Telephone Survey to Assess Substitution of Electronically Prescribed Branded Levothyroxine: The e-Rx LT4 Study
Jill M Paulson1* and James V Hennessey2
1Division of Endocrinology and Metabolism, George Washington University, USA
2Division of Endocrinology, Diabetes and Metabolism, Beth Israel Deaconess Medical Center, USA
Submission: March 20, 2017; Published: April 19, 2017
*Corresponding author: Jill M Paulson, Division of Endocrinology and Metabolism, George Washington University, Medical Faculty Associates,Washington DC, USA, Email: jmpaulson@mfg.gwu.edu
How to cite this article: Paulson JM, Hennessey JV. Telephone Survey to Assess Substitution of Electronically Prescribed Branded Levothyroxine: The e-Rx LT4 Study. J Endocrinol Thyroid Res. 2017; 1(4): 555569. DOI:10.19080/JETR.2017.01.555569
Abstract
Consistent dosing of thyroid hormone is important given the narrow therapeutic index of the medication. Each levothyroxine preparation is uniquely formulated to provide a consistent vehicle of levothyroxine delivery. Review of each products package insert clearly delineates the variety of excipients used in each brand while the active ingredient is consistently Na-L-thyroxine. The FDA process of determining interchangeability of the available preparations is based upon pharmacokinetic standards which may allow as much as 12.5% difference in the overall absorption of thyroxine depending on the preparation ingested. Because of this, a branded thyroxine replacement is often prescribed to ensure consistency of sourcing. We describe a prospective pilot study of 100 subjects who were prescribed branded thyroxine products through electronic prescribing (e-Rx). Subjects were called three times over seven months to assess the brand and dose of thyroxine therapy they had received at the pharmacy based on markings on the pill. The goal was to see if the pharmacy substituted branded thyroxine products.
Keywords: Hypothyroidism; Levothyroxine therapy; Branded therapy; Drug formulations; Drug Substitution; Electronic prescribing
Introduction
Levothyroxine is a narrow therapeutic index drug [1]. Each levothyroxine preparation is uniquely formulated to provide a consistent vehicle of levothyroxine delivery. Review of each products package insert clearly delineates the variety of excipients used in each brand while the active ingredient is consistently Na-L-thyroxine. The FDA process of determining interchangeability of the available preparations is based upon pharmacokinetic standards which may allow as much as 12.5% difference in the overall absorption of thyroxine depending on the preparation ingested [2]. Consistent dosing is necessary to avoid the potential side effects and morbidity of under- or overdosing [3]. This is of particular importance in high-risk populations including those with advanced age[4], advanced thyroid cancer[5], children[6], and pregnant women [7].
During the active study time, there were four brand-name and four generic levothyroxine preparations on the US market. As the FDA requires the determination of bioequivalence and determines the interchangeability of products considered therapeutically equivalent by pharmacokinetics, no clinical verification of these pairings are accomplished. Importantly, these methods do not take into account TSH measurements as are commonly utilized in the clinical evaluation of hypothyroid patients. Moreover, clinical outcomes are not utilized in the process of determining the equivalence of the various preparations, although clinical experience suggests that substitution of one preparation for another results in adverse clinical events [3,2]. For many years the consistency of LT4 dosing was routinely facilitated by using brand name thyroxine preparations as the L-thyroxine market was unregulated, and each preparation was considered unique [8-11]. With the introduction of FDA regulation in 1997, the quality of individual LT4 products was mandated but standards for assessment of therapeutic equivalence had not been initially determined [12]. There was speculation that introduction of bioequivalence standards would assure safe interchangeability if FDA criteria were met [2]. This would have resulted in a clear justification of a free substitution requirement mandated by most state boards of pharmacy when products were considered therapeutically equivalent. The observation that there are potentially significant differences in absorption between preparations deemed equivalent [13,14] underscores the concept that maintaining sole source use was a reasonable strategy to maintain consistent outcomes. Designating a particular brand to be dispensed with state specific language is a frequently utilized approach to maintain consistent LT4 performance from refill to refill with the intention of optimizing clinical outcomes. Brand name prescribing generally requires a handwritten message to the pharmacist that generic dispensing is not appropriate and consistent refills are expected. With the introduction of electronic prescribing, requirements for designating protection of name brand prescriptions varied from state to state and system to system. Our institution adopted an e-prescribing program which incorporated a check box which clearly marked each protected prescription. Experience with this system was variable over the initial interval of use, including frequent interactions with area pharmacies, inconsistent expectations to document protection and frequent misinterpretation of the physician’s expectation for name brand dispensing. This confusion resulted in generic substitution of the original prescription or subsequent refills, patient concern over the variable appearance of their pills, unnecessary phoning and excessive laboratory testing as prevailing clinical practice guidelines were followed [15]. There has been no study published that investigates the impact of electronic prescribing on dispensation of branded medications when requested.
Methods
We conducted an IRB approved, prospective telephone survey at a single site (academic medical center). We followed 100 subjects over a period of 7 months who were electronically prescribed a branded levothyroxine product. Subjects were recruited from our endocrinology practice. When a patient was seen who was on a brand name levothyroxine, they were handed a flier by the endocrinologist seeing the patient with information about the study and contact information. The endocrinologist emailed the patient information to the study team. Charts were reviewed to confirm that the patient did receive an appropriately annotated, electronically prescribed levothyroxine brandedprescription at the time of the visit. Additionally, we confirmed this prescription had been sent to the pharmacy (e-Rx) and verified transmission.
Three telephone interviews were conducted with each enrolled subject over a 7-month period to assess dispensation (Figure 1):
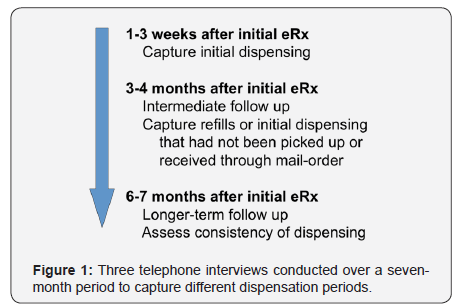
- One to three weeks after initial e-prescribing of the branded levothyroxine to assess what had been initially dispensed.
- Three to four months after the initial e-prescribing of branded levothyroxine to capture the nature of early refills.
- Six to seven months after the initial e-prescribing to evaluate the pattern of chronic therapy.
During each call, subjects were asked to describe the pill (shape, color, and letters/numbers on both sides). This description was then compared to a standardized pill identifier to identify manufacturer and which brand of levothyroxine the patient was dispensed.
Results
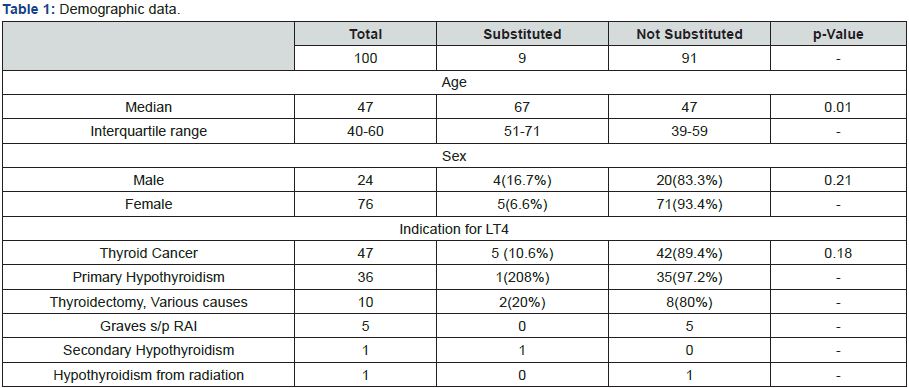
128 patients were called to obtain consent for participation of the planned 100 subjects needed for the study (79% response rate). Demographic data is presented in Table 1. Of the 100 subjects, the median age was 47 years old. 76 of the subjects were female. Indication for being on levothyroxine was obtained through review of the patient’s chart. 47% of the subjects were on levothyroxine for post-surgical hypothyroidism from thyroid cancer, 36% for primary hypothyroidism, 10% for post-surgical hypothyroidism due to non-cancerous cause, 5% for postablative hypothyroidism due to Graves’ disease, 1% for secondary hypothyroidism, and 1% for radiation-induced hypothyroidism. The average time it took for contact after the initial electronic prescription is shown in Table 1. 156 unique new prescriptions were electronically prescribed and dispensed over the studyperiod (86 Synthroid ®, 64 Levoxyl ®, 2 Unithroid ®, 4 Levothroid ®). Note that the study was done prior to Tirosint ® being on the US market.
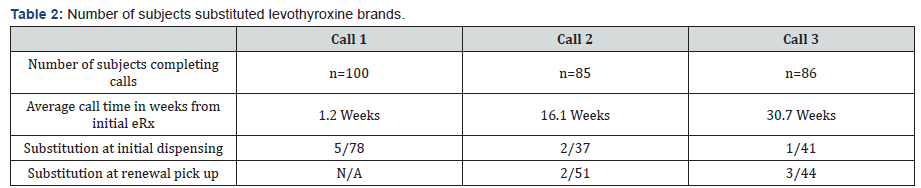
Note: The bottom two rows show how many subjects were substituted per call for both prescriptions that were picked up as a new eRx and those that were refills. Note that the denominator only includes those subjects who had picked up a new eRx or refill since the previous call. Many subjects received a new eRx during the study (e.g. dosage change) and may have had more than opportunity to pick up an initial eRx.
Table 2 shows the breakdown of calls, initial e-Rx pickup and renewal pick up. Note that some subjects did not pick up their initial e-Rx for more than a few weeks or months, using their prior prescription. Other subjects had multiple new e-Rxs throughout the study, either due to dosage change, or due to another reason.
During the first call, which took place between one to three weeks after the electronic prescription was sent to the pharmacy, the subject was asked if they had picked up a new prescription for the branded levothyroxine since they saw their endocrinologist (at the time of the e-Rx). Of the 100 subjects contacted, only 78 had picked up a new prescription of branded levothyroxine since seeing their endocrinologist. Of those 78, five were generically substituted at the initial pick up (6.4%). During the second call, which took place a mean time of 16 weeks after the initial e-Rx for branded levothyroxine, 85 of the 100 total subjects completed calls in the study. Thirty-seven of those subjects had a new e-Rx since the first telephone call resulting in them picking up a new prescription of branded levothyroxine. Two of the 37 (5.4%) were substituted. Additionally, 51 subjects had a renewal prescription pick up, and two of these 51 (3.9%) were also substituted.
During the third call, which took place a mean time of 30.7 weeks after the initial e-Rx of branded levothyroxine, 86 of the 100 subjects completed the call. There were 41 pick-ups of new e-Rxs, and one subject was substituted. There were 44 renewal prescriptions, and there were three substitutions. Because ofthe new e-Rxs that were picked up by review of the chart during our study period, some subjects had the opportunity for their branded levothyroxine to be substituted more than once.
Nine out of the 100 subjects received a generic formulation in lieu of the prescribed branded levothyroxine at the initial filling (Table 2). There was no difference between those substituted and those not substituted in regards to sex and indication (Table 1). Those who were substituted were older than those who were not substituted- 67 years old vs. 47 years old, respectively (p-value 0.01). Out of the 24 subjects who obtained their prescription via mail-order pharmacy, none were generically substituted.
Eight of the 91 subjects that had initial e-Rx that indicated ‘no substitution’ by clicking the ‘no substitution box’ were switched compared to 1/9 e-Rx where the prescriber also wrote additional text to the pharmacy.
Discussion
In this 7-month pilot study involving 100 subjects electronically prescribed brand-name levothyroxine, 9 were generically substituted, some more than once. If the subjects who did not follow up in the telephone calls were accounted for, that number may have been higher. An educational bias was identified as 2 subjects reported that the pharmacy attempted to substitute their prescription; they insisted on brand name prior to purchase. Other subjects may have had similar experiences and not offered that information. In our practices, we teach our patients how to identify branded levothyroxine preparations when they are prescribed. Therefore, due to heightened vigilance by some patients, the substitution rate may appear to be lower than it would have otherwise. This study was underpowered to determine if there was a difference in those substituted based on type of pharmacy.
Conclusion
Substitution of branded levothyroxine products contrary to specific physician orders occurs. Further work with medical boards and pharmacies is required to ensure patients are able to obtain prescriptions as written so that consistency in dosing can be assured.
References
- (1997) Prescription Drug Products; Levothyroxine Sodium, Docket Number 97N-0314. Federal Register 62(157): 43535-43538.
- Hennessey JV (2005) Limitations of Current Bioequivalence Standards. Paper presented at the Equivalence of Levothyroxine Sodium Products Joint Public Meeting, Board Room of the National Transportation Safety Board, L’Enfant Plaza, Washington DC, USA.
- Hennessey JV, Malabanan AO, Haugen BR, Levy EG (2010) Adverse event reporting in patients treated with levothyroxine: results of the pharmacovigilance task force survey of the american thyroid association, american association of clinical endocrinologists, and the endocrine society. Endocr Pract 16(3): 357-370.
- Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, et al. (2012) Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid 22(12): 1200-1235.
- Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, et al. (2016) 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer:The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26(1): 1-133.
- Carswell JM, Gordon JH, Popovsky E, Hale A, Brown RS (2013) Generic and Brand Name L-Thyroxine are not Bioequivalent for Children with Severe Congenital Hypothyroidism. J Clin Endocrinol Metab 98(2): 610- 617.
- Alexander EK, Pearce EN, Brent GA, Brown RS, Chen H, et al. (2017) 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid 27(3): 315-389.
- Sawin CT, Surks MI, London M, Chingleput R, Larsen PR (1984) Oral Thyroxine: Variation in biologic action and tablet content. Ann Intern Med 100(5): 641-645.
- Stoffer SS, Szpunar WE (1988) Potency of Current levothyroxine preparations evaluated by High-Performance Liquid Chromatography. Henry Ford Hosp Med J 36(1): 64-65.
- Hennessey JV, Burman KD, Wartofsky L (1985) The Equivalency of Two L-Thyroxine Preparations. Ann Intern Med 102(6): 770-773.
- Escalante DA, Arem N, Arem R (1995) Assessment of interchangeability of Two Brands of Levothyroxine Preparations with a Third-Generation TSH Assay. Am J Med 98(4): 374-378.
- Hennessey JV (2003) Levothyroxine a New Drug? Since When? How could that be? Thyroid 13(3): 279-282.
- Blakesley VA, Awni W, Locke C, Ludden T, Granneman GR, et al. (2004) Are bioequivalalence studies of levothyroxine sodium formulations in euthyroid volunteers reliable? Thyroid 14(3): 191-200.






























