Effect of Lycopene on Pharmacokinetic and Pharmacodynamics of Gliclazide in Diabetic Animal Models
Umachandar Lagisetty1*, Habibuddin Mohammed2 and Sivakumar Ramaiah3
1Pharmaceutical Sciences, Jawaharlal Nehru Technological University, India
2Shadan College of Pharmacy, India
3Geethanjali College of Pharmacy, India
Submission: August 02, 2018; Published: September 28, 2018
*Corresponding author: Umachandar Lagisetty, Research Scholar, Pharmaceutical Sciences, JNTU, Hyderabad, Telangana state, India, Email: umachandra.lagisetty@gmail.com
How to cite this article: Umachandar L, Habibuddin M, Sivakumar R. Effect of Lycopene on Pharmacokinetic and Pharmacodynamics of Gliclazide in Diabetic Animal Models. J Complement Med Alt Healthcare. 2018; 8(1): 555730. DOI: 10.19080/JCMAH.2018.08.555730
Abstract
Background: Diabetes mellitus is one of the metabolic disorders associated with high blood sugar levels. Utilization of natural drugs among patients under diabetes mellitus pharmacotherapy is across the board.
Objective: To examine the pk-pd(pharmacodynamic-pharmacokinetic) interactions of lycopene and gliclazide in animal models and to understand the safety & effectiveness.
Methods: Single and multiple dose interaction studies were carried out in normal rats, diabetes induced rats and rabbits to evaluate the effect of lycopene on the gliclazide activity. Blood samples from the study animals were used for the estimation insulin and glucose levels by using radioimmunoassay method and chemistry analyzer (automated) respectively. Homeostasis model assessment used for determination of β-cell function. Additionally, sophisticated HP-LC technique used for analysis of diabetic rabbits serum samples for gliclazide.
Results: Gliclazide produces significant reduction in blood glucose levels in diabetic animals. However, tests examined from gliclazide in blend with lycopene indicated more prominent diminishment in blood glucose concentration in animals with diabetes.
Conclusion: The study concludes that the lycopene along with gliclazide shows the significant pharmacodynamics interaction but doesn’t establish pharmacokinetics interaction up on single and multiple-dose treatment in animals.
Keywords: Lycopene; Gliclazide; Diabetes mellitus; Pharmacokinetics; Pharmacodynamics
Abbreviations: AUC: Area Under the Concentration Time Curve; AUMC: Area Under First Moment Curve; CL: Clearance; Cmax: Peak Serum Concentration; Kel: Elimination Rate Constant; MDT: Multiple-Dose Treatment; MRT: Mean Residence Time; SD: Standard Deviation; SDT: Single-Dose Treatment; Tmax: Peak Time; T1/2: Terminal Half-Life
Introduction
Multiple drug therapy is the simultaneous utilization of different drugs. It can be related to the solution and additionally utilization of excessive pharmaceuticals at measurements or frequencies higher than remedially basic. These restorative combinations might be lethal [1] or favorable [2] at the given therapeutic dose. Diabetes Mellitus (DM) is a metabolic disorder signifies with blood sugar level is abnormally high due to insulin insufficiency and function or both [3]. Diabetic patient’s shows decreased antioxidant levels and increased oxidative stress [4]. Gliclazide (second era sulfonylureas) is the favored decision of medication which is accounted for to have to have antioxidant properties [5] diminished inclination to prompt serious hypoglycaemia and cell reinforcement properties [6]. The mechanism of action includes K+ adenosine triphosphatase channel inhibition in pancreas [7,8] and gliclazide predominantly metabolized by CYP2C9 and moderately by CYP3A4 [9]. Indeed, phyto chemical extracts from herbs either alone or as combination have been guaranteed to avert DM complications [10]. Of these plants, mulberry (Morus alba L.) leaf, fenugreek (Trigonella foenumgraecum) seed [11], and American ginseng (Panaxquinquefolius) root [12] are much of the time proclaimed as worthy. Lycopene is a carotenoid, richest source in tomato fruits (Solanumlycopersicum) [13]. It is also found in watermelon, papaya, pink grapefruit, and pink guava [14]. A wide-ranging literature collection from all scientific references revealed that lycopene has antioxidant [15], antidiabetic activity and also a CYP3A4 enzyme inhibitor [16]. Reference gives evidence for lycopene, most likely to prevent hepatocellular carcinoma development [17], improvement in sperm quantity [18], motivates cell proliferation to bone growth [19]. Nevertheless, there is limited information about lycopene activity on blood glucose levels and interaction with anti-diabetic drug gliclazide in animal models. Therefore, this study innovates the hypoglycemic activity of lycopene on gliclazide in animal models.
Methods
Drugs
Gift samples of gliclazide and lycopene acquired from DRL, Hyderabad, India, and Parry Phytoremedies private limited, Pune respectively. Alloxan (monohydrate) be procured from Loba Chemie, Mumbai, India. Analytical grade materials and reagents used for present study.
Lycopene solution
lycopene powder 20mg weighed and dissolved in distilled water 10ml and make to 2 mg/ml solution. A dose of 4mg/kg of body weight was administered by using clean and dry oral feeding needle for 21 days [20].
Gliclazide solution
Gliclazide in a small amount of 0.1 N sodium hydroxideused to prepare gliclazide solution and water used for final volume makeup [21].
Preparation of alloxan solution
A dose of 110 mg/Kg alloxan monohydrate in sterile saline prepared and injected by s.c. route instantly within few minutes to evade degradation [20].
Animals
Albino rats (8-9 weeks aged) & albino rabbits (3 months old) selected gender male aged having weight between 170 and 250g and between 1 and 1.5kg respectively were obtained from M/s Mahavir Enterprises, Hyderabad. They were maintained under controlled room temperature (24±20C; relative humidity 60-70%) in a 12h light - dark cycle. The animals were provided a standard laboratory diet and water ad libitum. Before performing the experiment, the animals were acclimatized. The experimental protocol was approved by the Institutional Animals Ethics Committee (IAEC). Reference # GBN/GQ/2014.
Experimental study design
Five groups of male albino rats/rabbits were made and each consisting of six animals. Based on the gliclazide doses of 2 and 4mg/kg per body from dose-effect association learning in normal rats and rabbits, the weight considered for oral administration. Study designed as follows:
Group I: Normal control
Group II: Diabetic control
Group III: Gliclazide (2mg/kg for rats/; 4mg/kg for rabbits) body weight, po.
Group IV: 4mg/kg, po of Lycopene on body weight.
Group V: Lycopene (4mg/kg) + Gliclazide (2mg/kg for rats/4mg/kg for rabbits) body weight, po.
Diabetic rats - pharmacodynamic interactions
Male albino rats weighing (170-250g) were fasted for overnight prior to administration of freshly prepared alloxan monohydrate solution and injected within 5min of preparation to avoid solution degradation at a dose of 110mg/kg. Immediately, 5% glucose solution was orally administered for 72h to prevent any chances of hypoglycemia shock. Animals had provided with continuous with water and feed. Animals were confirmed for development of hyperglycaemia by analysis of fasting serum glucose levels after 72h of alloxan monohydrate solution injection where animals were fasted for a second time of 14h and blood samples collected from retro orbital plexus.
The rats having a fasting blood glucose level of 200mg/dl or above at 72h were included in the study as diabetic subjects. Gliclazide (2mg/kg, po.) was administered after 30min of lycopene administration (4mg/kg, po.). Blood glucose levels were estimated on initial, 1st day, 3rd day, 7th day, 14th day, and 21st day of the treatment.
Normal rats - pharmacodynamic interactions
Six rats were chosen for the investigation. Gliclazide (2mg/ kg of animal body weight) was given orally to Group III rats and withdrawn blood at programmed time points. A similar procedure was followed with either lycopene 4mg/kg, po. (Group IV) alone or combination of lycopene and gliclazide (Group V) as per the designed doses. Consequently, the treatment was sustained for 20 days with standard feeding. The animals were dosed with gliclazide (2mg/kg) after 30minutes of each lycopene treatment. Samples withdrawn at programmed time points for each treatment [9].
Diabetic rabbits - pharmacodynamic & pharmacokinetic interactions
Each group has six rabbits. Alloxan (100mg/kg) in normal saline used to induce diabetes on single intravenous iv injection [22]. Gliclazide (4mg/kg) was given orally to Group III rabbits and their samples withdrawn at planned time points. A similar procedure was followed with either lycopene only (Group IV) or combination of lycopene and gliclazide (Group V) at the specified doses.
After this single-dose interaction study, the same animals received daily treatments with lycopene for the next 20 days with regular feeding. Before 30minutes of gliclazide treatment the animals were dosed with lycopene for each treatment. Samples withdrawn at scheduled time intervals for each treatment of drugs gliclazide, lycopene, or combination [23].
Collection of serum samples
Under light ether anesthesia, marginal ear vein and retro orbital plexus punctured and the blood withdrawnfor14h fasted rats and rabbits respectively on different occasions i.e., day 0, 1st day, 3rd day, 7th day, 14th day, and 21st day. On day 0 (SDT) and day 21st (MDT) blood samples collected at different time intervals as 0h, 1h, 2h, 4h, 8h, 10h and 12h for pharmacokinetic study experiment and to estimate insulin levels and glucose concentration by using radioimmunoassay and chemistry analyzer (automated), respectively. Homeostasis model assessment was used to determine β-cell function. Additionally, high-performance LC (Liquid chromatography) technique was used and determines the serum gliclazide concentration in rabbits.
Determination of β-cell function
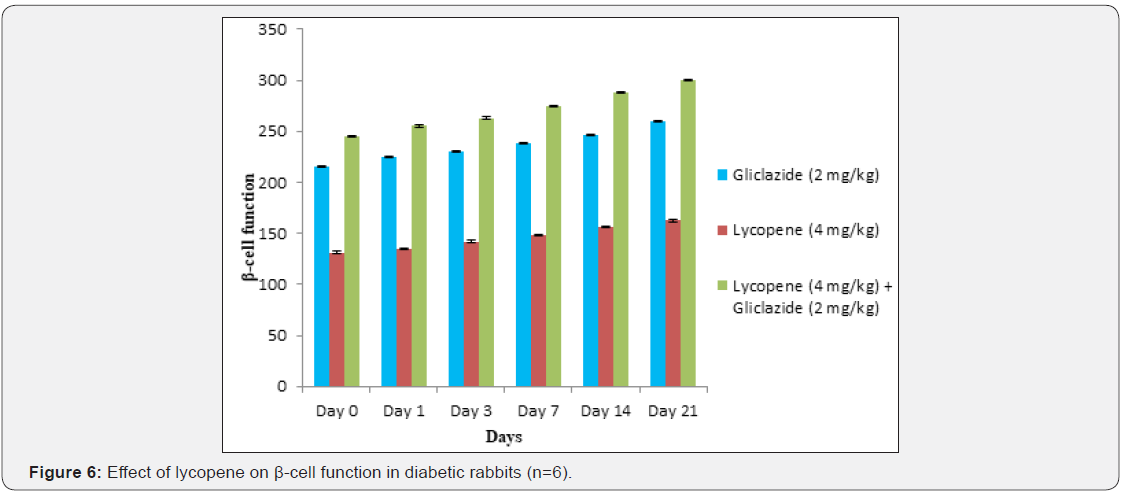
β-cell function was assessed by the homeostatic model assessment protocol and calculated [21,24,25].
β-cell function = (FSI x 20) / (FSG - 3.5) ×100

Where FSI=fasting serum insulin (μIU/ml) and
FSG= fasting serum glucose(mg/dL).
Pharmacokinetics: Gliclazide pharmacokinetics estimated by Kinetica 5.0 software, Alfa soft Limited.
Statistical analysis: The data were analysed using one-way analysis of variance (ANOVA), followed by Dunnett’s test (Sigma Plot version 11) and p<0.05 was considered as statistically significant. The data were expressed as mean ± Standard Deviation (SD).
Results
Pharmacodynamic interaction between lycopene and gliclazide
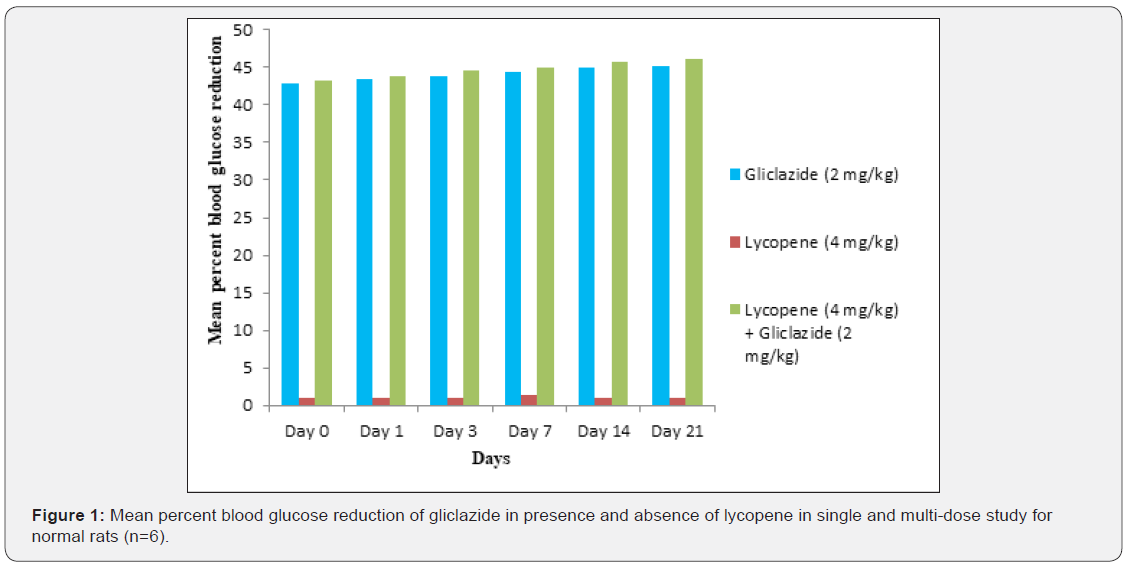
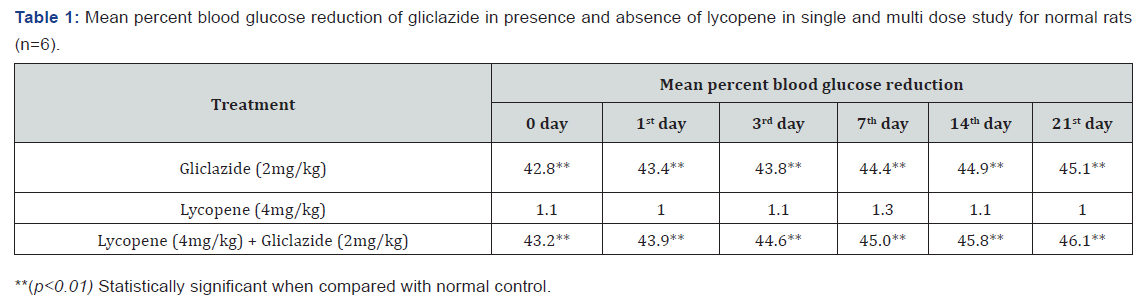
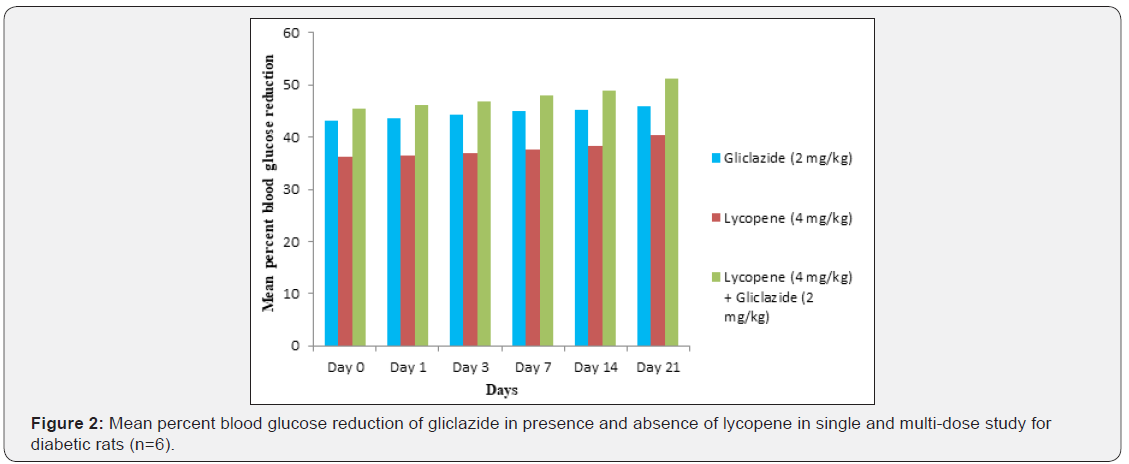
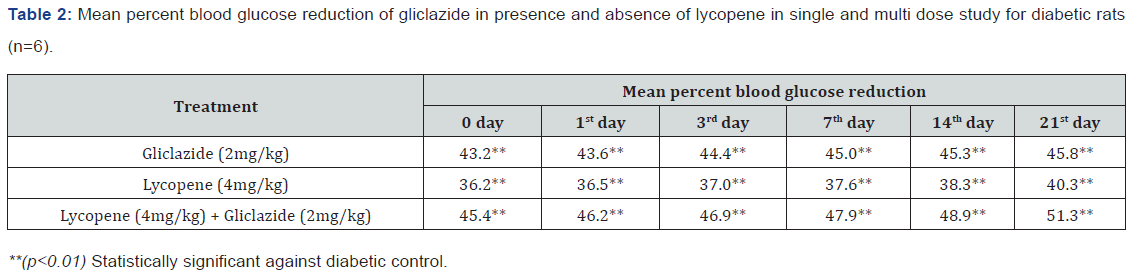
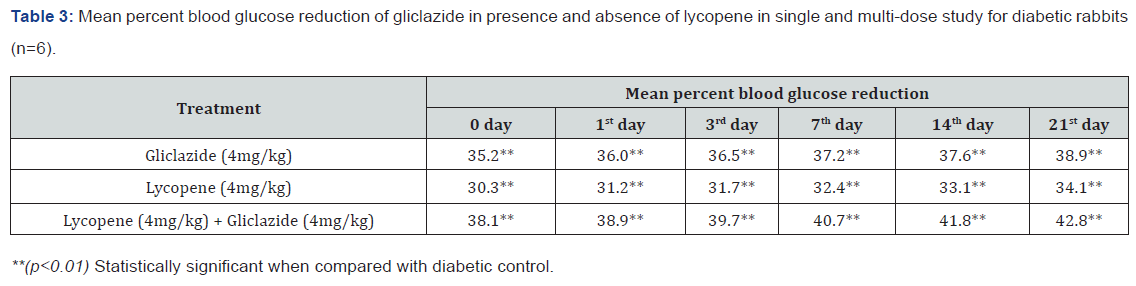
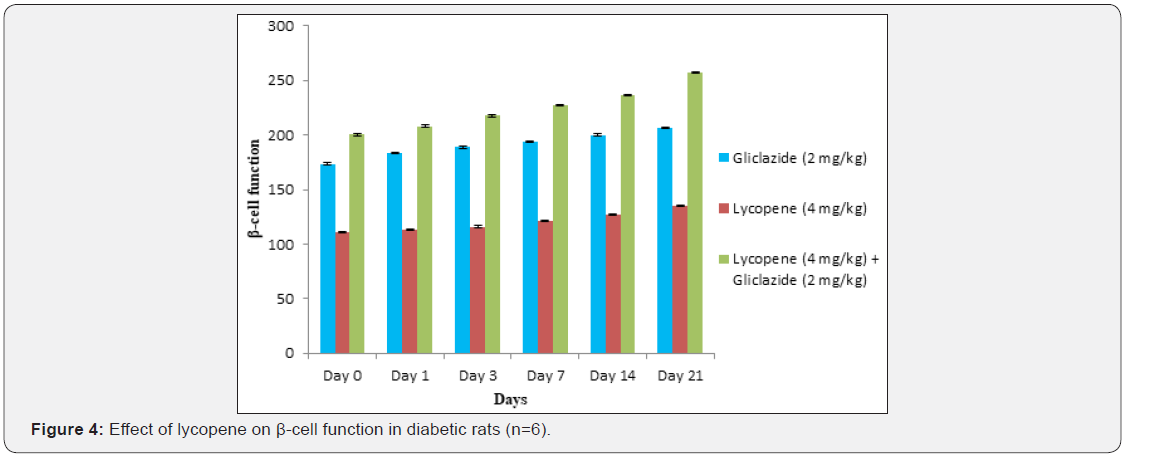
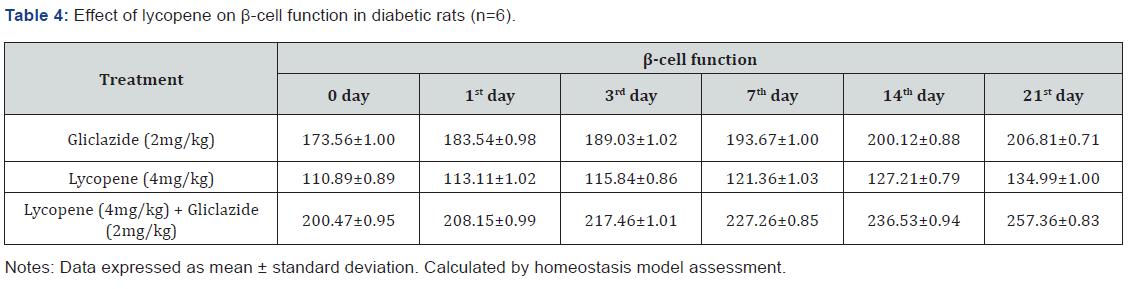
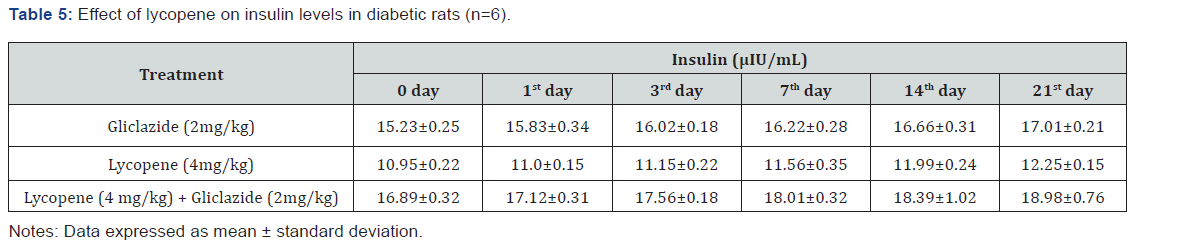
Gliclazide results: Gliclazide created the hypoglycemic effect in normal rats. The observations from Table 1 & Figure 1 of lowering of blood glucose levels by improvement in mean percent blood glucose reduction from initial day 42.8% to 45.1% for the day 21 treatment. The results from Table 2 & Figure 2 in diabetic rats the activity is more extreme as per record mean percentage blood glucose reduction from initial day 43.2% to 45.8% on day 21 treatment. In diabetic rabbits the anti-hyperglycemias action was shown in Table 3 and Figure 3 with maximum percent blood glucose reduction of 35.2% to 38.9% from the initial day to 21st day respectively. The anti-hyperglycaemic activity evidenced by improvement in β-cell function and insulin levels as recorded in (Table 4 & 5) & (Figure 4 & 5) for the initial dose in the 21st day study are 173.56±1.00 to 206.81±0.71 and 15.23±0.25 to 17.01±0.21 respectively in diabetic rats.
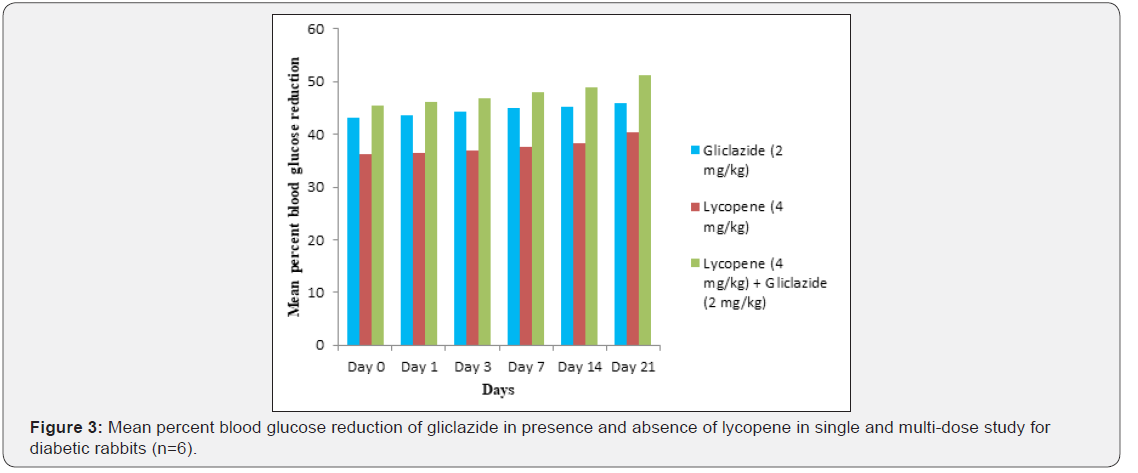

Lycopene results
The blood glucose levels from Table 1 and Figure 1 in normal rats doesn’t alter by lycopene treatment alone, but significant mean percentage blood glucose reduction observed in diabetic rabbits as 36.2% to 40.3% from day initially to 21st day treatment from Table 2 & Figure 2.This is the similar fashion as with gliclaizde the anti-hyperglycaemic activity probably with improvement in β-cell function and insulin levels as recorded in (Table 4&5) & (Figure 4&5) for the initial dose to 21st day study are 110.89±0.89 to 134.99±1.00 and 10.95±0.22 to 12.25±0.15 respectively.
Combination treatment results
Single and multiple dosed lycopene along with gliclazide proved to significant percent blood glucose improvement as 43.2% of initial dose to 46.1% of 21st day treatment in normal animal models (Table 1 & Figure 1). Further, numerous measurement combination of lycopene with gliclazide created fundamentally more prominent decreasing in blood glucose levels after treatment in diabetic rats and rabbits when contrasted and diabetic control. The values recorded from an initial dose to 21st day treatment of mean percentage blood glucose reduction are 45.4% to 51.3% and 38.1% to 42.8% of diabetic rats from Table 2 & Figure 1 and rabbits Table 3 & Figure 3 respectively.
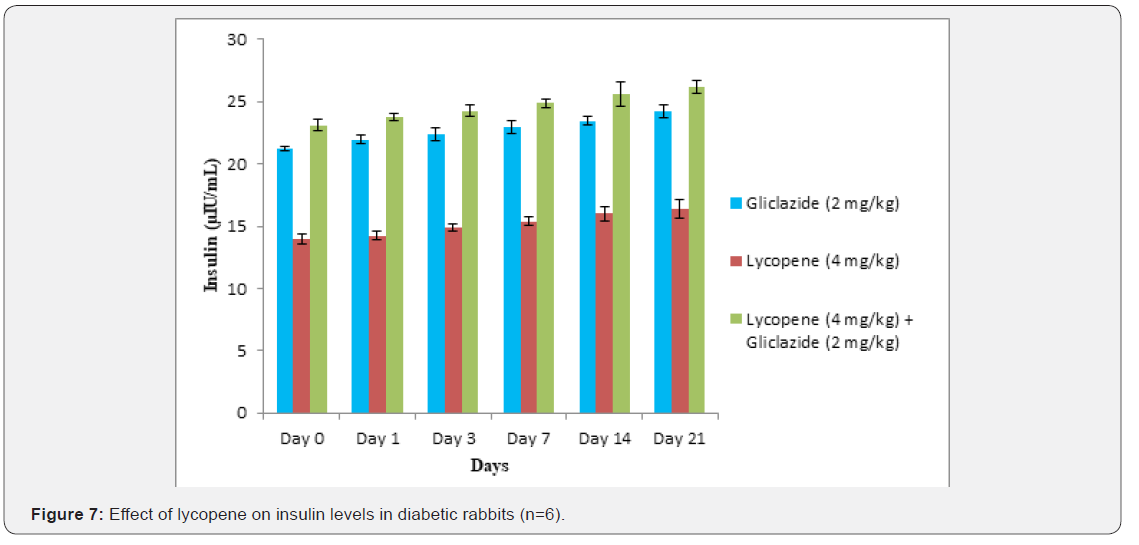
Lycopene exhibited supportive effect by escalating the activity of gliclazide and also significant changes in β-cell function 200.47±0.95 to 257.36±0.83 and insulin levels16.89±0.32 to 18.98±0.76μIU/mL in diabetic rats (Table 4&5 and Figure 4&5). Whereas the results validated from diabetic rabbit study the significant changes in β-cell function 200.47±0.95 to 257.36±0.83 and insulin levels 23.12±0.42 to 26.18±0.56μIU/mL from initial dose of single dose study to 21st day dose of multiple dosed study represented in Table 6 & 7 and (Figure 6&7).
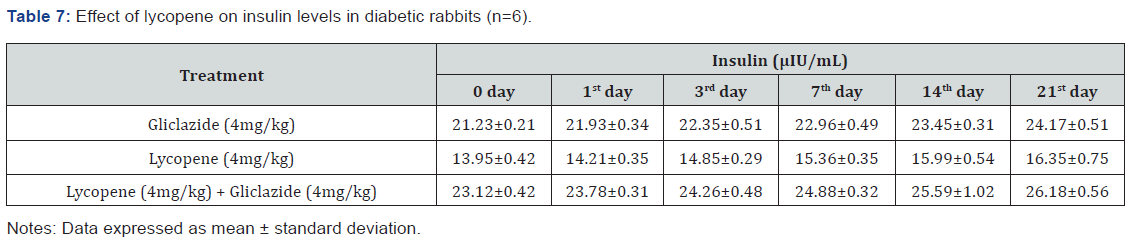
Pharmacokinetic interaction between lycopene and gliclazide
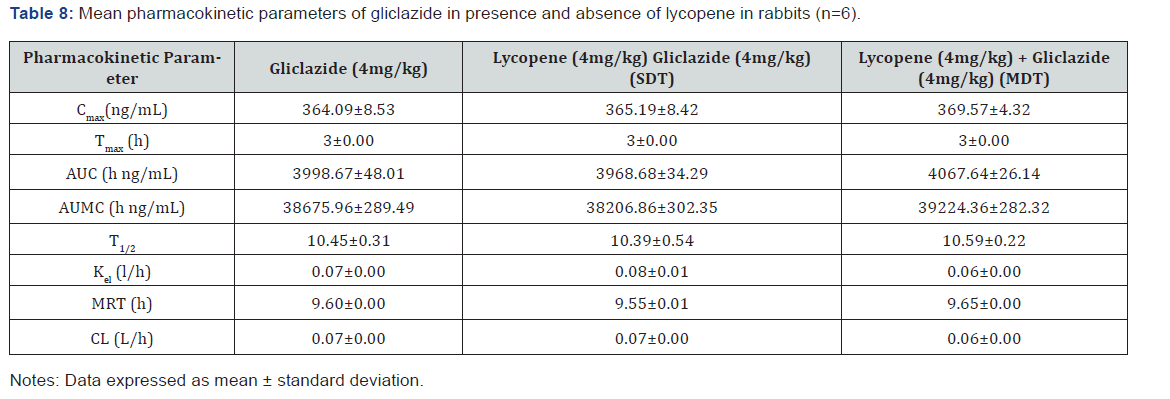
Pharmacokinetic parameters for gliclazide alone and along with lycopene on single and multiple-dose administration were recorded as given Table 8. There are no major fold changes with the combination treatment in diabetic rabbits.
Discussion
Medication combinations believe about being an imperative part of pharmacology research, and such communications are normally assessed in animal models [26]. Even though animal models can never swap the requirement for far reaching thinks about in human subjects, they help in understanding the systems of medication collaborations. The present investigation is intended to assess the impact of lycopene on the action of gliclazide in animal models. Animal model using normal rats was used to identify the interaction where as that of diabetic rats was used to validate the interaction. The experiment further validated by using dissimilar species, rabbit model [23]. The hypoglycemic action of gliclazide in rats is interceded by blocking K+channels in β-cells of pancreas [27], in this way empowering insulin emission and as well as improvement in β-cell function by the way expanding tissue take-up of glucose [28]. Insulin levels were estimated at time intervals, where greater improvement in mean percent blood glucose levels were observed both in rats and in rabbits under diabetes upon gliclazide treatment. Any medication or herbal active component may change the pharmacokinetic and pharmacodynamic movement of the substrate when it is a potential inducer or inhibitor of that specific medication utilizing proteins such as metabolizing enzymes. Lycopene has the potential to cause herb-drug interaction when administered with other drugs. This study revealed the influence of lycopene on the pharmacodynamic activity of gliclazide alone and in combination using single and multiple-dose treatments in rats and rabbits. The end determinations were assessed as far as glucose level (% mean glucose reduction), insulin level and β-cell work utilizing homeostatic model assessment and pharmacokinetics of gliclazide in rabbits. In the present investigation, single and multiple-dose treatment of lycopene obtained about significant change via improvement with percent blood glucose reduction in diabetic rats and diabetic rabbits when contrasted and normal and diabetic controls individually. Here lycopene shows a supportive effect when combined with gliclazide. Liver has been appeared to be an insulin subordinate tissue, and is apparently engaged with glucose and lipid homeostasis, which is normally extremely influenced amid diabetes [29]. Insulin impacts the intracellular usage of glucose in various ways. Glucokinase catalyzes the change of glucose to glucose - 6-phosphate and assumes a focal part in the preservation of glucose homeostasis. In the liver, this compound is an imperative controller of glucose stockpiling and transfer [30]. Lycopene may exert its hypoglycemia activity via increased hepatic glucokinase activity and probably by stimulating insulin release from pancreatic β-cells as evidenced by elevated serum insulin level [15]. Lycopene also evidenced for potent inhibitory effect on CYP3A4 enzyme [16]. One compartment open model was utilized to assess the pharmacokinetic parameters. Lycopene might not have any huge impact on the metabolism of gliclazide, which is fundamentally used by CYP2C9 and moderately by CYP3A4. This could most likely clarify the outcomes got in this examination where the pharmacokinetics of gliclazide was not changed by lycopene in both single-and different measurements treatment considers.
Conclusion
The examination affirms that the interaction of lycopene with gliclazide is pharmacodynamic in nature as the glucose levels in animal models are significantly reformed and no correction in pharmacokinetics of gliclazide is observed. Since the interaction is grasped in two different species, it is likewise liable to happen in people. Hence, this combination needs observing of glucose levels occasionally when managed for their clinical advantages in diabetic patients.
References
- Zheng Lin Zhao, Sang Chan Kim, Hong Feng Liu, Yi Yan Wu, Li Bo Li, et al. (2017) Blockade of nicotine sensitization by methanol extracts of Glycyrrhizae radix mediated via antagonism of accumbal oxidative stress. BMC Complement Altern Med 17(1): 493.
- Soudeiha MAH, Dahdouh EA, Azar E, Sarkis DK, Daoud Z (2017) In vitro Evaluation of the Colistin-Carbapenem Combination in Clinical Isolates of A. baumannii Using the Checkerboard, Etest, and Time-Kill Curve Techniques. Front Cell Infect Microbiol 7:209.
- Ozougwu JC, Obimba KC, Belonwu CD, Unakalamba CB (2013) The pathogenesis and pathophysiology of type 1 and type 2 diabetes mellitus. J Physiol Pathophysiol 4(4): 46-57.
- Ganjifrockwala FA, Joseph JT, George G (2017) Decreased total antioxidant levels and increased oxidative stress in South African type 2 diabetes mellitus patients, Journal of Endocrinology, Metabolism and Diabetes of South Africa 22(2): 21-25.
- Brien RC, Luo M, Balaza N (2000) In vitro and in vivo antioxidant properties of gliclazide. J Diabetes Complications 14(4): 201-206.
- Kimoto K, Suzuki K, Kizaki T, Hitomi Y, Ishida H, et al. (2003) Gliclazide protects pancreatic beta-cells from damage by hydrogen peroxide. Biochem Biophys Res Commun 303(1): 112-119.
- Ashcroft FM, Rorsman P (2013) KATP channels and islet hormone secretion: new insights and controversies. Nat Rev Endocrinol 9(11): 660-669.
- Proks P, De Wet H, Ashcroft FM (2014) Sulfonylureas suppress the stimulatory action of Mg-nucleotides on Kir6.2/SUR1 but not Kir6.2/ SUR2A KATP channels: a mechanistic study. J Gen Physiol 144(5): 469- 486.
- Proks P, De Wet H, Ashcroft FM (2014) Sulfonylureas suppress the stimulatory action of Mg-nucleotides on Kir6.2/SUR1 but not Kir6.2/ SUR2A KATP channels: a mechanistic study. J Gen Physiol 144(5): 469- 486.
- Chang CL, Lin Y, Bartolome AP, Chen YC, Chiu SC, et al. (2013) Herbal therapies for type 2 diabetes mellitus: chemistry, biology, and potential application of selected plants and compounds. Evid Based Complement Alternat Med 2013: 378657.
- Hamden K, Keskes H, Elgomdi O, Feki A, Alouche N (2017) Modulatory Effect of an Isolated Triglyceride from Fenugreek Seed Oil on of α-Amylase, Lipase and ACE Activities, Liver-Kidney Functions and Metabolic Disorders of Diabetic Rats. J Oleo Sci 66(6): 633-645.
- Xie JT, Wang CZ, Ni M, Wu JA, Mehendale SR, Aung HH, et al. American Ginseng Berry Juice Intake Reduces Blood Glucose and Body Weight in ob/ob Mice. J Food Sci 72(8): S590-S594.
- Anurag K, Richa S, Dethi, Chopra K (2008) Lycopene attenuates diabetes associated cognitive decline in rats. Life sci 83(3-4): 128-34.
- Seren S, Milton Mutchnick, Daryl Hutchinson, Ozgur Harmanci, Yusuf Bayraktar, et al. (2008) Potential role of lycopene in the treatment of hepatitis C and prevention of hepatocellular carcinoma. NutrCancer 60(6): 729-35.
- Daniel EE, Mohammed A, Yusuf T, Ahmed A, Mohammed KR (2015) Hypoglycemia effect of Lycopene in Streptozotocin-Induced Diabetic Wistar Rats. Br J Med Med Res 7(9): 762-770.
- Sunaga K, Ohkawa K, Nakamura K, Ohkubo A, Harada S, et al. (2012) Mechanism-Based Inhibition of Recombinant Human Cytochome P450 3A4 by Tomato Juice Extract. Biol. Pharm. Bull 35(3): 329-334.
- Vrieling A, Voskuil DW, Bonfrer JM, Korse CM, Van Doorn J, et al. (2007) Lycopene supplementation elevates circulating insulin-like growth factor binding protein-1 and -2 concentrations in persons at greater risk of colorectal cancer. American Journal Clinical Nutrition 86(5): 1456-1462.
- Durairajanayagam D, Agarwal A, Ong C, Prashast P (2014) Lycopene and male infertility. Asian J Androl 16(3): 420-425.
- Kim L, Rao AV, Rao LG (2003) Lycopene II-effect on osteoblasts: The carotenoid lycopene stimulates cell proliferation and alkaline phosphatase activity of SaOS-2 cells. J Med Food 6(2): 79-86.
- Haribabu T, Divakar K, Goli D (2013) Evaluation of anti-diabetic activity of Lycopene and its synergistic effect with Metformin hydrochloride and Glipizide in Alloxan induced diabetes in rats. SchAcad J Pharm 2(2): 119-124.
- Mastan SK, Kumar KE (2010) Effect of antiretroviral drugs on the pharmacodynamics of gliclazide with respect to glucose-insulin homeostasis in animal models. J ExpPharmacol 2: 1-11.
- Gumieniczek A, Hopkała H, Wójtowicz Z, Nikołajuk J (2002) Changes in antioxidant status of heart muscle tissue in experimental diabetes in rabbits. Acta Biochim Pol 49(2): 529-35.
- Kumar KE, Mastan SK (2011) Influence of efavirenz and nevirapine on the pharmacodynamics and pharmacokinetics of gliclazide in rabbits. J EndocrinolMetab 1(3): 113-24.
- Bonora E, Targher G, Alberiche M, Bonadonna RC, Saggiani F, Zenere M B, et al. (2000) Homeostasis model assessment closely mirrors the glucode clamp technique in the assessment of insulin sensitivity: studies in subject with various degrees of glucose tolerance and insulin sensitivity. Diabetes Care 23(1): 57-63.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, et al. (1985) Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7): 412-419.
- Gupta RC, Atul BV (2000) Drug metabolism studies in animal models. Ind J Pharmacol 32(4): 562-6.
- Ma A, Kamp M, Bird B (1989) The effects of long term gliclazide administration on insulin secretion and insulin sensitivity. Aus N Z J Med 19(1): 14-49.
- Vanhaeften TW, Vaneman TF, Gerich JE (1991) Influence of gliclazide on glucose stimulated insulin release in man. Metabolism 40(7): 751- 755.
- Shetti AA, Sanakal RD, Kaliwal BB (2012) Anti diabetic effect of ethanolic leaf extract of Phyllanthusamarus in alloxan induced diabetic mice. Asian J Plant SciRes 2(1): 11-15.
- Gomis RR, Favre C, García-Rocha M, Fernández-Novell JM, Ferrer JC, et al. (2003) Glucose 6-Phosphate Produced by Gluconeogenesis and by Glucokinase Is Equally Effective in Activating Hepatic Glycogen Synthase. J Biol Chem 278(11): 9740-9746.






























