A Multi-Disciplinary Management of a Rare Case of a Complete Lung Congenital Pulmonary Airway Malformation in a Newborn from a Developing Country
Wasif Ilyas Vohra1, Muhammad Shakir1, Rubaid Azhar Dhillon2*, Omar Irfan1,3 and Adnan Mirza1,
1The Aga Khan University, Department of Pediatrics, Pakistan
2Riphah Medical College, Pakistan
3Amaris Consulting, Canada
Submission: November 18, 2022; Published: November 29, 2022
*Corresponding author: Rubaid Azhar Dhillon, Riphah Medical College, Pakistan
How to cite this article: Wasif I V, Muhammad S, Rubaid Azhar D, Omar Irfan, Adnan M. A Multi-Disciplinary Management of a Rare Case of a Complete Lung Congenital Pulmonary Airway Malformation in a Newborn from a Developing Country. Int J Pul & Res Sci. 2022; 6(2): 555683. DOI: 10.19080/IJOPRS.2022.06.555683
Abstract
Congenital pulmonary airway malformation (CPAM) is a pulmonary malformation in which parts of the lung are replaced by hamartomatous, cystic, or non-functional abnormal tissue. Herein, we present a case of life-saving pneumonectomy in an infant with type II CPAM which affected entire right lung. A 1-day-old male infant who was a known case of CPAM presented with progressive respiratory distress requiring intubation. There is a paucity of evidence regarding the management protocol of entire lung CPAM. After multi-disciplinary input, right-sided pneumonectomy was performed. Thereafter, he had a prolonged, complicated hospital stay due to prolonged intubation, complicated by pneumonia, sepsis, and metabolic abnormalities. In this report we discuss the etiology, surgical procedure, management strategies, challenges faced in decision making of management, as well as potential complications, which make this case report a valuable asset to the scientific literature. Our case report contributes towards the scare literature present with regards to management of CPAM in the presence of respiratory distress. After receiving appropriate post-operative care, the patient was eventually discharged home in a stable condition. With the advancements in modern obstetric practice, CPAM is detected prenatally and early surgical resection of the affected area is considered the best management option in symptomatic cases of CPAM. A multidisciplinary approach with CPAM diagnosed prenatally and early surgical resection of the affected area is the gold standard evidence-based management protocol that exists.
Keywords: Congenital pulmonary airway malformation; Pneumonectomy; Respiratory distress; Pneumonia; Sepsis
Abbreviations: CPAM: Congenital Pulmonary Airway Malformation; BPS: Bronchopulmonary Sequestration RDS: Respiratory Distress; NICU: Neonatal Intensive Care Unit; CT: Computed Tomography; PDA: Persistent Ductus Arteriosus
Introduction
Congenital pulmonary airway malformation (CPAM) can present itself from the prenatal period all the way to childhood. This has an estimated frequency of one in 30,000 live births [1]. Due to abnormalities during embryogenesis, CPAM can develop at any stage during lung development. However, due to advancements in technology, the malformation can be diagnosed antenatally [1]. Ultrasonography is essential in getting a prenatal diagnosis and classifying it as microcystic or macrocystic [2]. Furthermore, when considering CPAM, MRI is conducted to rule out the differential diagnosis of bronchopulmonary sequestration (BPS) [2]. Symptoms usually begin to appear later in childhood, but sometimes respiratory distress (RDS) may develop in symptomatic neonates. Surgical resection is considered the best modality in the management of symptomatic disease. There is a small chance of malignant transformation in CPAMs (0.7%), which makes it important to establish an early diagnosis in asymptomatic patients as well [1]. Based on histological features and gross examination, CPAM has been classified into five subtypes [3]. In this case report, we present a full-term infant with extensive type II CPAM complicated by the development of pneumothorax who underwent right-sided pneumonectomy.
Case Presentation
The case is a male infant who was delivered at 37 weeks of gestation without any complications via elective lower segment caesarean section. He had a total birth weight of 2.6kg with an APGAR (appearance, pulse, grimace, activity, respiration) score of 9 at 1 and 5 minutes of age. The mother, gravida 3 para 2+0, had no known comorbidities throughout pregnancy. However, prenatal ultrasonography at gestational age of 22+0 weeks revealed polycystic lesions on the right lung of the fetus accompanied by a mediastinal shift and diaphragm inversion. On presentation at day 1, the baby was tachypneic with subcostal and intercostal recession. The neonate was intubated and shifted to the Neonatal Intensive Care Unit (NICU). A chest X-ray was carried out which showed multiple ill-defined lucent areas in the right lung fields, causing a mass effect resulting in mediastinal shift towards the left (Figure 1).
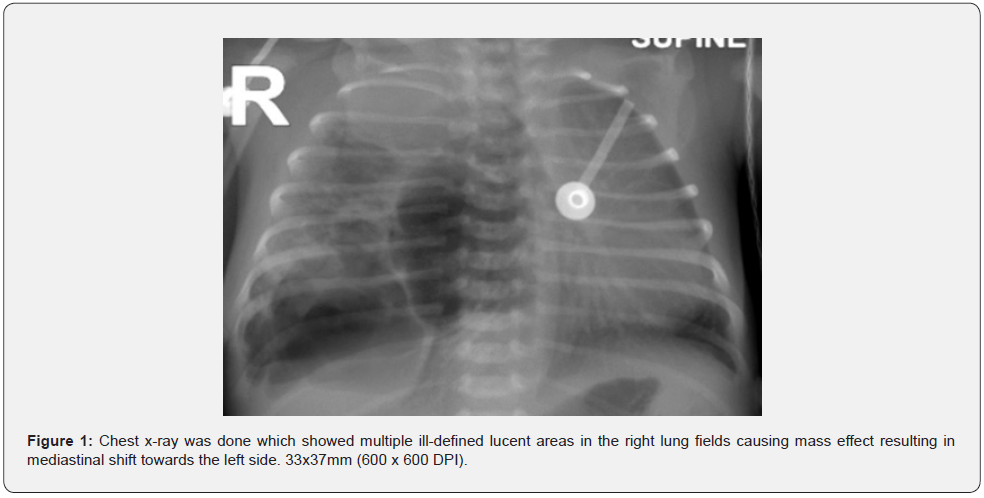
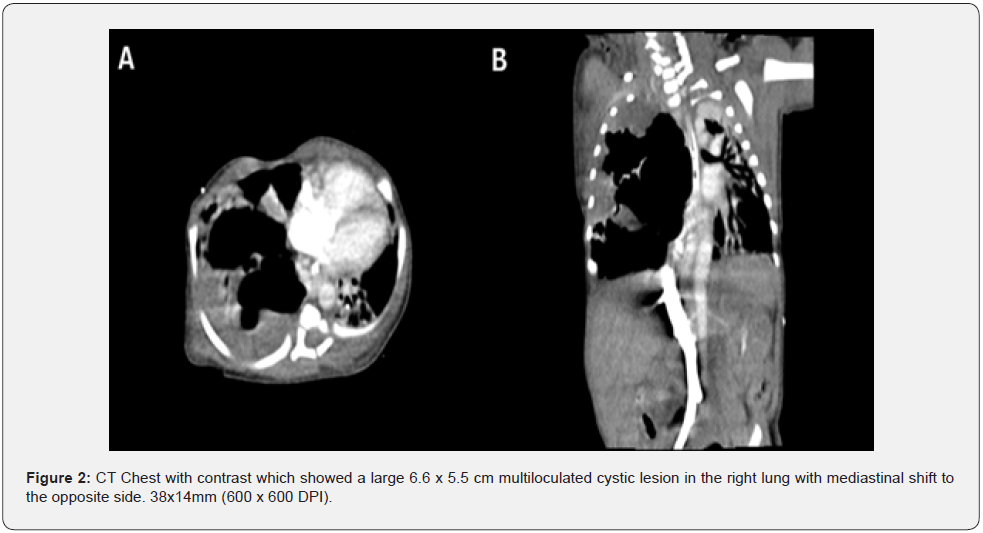
This was suggestive of CPAM. However, a thoracic ultrasound was also conducted to confirm this diagnosis. Due to deterioration in blood gases and failed extubation on the 4th day of admission, the Cardiothoracic Surgery team was contacted. A pneumonectomy was advised after a right-sided thoracotomy revealed the entire lung parenchyma to be replaced by an adenomatoid process. A Computed Tomography (CT) scan of the chest with contrast was obtained for pre-operative evaluation of lung anatomy, which showed a large 6.6 x 5.5 cm multi-loculated cystic lesion in the right lung with mediastinal shift to the opposite side (Figure 2). In addition to this, since the lesion was occupying approximately more than 20% of the pneumothorax, surgical resection is generally considered the treatment of choice [2].
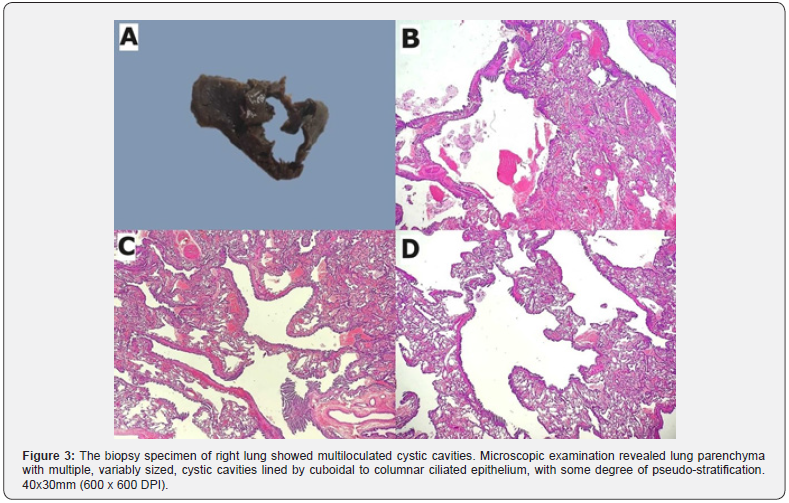
The biopsy specimen of the right lung showed multiloculated cystic cavities (Figure 3). Microscopic examination revealed lung parenchyma with multiple, variable sized, cystic cavities lined by cuboidal to columnar ciliated epithelium with some degree of pseudo-stratification. This bronchiolar type of epithelium showed intervening alveoli. There was no evidence of mucinous epithelium or any other mesenchymal component such as blastoma components or cartilage. These features were suggestive of type II congenital pulmonary adenomatoid malformation. An attempt was made to deliver the lung through the thoracotomy incision. However, this caused hemodynamic disturbance. After clamping the hilum, the procedure was carried out, and an improvement was seen in arterial blood gases. Following the procedure, the infant was kept in a negative balance, serial blood gases were monitored, and ventilator parameters were adjusted accordingly.
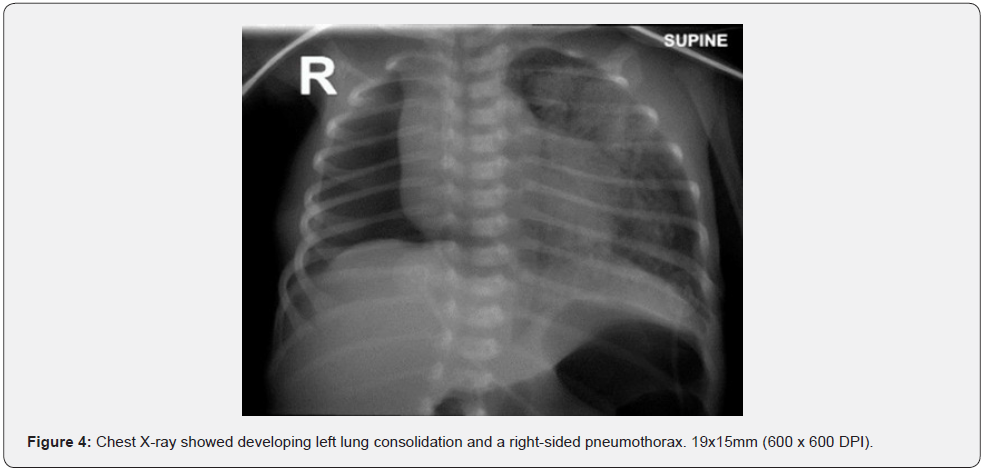
On the 6th day of life, an echocardiogram was conducted due to persistent tachycardia. The echocardiogram showed a small patent foramen ovale and persistent ductus arteriosus (PDA). The neonate was given propranolol for tachycardia, which slowly resolved. Following pneumonectomy, the neonate was extubated but developed pneumonia and septicemia. Pathology results showed the growth of Pseudomonas aerogenosa in samples obtained from tracheal aspirates and blood cultures. The neonate was resuscitated since no improvement was observed after giving antibiotics. A chest X-ray was conducted, which revealed a developing opacification on the left lung and a right-sided pneumothorax (Figure 4). After suspicion of CPAM in the left lung, CT scans were carried out, revealing two cysts which were most likely representative of CPAM. Upon a multidisciplinary discussion, it was decided to closely monitor the radiological changes. After some time, it was revealed to be an underlying pulmonary interstitial emphysema, which slowly resolved. The condition slowly stabilized as the neonate was extubated on the 30th day of life. The baby was sent home with 0.25 liters of oxygen administered through nasal prongs and IV meropenem. At the last follow-up of the patient, he was feeding well and experiencing normal growth along with minor respiratory symptoms.
Discussion
CPAM is an embryologic developmental disorder that commonly manifests as progressive respiratory distress in newborns or recurrent respiratory tract infections in children under the age of two years [2]. A study conducted by Ruchonnet-Metrailler et al. reported that only 25% of CPAM patients exhibit symptoms at birth and of those who do have symptoms, 25% are respiratory symptoms, where 11% of patients require mechanical ventilation and 13% most often require inhaled oxygen [4]. These respiratory symptoms may arise due to the presence of cystic lesions in the lung, mediastinal shift to the opposite side, pneumothorax, and inverted diaphragm [4]. CPAM most often affects only a certain lobe of the lung, and very few cases have been reported in literature discussing the involvement of the entire lung [5]. In our case report, several problems arose during management due to the extent of the disease.
CPAM is considered a hamartomatous congenital lung lesion which halts broncho-alveolar maturation in the first trimester and leads to overgrowth of mesenchymal cells, causing abnormal pulmonary tissue proliferation at various sites [2]. It has been characterized as five types of lesions. Type 0 is generally incompatible with life and type 1 is characterized by single, or multiple, large (more than 2cm) cysts lined with ciliated pseudo-stratified columnar epithelium which can cause mediastinal herniation. Type 2 has the worst prognosis, with numerous sub-centimeter cysts lined with ciliated cuboidal to columnar epithelium. Type 3 is characterized by a solid lung mass composed of numerous small cysts, whereas type 4 is distinguished by large peripheral cysts with thin walls lined by type 1 and type 2 pneumocytes [1, 2].
In neonatal respiratory failure, the definitive way to establish a diagnosis is through a chest X-ray. However, if multiple cystic lesions are reported on X-ray, the appearances can easily be confused with congenital diaphragmatic hernia, pulmonary sequestration, or bronchogenic cysts [5]. A thoracic ultrasound and CT scan provide a clearer diagnosis of CPAM. However, due to time constraints, a thorough analysis is not possible and in the post-operative stage, it may cause anxiety if further lesions are suspected. This was the case in our patient when left lung involvement was also suspected on CT scans. Antenatal ultrasonography can be helpful in establishing a prenatal diagnosis, especially when conducted at 16 to 22 weeks of gestation [5].
According to a multicentric study conducted in Japan, it was reported that on prenatal examination, lesions produced by bronchial atresia and bronchopulmonary sequestration are generally much smaller as compared to CPAM lesions. In addition to this, there is a reduced tendency for CPAM lesions to shrink towards the end of gestation [6]. For this reason, in CPAM patients, the normal lungs in neonates undergo pulmonary hypoplasia and develop pulmonary hypertension, which leads to respiratory issues [6]. This may explain why our patient developed respiratory distress despite being born at full term with no prenatal complications in either the mother or the fetus. The development of pulmonary hypertension may have contributed to the tachycardia that we observed in the patient. The echocardiogram showed a small patent foramen ovale and persistent ductus arteriosus. Due to the increased pulmonary vascular resistance, right to left shunting via the ductus arteriosus may have occurred, leading to hypoxemia and acidosis. Based on the pathophysiology, CPAM lesions tend to become infected due to the communication established between the normal airways and the cystic lesions [7]. Due to this communication, lesions can easily become infected by the deposition of external pathogens. Hence, this is the reason why our patient developed pneumonia and sepsis post-operatively.
Early surgical resection of the affected area is considered the best management option in symptomatic cases as well as asymptomatic cases [8]. It has been reported by studies that asymptomatic lesions tend to get infected later on in life in 23–89% of the cases [9, 10]. The resection should be clear of any remnants to promote the development of normal lung tissue. During surgery, the cystic lesion has the potential to become enlarged due to the influx of air from positive pressure ventilation and hence, pulmonary resection should be performed as soon as possible without further delay [7]. Some guidelines recommend performing total pneumonectomy in stages to prevent complications such as right pneumonectomy syndrome, reported in a case by Stolar et al. [11]. Moreover, post-operative mechanical ventilation must be carefully monitored to prevent injury to susceptible lungs, especially in infants [11]. A widespread or bilateral disease and fetal hydrops are considered important factors indicating a poor prognosis [12].
Conclusion
There are only a handful of cases reported with extensive disease that required pneumonectomy for CPAM in the neonatal period. Major lung resections, especially pneumonectomy, at neonatal age are associated with potential post-operative complications such as significant pulmonary hypertension, prolonged ICU course, and a complicated hospital stay [8]. Our case report describes a successful pneumonectomy in a neonate for entire lung CPAM despite the development of complications which were resolved through a multidisciplinary approach. Furthermore, there is no definitive protocol of care available for patients in the neonatal age group who undergo a pneumonectomy procedure. Our patient had a prolonged, complicated hospital course and developed sepsis, but eventually was discharged home in stable condition after receiving appropriate management.
References
- Bolde S, Pudale S, Pandit G, Ruikar K, Ingle SB (2015) Congenital pulmonary airway malformation: A report of two cases. World J Clin Cases 3(5): 470-473.
- Akcan AB, Ilgun D, Ozkisacik S, Erdem AO, Meteoglu I, et al. (2020) Should Come to Mind with a Newborn with Respiratory Distress-Congenital Cystic Adenomatoid Malformation? Acibadem University Journal of Health Sciences 11(1): 167-170.
- Stocker JT, Madewell JE, Drake RM (1977) Congenital cystic adenomatoid malformation of the lung: classification and morphologic spectrum. Hum Pathol 8(2): 155-171.
- Ruchonnet-Metrailler I, Leroy-Terquem E, Stirnemann J, Cros P, Ducoin H, et al. (2014) Neonatal outcomes of prenatally diagnosed congenital pulmonary malformations. Pediatrics 133(5): e1285-e1291.
- Prendergast B, Fernando AM, Mankad PS (1998) Congenital cystic adenoid malformation in a pre-term infant: management considerations. Pediatr Surg Int 14(1-2): 92-93.
- Kuroda T, Nishijima E, Maeda K, Fuchimoto Y, Hirobe S, et al. (2016) Perinatal features of congenital cystic lung diseases: results of a nationwide multicentric study in Japan. Pediatr Surg Int 32(9): 827-831.
- Usui N (2020) Surgical Treatment for and Prognosis of Congenital Pulmonary Airway Malformation. Congenital Cystic Lung Disease Pp: 63-71
- Ben-Ishay O, Nicksa GA, Wilson JM, Buchmiller TL (2012) Management of giant congenital pulmonary airway malformations requiring pneumonectomy. Ann Thorac Surg 94(4): 1073-1078.
- Wong A, Vieten D, Singh S, Harvey JG, Holland AJ (2009) Long-term outcome of asymptomatic patients with congenital cystic adenomatoid malformation. Pediatr Surg Int 25(6): 479-485.
- Durell J, Thakkar H, Gould S, Fowler D, Lakhoo K (2016) Pathology of asymptomatic, prenatally diagnosed cystic lung malformations. J Pediatr Surg 51(2): 231-235.
- Stolar C, Berdon W, Reyes C, Dillon P, Collins M, et al. (1988) Right pneumonectomy syndrome: a lethal complication of lung resection in a newborn with cystic adenomatoid malformation. J Pediatr Surg 23(12): 1180-1183.
- Ierullo AM, Ganapathy R, Crowley S, Craxford L, Bhide A, et al. (2005) Neonatal outcome of antenatally diagnosed congenital cystic adenomatoid malformations. Ultrasound Obstet Gynecol 26(2): 150-153.






























