- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Optimization of an Ion Exchange Process for the Selective Recovery of Cobalt and Copper from Heap Leaching Waste Materials
Janith Abeywickrama*, Nils Hoth and Carsten Drebenstedt
TU Bergakademie Freiberg, Institute for Mining, Gustav-Zeuner-Straße 1a, Germany
Submission: January 19, 2022; Published: February 25, 2022
*Corresponding author: Janith Abeywickrama, TU Bergakademie Freiberg, Institute for Mining, Gustav-Zeuner-Straße 1a, Germany, Email: Janith.Abeywickrama@mabb.tu-freiberg.de
How to cite this article: Janith Abeywickrama, Nils Hoth, Carsten Drebenstedt. Optimization of an Ion Exchange Process for the Selective Recovery of Cobalt and Copper from Heap Leaching Waste Materials. Insights Min Sci technol.2022; 3(1): 555605. DOI: 10.19080/IMST.2022.03.555605
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Abstract
Despite eliminating toxic metals in mine wastes and mine waters, reusing them in the circular economy could minimize the negative environmental footprints of former mining activities. Therefore, developing eco-friendly techniques for making products from mine waste is an important problem for which innovation is required. In this study, a series of laboratory-scale fixed-bed ion-exchange experiments were conducted to evaluate chelating functional group of bis-picolylamine (Lewatit TP 220 resin) for selective recovery of cobalt and copper from mine waters generated from heap leaching waste materials at a Chilean copper mine. Considerable separation of cobalt and copper was observed during the loading process, and the resin showed no affinity towards earth alkali and alkali metal ions. Moreover, no significant reduction in resin capacity was observed in the second cycle.
Ammonium hydroxide (3.5%) led to the recovery of more than 80% of the copper with a peak concentration of 18g/L. During the second stage, cobalt concentrations were increased up to 5.7g/L. Overall, more than 96% of the cobalt in the mine water was recovered selectively, increasing the concentration nearly 190 times. Our findings emphasize optimized parameters and strategies to produce intermediate products of heavy metal solutions from mine wastes based on ion-exchange technology.
Keywords: Ion exchange, Fixed-bed columns; Copper mine waste; Cobalt and copper, Selective recovery; Sulfuric
Abbreviations: BV: Bed Volume; BV/H: Bed Volumes Per Hour; Cu: Copper; Co: Cobalt; Eq/L: Equivalent Per Liter; G/L: Gram Per Liter; IX: Ion Exchange; Kgs: Kilo Grams; MP-AES: Microwave Plasma Atomic Emission Spectrometer Mg/L: Milligram Per Liter; Ni: Nickel; TP 220: Lewatit Monoplus TP 220; PVC: Polyvinylchloride
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Introduction
Cobalt is globally recognized as a strategic and critical raw material due to its high-tech low carbon applications [1-3] . However, in terms of supply, cobalt is only found in potentially exploitable quantities in a few countries and is mainly produced as a co- or byproduct of the copper and nickel industry [4]. Due to revolutions in the rechargeable battery market, cobalt demand is expected to increase exponentially within the next decade [5].
Therefore, it is essential to find secondary sources and process mechanisms to overcome current mining limitations and increase the supply. On contrary, most of the mine wastes in the world has greatly exceeded the maximum permissible heavy metal concentration on their materials, suggesting many possible social and ecological threads to the future [6].
Due to the deterioration of aquatic environments, industries all over the world are forced to reduce the heavy metal content of their discharge streams to acceptable levels [7]. Therefore, making products from heavy metal waste is an important problem where innovation is required. Novel approaches should be developed so that the environmental footprint of modern industry is strongly reduced compared with the situation today.
Heap leaching material and tailings of copper and nickel mines could be potential sources for experimentation on the recovery of heavy metals as secondary sources. This study focuses on the selective recovery of cobalt and copper from mine water generated by heap leaching materials in the northern part of Chile. In the last century, nearly 150 million tons of untreated mine tailings were dumped directly onto the shoreline of the Pacific Ocean [8,9].
This has resulted in the formation of large tailings deposits in this area [10]. With time, the liberation of divalent metal cations has formed zones with enriched concentrations of metals such as Cu (II), Ni (II), Co (II), and Zn (II) [9]. Over the last two decades, many techniques have been implemented to recover dissolved metals from heavy metal pregnant solutions. Among them ion exchange (IX) technology has unique advantages such as, recovery of metal value, selectivity of ions, less sludge volume produced and the meeting of strict discharge specifications [11-13].
Recently chelating resins and solvent impregnated resins have brought up more attractive options to enhance the efficiency of ion exchange process in metal recycling. Nevertheless, many of these resins do not have higher affinity towards cobalt than the other heavy metals and are directly inadequate in their capability to selectively recover cobalt from complex water matrices [13-15].
Especially if the concentration of other elements is very high (e.g., Cu, Ni, and Zn), it might be best to first separate the complex solution, followed by selectively extract targeting specific metals with an additional step [16,17]. A proper extraction of metal ions with an IX dominated process could decrease the energy consumption and waste production in the mine water treatment, while minimize/prevent the formation of acid mine drainage. Nevertheless, these aspects are rarely investigated with complex water matrices and poorly understood.
Recent studies with iminodiacetic acid (Lewatit TP 207) and amino phosphonic acid (Lewatit TP 260) have shown that these two resins can remove low concentrations of copper, nickel, zinc, and lead in aqueous solutions [16,18,19]. But both these resins have very low affinity towards the cobalt. Therefore, in the presence of copper, zinc, nickel, with earth alkali elements, cobalt acquires a very small chance to get adsorbed on to the resin.
Littlejohn and Vaughan [15] studied the ability of chelating functional group of bis-picolylamine (Lewatit TP 220) for selective elution of nickel and cobalt from laterite leach tailings. They were able to effectively recover 90–95% of nickel and 75–85% of cobalt using 2–4 bed volumes of 6.8% w/w NH4OH solution. Since the elution kinetics were rapid, copper as zinc were also co eluted in the same bed volume ranges.
Thus, the purpose of this study is to optimize the process parameters for lab-scale production of selected modern chelating functional group of bis-picolylamine for selective removal and extraction of cobalt and copper from mine water generated from Chilean heap leaching materials. Proper separation of these metal ions could produce an intermediate product that could use as recycled metals in circular economic concept.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Experimental Setup
Chelating Ion Exchange Resin
Lewatit MonoPlus TP-220 (TP 220) is a resin with bispicolylamine (bis-(2-pyridylmethyl) amine) functional groups provided by LANXESS Deutschland GmbH. The specifications and properties of these resins are shown in Table 1. TP 220 uses bis-picolylamine functional groups in a matrix of polystyrene crosslinked with divinylbenzene. This resin type has been previously studied by many researchers for removal of selected metal ions from acidic solutions [20-22].
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Generation of the Mine Water
The material used in this study to generate mine water was collected from a heap-leached stockpile at a Chilean surface copper mine in the northern part of Chile. These materials were mixtures of coarse- to medium-grained (60%) and fine-grained (40%) ore chunks. They were already subjected to leaching with sulfuric acid to extract Cu (II) and then dumped into a stockpile without further treatment. This has led these materials to weather under normal atmospheric conditions over time.
Two types of mine waters were made by separately mixing each heap leaching waste material (ES2 and ES3) with doubly distilled water. Generally, 3.5Kgs of heap leaching waste materials and 4L of doubly distilled water were mixed. The mixture was stirred several times during the day and allowed to settle overnight. Then, the suspension was allowed to settle until a clear separation of materials and water was observed (Figure 1).
The pregnant heavy metal solution had a unique blue color, indicating a high Cu (II) concentration. It was possible to move nearly 3 L of water to a new container without agitating the settled-out materials below the water. Then, a 1:1 dilution was made to obtain a concentration that resulted in a better loading curve and daytime Cu (II) breakthrough point. In the middle of the first column experiments, additional synthetic water was generated using cobalt (CoSO4.7H2O) and copper (CuSO4.5H2O) salts. The final experiment (column B1 and B2) was conducted using Cu (II) free outflows separately extracted during the loading of column A1 and column A2. Compositions of the generated mine waters used for various tests are presented in (Table 2).

- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Apparatus
>A microwave plasma atomic emission spectrometer (MP-AES - 4200 Agilent) was used to determine cations in the samples. A photometer (HACH LANGE, DR 3900) was used to quickly measure Cu (II) concentrations. pH values, conductivities, and redox potentials of effluent samples were measured with a multimeter (WTW 3320). A Peristaltic pump (IPC- ISMATEC) was used to pump water into the columns.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Fixed Bed Column Experiments
Four column experiments were conducted in two stages (Table 3). In the first stage, column A1 and A2 were loaded with mine water to separate Cu (II) from Co (II). Then the Cu (II) free Co (II) slightly enriched solution that was used with column B1 and B2 to further selectively recover Co (II). A2 represents the second cycle of A1 column whereas B2 represents the second cycle of B1 column.
All ion exchange column trials were conducted using clear polyvinylchloride (PVC) columns with 1.5cm diameters and 12cm heights (Figure 2). Pipework and fittings were made of polyethylene. Each column contained 12mL of resin and was considered a one-bed volume (BV- the volume of wet settled resin in the column). The resins were soaked with slightly acidified (pH 4.5) doubly distilled water overnight to make them fully hydrated before they were inserted into the columns.

Feed mine water solutions were pumped through the column in the upward flow direction at a controlled rate of 3BV/h using a peristaltic pump. Effluent samples were collected at regular intervals, and cations were analyzed using MP-AES. Columns A1 and A2 were loaded until the exhaustion point of copper. Column B was loaded until the exhaustion point of cobalt. All exhaustion points were determined via spectrophotometry. After the loading cycle, the columns were backflushed with at least 4BVs of doubly distilled water at a flow rate of 3BV/h to eliminate surplus feed solution in the IX system. The pH of the distilled water was adjusted to 2 prior to use to avoid leakage of organic reagents.
Regenerant solutions contained either ammonium hydroxide (3.5% or 5%) or sulfuric acid (5% or 10%). Regeneration was performed through the system in co-flow mode and with the same flow rate. Again, effluent solutions were collected regularly for MP-AES analysis. In each case, bed volumes were based upon the initial volume of the resin loaded in the column. The regenerant concentration and flow rate ranges were chosen by consulting the resin product sheet provided by LANXESS.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Chemical Analysis
Samples were filtered through 0.45μm filters into 10mL plastic sample test tubes. The samples from the loading experiments were acidified using 0.1mL of nitric acid. Depending on the assumed Cu (II) concentration in the samples, 1:10, 1:100, 1:500, 1:1000, or 1:10000 dilutions were made for MP-AES analysis. The main reason for the dilution was to avoid the blockage of the torch and avoid destroying the nebulizer with the accumulation of salts [23].
Then, samples were stored at 4°C in a refrigerator until MP-AES analysis was performed. During the analysis, the concentrations of Al, Ca, Cd, Ce, Co, Cu, Cr, Fe, K, La, Mg, Mn, Na, Ni, Pb, Sr, Y, and Zn were measured. Random samples were also checked with a spectrophotometer to determine the Cu (II) concentration. The Cu (II) 8506 (CuVer 1) method was used in the photometric analysis.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Results and Discussion
First Loading Cycle
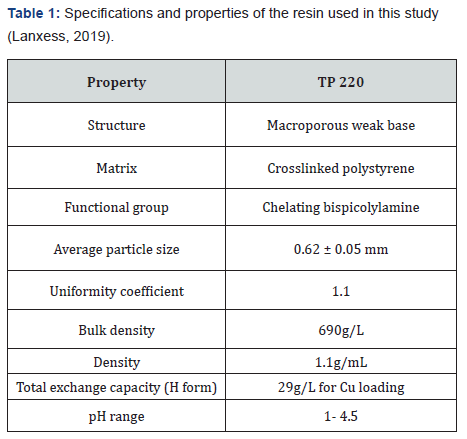
A column filled with 12mL of TP 220 resin (column A1) was loaded with mine water prepared from ES2 materials. The feed mine water had a Cu/Co ratio of 1:1. A flow rate of 36mL/h was used during the column loading process, which was equivalent to 3BV/h. It should be noted that synthetic water introduced to the column after 574BV had slightly different Co (II) and Cu (II) concentrations than real mine water. However, since the purpose of the test was to achieve a qualitative comparison of the resin performance with those from similar studies [16], the concentration difference was tolerable for the study.
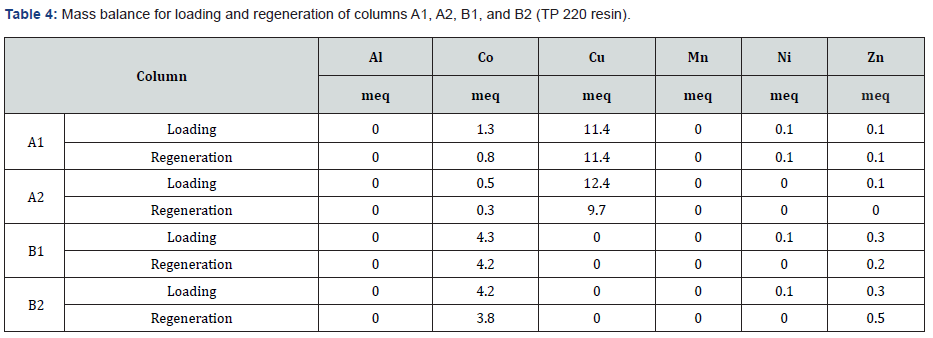
The breakthrough curve shown in (Figure 3a&b) plots the amount of the metal ions loaded on the resin against the bed volumes of feed mine waters passed through the column. According to the results, the affinities of TP 220 resin followed the order Cu> Ni> Zn> Co. As depicted in (Figure 3a&b), all metal ions were adsorbed until 230BV, and then accelerated breakthrough of cobalt reached exhaustion at approximately 400BV. It was subsequently observed that Ca, Mg, K, Na, Mn, and Al do not show any affinity toward the resin.
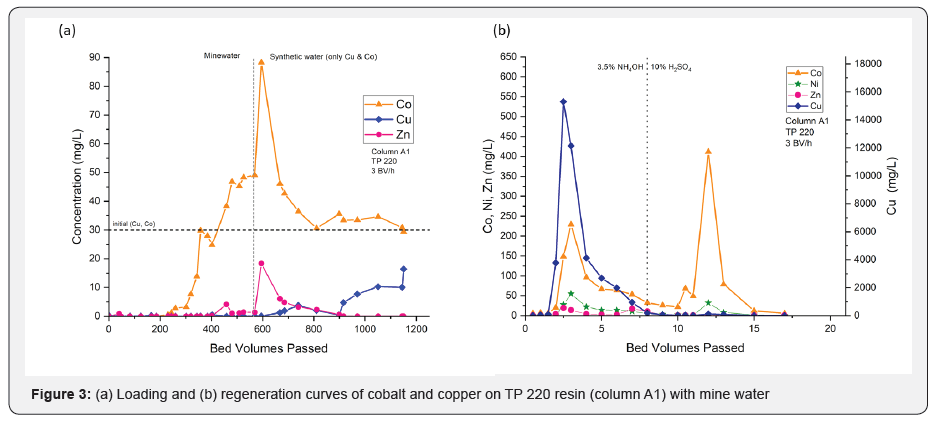
Soon after the feed changeover to the synthetic inflow, the ions with lower affinities (Co, Ni, and Zn) began to desorb substantially. Since one of the main objectives was to enhance the Co (II) and Cu (II) separation, switching solutions after the exhaustion of law affinitive ions could be further investigated as a tactical approach to enhance the ion separation during the loading process. Copper breakthrough started at approximately 600BV. Therefore, the loading process generated nearly 600BVs of Cu (II) r-free Co (II) solution that was slightly enriched and could be used to further refine cobalt.
However, photometric analyses only indicated the presence of copper in the effluent at 668BVs. Therefore, Cu (II) -free effluent was collected up to 668BV. According to the mass balance, 6.3meq of Co (II) had travelled through the column in the effluent at that time. This solution was later used in column B1 and B2 to further purify and extract Co (II). At the end of the loading (1151BV), the majority of available exchange sites appeared to have been occupied by copper (89%) and cobalt (10%) (Table 3). These values are in agreement with resin capacity data provided by Lanxess.
Regeneration was started with 7.5BVs of 3.5% ammonium hydroxide, followed by another 8BVs of 10% sulfuric acid, in an attempt to determine the utilities of the regenerants. The regeneration curve is shown in (Figure 3b). Cu (II) r started to elute with ammonium hydroxide and reached a peak concentration of 15.3g/L, while Co (II) reached a peak concentration of 0.2g/L. However, Ni (II) and Zn (II) were also coeluted with ammonium hydroxide. Once the regenerant solution was changed to 10% sulfuric acid, Co (II) was eluted again with a peak concentration of 0.4g/L and a minimal amount of Ni (II). According to mass balance, ammonium hydroxide removed most of the adsorbed Cu (II), and sulfuric acid did not elute any Cu (II). After the column was regenerated, it was conditioned with 5 bed volumes of 1% sodium hydroxide before the 2nd cycle.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Second Loading Cycle
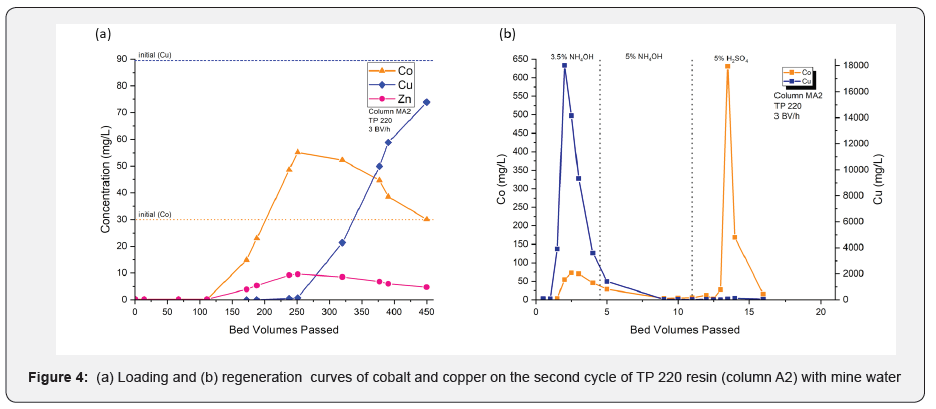
After conditioning, column A1 was again loaded (column A2) with MW3 water, which had a Cu/Co ratio of 3:1. Column A2 was also intended to load until the complete exhaustion of copper (Figure 4a&b). However, due to the limited availability of mine water, the test was terminated at 450BV. At the end of loading, Cu (II) utilized most of the resin capacity (> 98%). No significant difference in resin capacity was observed between the first and second loading cycles, suggesting that previously used regenerants did not make any reactions to clog the resin capacity. The mass balance showed that a higher Cu/Co ratio increased the amount of copper adsorption observed at the end of the loading process (Table 3).
Regeneration of column MA2 was started with 4.5 bed volumes of 3.5% ammonium hydroxide, followed by 5 bed volumes of 5% ammonium hydroxide and another 10 bed volumes of 5% sulfuric acid. According to the elution profiles, Cu (II) eluted with a peak concentration of 18g/L in 3.5% ammonium hydroxide solution. However, Co (II) coeluted with a peak concentration of 73mg/L.
It is interesting to note that changing the ammonium hydroxide concentration to 5% did not cause elution of any metal ions. Nevertheless, after changing the regenerant solution to 5% sulfuric acid at the 10thBV, Co (II) started to elute with a peak concentration of 0.7g/L. In the 2nd cycle, it was only possible to recover nearly 80% of the Cu (II) from the total adsorption. The elution profiles of columns A1 and A2 showed that 3.5% ammonium hydroxide removed more than 99% of the Cu (II) in 5-7 bed volumes. In addition, Co (II) was better eluted with 5% sulfuric acid after Cu (II) was eluted with 3.5% ammonium hydroxide.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Cobalt Enrichment Experiment
In the previous loading experiments involving TP 220 resins, almost 7 liters of Cu (II)-free Co (II)-enriched solution was produced. This water was loaded into a different column with TP 220 resin (column B) to concentrate the cobalt in the regeneration process. The loading was carried out with a flow rate of 3BV/h, and 5% sulfuric acid was used for regeneration. The experiment was continued for two cycles. Since both columns (B1 and B2) show similar results, only column B1 is depicted and discussed in the paper. (Figure 5a&b) shows the breakthrough curves of all ions available in mine water based on their initial concentration fractions (C/C0).
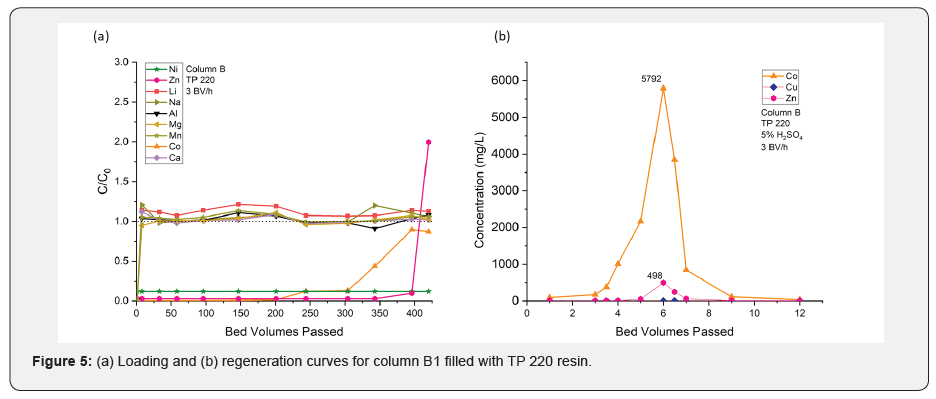
It can be clearly seen that neither alkali metals nor alkaline earth metals were adsorbed on the resin during the loading process. The column was mainly loaded with Co (II) and a smaller amount of Zn (II). The loading was terminated after 450BVs due to complete exhaustion of the cobalt (photometrically tested).
As iminodiacetic acid (e.g.-Lewatit TP 207) and amino phosphonic acid (e.g.-TP 260) show a considerable affinity towards earth alkali elements (Ca, Mg, and Al), some of the resin capacity is always lost due to the adsorption of unnecessary ions. As TP 220 did not show any affinity towards earth alkali and alkali elements most of the resin capacity can be used for the metal ion of interest According to mass balance, the column adsorbed 4.3meq cobalt and 0.3meq zinc. It should be noted that this only accounts for 35% of the operating capacity of TP 220 resin (Table 4) based on the results of the previous experiment.
Regeneration was continued at a fixed flow rate of 3BV/h, and eluant was collected at each half bed volume (Figure 3b). It was observed that the cobalt concentration increased with increasing bed volume and reached a peak concentration of 5.7g/L with 6 bed volumes. Then, the Co (II) concentration decreased, and elution was completed after 12 bed volumes. More than 96% of the Co (II) was recovered from the total adsorption, and the recovery also greatly enhanced with no earth alkali contaminants in the solution.
Nevertheless, a smaller amount of Zn (II) also coeluted with Co (II)and reached a maximum concentration of 0.5g/L. Recently, Vecino et al. [19] archived a better Zn (II) and Co (II) separation from an acidic mine water using Lewatit TP 207 and VP 1026 resin. Therefore, as the next step, additional column studies are planned to separate the Zn (II) and Co (II) aiming at producing an intermediate cobalt concentrate from the heap leaching mine waste.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Summary and Conclusion
These results demonstrate a positive outlook for utilization of Lewatit MonoPlus TP 220 (bis-picolylamine) resin for the selective recovery of cobalt and copper from mine water generated using heap leaching materials from a Chilean Cu mine. Loading mine water into the TP 220 resin at a flow rate of 3BV/h resulted in effective Cu-Co separation by adsorbing most of the copper onto the resin. Separate collection of the effluent produced before copper breakthrough enabled the recovery of a copper-free solution with a slightly enriched cobalt concentration, which was later used for further cobalt enrichment.
The rest of the effluent may well be recycled for Cu-Co separation. Switching the feed solution after the Co exhaustion point enhanced the Co-Cu separation and could be further investigated for the improve the metal ion separation by steering the mine water solutions at different levels of treatment. Regeneration of the lead column with 3.5% ammonium hydroxides resulted in desorption of all copper ions and generation of 15-18g/L peak concentrations.
In all cases, it was possible to recover more than 80% of the copper content during regeneration. However, cobalt, nickel and zinc were also coeluted. Once copper was removed, the TP 220 resin adsorbed more cobalt. Regeneration resulted in 96% cobalt recovery with a 5.7g/L peak concentration. This is a nearly 190-fold increase relative to the initial cobalt concentration in the mine water. Additional tests with column series combining different resin types are planned to further increase the concentration and enhance Cu-Co separation.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
Acknowledgments
The investigations were part of the SECMINTEC project funded by the Federal Ministry of Education and Research in Germany (BMBF) and the CLIENT program (022R186A). The authors would like to acknowledge LANXESS Deutschland GmbH for providing the resin samples. We also want to thank Chilean colleagues and companies for their cooperation. The authors thank Mr. Ozan Aydin and Mrs. Marlies Grimmer for their assistant in laboratory experiments and analyses.
- Research Articlec
- Abstract
- Introduction
- Experimental Setup
- Generation of the Mine Water
- Apparatus
- Fixed Bed Column Experiments
- Chemical Analysis
- Results and Discussion
- Second Loading Cycle
- Cobalt Enrichment Experiment
- Summary and Conclusion
- Declaration of Competing Interest
- Acknowledgments
- References
References
- Butu A, Butu M, Fidler G, Rodino S (2017) Recovery and Recycling of Critical Metals-A Strategic Priority for European Union. International Multidisciplinary Scientific GeoConference: SGEM 17(1.3): 725-732.
- Tkaczyk AH, Bartl A, Amato A, Lapkovskis V, Petranikova M, et al. (2018) Sustainability evaluation of essential critical raw materials: Cobalt, niobium, tungsten and rare earth elements. Journal of Physics D: Applied Physics 51(20): 203001.
- European Union (2017) Study on the review of the list of critical raw materials: Final report. Publications Office of the European Union.
- Olivetti EA, Ceder G, Gaustad GG, Fu X (2017) Lithium-Ion Battery Supply Chain Considerations: Analysis of Potential Bottlenecks in Critical Metals. Joule 1(2): 229-243.
- Beatty D, Fu X, Bustamante M, Gaustad G, Babbitt C, et al. (2019) Cobalt Criticality and Availability in the Wake of Increased Electric Vehicle Demand: A Short-Term Scenario Analysis. In: Gaustad G. et al. (eds) REWAS 2019. The Minerals, Metals & Materials Series. Springer, US, pp: 355-357.
- Kumar V, Parihar RD, Sharma A, Bakshi P, Singh Sidhu GP, et al. (2019) Global evaluation of heavy metal content in surface water bodies: A meta-analysis using heavy metal pollution indices and multivariate statistical analyses. Chemosphere 236: 124364.
- Jordan YC, Ghulam A, Hartling S (2014) Traits of surface water pollution under climate and land use changes: A remote sensing and hydrological modeling approach. Earth-Science Reviews 128: 181-195.
- Correa JA, Ramírez MA, Jean-Paul DLH, Román D, Rivera L, et al. (2000) Copper, Copper Mining Effluents and Grazing as Potential Determinants of Algal Abundance and Diversity in Northern Chile. Environmental Monitoring and Assessment 61(2): 267-283.
- Dold B (2006) Element flows associated with marine shore mine tailings deposits. Environmental Science & Technology 40(3): 752–758.
- Lee MR, Correa JA (2007) An assessment of the impact of copper mine tailings disposal on meiofaunal assemblages using microcosm bioassays. Mar Environ Res 64(1): 1-20.
- Abo-Farha SA, Abdel-Aal AY, Ashour IA, Garamon SE (2009) Removal of some heavy metal cations by synthetic resin purolite C100. Journal of Hazardous Materials 169(1-3): 190–194.
- Ali SW, Waqar F, Malik MA, Yasin T, Muhammad B, et al. (2013) Study on the synthesis of a macroporous ethylacrylate-divinylbenzene copolymer, its conversion into a bi-functional cation exchange resin and applications for extraction of toxic heavy metals from wastewater. Journal of Applied Polymer Science 129(4): 2234–2243.
- Deventer JV (2011) Selected ion exchange applications in the hydrometallurgical industry. Solvent Extraction and Ion Exchange 29(5-6): 695-718.
- Lanxess (2019) Lewatit-MonoPlus-TP-220-L.pdf. https://www. lenntech.com/Data-sheets/Lewatit-MonoPlus-TP-220-L.pdf.
- Littlejohn P, Vaughan J (2013) Recovery of nickel and cobalt from laterite leach tailings through resin-in-pulp scavenging and selective ammoniacal elution. Minerals Engineering 54: 14-20.
- Abeywickrama J, Hoth N, Ussath M, Drebenstedt C (2020) Selective Extraction of Cobalt and Copper from Chilean Mine Water by Ion Exchange Resin. Gornye Nauki i Tekhnologii = Mining Science and Technology (Russia) 5(1): 25-29.
- Nekouei RK, Pahlevani F, Assefi M, Maroufi S, Sahajwalla V, et al. (2019) Selective isolation of heavy metals from spent electronic waste solution by macroporous ion-exchange resins. Journal of Hazardous Materials 371: 389-396.
- Pedregal MAI, Abeywickrama J, Hoth N, Grimmer M, Drebenstedt C, et al. (2021) Modeling of Ion Exchange Processes to Optimize Metal Removal from Complex Mine Water Matrices. Water 13(21): 3109.
- Vecino BX, Reig i Amat M, López RJ, Valderrama ACA, et al. (2019) Recovery and separation of valuable metals (copper and zinc) from acidic mine waters by ion-exchange resins. 16th International Conference on Environmental Science and Technology CEST2019, 4-7 September 2019, Rhodes, Greece.
- Botelho AB, Espinosa DCR, Dreisinger D, Tenório JAS (2019) Effect of PH to recover cu(ii), ni(ii) and co(ii) from nickel laterite leach using chelating resins. Tecnologia Em Metalurgia Materiais e Mineração 16(1): 135–140.
- Kołodyńska D, Sofińska-Chmiel W, Mendyk E, Hubicki Z (2014) DOWEX M 4195 and LEWATIT® MonoPlus TP 220 in Heavy Metal Ions Removal from Acidic Streams. Separation Science and Technology 49(13): 2003-2015.
- Wołowicz A, Hubicki Z (2012) The use of the chelating resin of a new generation Lewatit MonoPlus TP-220 with the bis-picolylamine functional groups in the removal of selected metal ions from acidic solutions. Chemical Engineering Journal 197: 493-508.
- Ozbek N, Akman S (2016) Method development for the determination of calcium, copper, magnesium, manganese, iron, potassium, phosphorus and zinc in different types of breads by microwave induced plasma-atomic emission spectrometry. Food Chemistry 200: 245-248.






























