Has Faetal Whole Faetal Pancreas Grafting Some Perspectives among the Surgical Methods of Diabetes Mellitus Treatment?
Coulic V1* and Novikov VK2
1Translational Medicine Laboratory of the Brussels Free University, Russia
2NV Sklifosovsky Research Institute of Emergency Medicine, Russia
Submission: February 22, 2017; Published: March 23, 2017
*Corresponding author: Coulic V, Translational Medicine Laboratory of the Brussels Free University, Russia, Tel: 32-0-71-364252; Fax: 32-2-4772320; Email: coulic.very@belgacom.net
How to cite this article: Coulic V, Novikov V.Has Faetal Whole Faetal Pancreas Grafting Some Perspectives among the Surgical Methods of Diabetes Mellitus Treatment?. J Endocrinol Thyroid Res. 2017; 1(3): 555562. DOI:10.19080/JETR.2017.01.555562
Abstract
Background: Presently the best results in diabetes mellitus treatment are obtained after pancreas transplantation and bariatric surgery which have not negligible consequences. Implantation of pancreatic adult beta cells, islets, stem cells up to now has only temporary effect.
Material and methods: In experiments on 148 rats (including control series) Streptozotocin® diabetes mellitus was induced and followed by subcutaneous syngeneic foetal whole pancreas syngeneic implantation. In clinics, between 1982 and 1990, 59 patients with type 1(53) and insulin requiring type 2(6) diabetes mellitus have underwear allogeneic implantation of foetal pancreas without immunosuppressive drug use after obtaining their and the foetus’ parents’ informed consent , as well as the agreement of the USSR Ministry of Health Care.
Results: Long lasting correction of Streptozotocin® induced diabetes was obtained in 30% of the rats with syngeneic implantation of foetal pancreas. The best results- in 7 out of 10 animal series were observed when 4 and more foetal pancreas was implanted. In clinics, complete healing of the patients could not be observed, but significant and long lasting (up to 2-10 years, 70% of the cases) improvement of general condition including remission of retinopathy and neuropathy, decrease of glycaemia levels and of the daily delivered insulin amount accompanied by an increase of blood C-peptide were noted in 70% of the humans concerned.
Discussion and conclusion: The positive aspects and difficulties of foetal pancreas implantation are analyzed comparing this experience with literature data. Perspectives of adaptation to contemporary techniques and ethic conditions are suggested.
Keywords:Diabetes mellitus surgical treatment; Pancreas transplantation; Foetal pancreas; Foetal pancreas implantation; Beta cells implantation; Pancreas stem cells endocrine pancreas regeneration
Abbreviations:APD: Animal Protein Deprivation; APDD: Animal Protein Deprivation Diabetes; DM: Diabetes Mellitus; DM1: Type 1 Diabetes Mellitus; DM2: Type 2 Diabetes Mellitus; EEG: Electroencephalogram; FPI: Foetal Pancreas Implantation; Ptx: Adult Pancreas Transplantation
Case Report
In spite of the remarkable development of drugs (insulin’s with various intensity and duration of action; oral antidiabetic preparations of different classes) and devices for their rationale administration up to artificial pancreas [1-18], conservative treatment of diabetes mellitus type 1 as well as type 2, only can significantly improve the condition of the patients and prolong their life, diminishing the complications of the disease, but does not completely cure it. Only surgical approach allows a total and definite healing in most cases pancreas Transplantation for DM1 and bariatric surgery, especially by-pass for DM2 patients [19-26]. However these operations are complex not easily tolerated and followed by severe consequences: immunosuppressivetreatment with its infectious and oncological complications on one side, metabolic disorders of nutriments and vitamin absorption/ assimilation on the other one [20,25,26].
It is the reason why since decades another solution to the problem has been considered: a kind of reduced and targeted transplantation of the endocrine part - cells or islet - implantation: acceptable for treatment of the not too severe DM [27-38]. The first results were promising; but the effect duration was limited; collecting, storage and immunologic (if allo- or xenogeneic) remain. In the 80ties foetal pancreas implantation was studied in animal experiments and even in clinics [39-42]. The results of these studies have shown that foetal implants are able to surviveand develop in adult recipient and even to function. Because of the great 30-year moratorium of the researches on embryonic and foetal material [43-44], this way was practically abandoned.
New trials were started with pancreatic stem cells, first cultivated in vitro and later implanted into the patient organism [45-46]. But cultivated cells have great difficulties to adapt, develop and work in the receptor organism. The aim of the present work is to come back to our own investigations [40,42,47- 55], to analyse their results taking into account more recently obtained experimental data, to confront our conclusions with literature and answer to the question: has the foetal pancreas grafting a place, a perspective in the DM surgical treatment, at what conditions ?
Material and Methods
In this work we have put together for a new analysis animal experiences (part 1) and clinical trial (part 2). Both parts have been the subject of separate and partial publications, to which we refer when necessary.
Part 1
Experiments were carried on 148 rats (Fischer, Lewin and Wistar, males and females, aged 2-4 months at the beginning of the observation).
They were dispatched into the following series:
- Control 1 -5 sham operations - creating a site for implantation without performing the FPI (NB. data obtained during several day observation before the STZ injection were also used as control).
- Control 2 - FPI in healthy animals -10 animals.
- Streptozotocin diabetes - total 59 - without treatment 25 with implantation of foetal pancreas at days 1, 4, 7, 12, 42 after the STZ injection: 34 Animal protein deprivation and foetal pancreas implantation.
- -75 Streptozotocin (75 or 100mg/kg) was injected intra peritoneum. In experimental series the injection was provided 4 days before syngeneic implantation of foetal pancreas (FPI).
Animal protein deprivation (APD) consisted in exclusion from the food any animal protein (milk, cheese, meat) during the female gravidity and the first 4-6 weeks after birth. Drink water and other food such as vegetables and corns were delivered ad libido. At this moment animal protein deprivation was stopped, usual diet was restored and implantation of allogeneic foetal pancreas was provided, in order to imitate the possible human situation in cases of chronic hunger [40] (see also “Diabetes Mellitus” Report of the WHO research group- 1975). NB. For STZ diabetes treatment Syngeneic grafting was adopted in order to test the graft potential capacities without the bias of biological incompatibility. In all the series, donors were rat foetus es collected after 14-18 days of intrauterine development. Recipients were adult rats with a body weight of 200±10g (female rats) or 250±30g (male rats). All the operations wereperformed under general anesthesia (0.1ml of 0.015% Nembutal solution/100g BW injected intra peritoneum).
For implant collect, after extracting a foetus from the mother’s womb the foetal organ was removed and placed in a saline solution. The implantation site was: ear (104 rats), spleen hilum (14 rats). In the first experimental group, the foetal tissue (1 or 2 foetal pancreas were introduced into a subcutaneous pouch of both ear pavilions of the recipient. The skin wound was closed with “No becutane”® spray. In the second group, laparotomy was performed and 3- 8 pancreases were introduced under the peritoneum and the spleen capsule. Abdominal wound was closed by 2-layer running suture with Ethilon 4 °C (Figure 1). After the operation, the recipient was placed in a separate standard cage and fed with a standard maintenance diet in an agreed animal husbandry. Some of them received 1 or more IU of Actrapid® subcutaneously to maintain glycaemia within 250 and 350mg/dl.
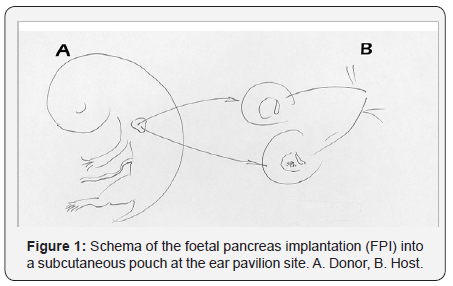
The following investigations were provided: BW, amount of daily drunk water, measure of capillary glycaemia and of glycosuria, proteinuria, ketonuria, haematuria and pH by strips (“Acutrends”, USA). In some cases IGF 1 was determined in blood serum .Observation delays have run up to 9 months. Post-operative animals were euthanized by anesthetic overdose at varying time points (days 2, 4, 7, 14, 58, 80, 120, 397) for morphological investigations of the implant and main organs of the recipient. The material was fixed in 12% formaldehyde, and paraffin inclusion was performed. Subsequently, embedded tissue slices 3-4 microns in thickness were stained with hematoxylin eosin, in some cases determination of insulin, glucagon and somatostatin by immune histochemistry methods, and electron microscopy was performed after glutaraldehyde fixation and araldite embedding. All experiments were conducted in conformity the Helsinki Bioethics Convention and recent experiments were approved by the Brussels Free University Ethics Committee. (Protocol 50)
Part 2
The clinical study was performed in Moscow Hospital N °64 at the Gynecology Unit under the supervision of the head and medical staff of this department, with the agreement of the USRR Public Health Ministry from 1980 till1990 years.Pregnant women, exempt of local infection, tumor, and infectious diseases, with spontaneous or induced for medical reasons early delivery of not viable foetus (aged 14-22 weeks, BW<500g), both sexes), were informed and gave their consent to the foetal pancreas collecting. In sterile conditions of operation room the donor’s laparotomy was performed and the pancreas isolated and placed in a small amount (1-2ml) of conserving solution used for tissue culture, minced with scissors (whole pancreas dimensions: 2x2x20 to 4x5x25mm, fragment volumes: 1-2mm³). After operation the donor‘s wound was closed and the foetus returned to the family or to the hospital service for engraving. Recipients were patients with compensated (at the moment of the operation) but severe or middle complicated type
1 diabetes (53) and type 2 insulin requiring DM(6) aged 16-49 years, having given their informed consent. During to the implant collection, the recipient was prepared, abdominal skin disinfected. In the Para umbilical region a small incision of 0.5cm was performed and the foetal material was injected through a special trocar (diameter 2.5mm) into the subcutaneous fat at a depth of 2-3cm. This site was supposed to ensure a venous drainage partly in direction of the portal vein through the Para umbilical venous web. One silk ditch closed the wound.
After operation usual investigations were provided: blood glucose, urine glucose and acetone, blood creatinine, urea and other routine indices determination [42]. In some cases blood c-peptide and insulin were determined, as well as some immunologic investigations (CD4, CD8, global amount of Theophylline sensible lymphocytes and active lymphocytes). The follow up was realized by family polyclinics physicians and under the control by endocrinologists and by our team. Only 3 patients have systematically received higher doses of insulin during the first post operation 3 days. For the others insulin was given according to the usual schema of glycaemia correction needs. Ultrasound investigation of the site of FPI and abdomen was performed to follow the morphological evolution of the implant in 16 patients. EEG was also performed in patients with severe neuropathy and encephalopathy.
The observation delay varied from 6 months to 10 years and more. Repeated implantation were performed 6 times according to the patients’ requests. Biopsies of the implant were not performed.*NB. Since 1990 the study has been interrupted: the implant harmlessness was further impossible to warrant, due to the appearance and increase of HIV, hepatitis B and C among the population. Statistics: Mean values and standard deviations (MSD) were calculated, liability, (p) was determined using Td against Tst Student criterion, correlation analysis of factors influencing FPI issue in clinics was provided.
Results
Part 1
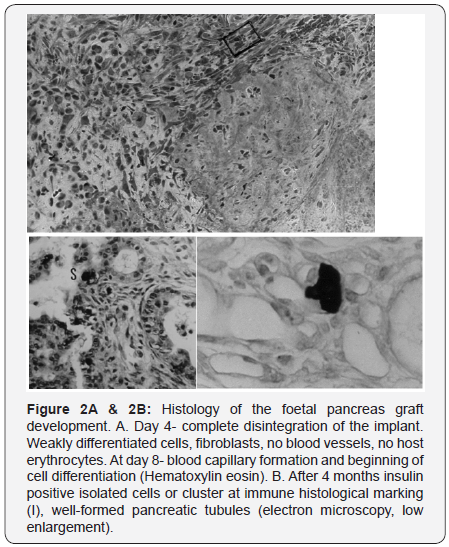
Syngeneic FPI at the ear site has allowed the following observations: during the first week, complete destruction of the organ structure: only weakly differentiated cells, fibroblastsand necrotic or apoptotic cells may be observed (Figure 2A). At the end of the first post implantation week some lacunas and capillaries appeared and the pancreas development has started (Figure 2A). But though the volume of the new-formed organoids was much more the initial one (5x3x2mm versus 2x1x1, 5mm) the typical structure of adult pancreas with well-structured exocrine and endocrine parts were never observed. Nevertheless after 4 months a pancreas-like formation was observed with tubules, endocrine cells positive to insulin, glucagon and somatostatin (Figure 2B). These endocrine cells might be functional, that was verified in series with STZ diabetes.
In control healthy rats glucose blood levels varied between 80 and 130mg/dl and daily drunk water volume was between 20 and 40 ml; urine was free of blood, protein, glucose, urea and pH was 5-6. We considered as normalized treated diabetic animals whose glycaemia had come back to 100±20mg/dl, daily drunk water amount - to 30±6ml polyuria, urine ketone have disappeared, and BW has reached normal level according to the strain evolution curve depending on age and sex. After STZ injection without any treatment the mean survival delay was 12±7 days, glycaemia reached 500 and more mg/dl with polyuria, high glycosuria, presence of ketone in urine, severe decrease of BW and general condition worsening.
FPI has given positive results in 8 of the 24 animals concerned (only 30%), that was complete and stable during several months, normalization of glycaemia, disappearance of glycosuria and improvement of general condition including BW increase and normalization (compared to standard BW curves by strain, sex and age). A whole series of 16 STZ rats has failedafter FPI of 1 or 2 pancreases and no other intervention. The best results were obtained when glycaemia level during the first days and weeks after grafting was kept between 250 and 350mg/dl by insulin injection of 2-4UI daily at the beginning, and then slowly decrease in dose and injection frequency.
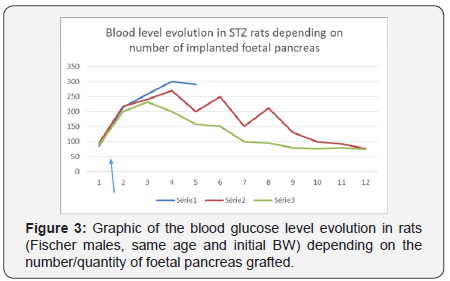
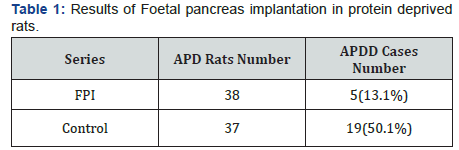
Insulin supplement was stopped when glycaemia has fallen under the end point 250mg/dl (Figure 3). The result of APD was a slowing of the development: BW gain, sexual differentiation and in 50.1% of the animals the development of diabetes some months after the nutrition normalization. The allogeneic FPI has hastened the correction of the animal development after restoration of a normal diet and significantly decreased the incidence of post APD diabetes (Table 1). Interesting that the allogeneic implant acute rejection was observed only at the end of the restoration process, i.e. about 1 month after FPI, when animals have practically reached their normal BW.
Part 2
Among the 59 patients undergoing FPI, 6 of them twice or even thrice, neither worsening of the DM course, nor implant acute rejection was observed. According to the results of inquiries after FPI an increase of hypoglycaemia events was noted (65±7.9% of the patients after FPI versus 35±6.7% before it)*, despite exogenous insulin doses daily injected were decreased from 61±17IU before operation to 41±14IU after FPI. This tendency represented an average of 25% decrease and has lasted during 4-7 years. Presence of acetone in urine was diminished (18±6.8% of the patients after FPI versus 43±7.3% before it).
4 kinds of reaction to the FPI were revealed 1/ no influence at all (5 patients), 2/ slowly developing but long lasting (up to 10 years) improvement of general condition with decrease of necessary daily insulin amount, improving of vision and disappearance of neuropathy, 3/ temporary improvement ofall indices as in group 2, but during from 2 to 24 months with come-back to a situation nevertheless better than before FPI and lasting during years, from the points of view of complication development and insulin doses necessary to compensate the DM, 4/ temporary - from 4 to 14 months improvement as in group 2 and 3 but with further worsening of the disease symptoms.
Ultrasound investigation has shown a significant increasing of the implant dimensions, especially in patients with a type 2 effect and a fibrous reduction in late delays. A temporary increase of the patient’s own pancreas dimensions was also noted. Encephalography has shown normalization of the brain electric activity in 15 out of 16 investigated patients. As to hormonal investigation results increase of blood C-peptide (0.4- 4ng/ml) in 10 out of 20 investigated patient. An increase of blood somatomedin (from 52±10 versus 91±20ng/ml) and IGF-1, normalization to control levels of increased or decreased glucagon were also observed in isolated patients investigated during the first year after FPI. Results of blood lymphocytes investigation have shown some changes correlated in time to the FPI effect, but not significant enough.
According to the results of the inquiries, different improvements in the disease development maintained during 4-7 years. Stability of the DM was noted during 4-10 years after FPI in 74±1, 3% of the patients (interval of liability 95%). The second and third FPI has a less expressed positive effect, including a shorter restoration of the patient’s potency. In order to diminish the hypoglycemia and yo-yo events, Biostator and Apparatus of Diabetes Diagnosis and complex insulin treatment [7,12-15] were first used insulin delivery monitoring with positive stabilizing effect not only during the sessions but during several days after them.
Discussion
Our results have confirmed that foetal pancreas grafting has proved to be possible, to give growth to adult organ-like formations with specific morphology and endocrine function [39,41] and others. Complete correction of STZ induced Diabetes in tats under certain conditions as well as significant improvement of diabetic patients’ condition and Indices during several years in 70% of the cases. Some remarks are necessary. Considering the syngeneic implant structure evolution in rats, we never have observed the development of an adult structured exocrine and endocrine pancreas. Both types of tissue coexisted but without the connexion they have in the foetal organ before grafting, or in the normal adult organ. This could be due to the implantation site - subcutaneous ear pouch - which did not ensure the place enough for a normal expansion and no possibility for evacuation/excretion of the secreted juice that could inhibited its secretion.
It could explain that other authors have described the restoration of both endo and exocrine parts of the implant placed under liver or spleen capsule [36]. Another explanation is theabsence in the pancreas of a well-organized intramural nervous system, the compact character of the gland. The same observations were obtained with foetal liver transplantation [56-57]. This makes the difference with the hollow organs of the digestive tract which give growth to a normal adult oesophagus, stomach or intestines [51-52] and with the heart [58-59]. However the very presence pancreatic tubules in the developed FPI can explain the long lasting effect of the graft in STZ rats and even in patients The role of these formations is known in the endocrine cells production and endocrine pancreas natural regeneration. Hence the existence and function of FPI were found to be significantly more important that the one of differentiated endocrine cells or clusters implants. In rats, the success of FPI was conditioned by several factors such as: no less than 4 foetal pancreas implanted, warm ischaemia no more than 1 hour, a well vascularized site (ear pavilion, organ parenchyma - spleen or liver), regulation of glycaemia between 250 and 350 mg/dl by insulin injections, that is high enough to stimulate insulin production by the graft, but not too high, that can inhibit or paralyze it. We have also noted this inhibitory influence of maintaining high insulin doses after FPI on the early post operation evolution of the patients.
An important question is the FPI immunogenicity. In our experiments, the foetal material used was syngeneic that has allowed to avoid graft rejection problems and be sure of the FPI potentiality. We have proved that allogeneic foetal organ graft is rejected as well as adult organ one (1994). Maybe embryonic material is less antigenic [46], but we have not used it. It is also possible that DM1 patients are immunologically slightly modified as shown by altered ratio CD4/CD8 lymphocytes (not published own observations) and other literature data [60-63]. If true, this can explain why our FPI underwent no acute, but more or less quick chronic rejection.
Interesting that in APPD series a similar phenomenon could be observed: the implanted was tolerated by weak dystrophic rats, but the implant was acutely rejected as soon as the animal condition was improved. This may be important when considering the complex treatment of serious malnutrition. The possible origin of the so called ‘before 1975 “DM type 3” probably lays in the lack of protein for the adequate insulin synthesis corresponding to the needs of an adult organism. So, for DM treatment FPI has the following advantages:
- No need to pass by an expensive culture in vitro, long lasting effect on the different aspects of the disease including its vascular, neurologic and other complications.
- organization : (the potential recipient has to be ready to answer the calling as soon as possible when a potential donor is considered),
- storage (in our cases the maximal warm storage between the donor’s death and the implantation at 19-20 °C was 60-90 min, more often less than 30 min, that was considered as a factor of success), tolerance problem between host and implant,,
- ethic problems which can be overcome if considering the foetus as a child and, as for the children)donors, obtain the informed consent of the parents or the mother if no father declared.
Coming back to the development of FPI within the adult organism, our experimental observation s has shown that the first post operation week presents a certain interest. During this period the implant loses its morphologic structure: part of already differentiated cells die (apoptosis, ischemic necrosis), part seems to dedifferentiate, part of weakly differentiated cells - precursor stem cells - survive.
The cause might be the diffusion conditions of “feeding” which end only after 5-7 days when host capillaries penetrate the graft and blood elements can be observed within it. It marks the beginning of re-differentiation of the implant elements which corresponds to the start of IGF1 elevation in the host blood. The process is the same in all the foetal organ implants we have observed [47, 55]. The phenomenon has not be systematically investigated as for the nature of these not differentiated cells, the presence of growth factors in this medium, the role of the host own stem cells and growth factors (base level or induced by FIU) in the implant development. Among the observed cells of the implant there are probably precursor cells specific for the grafted organ, as far as from a FPI only pancreas cells and tissue will grow, and the same is observed with other foetal organ implants. Teratoma growth is an exception [64]. So, may be, FPI and other foetal organ implantation constitutes a natural short cut for in vivo production of these organoids which are going to revolutionize the regenerative medicine? [65-68]
Conclusion
- FPI for DM treatment remains promising but under certain conditions of collect, storage, material quantity implanted and host adequate management and follow up.
- The presence of pancreatic tubules in the grown FPI is probably the warrant of beta-cell further production which allows long lasting effect of the procedure.
- Implantation of fetal organs could also be applied to clinical purposes, the main problems to resolve being: a) development of acquired immunological tolerance rather than immune suppression and b) constitution of fetal tissue/organ banks with in vitro as well as in vivo storage.
- Syngeneic implantation of foetal organs with its features of regression followed by ontogenetic development seems to be an interesting model for the IN VIVO study of such phenomena as possible de-differentiation of tissues and managing re-differentiation growth factors.
- The organ-like development of FPI (and other foetal organ grafting) could be an effective short cut for obtaining IN VIVO well-formed and vascularized organoids able to repair or replace altered adult organ.
Acknowledgement
The authors express their thankfulness to: Drs Furugyan P, Mbulava Mava K, Ms Pisareva T from People Friendship University for their participation to some parts of the work, to Dr VN Kukushkin and SV Malygina from the Hospital N° 64, for their support, control and help,to Drs Hustin J and Delrée P from IPG (Gosselies; Belgium) for their precious consultation in optic and electron microscopy, to Dr C De Prez and her technician team from CHU Brugmann Dpt of Pathology (Brussels, Belgium) for their support and help in managing biopsy material; to Professors V Shumakov (presently deceased, Institute of Transplantation, Moscow, Russia), Bergmann P and Corazza F from the Experimental/Translational Medicine Laboratory of the Fre Brussels University, for their scientific support and help to Ms M. Leroy (IPG, Gosselies, Belgium) for logistic help.
References
- Répertoire commenté des medicaments CBIP (Centre belge d information pharmaco thérapeutique) (2016) Diabète CBIP (Ed.), Brussels, Belgium pp: 181-196.
- Woo VC (2015) New insulin’s and new aspects in insulin delivery. Can J Diabetes 39(4): 353-433.
- Lastow O (2014) Conference report: 2nd Medicon Valey Inhalation Sympoium. Ther Deliv 5(2): 119-121.
- Heinemann L (2011) New ways of insulin delivery. Int J Clin Pract Suppl 170: 31-46.
- Farmer TG, Edgar TG, Peppas NA (2008) The future of open and closedloop insulin delivery for Diabetes Mellitus. J Pharm Pharmacol 60(1): 1-13.
- Matsuda E, Brennan P (2012) The effectiveness of continuous subcutaneous insulin pumps with continuous glucose monitoring in outpatient adolescents with type 1 diabetes: a systematic review. JBI Libr Syst Rev 10(Suppl 42): 1-10.
- Sparacino G, Zanon M, Facchinetti A, Zecchin C, Maran A, et al. (2012) Italian contribution to the development of continuous glucose monitoring sensors for diabetes management. Sensors Basel 12(10): 13753-13780.
- Haidar A, Legault L, Dallaire M, Rabasa Loret R (2013) Glucoseresponsive insulin and glucagon delivery (dual hormone artificial pancreas) in adults with type 1 diabetes: a randomized crossover controlled trial. CMAJ 185(4): 297-305.
- Blauw H, Keith HP, Koops R, De VJH (2016) A review of safety and design requirements of the artificial pancreas. Ann Biomed Eng 44(11): 3158-3172.
- Harvey RA, Dassau E, Zisser H, Seborg DE, Jovanovic L, et al. (2012) Design of the health monitoring system for the artificial pancreas: low glucose prediction module. J Diabetes Sci Technol 6(6): 1345-1354
- Facchinetti A, Sparacino G, Guerra S, Lujif YM, Cobelli C, et al. (2013) Real-time improvement of continuous glucose-monitoring accuracy: the smart sensor concept. Diabetes Care 36(4): 793-800.
- Novikov VK, Coulic VP (1987) Méthode/Procédé de diagnostic du diabète sucré Moscou 15.11.1991 Brevet n ° 1718822.
- Simonov VA, Kluchko AV, Novikov VK, Coulic VP (1992) Electronic device for control of the energetic balance in man and animals (appliances in Diabetes Mellitus). Proceedings of the 14th Annual International Conference of the IEEE Engineering in Medicine and Biology Society. Paris, France.
- Coulic V, Novikov V, Devriendt J, Gillet C, Bodson A, et al. ( 2007) Use of temperature gradient measuring device in monitoring of diabetic and critically ill patients” in “Sciences and Technologies for Health”, SFGMB & IEEE, Lyon, France, pp. 372-375.
- Coulic V, Novikov V, Devriendt J (2013) A new feedback for monitoring insulin therapy? (First experimental and clinic tests). Pancreatic disorders and Therapy S5: 2165-7092.
- Tarabarko NV, Novikov VK, Rjevskaya ON, et al. (2006) Surgical treatment of Diabetes Mellitus. Viestnik Transplantologyi y Iskustvennykh Organov 8(4): 23-30.
- Novikov VK, Vietluguina MA, Maïssiouk IaG (2012) Correction of carbohydrate metabolism by using the apparatus of insulin therapy in patients with type I Diabetes Mellitus after renal transplantation. Viestnik Transplantologyi y Iskustvennykh Organov (14): 72-6.
- Coulic V, DeVuyst M, DePrez C, Hustin J, Sidi B, et al. (2001) Combined implantation of several organs into syngenic adult animals (first results) Acta Gastro-enterologica Belgica 64(6): B01.
- Stites E, Kennealey P, Wiseman AC (2016) Current status of pancreas transplantation. Curr Opin Nephrol Hypertension 25(6): 563-569.
- Lucan VC, Berardinelli L (2016) Gastrointestinal side effects of posttransplant therapy. J Gastroint Liver Dis 25(3): 367-373.
- Gruessner RW, Gressner AC (2013) The current status of pancreas transplantation. Rev Endocrilol 9(9): 555-562.
- Squifflet JP, Gruessner RW, Sutherland DE (2008) The history of pancreas transplantation; past, present and future. Acta Chir Belg 108(3): 367-378.
- Kimelman M, Brandacher G (2013) Trends in immune suppression after pancreas transplantation: what in the pipeline? Curr Opin organ transpl 18(1): 76-82.
- Schauer PR, Bhatt DL, Kirwann JP, Kashyap SR, steven E (2017) Bariatric Surgery versus intensive medical therapy for Diabetes- 5-year outcomes. N Engl Med J 376(7): 641-651.
- Crawford MR, Bena JF, Schauer PR, Kashyap SR (2017) Long term outcomes of bariatric surgery on bone density in obese patients with type 2 diabetes. J Diabetes Complications 31(4): 780-781.
- Magain AD, Richard T, De Becker B, Rorive S, Benahmed A, et al. (2016) Nonsurgical complications of bariatric surgery: about four cases and review of the literature. Rev Med Brux 37(1): 26-34.
- Ballinger WF, Lacy PE (1972) Transplantation of intact pancreatic islets in rats. Surgery 72(2): 175-186.
- Ballinger WF, Lacy PE, Scharp DW, Kemp DB, Knight M (1973) Isografts and allografts of pancreatic islets in rats. Br J Surg 60(4): 313.
- Scharp DW, Lacy PE, Santiago JV, coucollugh MC, Weide LG, et al. (1990) Insulin independence after islet transplantation into type I diabetic patient. Diabetes 39(4): 515-518.
- Houwing H, Van ARM, Vander ZEA, Strubble J, Paul TR, et al. (1996) Noradrenergic and cholinergic reinnervation of islet grafts in diabetic rats. Cell Transplant 5(1): 21-30.
- Noda N, Ina K, Shimada T, Yasunami Y, Ryu S, et al. (1994) Reinnervation of allografted pancreatic islets in the rat liver. Proc Soc Exp Biol Med 207(1): 76-80.
- Carlsson PO, Andersson A, Carlsson C, Hekkestrom C, Hoglusnd E, et al. (2000) Engraftment and growth of transplanted pancreatic islets. Ups J Med Sci 105(2): 107-123.
- Reinholt FP, Hultenby L, Tibell A, Lorsgren O, Groth CG (1998) Survival of fetal porcine pancreatic islet tissue transplanted to a diabetic patient: findings by ultrastructural imunocytochemistry. Xenotransplantation 5(3): 222-225.
- Groth CG, Tibell A, Wennberg L, Korsgren O (1999) Xenoislet transplantation: experimental and clinical aspects. J Mol Med 77(1): 153-154.
- Wennberg L, Goto M, Maeda A, Song Z, Benjamin C, et al. (2005) The efficacy of CD40 ligand blockade in discordant pig-to-rat islet xenotransplantation is correlated with an immuno suppressive effect of immunoglobulin. Transplantation 79(2): 157-164.
- Plesner A, Verchere CB (2011) Advances and challenges in islet transplantation: islet procurement rates and lessons learned from suboptimal islet transplantation. J Transplant 2011: 979527.
- Zahradnicka M, Girman P, Kriz J, Berkova Z, Saudek F, et al. (2016) Islet transplantation as a treatment for hypoglycemia unawareness syndrome. Evaluation of the pilot program and comparison with pancreas transplantation. Cas Lek Cezsk 155(7): 349-356.
- Hatipoglu B (2016) Islet cell transplantation and alternative therapies. Endocrinol Metab Clin North Am 45(4): 923-931.
- Garvey JFW, Millard PR, Morris PJ (1980) Experimental transplantation of fetal pancreas and isolated islets in the rat: srudies of the donor pretreatment and recipient immune suppression. Transplant Proc 12 (4suppl2): 186-189.
- Furugyan P, Diallo, Coulic V (1981) Experimental protein deprivation diabetes and method of its correction. Bul Exptl Biol Med n °10 p. 350- 353.
- Hardy MA, Lau HT, Weder C, Reemstma K (1984) Foetal pancreatic islet transplantation: immune-alteration with ultraviolet irradiation. World J Surg 8(2): 207-213.
- Kulik CVP, Novikov VK, Pisareva TP, Ignatenko SN, Kukushkin VN, et al. (1987) Fetal pancreatic implantation without conservation in diabetic patients. J Sovietskaya Medicina (7): 16-22.
- De WG, Mannery C (2003) Human embryonic stem cells: research, ethics and policy. Hum Reprod 18(4): 672-682.
- Rowley JD, Blackburn E, Gazzaniga MS, Foster DW (2002) Harmful moratorium on stem cell research. Science 297(5589): 1957.
- Borissov MA, Petrakova OS, Gvazava IG, Kalistratova EN, Vasiliev AV (2016) Stem cells in the treatment of insulin-dependent diabetes mellitus. Acta Naturae 8(3): 31-43.
- Ishii T, Eto K (2014) Foetal stem cell transplantation: past, present and future. WJSC 6(4): 404-420.
- Coulic V, Collette J, Gillis V, Novikov VK (1993) IGF-1 in fetal pancreas transplantation Chinese. Med Sci Journal 8(62): A 004.
- Coulic VP, Novikov VK, M Bulava K, Furugyan P, Iskrenko IA (1994) Transplantation of the foetus pancreas to adult animals . Sechenov Physiological Journal 80(10): 111-118.
- Coulic V, Hustin J, Novikov V (1996) Morph functional peculiarities of ectopic foetal pancreatic growth. Acta Gastro enterologica Belgica 59(suppl 1): 62.
- Coulic V, De Vuyst M and Hustin J, Otero J, De Koster E, et al. (1996) Factors influencing fetal digestive organs ectopic growth. In: Eurem, Dublin (Eds.), Abstracts 1996, B 12-4. Proceedings of EUREM 11, 3: 327.
- Coulic V, Otero J, Hustin J, Hoang A, Longheval G, et al. (1997) Some laws of fetal endodermal organ ectopic growth. Acta Gastroenterologica Belgica 60(suppl 1): C77.
- Coulic V, Hustin J, DeVuyst M, Hoang A, Otero J, et al. (1998) Digestiveorgans culture in vivo: delayed observations. Acta Gastroenterologica Belgica 61(suppl 1): B18.
- Coulic V, Hustin J, De Vuyst M, De Prez C, Staroukine M, et al. (1999) Degenerative and regenerative processes in fetal organ ectopic growth. Analytical Cellular Pathology 18(1): A046.
- Coulic V, Collette J, DeVuyst M, Hustin J, DeKoster E (2001) The first phase of the syngeneic foetal organ implant develompent: crucial and mysterious process. Abstracts 14th Heidelberg Cytometry Symposium (HCS) October 18-20, Germany.
- Coulic V, Collette J, Hustin J, DeKoster E, Deltenre M (2001) Growth factors and fetal digestive organ implants development. Acta Gastroenterologica Belgica 64(1): B2.
- Coulic V, Delrée P, DePrez C, Bakari S, Lasser L, et al. (2003 ) Foetal liver syngenic transplantation (First report), XVth Belgian Week of Gastroenterology (Abstracts), Acta Gastro-enterologica Belgica, 66, n°1, B01.
- Coulic V, Delrée P, DePrez C, Sidi B, Lasser L, et al. (2003) Syngenic transplantation of fetal liver. European Journal of Internal Medicine 14(Suppl 1): S23.
- Coulic V, Deltenre P, Beckers E, DeReuck M, Krzemien M, et al. (2001) Morphological and physiological features of ectopically grown foetal heart implants. Acta Clinica belgica.
- Coulic V, Collette J, Deltenre P, DePrez C, DeVuyst M, et al. (2001) Cardiomyoblasts implantation (First results) // XIVth ISDQP International Congress diagnostic molecular pathology : from genomics to proteomics. Proceedings p. 66.
- Kempura J, Caraffa A, Ranconi G, Lessiani G, Conti P (2016) Are mast cells important in diabetes ? Pol J Pathol 67(3): 199-206.
- El-Samahy MN, Tatawy AA, Adly AA, Habeeb NM, Ismail EA, et al. (2017) Expression of CD4(+) CD29(nul) I lymphocytes in children and adolescents with type I diabetes mellitus: relaton to microvascular complications, aoertic elastic properties and carotid intima media thickness/ Pediatr Diabetes.
- Thorsen SU, Pipper CB, Mortensen HB, Swensson J (2016) Level of soluble TREM-1 in children with newly diagnosed type 1 diabetes and their siblings without typed 1 diabetes; a Danish case-control study. Pediatr Diabetes, doi: 10.1111/pedi.12464.
- Torres-Castro, Arroyo-Camarena UD, Martinez-Reyes CP, Gomez-Arauz, Beja TY, et al. (2016) Human monocytes and macrophages undergo M1) type inflammatory polarization in response to high levels of glucose. Immunol Lett 176: 81-89.
- Coulic V, De Prez C, Abdulsater F, De Koster E, Gielen I, et al. (2013) Is incidental teratoma growth possible after syngeneic foetal organ implantation in the adult rat? JMRP 2(5): 7-13.
- Ruidant L (2017) La culture d’organoïdes humains est en plein essor. Le Journal du médecin (2482): 28.
- Wang W, Jin S, Ye K (2017) Development of islets organoids from H9 human embryonic stem cells in biomimetic 3D scaffold. Stem Cell Dev, doi:10.1089/scd.2016.0115.
- Kim Y, Kim H, Ko UH, Oh Y, Lim A, et al. (2016) Islet-like organoids derived from human pluripotent cells efficiently function in the glucose responsiveness in vitro and in vivo. Sci Rep 12(6): 35145.
- Raikwar SP, Kim EM, Sivitz WI, Allamargot C, Thedens DR, et al. (2015) Human iPS cell-derived insulin producing cells from vascularized organoid under kidney capsules in diabetic mice. PloS One 10(1): e0116582.






























