NG2 Proteoglycan in the Diagnosis, Prognosis and Therapy of Gliomas
Davide Schiffer*, Laura Annovazzi, Enrica Bovio and Marta Mellai
Research Center, Policlinico di Monza Foundation, Italy
Submission: February 23, 2017; Published: April 27, 2017
*Corresponding author: Davide Schiffer, Research Center, Policlinico di Monza Foundation, Vercelli, Italy, Tel: +39 -0161-3691;Fax: +39-0161-369109; Email: davide.schiffer@unito.it
How to cite this article: Schiffer D, Annovazzi L, Bovio E, Mellai M. NG2 Proteoglycan in the Diagnosis, Prognosis and Therapy of Gliomas. Int J cell Sci & mol biol. 2017; 2(2) : 555582. DOI : 10.19080/IJCSMB.2017.02.555582
Abstract
The review concerns the state of art of NG2 proteoglycan from its first description to its utilization in glioma therapy. NG2 protein is expressed during development in glia cells committing themselves to a differentiation. It regulates cell proliferation, migration, invasion and neuronal function through a crosstalk with neurons by its extracellular domain. It marks the oligodendrocyte precursor cells, differentiate into mature oligodendrocytes, and also astrocytes. It is expressed also in pericytes and it is involved in their relationship with that endothelial cells.
NG2 distribution in normal central nervous system and in gliomas is discussed, together with its association with Olig2 and PDGFRa.
Recently, it became the focus of attention as a therapeutic target. Various attempts have been made using monoclonal antibodies or RNAi, both in animals and in cell lines. Its ablation resulted in the reduction of glioblastoma cell viability. In children gliomas NG2 distribution is different, but children's brains are very rich in NG2 cells, so that the possibility of a tumor prevention might be hypothesized.
Keywords:NG2; Gliomas; Diagnosis; Therapy
Introduction
Glia cells expressing chondroitin sulfate proteoglycan 4 (CSPG4) are called NG2 (neuron-glia antigen 2) cells and occur in developing as well as in adult brain. They are oligodendrocyte progenitor cells (OPCs) that differentiate into mature oligodendrocytes. Beside supporting neurons and synaptic signaling, they play a role in gliomagenesis and progression and, therefore, they appear as a potential target for diagnosis and therapy of gliomas [1]. In the past, the problem was merely a curiosity, that of glycosaminoglycans in human and in ENU transplacentally induced gliomas [2-4], but today with the description of NG2, Olig2 and platelet-derived growth factor receptor alpha (PDGFRα), it is refined pinning on the origin of gliomas and their therapy.
Discussion
NG2 protein, highly expressed during development and down-regulated during differentiation, beside glia cells, is expressed in pericytes, independently of vasculogenesis and angiogenesis, giving a peculiar significance to their relationship with endothelial cells [5]. NG2 protein is a membrane proteoglycan consisting of a large extracellular, a transmembrane and a short cytoplasmic domain, important for NG2 function [6]. It is activated by ligands through focal adhesion kinase(FAK) and mitogen-activated protein kinase(MAPK) and it regulates cell proliferation, migration, invasion and neuronal function [7] co-acting with PDGFRα. NG2 cleavage by the α-secretase ADAM10 produces in an ectodomain in the extracellular matrix and a C-terminal fragment that is further processed by the γ-secretase to release an intracellular domain. There would be a bi-directional crosstalk between OPCs and the surrounding neuronal network: a novel physiological role for OPCs in regulating information processing at neuronal synapses [8], being the extracellular domain endowed with neuromodulatory properties [9].
NG2 marks OPCs together with PDGFRα; these cells are distinct from astrocytes and mature oligodendrocytes and for them the term polydendrocytes has been coined [10]. They arise from foci in the wall of the lateral ventricle, including the neurogenic niches of subventricular zone (SVZ) and the hippocampal dentate gyrus [11-13], but it remains to be established whether they give origin to all of oligodendrocyte lineages. They occur, therefore, in the gray and white matter in all brain regions in postnatal development and in adulthood [14]. In the postnatal and adult brain, NG2-glia represents the largest population of endogenous/resident progenitor cells (4-8% of total cells, depending on the brain region), rapidly reacting, to any type of injury and with a strong potential to repopulate areas of lesion [15-19]. NG2-glia morphology and its functional properties (e.g. membrane channels and receptors) have also been found to be distinct in different brain regions [20-21] as well as the interactions between NG2-glia and other neural cell types [22-25].
Our knowledge on the distribution and functions of NG2 has tremendously increased, but many unresolved questions remain [26]. For example, it is known that, depending on the developmental stage and the brain area, NG2 cells can also produce astrocytes [27-28] or they remain as self-renewing NG2-glia [16]. It has been discussed whether the differentiation of NG2 cells depend on the microenvironment that could condition their ability to form oligodendrocytes or astrocytes [27]. NG2 cells can originate GFAP- and A2B5+ type 2 astrocytes developing later than type 1 astrocytes. Of particular interest is the behavior of NG2 cells as reactive glia after injuries, demyelination, neurodegeneration and as carriers of voltage- and ligand-activated channels [26]. NG2 would not be expressed by multipotent stem cells, but it is up-regulated when stem cells commit themselves to a particular cellular lineage. It is then strongly expressed on partially-committed progenitors that are still proliferative, motile and retain a certain degree of developmental plasticity. Upon terminal differentiation of these progenitors, NG2 expression is down-regulated [6].
Gliomas. NG2+ OPCs have been considered as precursors in glioma development in the adult [29-31]. Only one daughter cell inherits NG2 [30], that is co-expressed with PDGFα for tumor transformation. It is not a marker for a specific cell type, but for an "activated" (as opposed to quiescent) status of cells [6]. Diffuse gliomas express markers characteristic of OPCs, such as NG2, PDGFRα and Olig2 [32-34]. OPCs remain widespread in the adult brain [35,36] and they represent the most abundant population of cycling cells in the adult central nervous system (CNS) [35,37] as a pool of cells in which accumulating mutations can lead to gliomagenesis [6,38], as it has been observed in glioma models [39-41]. As a matter of fact, NG2 correlates with the degree of malignancy of gliomas [29,32,42,43].
No pattern of co-expression of NG2, Olig2 and PDGFRα has till now been found in glioblastoma (GB), so that it is not known whether the co-expression pattern found in the CNS is recapitulated in GB [44,45]. NG2 expression is widespread in murine and variable in human gliomas [44,45]. Conflicting results were obtained for the distribution of Olig2 [12,46], whereas PDGFRα is regularly expressed in II-IV grade gliomas and amplified in GB [47]. Attempts to obtain diagnostic tools [29,48] or to conclude on the GB histogenesis [48] have been made. All the observations are in line with the origin of most gliomas from the subcortical white matter containing oligodendrocyte precursors expressing NG2, PDGFRa and Olig2, recruited experimentally in animals growth factors and in human gliomas [41].
As in normal tissue, NG2 is strongly expressed in pericytes of tumor vasculature with a disruption of the relationship with endothelial cells [32,42,49], even to form tubes without endothelial cells [50]. It has a role in blood vessel development and glioma progression; in its absence the proliferation of pericytes is reduced [51] and the recruitment of endothelial cells is affected [52]. However, in other's experience it has been observed that all PDGFRβ+ pericytes are NG2+, but a small number of NG2+ cells express PDGFRβ [53].
NG2 cells are found in gliomas conditioning a poor survival. In GB, NG2 would favor cell migration by binding to vascular collagen VI [54] and in pericytes it would favor angiogenesis by sequestering angiostatin [32]. In pediatric gliomas, however, the role of NG2 is not sufficiently known [1].
The main function of NG2 is undoubtedly the promotion of cell proliferation and motility, via integrins and growth factors [6]. As for cell motility, beside collagen VI and laminin 2, the contact itself with vessels and neuronal processes seems to be important [55]. In cell proliferation, NG2 intervenes binding fibroblast growth factor 2 (FGF2) and PDGF-AA [56]; as a matter of fact, using anti-NG2 antibody, the proliferation of OPCs is inhibited [6]. The effect on survival would also be supported by α3βiintegrin and by the phosphoinositide-3-kinase-protein kinase (PI3K)/Akt pathway [6,43]. NG2-transfected U251 glioma cells are resistant to treatment with tumor necrosis factor alpha (TNFα) and doxorubicin, etoposide and vincristine [43], There is a relationship between NG2 expression and apoptosis resistance.
By NG2 as biomarker, a NG2+ cell population has been identified with strong proliferative, clonogenetic and tumorigenetic capacity. It has been shown that 83% of proliferating cells are NG2+ and 50% of NG2+ cells proliferate. NG2+ GB over express genes of mitosis and cell cycling [53].
Recently, therapies based on monoclonal antibodies (mAbs) have gained in popularity given their clinical and commercial success for a variety of malignant diseases [57]. This approach has been applied especially in breast cancer and metastatic melanoma therapy. In gliomas, by targeting NG2 cells by antibodies or RNAi, apoptosis, reduced angiogenesis and invasion have been obtained [58-60]. In experiments on GB animal models, targeting NG2 with the monoclonal antibody mAb9.2.27 and activated natural killer (NK) cells abrogated the tumor growth and prolonged the survival of animals by favoring the establishment of a pro-inflammatory microenvironment [59,60]. Ablating NG2 and GD3A, a ganglioside expressed on developing migratory glia, using a Mab-Zap saporin immunotoxin system, resulted in significant reduction in GB cell viability compared to single epitope targeting and controls [61] In Ethylnitrosourea (ENU) transplacentally induced gliomas of the rat, NG2 was found to be diffusely distributed in oligodendrogliomas and not in cells differentiating into mature oligodendrocytes [62].
All these procedures would be of particular interest in pediatric gliomas, because the child's brain contains a large number of OPCs expressing NG2 [63]. An interesting study demonstrated that 14 mAbs, directed against the ectodomain of human NG2, specifically identify NG2 isoforms in foetal brain and GB tissue, while they are absent in the adult brain [64]. Interestingly, one isoform seems to be exclusive of tumor cells. These results allow to discriminate between phenotype-specific and neoplastic versus non-neoplastic variants of proteoglycans, offering the possibility for more selective immunotherapeutic targeting of brain tumors (Figure 1).
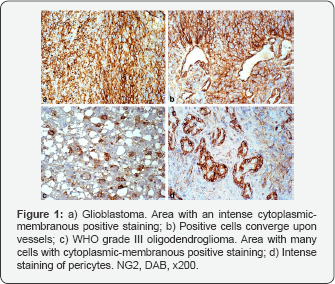
Conclusion
Till now experiments in animals demonstrated the efficacy of NG2 blocking in reducing tumor cell viability and animal survival. The experimentation did not yet transfer to the clinical level, but, since the premises, the possibility that this will happen soon is high.
Acknowledgements
This work was supported by Cassa di Risparmio di Vercelli Foundation.
References
- Yadavilli S, Hwang EI, Packer RJ, Nazarian J (2016) The Role of NG2 Proteoglycan in Glioma. TranslOncol 9(1): 57-63.
- Bertolotto A, Goia L, Schiffer D (1986) Immunohistochemical study of chondroitin sulfate in human gliomas. ActaNeuropathol 72(2): 189196.
- Mauro A, Bertolotto A, Giordana MT, Magrassi ML, Migheli A, et al. (1983) Biochemical and histochemical evaluation of glycosaminoglycans in brain tumors induced in rats by nitrosourea derivatives. J Neurooncol 1(4): 299-306.
- Giordana MT, Bertolotto A, Mauro A, Migheli A, Pezzotta S, et al. (1982) Glycosaminoglycans in human cerebral tumors. Part II. Histochemical findings and correlations. ActaNeuropathol 57(4): 299-305.
- Ozerdem U, Grako KA, Dahlin-Huppe K, Monosov E, Stallcup WB, et al. (2001) NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn 222(2): 218-227.
- Stallcup WB, Huang FJ (2008) A role for the NG2 proteoglycan in glioma progression. Cell AdhMigr 2(3): 192-201 Adh, Migr.
- Stallcup WB (2002) The NG2 proteoglycan: past insights and future prospects. J Neurocytol 31(6-7): 423-435.
- Sakry D, Neitz A, Singh J, Frischknecht (2014) Oligodendrocyte precursor cells modulate the neuronal network by activity-dependent ectodomain cleavage of glial NG2. PLoSBiol 12(11): e1001993.
- Sakry D, Totter J (2016) The role of the NG2 proteoglycan in OPC and CNS network function.Brain Res 1638 (Pt B): 161-166.
- Nishiyama A, Watanabe M, Yang Z, Bu J (2002) Identity, distribution, and development of polydendrocytes: NG2-expressing glial cells. J Neurocytol 31(6): 437-455.
- Aguirre A, Gallo V (2004) Postnatal neurogenesis and gliogenesis in the olfactory bulb from NG2-expressing progenitors of the subventricular zone. J Neurosci 24(46): 10530-10541.
- Aguirre AA, Chittajallu R, Belachew S, Gallo V (2004) NG2-expressing cells in the subventricular zone are type C-like cells and contribute to interneuron generation in the postnatal hippocampus. J Cell Biol 165(4): 575-589.
- Passlick S, Grauer M, Schäfer C, Jabs R, Seifert G et al. (2013) Expression of the γ2-subunit distinguishes synaptic and extrasynaptic GABA(A) receptors in NG2 cells of the hippocampus. J Neurosci 33(29): 12030 40.
- Nishiyama A, Komitova M, Suzuki R, Zhu X (2009) Polydendrocytes (NG2 cells): multifunctional cells with lineage plasticity. Nat Rev Neurosci 10(1): 9-22.
- Aguirre A, Dupree JL, Mangin JM, Gallo V (2007) A functional role for EGFR signaling in myelination and remyelination. Nat Neurosci 10(8): 990-1002.
- Simon C, Götz M, Dimou L (2011) Progenitors in the adult cerebral cortex: cell cycle properties and regulation by physiological stimuli and injury. Glia 59(6): 869-881.
- Whittaker MT, Zai LJ, Lee HJ, Pajoohesh GA, Wu J, et al. (2012) GGF2 (Nrg1-ß3) treatment enhances NG2+ cell response and improves functional recovery after spinal cord injury. Glia 60(2): 281-294.
- Filous AR, Tran A, Howell CJ, Busch SA, Evans TA, et al. (2014) Entrapment via synaptic-like connections between NG2 proteoglycan+ cells and dystrophic axons in the lesion plays a role in regeneration failure after spinal cord injury. J Neurosci 34(49): 16369-16384.
- Scafidi J, Hammond TR, Scafidi S, Ritter J, Jablonska B, et al. (2014) Intranasal epidermal growth factor treatment rescues neonatal brain injury. Nature 506(7487): 230-234.
- Chittajallu R, Aguirre A, Gallo V (2004) NG2-positive cells in the mouse white and grey matter display distinct physiological properties. J Physiol 561(Pt 1): 109-122.
- Bakiri Y, Attwell D, Karadottir R (2009) Electrical signalling properties of oligodendrocyte precursor cells. Neuron Glia Biol 5(1-2): 3-11.
- Wigley R, Hamilton N, Nishiyama A, Kirchhoff F, Butt AM (2007) Morphological and physiological interactions of NG2-glia with astrocytes and neurons. J Anat 210(6): 661-670.
- Wu J, Yoo S, Wilcock D, Lytle JM, Leung PY, et al. (2010) Interaction of NG2(+) glial progenitors and microglia/macrophages from the injured spinal cord. Glia 58(4): 410-422.
- Hammond TR, Gadea A, Dupree J, Kerninon C, Nait OB, et al. (2014) Astrocyte-derived endothelin-1 inhibits remyelination through notch activation. Neuron 81(3): 588-602.
- Miron VE, Boyd A, Zhao JW, Yuen TJ, Ruckh JM, et al. (2013) M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci 16(9): 1211-1218.
- Dimou L, Gallo V (2015) NG2-glia and their functions in the central nervous system. Glia 63(8): 1429-1451.
- Zhu X, Bergles DE, Nishiyama A (2008) NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development 135(1): 145-157.
- Zhu X, Hill RA, Dietrich D, Komitova M, Suzuki R, et al. (2011) Age- dependent fate and lineage restriction of single NG2 cells. Development 138(4): 745-753.
- Shoshan Y, Nishiyama A, Chang A, Mork S, Barnett GH, et al. (1999) Expression of oligodendrocyte progenitor cell antigens by gliomas: implications for the histogenesis of brain tumors Proc Natl Acad Sci U S A 96(18): 10361-10366.
- Sugiarto S, Persson AI, Munoz EG, Waldhuber M, Lamagna C, et al. (2011) Asymmetry-defective oligodendrocyte progenitors are glioma precursors. Cancer Cell 20(3): 328-340.
- Chen Y, Wang Z, Dai X, Fei X, Shen Y, et al. (2015) Glioma initiating cells contribute to malignant transformation of host glial cells during tumor tissue remodeling via PDGF signaling. Cancer Lett 365(2): 174-181.
- Chekenya M, Pilkington GJ (2002) NG2 precursor cells in neoplasia: functional, histogenesis and therapeutic implications for malignant brain tumours. J Neurocytol 31(6-7): 507-521.
- Bouvier C, Bartoli C, Aguirre CL, Virard I, Colin C, et al. (2003) Shared oligodendrocyte lineage gene expression in gliomas and oligodendrocyte progenitor cells. J Neurosurg 99(2): 344-350.
- Ligon KL, Alberta JA, Kho AT, Weiss J, Kwaan MR, et al. (2004) The oligodendroglial lineage marker OLIG2 is universally expressed in diffuse gliomas. J Neuropathol Exp Neurol 63(5): 499-509.
- Dawson MR, Polito A, Levine JM, Reynolds R (2003) NG2-expressing glial progenitor cells: an abundant and widespread population of cycling cells in the adult rat CNS. Mol Cell Neurosci 24(2): 476-488.
- Mason JL, Goldman JE (2002) A2B5+ and O4+ Cycling progenitors in the adult forebrain white matter respond differentially to PDGF-AA, FGF-2, and IGF-1. Mol Cell Neurosci 20(1): 30-42.
- Gensert JM, Goldman JE (2001) Heterogeneity ofcyclingglialprogenitors in the adult mammalian cortex and white matter. J Neurobiol 48(2): 75-86.
- Uhrbom L, Hesselager G, Ostman A, Nistér M, Westermark B, et al. (2000) Dependence of autocrine growth factor stimulation in platelet- derived growth factor-B-induced mouse brain tumor cells. Int J Cancer 85(3): 398-406.
- Hu X, Holland EC (2005) Applications of mouse glioma models in preclinical trials. Mutat Res 576(1-2): 54-65.
- Dai C, Celestino JC, Okada Y, Louis DN, Fuller GN, et al. (2001) PDGF autocrine stimulation dedifferentiates cultured astrocytes and induces oligodendrogliomas and oligoastrocytomas from neural progenitors and astrocytes in vivo. Genes Dev 15(15): 1913-1925.
- Shih AH, Holland EC (2006) Platelet-derived growth factor (PDGF) and glialtumorigenesis. Cancer Lett 232(2): 139-147.
- Schrappe M, Klier FG, Spiro RC, Waltz TA, Reisfeld RA, et al. (1991) Correlation of chondroitin sulfate proteoglycan expression on proliferating brain capillary endothelial cells with the malignant phenotype of astroglial cells. Cancer Res 51(18): 4986-4993.
- Chekenya M, Krakstad C, Svendsen A, Netland IA, Staalesen V, et al. (2008) The progenitor cell marker NG2/MPG promotes chemoresistance by activation of integrin-dependent PI3K/Akt signaling. Oncogene 27(39): 5182-5194.
- Riemenschneider MJ, Koy TH, Reifenberger G (2004) Expression of oligodendrocyte lineage genes in oligodendroglial and astrocytic gliomas. Acta Neuropathol 107(3): 277-282.
- Johansson FK, Göransson H, Westermark B (2005) Expression analysis of genes involved in brain tumor progression driven by retroviral insertional mutagenesis in mice. Oncogene 24(24): 3896-3905.
- Yokoo H, Nobusawa S, Takebayashi H, Ikenaka K, Isoda K, et al. (2004) Anti-human Olig2 antibody as a useful immunohistochemical marker of normal oligodendrocytes and gliomas. Am J Pathol 164(5): 17171725.
- Guha A, Feldkamp MM, Lau N, Boss G, Pawson A (1997) Proliferation of human malignant astrocytomas is dependent on Ras activation. Oncogene 15(23): 2755-2765.
- Colin C, Virard I, Baeza N, Tchoghandjian A, Fernandez C, et al. (2007) Relevance of combinatorial profiles of intermediate filaments and transcription factors for glioma histogenesis. Neuropathol Appl Neurobiol 33(4): 431-439.
- Wesseling P, Schlingemann RO, Rietveld FJ, Link M, Burger PC, et al. (1995) Early and extensive contribution of pericytes/vascular smooth muscle cells to microvascular proliferation in glioblastoma multiforme: an immuno-light and immuno-electron microscopic study. J Neuropathol Exp Neurol 54(3): 304-310.
- Ozerdem U, Stallcup WB (2003) Early contribution of pericytes to angiogenic sprouting and tube formation. Angiogenesis 6(3): 241-249.
- Ozerdem U, Stallcup WB (2004) Pathological angiogenesis is reduced by targeting pericytes via the NG2 proteoglycan. Angiogenesis 7(3): 269-276.
- Fukushi J, Makagiansar IT, Stallcup WB (2004) NG2 proteoglycan promotes endothelial cell motility and angiogenesis via engagement of galectin-3 and alpha3beta1 integrin. Mol Biol Cell 15(8): 3580-3590.
- Al-Mayhani MT, Grenfell R, Narita M, Piccirillo S, Kenney HE, et al. (2011) NG2 expression in glioblastoma identifies an actively proliferating population with an aggressive molecular signature. Neuro Oncol 13(8): 830-845.
- You WK, Bonaldo P, Stallcup WB (2012) Collagen VI ablation retards brain tumor progression due to deficits in assembly of the vascular basal lamina. Am J Pathol 180(3): 1145-1158.
- Farin A, Suzuki SO, Weiker M, Goldman JE, Bruce JN, et al. (2006) Transplanted glioma cells migrate and proliferate on host brain vasculature: a dynamic analysis. Glia 53(8): 799-808.
- Goretzki L, Burg MA, Grako KA, Stallcup WB (1999) High-affinity binding of basic fibroblast growth factor and platelet-derived growth factor-AA to the core protein of the NG2 proteoglycan. J Biol Chem 274(24): 16831-16837.
- Campoli M, Ferrone S, Wang X (2010) Functional and clinical relevance of chondroitin sulfate proteoglycan 4. Adv Cancer Res 109: 73-121.
- Wang J, Svendsen A, Kmiecik J, Immervoll H, Skaftnesmo KO, et al. (2011) Targeting the NG2/CSPG4 proteoglycan retards tumour growth and angiogenesis in preclinical models of GBM and melanoma. PLoS One 6(7): e23062.
- Poli A, Wang J, Domingues O, Planaguma J, Yan T, et al. (2013) Targeting glioblastoma with NK cells and mAb against NG2/CSPG4 prolongs animal survival. Oncotarget 4(9): 1527-1546.
- Kmiecik J, Gras Navarro A, Poli A, Planaguma JP, Zimmer J, et al. (2014) Combining NK cells and mAb9.2.27 to combat NG2-dependent and anti-inflammatory signals in glioblastoma. Oncoimmunology 3(1): e27185.
- Higgins SC, Fillmore HL, Ashkan K, Butt AM, Pilkington GJ (2015) Dual targeting NG2 and GD3A using Mab-Zap immunotoxin results in reduced glioma cell viability in vitro. Anticancer Res 35(1): 77-84.
- Brianfon-Marjollet A, Balenci L, Estève F, Honnorat J, Farion R, et al. (2010) NG2-expressing glial precursor cells are a new potential oligodendroglioma cell initiating population in N-ethyl-N-nitrosourea- induced gliomagenesis. Carcinogenesis 31(10): 1718-1725.
- Higgins SC, Bolteus AJ, Donovan LK, Hasegawa H, Doey L, et al. (2014) Expression of the chondroitin sulphate proteoglycan, NG2, in paediatric brain tumors. Anticancer Res 34(12): 6919-6924.
- Girolamo F, Dallatomasina A, Rizzi M, Errede M, Wälchli T, et al. (2014) Diversified expression of NG2/CSPG4 isoforms in glioblastoma and human foetal brain identifies pericyte subsets. PLoS One 8(12): e84883. Erratum in: PLoS One 8(12): e95120.






























