Intermediate Species Accounts for more Than a Half of Sulfur Speciation in Mine Waters - A Case Study Revealed using Vis Spectrophotometry and Microwave Plasma Atomic Emission Spectrometry
Viktoria Rafique*, Marlies Grimmer, Nils Hoth, Janith Abeywickrama and Carsten Drebenstedt
TU Bergakademie Freiberg, Institute for Mining, Gustav-Zeuner-Straße 1a, Germany
Submission: January 19, 2022; Published: March 24, 2022
*Corresponding author: Viktoria Rafique, TU Bergakademie Freiberg, Institute for Mining, Gustav-Zeuner-Straße 1a, Germany
How to cite this article: Rafique V, Grimmer M, Hoth N, Abeywickrama J, Drebenstedt C. Intermediate Species Accounts for More Than a Half of Sulfur Speciation in Mine Waters - A Case Study Revealed Using Vis Spectrophotometry and Microwave Plasma Atomic Emission Spectrometry . Insights Min Sci technol.2022; 3(2): 555607. DOI: 10.19080/IMST.2022.03.555607
Abstract
Even though sulfate and sulfide concentrations do not adequately describe the total sulfur pool in mine waters influenced by microbial sulfate reduction, detailed speciation of sulfur and the significance of different sulfur species in mine waters are so far less poorly understood in acid mine drainage research. Therefore, this study offers small-scale research on different sulfur species in mine waters and their relevance in total mass balance of sulfur species. The conventional determination of sulfur species is usually carried out using ion chromatography and involves considerable sample preparation, incl. derivatization of the sulfur species.
Therefore, methods for easy, effective, and low-cost spectrophotometric determination of sulfur species other than sulfate and sulfide were tested. Additionally, the cross-reactions of organic sulfur species with the spectrophotometric methods were also investigated. Besides sulfate, thiosulfate, sulfite, and sulfide coexist in real mine waters and are determinable with easy and cheap methods suitable for smaller water laboratories.
These methods were applied on real mine waters originating from active and remediated mining sites. The results were evaluated regarding potential sulfur imbalances. Sulfur from sulfate usually represents most of the sulfur pool. Thiosulfate is present in considerable quantities; however, it constitutes yet a minor proportion of the total sulfur and ‘other-than-sulfate’ sulfur. It has become clear that some mine waters contain multiple inorganics and possibly, organic sulfur species that would require more advanced analytical methods.
Keywords: Sulfur species; Mine water; Total sulfur; Thiosulfate; Sulfate; Sulfite; Sulfide; Circumneutral pH
Abbreviations: AMD: Acid Mine Drainage; SOI: Sulfur Oxidation Intermediates; MIW: mining-affected waters; MP‑AES: Microwave-Plasma Atomic Emission Spectrometry; ORP: oxidation-reduction potential; EC: electrical conductivity
Introduction
The presence of sulfur related species in mine waters can cause a negative environmental impact, widely known as acid mine drainage (AMD). It is well known that sulfur redox is very important in acid generation and the mobilization of toxic metals [1,2] . Nevertheless, not all sulfur species are acid-forming, and the detailed speciation of sulfur and the significance of different sulfur species when evaluating potential AMD processes by means of reliable laboratory tests are poorly understood.
Despite their significant role as one of the major nutrients [3,4], different sulfur species in interacting system of resources such as water and biota, could also lead biochemical electron transfer under anaerobic environments [5]. Moreover, as organic sulfur species are dominated in soils [6,7] and shallow subsurface environments [8], AMD related processes could significantly alter the organic sulfur compounds in water.
Nevertheless, many mine water studies usually consider only the most oxidized (SO42-, sulfate) and/or most reduced sulfur species (S2-, sulfide) in their studies [2,9,10], due to lack of reliable techniques in measuring other available species. However, many mine matrices indicate a deficiency in sulfur mass balance solely based on the sulfate and sulfide content.
Therefore, in this study three different mine water types obtained from Saxon mining area, Germany, were analyzed for their sulfur speciation. The selected locations were affected by sulfate reduction; hence, the presence of inorganic sulfate reduction intermediates was expected. A typical argument against the inspection of sulfur oxidation intermediates (SOI) is their assumed instability and rapid oxidation to sulfate through Fe3+ [11,12].
Organic sulfur species that also influence the mine water quality are often forgotten. SOI and organic sulfur species can be the missing pieces in the sulfur mass balance equation. It is not elucidated whether SOI pose a toxicological risk. However, they use up dissolved oxygen and contribute to the acidification of receiving water bodies and therefore should be monitored [12,13].
The up-to-date literature regarding sulfur species in mine waters discusses mainly the dissolution of pyrite in an acidic medium; the kinetics and mechanisms are well elaborated in the literature [14-18]. It was identified that thiosulfate is a key product in pyrite oxidation; however, under acidic conditions reacts to tetrathionate [19]. Thus, polythionates are expected to be the prevailing SOI in an acidic environment, whereas thiosulfate is dominant at higher pH values [20]. Thermodynamic considerations could confirm the disproportion of thiosulfate to elemental sulfur and tetrathionate at pH of about 3 [21].
Little we know about the sulfur species in pH-circumneutral mining-affected waters (MIW). Pioneers in targeting pH-circumneutral MIW were investigating the microbial communities in pH-neutral mine waters, as well as the sulfur speciation. Unfortunately, a more detailed chemical composition beyond the sulfur species of the targeted MIW was not presented [22]. In their recent publication, the researching persons provided a tool for determining the sulfur mass balances in circumneutral mine waters.
Sulfide and sulfate were determined with spectrometry, whereas sulfite, thiosulfate and elemental sulfur were ascertained using HPLC‑UV/VIS. The total sulfur was determined by ICP-AES. Additionally, it was demonstrated that ‘thiosalts’ SnOx2- present only a minor component of the SOI sulfur pool, contrary to the assumption that thiosalts were the dominant SOI [23].
Here, the overall chemistry of the samples was not observed, too. Mine water samples, and generally, environmental samples, usually contain high amounts of inorganic ions (Fe2+, Cl-, NO3-, SO42-). HPLC and IC systems are susceptible to high ionic strength matrices [24] and thus require a pre-treatment or derivatization to avoid analytical problems. The analyses of such waters using chromatography are therefore strenuous and cost intensive.
In our paper, we want to propose a similar mass-balance tool for sulfur species in circumneutral mine waters using simple, available, and cost-effective spectrophotometric methods that do not require tedious pre‑treatment. The results will be brought into comparison with the total sulfur content measured via microwave-plasma atomic emission spectrometry (MP‑AES).
Methods And Procedures
Selection Of Methods
The literature pool was searched for spectrophotometric methods for the determination of SOI that are viable for water laboratories possessing basic low-cost equipment. Here, the spectrophotometer DR 3900 from company Hach® was used. Among the collected methods, procedures with the lowest toxicity reagents were selected and tested for their feasibility and practicability.
For the determination of sulfide, the conventional blue methylene method was applied, whereas a test kit for sulfide determination from the company Hach® was utilized [25]. Literature offers techniques for the spectrophotometric determination of sulfite in water samples containing sulfide and thiosulfate, as well as methods for simultaneous thiosulfate and dithionate determination in the presence of sulfide and sulfite [26,27].
A conventional method based on the barium sulfate BaSO4 precipitation was selected to determine the sulfate concentration [28]. To exercise the selected methods, standard solutions containing the corresponding sulfur species were prepared in the relevant concentration ranges and measured via spectrophotometry to investigate the workability of the method as well as the construction of calibration curves.
Procedures
Sulfide 8131
For testing the sulfide method in the default range, 1 L of 0.2g/L stock solution was prepared under a protective atmosphere with oxygen-free double-distilled water to prevent immediate sulfide oxidation. Aliquots were taken from the stock solution to prepare a set of standard sulfide solutions with concentrations of 40, 160, 240 and 320µg/ L.
The solutions were then transferred to an open atmosphere and tested as quickly as possible on the initial sulfide concentration (t=0). This method is based on the reaction of hydrogen sulfide and acid-soluble sulfides with N, N dimethyl p phenylenediamine to form methylene blue. The color intensity is proportional to the sulfide concentration. The measured wavelength is 665 nm for spectrophotometers [25].
A 50-mL sulfite stock solution of 0.6 g/ L SO32- was prepared from 0.05 g sodium sulfite Na2SO3 and nitrogen-purged redistilled water. An aliquot of the stock solution containing 6, 32, 64 and 102µg sulfite, thus 10, 50, 100 and 160 µl of the stock solution, respectively, was added to a solution of 10 mL fuchsine-reagent (400 mg fuchsine dissolved in 1 L 2.4 M H2SO4) in a 100mL-volumetric flask.
After 10 minutes, 1 mL of 10 M formaldehyde was added to the mixture and the volume was adjusted to 100 mL with redistilled water. Following the incubation time of 10 min at room temperature, the extinction of the color complex was measured in the range of 400‑700 nm. The expected maximum according to the literature is at 570 nm. As a reference, a sulfite‑free reagent‑mixture was used [27,29].
Thiosulfate
The thiosulfate stock solution was obtained by dissolving 0.06 g of sodium thiosulfate pentahydrate Na2S2O3∙ 5 H2O in 0.5 L of nitrogen-purged water with a small amount of sodium carbonate as a stabilizer, giving a stock solution with 50 mM thiosulfate concentration.
The procedure went as followed: 4 mL of 0.2 M alkaline maleate buffer, 4 mL of 15 mM formaldehyde, the task of which is to mask the potentially present sulfite, and 40 mL of a sample (an aliquot of the stock solution and water) was placed into a smaller beaker. The solution was left standing for one minute. Then, two drops of freshly precipitated suspended zinc bicarbonate hydroxide Zn (HCO3) (OH) were added (prepared by mixing 5 vol% zinc acetate Zn (OOCCH3)2 and 7.5 vol% sodium carbonate Na2CO3 in equal volume ratio). At this point, sulfide in the sample precipitates as white zinc sulfide ZnS and can be removed by filtration.
The precipitate was washed with distilled water, and the filtrate was transferred to a 100 mL volumetric flask. Subsequently, 8 mL of 2 M acetic acid and 9.6 mL of iodate-iodide solution were added to the volumetric flask. The iodate-iodide solution was prepared by dissolving 72.6 g potassium iodide KI, 0.2 g of sodium carbonate Na2CO3 and 0.0178 g potassium iodate KIO3 in 500 mL distilled water, which gives 1 mN (= 1.67 mM) standard iodate in 0.87 M iodide solution.
The volumetric flask was filled up to 100mL with distilled water, and the absorption spectrum was recorded in the range 320-600nm. According to literature, the expected extinction maximum is at 350nm [26]. The added aliquots of stock solution were of the following volumes: 4, 8, 12 and 16mL, which altogether gave synthetic samples with thiosulfate concentrations of 500µmol/ L, 1 mmol/ L, 1.5 mmol/ L and 2 mmol/ L, respectively.
As a reference for photometry, a thiosulfate‑free sample was used [26]. This method is based on the reaction of thiosulfate with iodine. The more thiosulfate is present in the sample, the more iodine is converted to colorless iodide. As follows, the more thiosulfate the sample contains, the less intense yellow color is observed.
Sulfate 8051
A set of standard solutions with sulfate concentrations of 2, 20, 40, 60 and 70 mg/ L, respectively, was prepared from a commercial 1000 mg/ L sulfate standard solution. The compact method for the determination of sulfate is based on the precipitation of barium salt with sulfate forming a milky precipitate. For that, a cuvette was filled with 10 mL of distilled water and a paired cuvette is filled with a sample.
The content of one Sulfa Ver® 4 powder pillow was added to the cuvette with a sample and swirled to dissolve the barium salt. After 5 minutes, the cuvette with the blank sample was cleaned, inserted into a spectrophotometer, and the blank value was recorded. Then, the sample cuvette was cleaned, inserted and the sulfate concentration was determined [28,29].
Cross-Reactions
Additional experiments were made to investigate possible cross-reactions of inorganic and organic sulfur species or overall interferences in the spectrophotometric measurements.
a) Thiosulfate and sulfite solutions containing 10, 20 and 50mg/ L thiosulfate and sulfite, respectively, were acidified with acetic acid to pH 4‑5. Their oxidation to sulfate was traced using the method mentioned in 5.2.4. This experiment was to show whether, how fast, and under which conditions do thiosulfate and sulfite react to sulfate. It was expected that the oxidation to sulfate will occur faster in acidified samples.
b) Thiosulfate, L-cysteine, thiourea, mercaptosuccinic acid and mercaptoethanol standard solutions in the range 5‑800µg/ L sulfur (in compliance with the range of Sulfide 8131 method) were tested with the Sulfide 8131 method to inspect possible interference of the named sulfur species with spectrophotometric sulfide determination giving a false positive sulfide result and thus biasing the sulfide determination.
c) Thiosulfate, L-cysteine, thiourea, mercaptosuccinic acid and mercaptoethanol solutions of 50mg/ L sulfur were tested with the Sulfate 8051 method to observe whether these species interfere with the BaSO4 precipitation and thus falsify the spectrophotometric determination of sulfate.
Stabilization Of the Targeted Sulfur Species in Real Mine Waters
Experiments for developing a stabilizing solution that would preserve sulfide, thiosulfate, and sulfite in a water sample, were carried out before sampling campaigns. Academic literature describes spectrophotometric determination techniques for sulfide, sulfite as well as thiosulfate along with corresponding suitable sampling and stabilizing instructions [27,30,31].
Based on the mentioned works and additional experiments, the following stabilizations have been applied for use on a sampling campaign:
a) Equal volumes of NH3-NH4NO3 buffer, 5% zinc acetate solution and sample in a 20mL brown-glass bottle with a screw cap containing PTFE septum for the sulfide stabilization.
b) 5 mL of fuchsine-reagent and 20mL of sample in a 50mL brown-glass bottle with a screw cap containing PTFE septum to fix sulfite.
Since thiosulfate and sulfate were proven to be stable in previous experiments, they were measured in native subsamples that were filled brimful into 100mL bottles with screw caps containing PTFE septa to avoid sample degassing and oxygen exposure. For total sulfur analysis measured via spectrometry, the samples were filtered through 0.45μm surfactant free cellulose acetate syringe filters purchased from Santorius, collected in 50 mL PE bottles, and immediately acidified with 1 v/v % supra quality concentrated HNO3.
Sampling Procedure
The sampling campaigns took place in December 2019 (samples MX and RX) and November 2020 (WX). The mine water was sampled at three different (former) mining sites. Samples MX originate from an open-pit lignite mine site, where the mine water is pumped and channeled towards a mine water treatment plant. The water arriving at the mine water treatment plant carries a strong sulfidic odor, whereas the hotspot of the sulfide oxidation is not known.
Therefore, some suspected points were selected and sampled to reveal the spacial origin of sulfide‑rich water. Samples RX also originate from a lignite mine, however in this case, sulfide-rich outflows from a former deep mine that penetrate the surface of the lignite mine were sampled. WX include mine water samples from decommissioned underground uranium mining sites.
A collection of subsamples was taken on each sampling point, correspondingly to the targeted parameter(s). Moreover, typical field values such as pH, temperature, oxidation-reduction potential (ORP) and electrical conductivity (EC) were measured on site. Subsamples for the laboratory measurements of thiosulfate, sulfite, sulfide, and sulfate were taken and stabilized as described in 2.5. All subsamples were kept refrigerated at 3 °C until the next day when they were analyzed at the water laboratory at the Institute of Mining and Special Construction Engineering at TU Freiberg.
Sulfur Mass Balance Calculations
Under the assumption that most of the sulfur in mine waters occurs in the form of sulfate (S from SO42-, shortly S‑SO42-), sulfite (analog S‑SO32-), thiosulfate (S‑S2O32-) and sulfide (S‑S2-), the sulfur content in mmol/ L was first calculated from the spectrophotometric analyses (output in mg/ L) as follows. The calculated sulfur content from the spectrophotometry was compared with the total sulfur content measured via MP-AES.
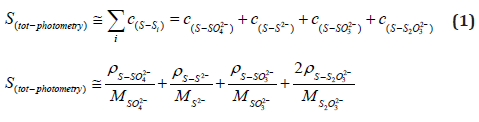
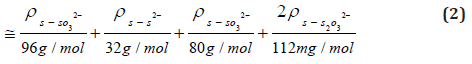
Result
Cross-Reaction I
Thiosulfate proved to be reasonably stable for at least a week, even when acidified. No sulfate was detected in the thiosulfate solutions with the Sulfate 8051 method. Therefore, we can assume that thiosulfate does not bias the spectrophotometric sulfate determination. Sulfite, on the other hand, formed a milky precipitate with BaSO4 immediately.
Acidified samples were oxidized almost at the same rate as the non‑acidified ones. The initial sulfite concentration was completely retrieved in the sulfate after 7 days. We can conclude that sulfite oxidizes rapidly in unpreserved samples or that the method gives a falsely positive result in the presence of sulfite. Considering very low sulfite content in typical mine waters, none of these conclusions is significant for further investigations.
Cross-Reaction Ii
The organosulfur species have partly reacted to methylene blue when inspected with the Sulfide 8131 method. Random amounts of sulfide in the lower method concentration range (5-20 µg/ L) were found in these solutions. Therefore, it is apparent that organic sulfur species may mildly positively bias the spectrophotometric determination of sulfide.
Cross-Reaction Iii
No sulfate was detected in the solutions of thiourea, mercaptoethanol, mercaptosuccinic acid and L‑cysteine hydrochloride monohydrate with sulfur content of 50 mg/ L. Therefore, we can conclude that organic sulfur species will not bias the determination of sulfate. The methods for spectrophotometric determination of thiosulfate and sulfite include steps for masking other SOI species. Altogether with the above-elaborated cross-reactions, the selected spectrophotometric methods will provide results with fair validity (Figure 1).
Real Mine Water Characteristics
General Aspects
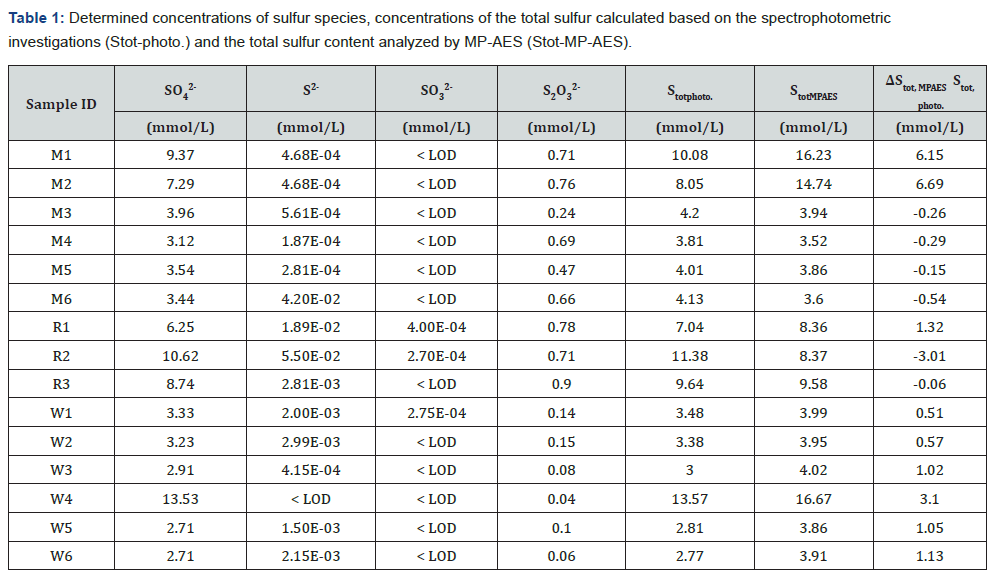
The sampled mine waters can be considered circumneutral since their pH ranged from 6.35 to 7.63 with a median value of 7.24. The redox potentials (EH) range between 113 and 305mV (median value 177mV) which is reasonable for mine water influenced by microbial sulfate reduction. The median value of present ferrous ion in the samples was 2.38mg/L Fe2+. The experience has shown that such ferrous ion content is common for mine waters of similar character. Further analyses results are placed in the Table 1.
Sulfur Imbalances
Table 1 provides a comparison of the total sulfur from the spectrometric and the spectrophotometric analysis. Here, a deviation Δ was calculated using to illustrate the difference between total sulfur contents found via MP‑AES and spectrophotometry.
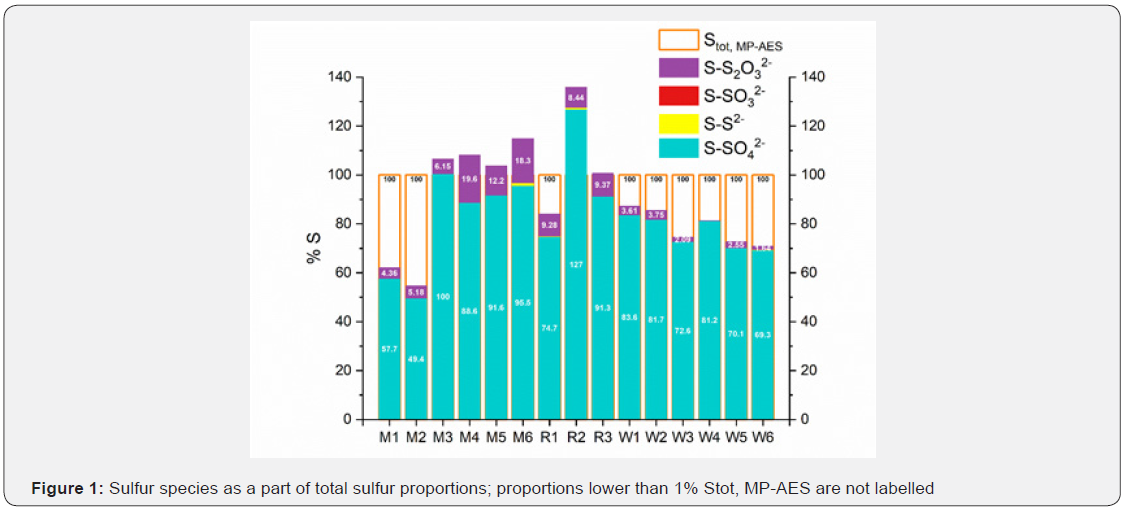
The sulfur from sulfate accounts for most of the sulfur in all samples. Sulfur from sulfide, sulfite and thiosulfate constitutes a minor part, whereas the most prevailing among these is thiosulfate sulfur. The positive sulfur deviation Δ implies that sample contains more sulfur than what is determinable via spectrophotometry. On the other hand, a negative deviation suggests that the sulfur measured via spectrophotometry was not retrieved in the spectrometric measurement.
In the following figure, the total S measured by MP-AES is set to be 100% S. The sulfur species measured via spectrophotometry are displayed as % from the Stot, MP-AES. This figure reaffirms that the proportion of sulfate sulfur is the biggest in all samples except for M2. In some samples, the summary of sulfur from spectrophotometry exceeds the total sulfur measured in MP AES. This analytical artifact will be addressed in the discussion section.
Discussion
The selected spectrophotometric methods are eligible to be applied independently since the experimental procedures ensure the exclusion of cross-reactions among the inorganic sulfur species. The rapid oxidation of sulfite to sulfate is insignificant in this context; since the sulfite is considered to be solely a side product of sulfide oxidation via the thiosulfate mechanism [8], we can assume that the amount of sulfite in mine water samples of acidic and circumneutral pH will be small, if not negligible, which would decrease its relevancy when looking into the sulfur balances.
The behavior of organic sulfur species in 3.1.2 cannot be explained – it is not clear whether some organic sulfur species reacted with the water or with the reagents to form sulfide – or the spectrophotometric reaction was interfered due to the presence of the organic sulfur species. However, if the investigated organic sulfur species are present in typical mine waters, they will not occur in such concentrations that would seriously impair the sulfide determination. Furthermore, the spectrophotometric determination of sulfate was not impaired in the presence of organic sulfur species.
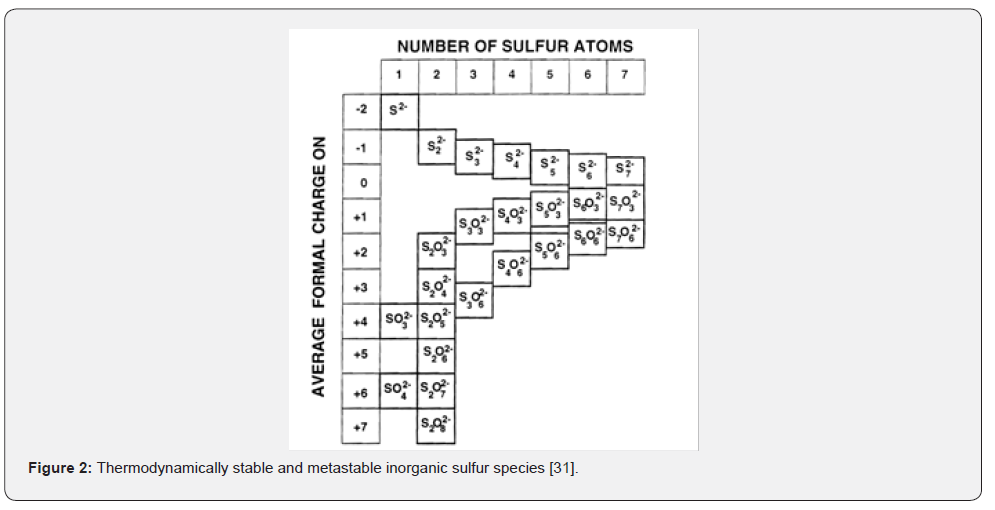
The balancing of sulfur in real mine water samples turned out to be a challenge; samples showing negative sulfur deviation suggest that the spectrophotometric results may be overestimated. Another possible scenario is an undetermination of sulfur in the MP-AES analysis. There is evidence that e.g., sample R2 contained very fine colloidal elemental sulfur. It is considered that the elemental sulfur, if not removed, raises additional iodine consumption in the determination of thiosulfate resulting in overestimation. Colloids would also contribute to an additional light scatter in a sample and hence increased light extinction in the spectrophotometer (Figure 2).
Since the samples for MP‑AES analysis was filtered before the measurement for equipment protection, we can assume that an unknown portion of the sulfur could have been withheld in the filter and consequently, we retrieved less sulfur than what was present. For future applications, the elemental sulfur should be removed by e.g., solvent extraction.
Addressing the opposite situation; samples with a positive sulfur deviation imply that not every sulfur species had been considered in the spectrophotometric analysis. Since the pool of inorganic species contains more than four species than those targeted, such a scenario is well probable. Not to forget about the universe of organic sulfur species that is unproportionally larger than the inorganic one. The following figure displays the pool of inorganic sulfur species.
Conclusion
It was confirmed that sulfur species other than sulfate and sulfide are present in mine waters and should not be disregarded when analyzing microbially reduced mine water samples since they may constitute more than half of the total sulfur amount. The literature search yielded methods for easy and low-cost spectrophotometric determination of sulfite and thiosulfate - these have been utilized to quantify the sulfite and thiosulfate in real mine water samples.
Observing the sulfur balance in real mine water samples, it has become obvious that a microbially reduced mine water system is complex and most probably contains a variety of inorganic and organic sulfur species. From among sulfide, sulfite and thiosulfate, thiosulfate was the prominent species. However, in most of the samples, thiosulfate constituted only a minor part of the ‘other-than-sulfate’ sulfur. The used spectrophotometric methods for the determination of sulfite and thiosulfate should be applied best after the exclusion of colloidal sulfur.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgment
The investigations were part of a master thesis research project funded by TU Berg Akademie Freiberg. The authors would like to acknowledge mining companies which allowed the sampling team to enter their sites and collect the samples under their guidance.
References
- Karimian N, Johnston SG, Burton ED (2018) Iron and sulfur cycling in acid sulfate soil wetlands under dynamic redox conditions: A review. Chemosphere 197: 803–816.
- Kassahun A, Hoth N, Paul M (2018) Mine water tracer substances for biogeochemical processes in flooded uranium mines.
- Cao W, Dang Z, Zhou XQ, Yi XY, Wu PX, et al. (2011) Removal of sulphate from aqueous solution using modified rice straw: Preparation, characterization, and adsorption performance. Carbohydrate Polymers 85(3): 571–577.
- Courbet G, Gallardo K, Vigani G, Brune MS, Trouverie J, et al. (2019) Disentangling the complexity and diversity of crosstalk between sulfur and other mineral nutrients in cultivated plants. Journal of Experimental Botany 70(16): 4183–4196.
- Fdz PF, Fdz PM, Fernández N, Urueña MA, García PA, et al. (2001) Combining the biological nitrogen and sulfur cycles in anaerobic conditions. Water Science and Technology 44(8): 77–84.
- Fuentes LLO, Medrano MJ, Pérez LF, Rivas MEN, García EEL, et al. (2019) From Elemental Sulfur to Hydrogen Sulfide in Agricultural Soils and Plants. Molecules 24(12): 2282.
- Wilhelm Scherer H (2009) Sulfur in soils. Journal of Plant Nutrition and Soil Science 172(3): 326–335.
- Antler G, Mills JV, Hutchings AM, Redeker KR, Turchyn AV (2019) The Sedimentary Carbon-Sulfur-Iron Interplay - A Lesson from East Anglian Salt Marsh Sediments. Frontiers in Earth Science p. 7.
- Abeywickrama J, Grimmer M, Hoth N, Grab T, Drebenstedt C (2021) Geochemical characterization of fouling on mine water driven plate heat exchangers in Saxon mining region, Germany. International Journal of Heat and Mass Transfer 176:
- Ren K, Zeng J, Liang J, Yuan D, Jiao Y, et al . (2021) Impacts of acid mine drainage on karst aquifers: Evidence from hydro geochemistry, stable sulfur and oxygen isotopes. Science of The Total Environment 761: 143223.
- Luther GW (1987) Pyrite oxidation and reduction: Molecular orbital theory considerations. Geochimica et Cosmochimica Acta 51(12): 3193–3199.
- Rickard D (2012) Sulfidic Sediments and Sedimentary Rocks. Developments in Sedimentology 65: 1-801.
- Miranda TJC, Pappoe M, Hawboldt K, Bottaro C (2013) The Importance of Thiosalts Speciation: Review of Analytical Methods, Kinetics, and Treatment. Critical Reviews in Environmental Science and Technology 43(19): 2013-2070.
- Moses CO, Kirk ND, Herman JS, Mills AL (1987) Aqueous pyrite oxidation by dissolved oxygen and by ferric iron. Geochimica et Cosmochimica Acta 51(6): 1561–1571.
- Sand W, Gehrke T, Jozsa PG, Schippers A (2001) (Bio)chemistry of bacterial leaching-Direct vs. Indirect bioleaching. Hydrometallurgy 59(2): 159-175.
- Schippers A (2004) Biogeochemistry of metal sulfide oxidation in mining environments, sediments, and soils. In J. P. Amend, K. J. Edwards, & T. W. Lyons, Sulfur Biogeochemistry-Past and Present. Geological Society of America.
- Schippers A, Sand W (1999) Bacterial Leaching of Metal Sulfides Proceeds by Two Indirect Mechanisms via Thiosulfate or via Polysulfides and Sulfur. Applied and Environmental Microbiology.
- Singer PC, Stumm W (1970) Acidic mine drainage: The rate-determining step. Science 167(3921): 1121-1123.
- Schippers A, Jozsa P, Sand W (1996) Sulfur chemistry in bacterial leaching of pyrite. Applied and Environmental Microbiology 62(9).
- Nordstrom DK (2011) Mine Waters: Acidic to Circmneutral. Elements 7(6): 393-398.
- Descostes M, Vitorge P, Beaucaire C (2004) Pyrite dissolution in acidic media. Geochimica et Cosmochimica Acta 68(22): 4559–4569.
- Whaley MK, Jessen GL, Nelson TC, Mori JF, Apte S, et al. (2019) The Potential Role of Halothiobacillus spp. In Sulfur Oxidation and Acid Generation in Circum-Neutral Mine Tailings Reservoirs. Front Microbiol 10: 297.
- Whaley MK, Marshall S, Nelson TEC, Twible L, Jarolimek CV, et al. (2020) A Mass-Balance Tool for Monitoring Potential Dissolved Sulfur Oxidation Risks in Mining Impacted Water 39: 291-307.
- Michalski A, Damoc E, Lange O, Denisov E, Nolting D, et al. (2012) Ultra-High Resolution Linear Ion Trap Orbitrap Mass Spectrometer (Orbitrap Elite) Facilitates Top-Down LC MS/MS and Versatile Peptide Fragmentation Modes*. Molecular & Cellular Proteomics 11(3).
- Hach Sulfide Method 8131 (2019) https://de.hach.com/asset-get.download.jsa?id=7639983901
- Koh T, Miura Y (1987) Spectrophotometric Determination of Micro Amounts of Sulfide, Sulfite and Thiosulfate. Analytical Sciences 3(6): 543-547.
- Pachmayr F (1960) Vorkommen und Bestimmung von Schwefelverbindungen in Mineralwasser. LMU, Germany.
- Hach Sulfide Method 8051 (2019) https://de.hach.com/asset-get.download.jsa?id=7639983902
- Bak F, Pfennig N (1987) Chemolithotrophic growth of Desulfovibrio sulfodismutans sp. Nov. By disproportionation of inorganic sulfur compounds. Archives of Microbiology 147(2): 184–189.
- Nor YM, Tabatabai MA (1975) Colorimetric Determination of Microgram Quantities of Thiosulfate and Tetrathionate. Analytical Letters 8(8): 537-547.
- Williamson MA, Rimstidt JD (1992) Correlation between structure and thermodynamic properties of aqueous sulfur species. Geochimica et Cosmochimica Acta 56(11): 3867–3880.






























