa EL S, Hussein O, Husam H. Diagnostic Value of High Resolution Neckultrasongraghy, Fine Needle Aspiration Cytology and BRAFV600E Mutation in Diagnosis of Malignant Thyroid Nodules. J Endocrinol Thyroid Res. 2017; 2(1): 555580. DOI:10.19080/JETR.2017.02.555580
Abstract
Background: Thyroid malignancy is rapidly raising, nearly 9-15% of the thyroid nodules are malignant nodules. Finding an optimal mean to diagnose malignant thyroid nodule preoperative is a challenge. Combination of molecular analysis, imaging and cytopathology may be helpful. This study aimed to evaluate the value of adding BRAFV600E analysis to nodules with suspicious ultra-sonographic criteria.
Patients and methodology: 50 patients from Kasr alainy endocrine outpatient clinic with solitary or multiple thyroid nodules are included, All patients subjected to full history and examination, thyroid profile, high resolution neck High resolution neck ultrasound (U/S), Fine needle Aspiration Cytology (FNAC), U/S guided FNAC and BRAFV600E analysis on FNAC using DNA sequencing then high resolution melt curve analysis (HRMA) for confirmation.
Results: The incidence of BRAFV600E mutation among papillary thyroid cancer (PTC) patients was 55.6%. it’s sensitivity 42.9%, specificity was 100% in diagnosing PTC. Sensitivity of high resolution neck ultrasonograghy in detecting malignancy was 88.2%, specificity 72.7%. Addition of ultrasonography to BRAFV600E analysis increased ultrasound sensitivity in detecting PTC preoperative to 92%. There was a positive correlation between most suspicious ultrasonography findings and presence of BRAFV600E mutation (increased AP/T diameter, Increase intra-nodular blood flow, cervical lymphadenopathy, absent or incomplete halo (all p value<0.001), irregular border p value 0.004, micro calcifications p value 0.007.
Conclusion: Adding BRAF-V600E analysis to work up of suspicious thyroid nodule would increase the sensitivity of preoperative diagnosis of PTC especially in cases of indefinite pathological findings. HRMA is simple, low cost tool for BRAF-V600E analysis in comparison to DNA sequencing method.
Keywords:Thyroid nodule; Thyroid cancer; High resolution neck ultrasound (U/S); Fine needle aspiration cytology (FNAC); BRAFV600E mutation
Abbreviation:(U/S): High Resolution Neck Ultrasound; FNAC: Fine Needle Aspiration Cytology, PTC: Papillary Thyroid Cancer; HRMA: High Resolution Melt Curve Analysis
Introduction
Thyroid nodules are a common problem their prevalence reaching 35% by ultrasound examination in some studies [1] and 8-65% in autopsy data [2] There is an increasing rate ofthyroid cancer incidence worldwide, There is an increasing rate of thyroid cancer incidence worldwide. 9~15 % of all thyroid nodules are malignant [3,4]. Accordingly the initial evaluation of thyroid nodule should focus on exclusion of malignancy [3].
High resolution U/S is the most sensitive diagnostic imagingtechnique for the detection of the thyroid nodules [4], it cancharacterize suspicious nodules by some sonographic [5], but itdepends on the skills and accuracy of the operator [6].
FNAC is the most important step in the work up of thethyroid nodule; Diagnostic accuracy has improved using USneedle localization due to a decreased number of inadequatespecimens and false negative results [7]. The pathology reportfrom FNAB according to Bethesda may be read as non-diagnostic,benign, indeterminate lesion (atypia or follicular lesion ofundetermined significance), Follicular neoplasm or suspiciousfor follicular neoplasm, Suspicious for malignancy and malignant[8,9]. Benign lesions on FNAB have an approximate 3% risk ofmalignancy (vary with patient population), and may be followedclinically with ultrasound or with a repeat FNAB [9], which, ifalso benign, decreases the risk of a false negative to 1.3% [10],the only malignant pathology reliably diagnosed through FNABis papillary thyroid carcinoma, as features such as ‘OrphanAnnie’ nuclei, nuclear grooves, intra-nuclear inclusions, andpsammoma bodies can be sufficient for a diagnosis [11]. Benignand malignant follicular neoplasms and oncocytic (Hurthle cell)adenomas and carcinomas cannot be distinguished on the basisof cytology alone, as tissue architecture is required to make thediagnosis of malignancy through observation of capsular orangio-lymphatic invasion [12].
Indeterminate cytology findings vary from 15% to 25%[13]. Indeterminate FNAC results and cyto-diagnostic errors areunavoidable due to overlapping cytological features particularlyamong hyperplastic adenomatous nodules, follicular neoplasmsand follicular variants of papillary carcinoma [14]. Thecorrelation of U/S features with FNAB results helps to overcomethe limitations of FNAB alone [15] even when cytological resultswere the same, the malignancy rate was higher when noduleshad suspicious U/S findings [16], So, U/S features and FNABresults are complementary to each other [17].
From that comes the importance of a new diagnostic toolto solve the problem, to overcome the limitations of FNA and toavoid unnecessary surgery. Several molecular factors have beenadded to improve the diagnostic accuracy of U/S-guided FNAB[2,18] one of the promising genetic factors is the analysis of theV600E mutation [19,20].
Aim of the Work
To detect an accurate and useful tool for early preoperativedetermination of thyroid malignancy in the patients with thyroidnodule and assess its value in addition to the previous modalities(ultrasonography, and FNAC) that may increase the diagnosticaccuracy and prevent unnecessary surgery
Patients and Methods
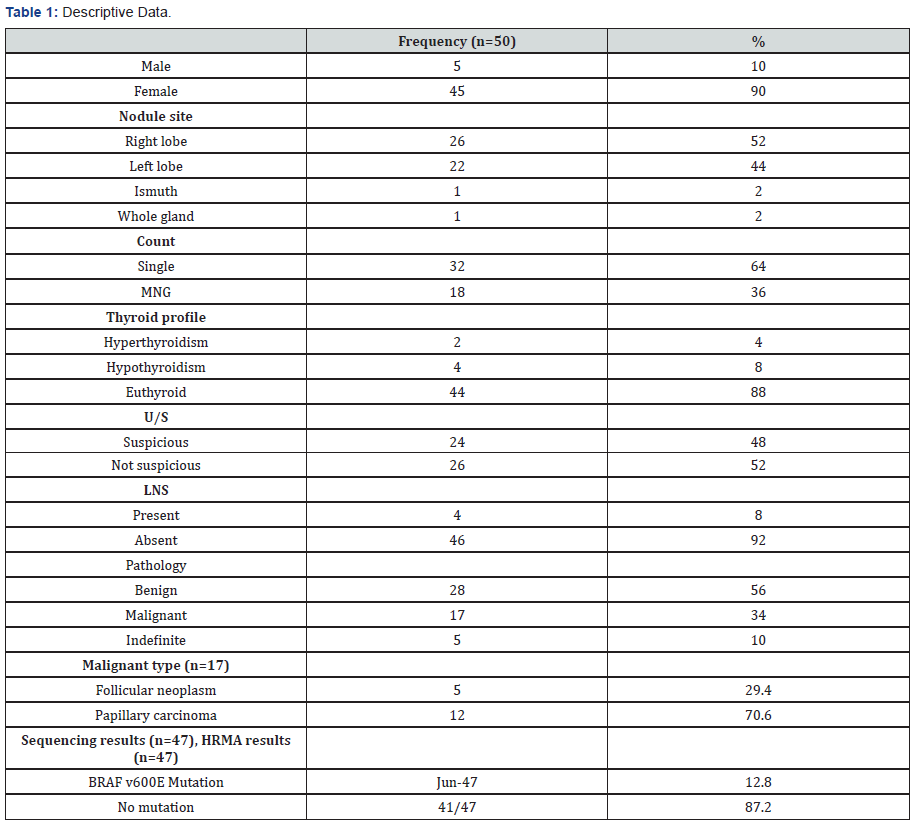
This single group comparative descriptive study included50 patients from Kasr Alainy endocrine outpatient clinic, CairoUniversity.The thyroid examined contained either solitaryor multiple nodules. Four patients with four nodules werehypothyroid (8%), and two nodules were in hyperthyroidpatients (4%), and the remaining part 44 nodules were ineuthyroid patients (88%).18 examined nodules were solitarynodule and 32 were a nodule in MNG. Nodules’ size ranges from0.6-28mm3, Mean 6.6mm3±5.8mm3 SD, Median 6.0mm3. Patients’ages ranged from 22-65 years old median 43.0±9.6 SD, Median43.5. five males (10%) and 45 females (90%). Demographic dataare included in Table 1.
All patients were subjected to
- Full clinical history including age, sex, past historyof head and neck irradiation and positive family history ofthyroid cancer.
- Thyroid examination.
- Thyroid profile (FT3, FT4, and TSH).
- High resolution neck ultrasound.
- U/S guided FNAC.
- BRAFV600E analysis.
Ethical considerations
All patients were informed about these types of investigationsand for confidentiality their names were omitted and replacedby numerical codes.
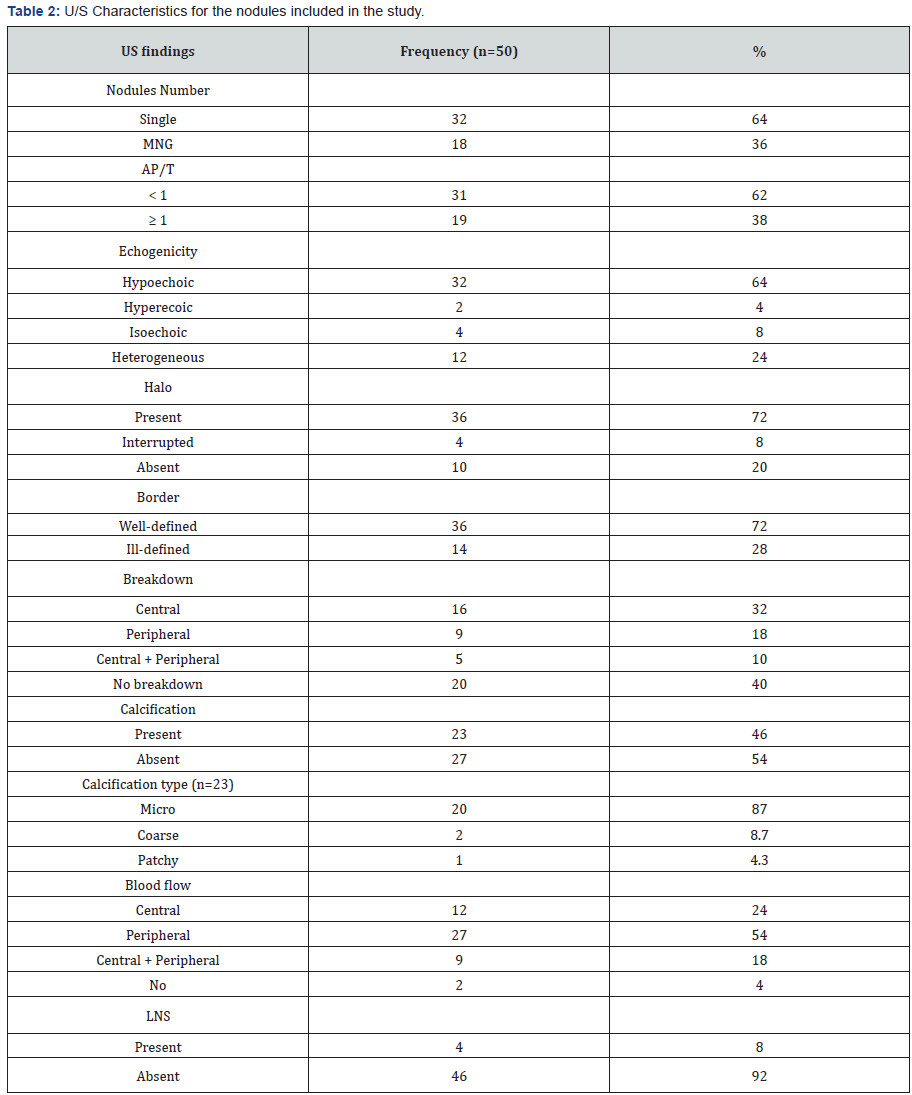
Sonographic criteria for thyroid nodules include
Echogenicity, Presence or absence of calcification and itstypes, A-P to Transverse diameter ratio, Blood flow in the noduleand its patterns whether central or peripheral or both, Solitaryor dominant nodules, Presence or absence of halo, Solid, cystic orpartially solid and partially cystic, Sharpness of border.
Signs of increase risk of malignancy in U/S include
- Hypo-echogenicity. Micro- or interrupted rimcalcifications, Irregular Margins.
- Absence of Halo or incomplete halo.
- Increased Intra-nodular flow.
- Increase antro- posterior to transverse diameter(AP/T) diameter.
- Significant increase in size over time.
- Invasion of anterior strap muscles
- Presence of abnormal cervical lymphadenopathy.
All cytology specimens were obtained under ultrasoundguidance using a 21-gauge needle attached to a plastic syringe,part of the smear obtained were wet fixed in 95% ethyl alcoholand stained with hematoxylin and eosin stain for routinecytological evaluation, and the other part put in test tube withsaline for genetic examination.
Molecular genetic testing
Specimen collection: Samples were collected from eachsubject in sterile containers for the genotyping technique.Samples were either processed fresh or were stored at 2-8 °C.
DNA extraction: This was performed using ThermoScientific Gene JET Whole Blood Genomic DNA Purification MiniKit supplied by Thermo Scientific Fermentas. The extractedDNA then subjected to the algorithm for molecular detectionof BRAFV600E mutation. The aim of the current study is thedetection of the most common mutation in the BRAF gene.V600E in DNA samples collected from thyroid nodules.
We used DNA sequencing method for BRAF V600E mutationdetection, unfortunately all samples were BRAF V600E mutationnegative, so we tried more accurate method (HRMA), then DNAsequencing for validation of the mutant BRAF results had done.
The extracted DNA were conducted to the work asfollows
Extracted DNA DNA sequencing of exon 15 HRMA DNAsequencing for validation of the mutant BRAF results
DNA amplification using the polymerase chainreaction
DNA amplification using the PCR: Enzymatic amplificationwas performed by PCR using HotStarTaq® Master Mix Kit (250units) supplied by QIAGEN® and BIO RAD T100TM ThermalCycler*.
Detection of PCR amplification products: Fluorescentstained DNA was detected by using Agarose Gel Electrophoresisand Ultra-Violet Light Trans-illumination.
Purification of PCR product: This was performed usingQIAquick® PCR Purification Kit supplied by QIAGEN® (Table 2).
Sequencing coding sequence and intronic boundaries ofExon 2 and Exon 3 in INS gene Cycle Sequencing: Using theApplied Biosystems* (ABI) PRISM® BigDye® Cycle SequencingReady Reaction Kit.
Second purification: Removal of excess Dye DeoxyTMterminators from sequencing reactions by using Centri-Sepcolumns (Princeton separations) Terminator v3.1 CompletedDNA long Read capillary electrophoresis on the ABI 3500.
Analysis of data
Sequences were compared to the published Referencesequence of Homo sapiens (BRAF), NCBI (National Centre ofBiotechnology Information): (NG_007873.3, NM_004333.4,and NP_004324.2) Analysis was done by BLAST (Basic LocalAlignment Search Tool) (www.ncbi.nlm.nih.gov) and the CLCBIOsequence viewer 6 program (www.clcbio.com ).
High resolution melt curve analysis: The HRMA reactionwas done according to the protocol of the kit MeltDoctor TMHRM Master Mix Applied Biosystems, USA.
DNA sequencing for validation of the BRAF mutantresults: The samples with mutant results were processed afterHRM analysis by pipetting the reaction from the reaction tubesfollowed by purification of the products.
Statistical analysis: Pre-coded data was entered on thecomputer using “Microsoft Office Excel Software” program(2010) for windows. Data was then transferred to the StatisticalPackage of Social Science Software program, version 21 (SPSS) tobe statistically analyzed.
Data was summarized using range, mean, standard deviationand median for quantitative variables and frequency andpercentage for qualitative ones. Comparison between groups wasperformed using independent sample t-test or Mann Whitneytest for quantitative variables and Chi square or Fisher’s exacttest for qualitative ones. P values less than 0.05 were consideredstatistically significant, and less than 0.01 were consideredhighly significant.
Results
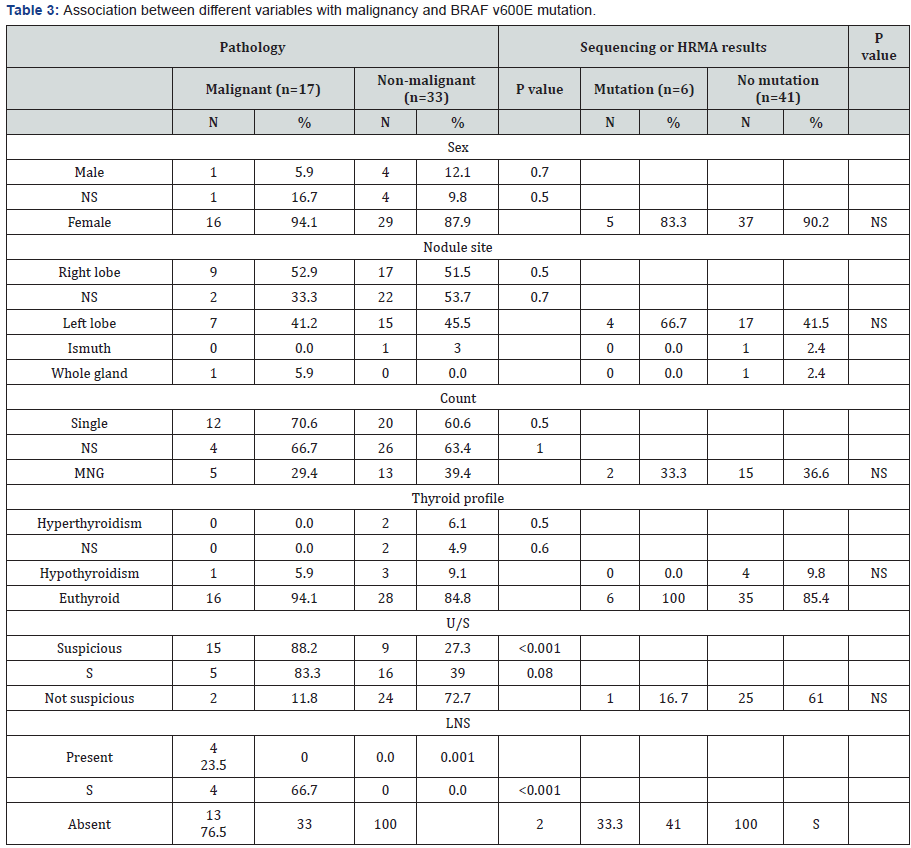
There is no statistically significant association betweendifferent sex, nodular site, count and thyroid functions withmalignancy or BRAFv600E gene mutation (Table 3). Pathologyresults showed 28 benign nodules, 17 malignant nodule (12papillary -5 follicular neoplasm) and 5 cases with indefinitenodules. The incidence of BRAF V600E gene mutation in ourpatients with PTC was 55.6%
50 nodules were included but unfortunately because ofrepetition of genetic analysis for BRAF V600E gene mutations byDNA sequencing then again by HRMA methods that leads to that3 samples were not enough to complete genetic analysis so BRAFV600E analysis completed only in 47 samples.
We found a positive correlation with significant p valuebetween some suspicious sonographic criteria and malignancyand some suspicious sonographic criteria and BRAF V600Emutation as increased (AP/T) diameter, absent or interruptedhalo, micro-calcifications, increase intra-nodular blood flow,presence of suspicious LNs and irregular border with increasedboth sensitivity and specificity (Table 4 & 5).
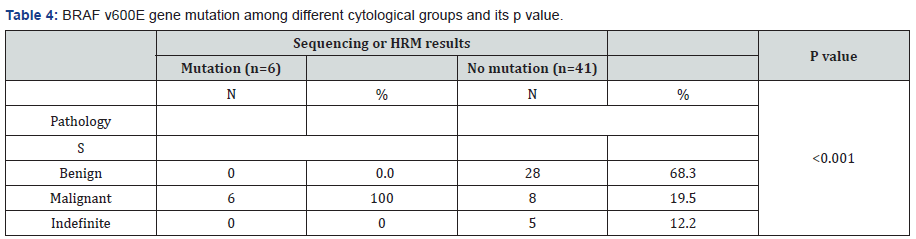
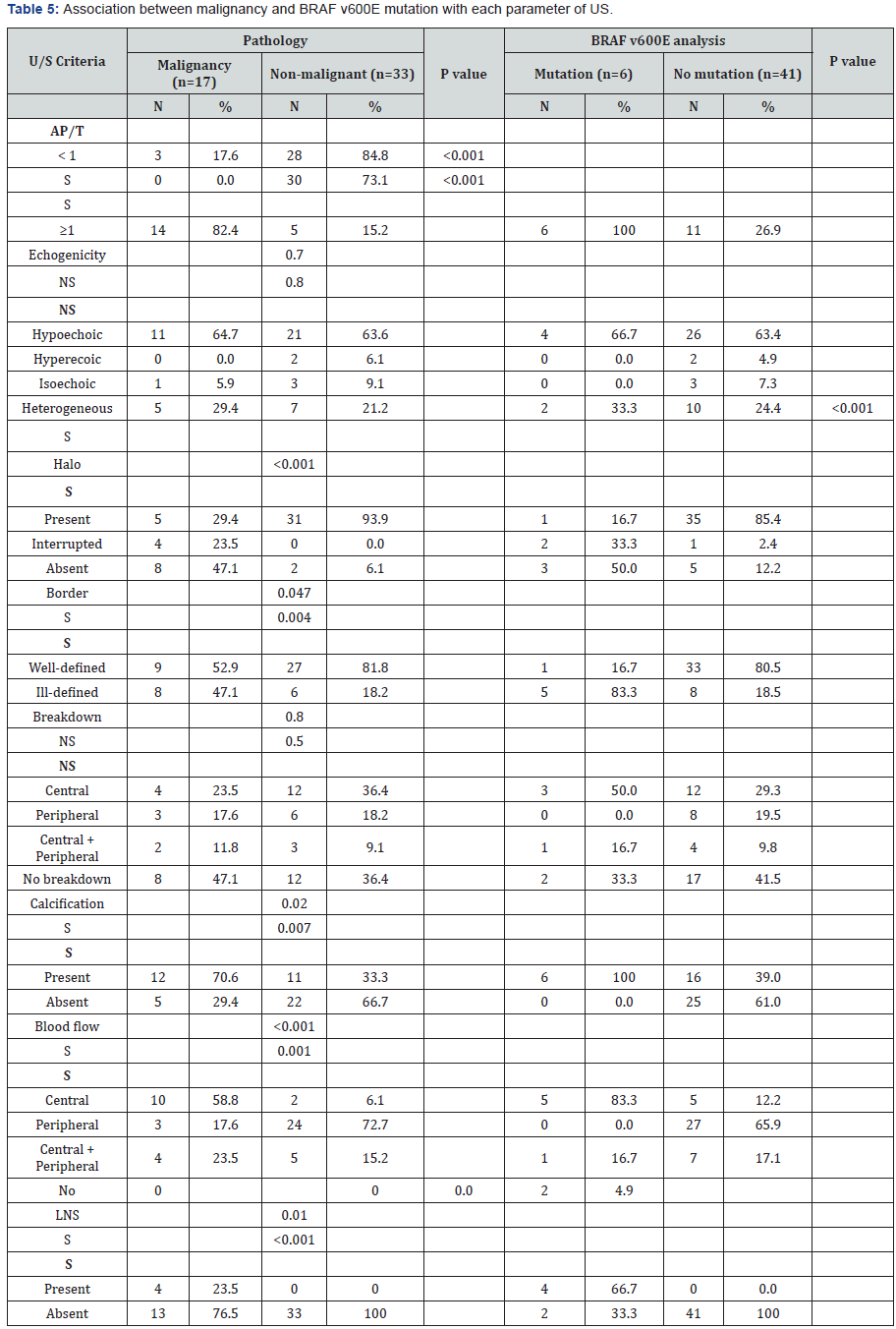
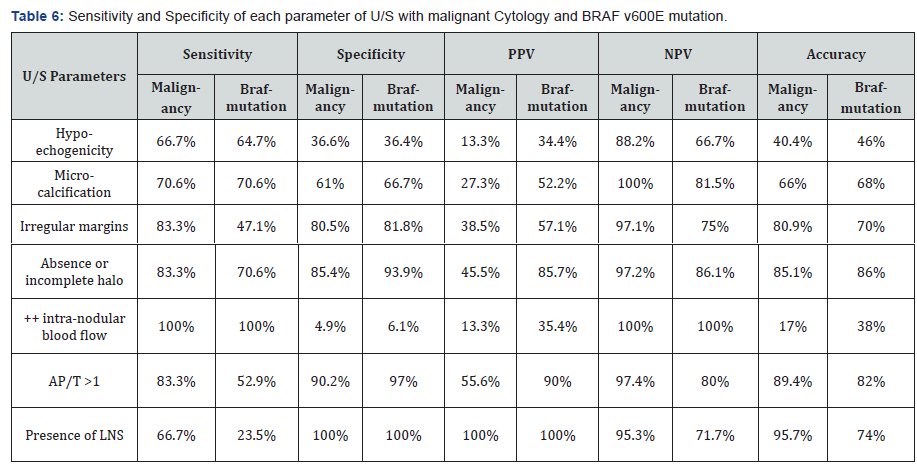
Six malignant nodules were found to harbor BRAF V600Egene mutation, five of them were papillary thyroid carcinomaand one was follicular neoplasm by FNAC with P value 0.001 andnothing was found in benign nodules so there was significantcorrelation between BRAF V600E gene mutation and papillarythyroid cancer. Overall, sensitivity of ultrasound in detectingmalignancy was found to be 88.2%, specificity 72.7%, Sensitivityof US and BRAF V600E mutation combined was 92% (Table 6).
Discussion
It is noticed that thyroid nodules more common finding infemales more than males, Framingham survey for thyroid nodulesshows that prevalence in females 6.4% and 1.5% in males, andthe Whickham study displays (6.6:1 ratio females /males)[21], This might explain why most of the patients in our studywere females representing 90%. The incidence of malignancyin our study was 34% this result is biased because 12 nodulesdiagnosed as PTC are chosen retrograde after diagnosis of PTCto assess BRAF v600E mutation status.
The incidence of BRAF-V600E gene mutation in our patientswith PTC was 55.6%; previous meta-analyses have publishedvalues for the overall prevalence of the BRAF-V600E mutationin PTC ranging from 29 to 83%. [10,22,23], this wide rangemay be due to variations in PTC subtype, subjects’ geographicalbackgrounds, and research methodology.
No difference in BRAFV600E gene mutation incidencebetween different age groups and this shows agreement withAh Young Park et al. [24], but regarding our result of absenceof significant association of BRAFV600E gene mutation withspecial sex that shows disagreement with Ah Young Park et al.study that shows increase mutation in male sex, this may be dueto the small number of cases involved in our study, also 90% ofour cases are females that cannot reflect the actual prevalence[24].
Our study contained five nodules (10%) with undeterminedcytology, four of them have suspicious US features and all werenegative regarding BRAF-V600E gene mutation, we can’t excludemalignancy in these cases by absence of BRAF-V600E genemutation alone as BRAF-V600E mutation is found only in nearly40% of PTC cases [25].
The presence of BRAF-V600E mutation in one case offollicular neoplasm out of five cases shows agreement with onlyone case report Pennelli et al. [26], this conflicting result maybe due to inadequate cytological diagnosis and interpretationas follicular variant of papillary cancer may be interpreted as amicro follicular lesion in FNA [27], so this case need follow upand pathological confirmation post thyroidectomy to make sureof this result.
In this present study BRAF-V600E mutation was negativein benign cases, this result shows agreement with other studies[10,11] and BRAF-V600E mutation is believed to be specific forPTC because no report has been issued of this mutation in benignnodules [28].
The sensitivity of BRAF-V600E gene mutation in detectingPTC was 42.9%, specificity was 100%, positive predictive value100%, and accuracy 83%, this result shows agreement with Kimet al that shows BRAF-V600E mutation alone has a specificityof 100% and a positive predictive value of 100% for diagnosingmalignancy in PTC [9,29].
Nam et al. [30] reported significant improvements in thesensitivity, negative predictive value, and diagnostic accuracy ofFNA cytology for diagnosing malignancy by adding BRAF-V600Eanalysis to both US and FNAC [30].
Several studies have found a positive correlation betweenthe BRAF-V600E mutation and suspicious sonographic featuresof thyroid nodules [7], other studies show inconsistent results;Kwak, et al. found that only marked hypo echogenicity wasassociated with BRAF-V600E positivity, Hwang et al. found thatonly calcification was associated with BRAF-V600E positivity[29,31] both these studies predominantly evaluated smallpapillary thyroid micro carcinomas, with mean tumor sizesof 6-9mm3, the small size was likely a significant confoundingfactor in characterizing these lesions by preoperative US.
Our study shows positive correlation between mostsuspicious US findings and BRAF-V600E gene mutation andshows that AP/T≥1, absent or incomplete halo, irregular border,micro-calcifications and Increase intra-nodular blood flow havepositive correlation with positivity of BRAF-V600E mutationthese results show agreement with Kabaker et al. [32] thatshowed association of BRAF-V600E positivity with most knownsuspicious U/S finding including taller than wide shape, illdefinedmargin, hypo-echogenicity, micro-calcifications andabsent halo (p value<0.001) [32].
This study also showed that BRAF-V600E gene mutation issignificantly present in cases with cervical lymphadenopathythis result shows agreement with many studies that correlateBRAFV600E gene mutation with the frequency of lymph nodemetastases [10,22] Park et al. [24] study concluded that theBRAF-V600E mutation was associated extra-thyroidal extension,central and lateral lymph node metastasis, and advanced tumorstage (P<0.0001, so BRAFV600E analysis preoperative may be ofvalue as prognostic marker and affect surgical decision regardingtype of surgery.
DNA sequencing method shows high cost, time consumingand low sensitivity for the clinical screening of BRAF V600Emutation and this shows agreement with other study [33] thatshows accuracy of DNA sequencing is low when dealing withcytological specimens where the cells in question may be aminor population among the vast majority of background nonneoplasticcells, Furthermore, this method needs expensiveequipment which may not be economical for all the patients[34], In contrast HRMA has been proved to be applicable, costefficientand very sensitive scanning method that allows rapiddetection of DNA sequence variations without cumbersomepost–polymerase chain reaction (PCR) methods, which is notachievable by direct sequencing [35]. Because targeted therapyfor thyroid cancers with multikinase inhibitors is under activedevelopment [36,37], the detection of mutations in the FNACmaterial may be helpful in the future to guide mutation-specifictargeted therapies that can be initiated preoperatively or inthose patients who are not surgical candidates. This study hassome limitations as small number of patients included, somegenetic mutations should be included as RAS and RET and RET/PTC rearrangement.
Conclusion
BRAFV600E positivity was associated with most knownsuspicious U/S finding, BRAFV600E gene mutation analysisis a new hopeful diagnostic and prognostic tool that helps toovercome limitations of high resolution neck ultrasonographyand fine needle aspiration biopsy, Adding BRAF-V600E analysisto U/S and FNA would increase the sensitivity of preoperativediagnosis of PTC especially in cases of indefinite nodules. HRMAis an excellent, simple, low cost tool for BRAF-V600E analysis incomparison to DNA sequencing method.
Acknowledgement
I’d like to Knowledge Dr. Marwa Abdel Naser (pathologist), Dr.Mohamed Mostafa for statistical support and also my institute.
Financial Disclosure
Fund and sponsorship for this research from Cairo University.
References
- Frates MC, Benson CB, Charboneau JW, Cibas ES, Clark OH, et al.(2005) Management of thyroid nodules detected at US: society ofradiologists in ultrasound consensus conference statement. Radiology237(3): 794-800.
- Yip L, Farris C, Kabaker AS, Hodak SP, Nikiforova MN, et al. (2012) Costimpact of molecular testing for indeterminate thyroid nodule fineneedleaspiration biopsies. The J Clin Endocrinol Metab 97(6): 1905-1912.
- Alam T, Khattak YJ, Beg M, Raouf A, Azeemuddin M, et al. (2013)Diagnostic accuracy of ultrasonography in differentiating benign andmalignant thyroid nodules using fine needle aspiration cytology as thereference standard. Asian Pac J Cancer Prev 15(22): 10039-10043.
- Doddi S, Chohda E, Maghsoudi S, Sheehan L, Sinha A, et al. (2015) Thefinal outcome of indeterminate cytology of thyroid nodules in a DistrictGeneral Hospital. G Chir 36(3): 122.
- Kim TY, Kim WB, Rhee YS, Song JY, Kim JM, et al. (2006) The BRAFmutation is useful for prediction of clinical recurrence in low‐riskpatients with conventional papillary thyroid carcinoma. Clinicalendocrinology (Oxf) 65(3): 364-368.
- Feng L, Li M, Zhang QP, Piao ZA, Wang ZH, et al. (2011) Utility of BRAFprotein overexpression in predicting the metastasis potential ofpapillary thyroid carcinoma. Oncol Lett 2(1): 59-63.
- Lee EJ, Song KH, Kim DL, Jang YM, Hwang TS, et al. (2011) TheBRAFV600E mutation is associated with malignant ultrasonographicfeatures in thyroid nodules. Clin Endocrinol (Oxf) 75(6): 844-850.
- Liu X, Medici M, Kwong N, Angell TE, Marqusee E, et al. (2016) Bethesdacategorization of thyroid nodule cytology and prediction of thyroidcancer type and prognosis. Thyroid 26(2): 256-261.
- Gharib H, Papini E, Garber JR, Duick DS, Harrell RM, et al. (2016)American Association of Clinical Endocrinologists, American CollegeOf Endocrinology, and Associazione Medici Endocrinologi MedicalGuidelines For Clinical Practice For The Diagnosis and Management ofThyroid Nodules-2016 Update. Endocr Pract 22(5): 622-639.
- Xing M (2005) BRAF mutation in thyroid cancer. Endocr Relat Cancer12(2): 245-262.
- Xing M (2007) BRAF mutation in papillary thyroid cancer: pathogenicrole, molecular bases, and clinical implications. Endocr Rev 28(7): 742-762.
- Vossen RH, Aten E, Roos A, den Dunnen JT (2009) High‐ResolutionMelting Analysis (HRMA)-More than just sequence variant screening.Hum Mutat 30(6): 860-866.
- Yoon JH, Kwak JY, Moon HJ, Kim MJ, Kim EK (2011) The diagnosticaccuracy of ultrasound-guided fine-needle aspiration biopsy andthe sonographic differences between benign and malignant thyroidnodules 3cm or larger. Thyroid 21(9): 993-1000.
- Moon HJ, Kwak JY, Kim EK, Kim MJ (2011) A taller-than-wide shapein thyroid nodules in transverse and longitudinal ultrasonographicplanes and the prediction of malignancy. Thyroid 21(11): 1249-1253.
- Moses W, Weng J, Sansano I, Peng M, Khanafshar E, et al. (2010)Molecular testing for somatic mutations improves the accuracy ofthyroid fine-needle aspiration biopsy. World J Surg 34(11): 2589-2594.
- Yoo MR, Gweon HM, Park AY, Cho KE, Kim JA, et al. (2015) RepeatDiagnoses of Bethesda Category III Thyroid Nodules: What To Do Next?PloS one 10(6): e0130138.
- Nikiforov YE, Nikiforova MN (2011) Molecular genetics and diagnosisof thyroid cancer. Nat Rev Endocrinol 7(10): 569-580.
- D’Souza MM, Marwaha RK, Sharma R, Jaimini A, Thomas S, et al. (2010)Prospective evaluation of solitary thyroid nodule on 18F-FDG PET/CTand high-resolution ultrasonography. Ann Nucl Med 24(5): 345-355.
- Xing M (2010) Prognostic utility of BRAF mutation in papillary thyroidcancer. Mol Cell Endocrinol 321(1): 86-93.
- Xing M (2016) Diagnostic and Prognostic Molecular Markers inThyroid Cancer. Thyroid Cancer pp. 281-292.
- Vander JB, Gaston EA, Dawber TR (1968) The significance of nontoxicthyroid nodules: final report of a 15-year study of the incidence ofthyroid malignancy. Ann Intern Med 69(3): 537-540.
- Lee JH, Lee ES, Kim YS (2007) Clinicopathologic significance of BRAFV600E mutation in papillary carcinomas of the thyroid. Cancer 110(1):38-46.
- Witt RL, Ferris RL, Pribitkin EA, Sherman SI, Steward DL, et al. (2013)Diagnosis and management of differentiated thyroid cancer usingmolecular biology. Laryngoscope 123(4): 1059-1064.
- Park AY, Son EJ, Kim JA, Youk JH, Park YJ, et al. (2014) Associationsof the BRAF V600E Mutation with Sonographic Features andClinicopathologic Characteristics in a Large Population withConventional Papillary Thyroid Carcinoma. PloS one 9(10): e110868.
- Fugazzola L, Puxeddu E, Avenia N, Romei C, Cirello V, et al. (2006)Correlation between B-RAFV600E mutation and clinic-pathologicparameters in papillary thyroid carcinoma: data from a multicentricItalian study and review of the literature. Endocr Relat Cancer 13(2):455-464.
- Pennelli G, Vianello F, Barollo S, Pezzani R, Merante Boschin I, et al.(2011) BRAF K601E Mutation in a Patient with a Follicular ThyroidCarcinoma. Thyroid 21(12): 1393-1396.
- Jain M, Khan A, Patwardhan N, Reale F, Safran M (2001) Follicular variantof papillary thyroid carcinoma: a comparative study of histopathologicfeatures and cytology results in 141 patients. Endocrine Practice 7(2)79-84.
- Pizzolanti G, Russo L, Richiusa P, Bronte V, Nuara RB, et al. (2007) Fineneedleaspiration molecular analysis for the diagnosis of papillarythyroid carcinoma through BRAFV600E mutation and RET/PTCrearrangement. Thyroid 17(11): 1109-1115.
- Kim SK, Hwang TS, Yoo YB, Han HS, Kim DL, et al. (2011) Surgicalresults of thyroid nodules according to a management guideline basedon the BRAFV600E mutation status. The J Clin Endocrinol Metab 96(3):658-664.
- Nam SY, Han BK, Ko EY, Kang SS, Hahn SY, et al. (2010) BRAF V600Emutation analysis of thyroid nodules needle aspirates in relation totheir ultrasongraphic classification: a potential guide for selection ofsamples for molecular analysis. Thyroid 20(3): 273-279.
- Jiyoung Hwang, Jung Hee Shin, Boo-Kyung Han, Eun Young Ko, SeokSeon Kang, et al. (2010) Papillary thyroid carcinoma with BRAF V600Emutation: sonographic prediction. American Journal of Roentgenology194(5): W425-W430.
- Kabaker AS, Tublin ME, Nikiforov YE, Armstrong MJ, Hodak SP, et al.(2012) Suspicious ultrasound characteristics predict BRAF V600Epositivepapillary thyroid carcinoma. Thyroid 22(6): 585-589.
- Pichler M, Balic M, Stadelmeyer E, Ausch C, Wild M, et al. (2009)Evaluation of high-resolution melting analysis as a diagnostic tool todetect the BRAF V600E mutation in colorectal tumors. J Mol Diagn11(2): 140-147.
- Ziai J, Hui P (2012) BRAF mutation testing in clinical practice. Expertreview of molecular diagnostics 12(2): 127-138.
- Simi L, Pratesi N, Vignoli M, Sestini R, Cianchi F, et al. (2008) Highresolutionmelting analysis for rapid detection of KRAS, BRAF, andPIK3CA gene mutations in colorectal cancer. Am J Clin Pathol 130(2):247-253
- Espinosa AV, Porchia L, Ringel MD (2007) Targeting BRAF in thyroidcancer. Br J Cancer 96(1): 16-20.
- Fagin JA (2007) The Jeremiah Metzger Lecture: intelligent design ofcancer therapy: trials and tribulations. Trans Am Clin Climatol Assoc118: 253-261.






























