Fabrication and Characterization of Electros Punned Eprosartan Loaded Biocompatible Polyvinyl Acetate/Povidone Based Nanofiber Mat: A Novel Approach as Transdermal Carrier for Therapeutic Actives
Mali AJ1*, Archana Kadam2, Bothiraja C1 and Pawar AP1
1Department of Pharmaceutics, Bharati Vidyapeeth (Deemed to be University), India
2Department of Quality Assurance, Bharati Vidyapeeth (Deemed to be University), India
Submission: July 11, 2023; Published: July 31, 2023
*Corresponding author: Mali AJ, Department of Pharmaceutics, Bharati Vidyapeeth University, Poona College of Pharmacy, Eran Dwane, Pune 411038, Maharashtra, India, Email: ashwinjmali@rediffmail.com
How to cite this article: Mali AJ, Archana Kadam, Bothiraja C, Pawar AP. Fabrication and Characterization of Electros Punned Eprosartan Loaded Biocompatible Polyvinyl Acetate/Povidone Based Nanofiber Mat: A Novel Approach as Transdermal Carrier for Therapeutic Actives. Glob J Pharmaceu Sci. 2023; 10(5): 555800. DOI: 10.19080/GJPPS.2023.10.555800.
Abstract
Introduction: Nanofiber (NF) represents one of the newest, promising and advanced technologies in transdermal and or mucosal drug delivery due to its unique advantages. The present study aims to explore the feasibility of Kollidon® SR (K-SR) to fabricate sustained release Eprosartan (EPR) loaded nanofiber (NF) by using electrospinning technology for transdermal drug delivery. The EPR-NF mat was prepared with sodium iodide (SI) along the backing membrane and compared with film cast mat. The fabricated NF mechanical and physicochemical properties were assessed. Further, NF evaluated for in-vitro and ex-vivo diffusion study, skin irritation study and for antihypertensive activity.
Result: The formed NF in presence of SI was attributed increased conductivity of polymer solution resulting in the uniform NF. The SEM and IR elucidated the thin, uniform, smooth surface of NF without bead formation and interaction between EPR and K-SR. The PXRD and DSC revealed the molecular dispersion with less crystalline nature of the NF. The diffusion and permeation study resulted in initial burst release with gradual diffusion with zero order kinetics. The skin irritation study revealed lacking erythema or edema reflecting its acceptability. Furthermore, developed EPR-NF mat efficaciously controlled the systolic blood pressure (SBP) in rats up to 24h.
Conclusion: As per the results, K-SR can be explored as a promising carrier for the fabrication of electros punned NF mat for successful transdermal delivery of therapeutic actives.
Keywords: Nanofiber; Electrospinning; Kollidon SR; Eprosartan; Sodium iodide; Systolic blood pressure
Abbreviations: SBP: Systolic Blood Pressure; EPR: Eprosartan; NF: Loaded Nanofiber; NICE: National Institute of Clinical Excellence; SLS: Sodium Lauryl Sulphate; SI: Sodium Iodide; DCM: Dissolving EPR and K-SR in Dichloromethane
Introduction
Hypertension (HTN) is one of the persistent disorders throughout the world having a systolic blood pressure (SBP) > 140 mmHg and diastolic blood pressure (DBP) > 90mmHg as per the National Institute of Clinical Excellence (NICE) [1]. It is the most prevalent risk factor for a variety of health conditions such as cardiovascular disease. stroke and kidney failure resulting 7.5 million deaths per year worldwide. Despite various advances in the field, it is projected that 1.56 billion people will suffer from HTN by 2025 [2,3].
Most of the drugs used for the hypertension are administered through oral route. Various novel drug delivery systems such as liposomes, nanoparticles, solid-lipid nanoparticles (SLNs), colloidal emulsions, or dispersible tablets, self-emulsifying drug delivery system (SEDDs) etc are explored to accomplish the correct concentration, rate and release of drug [4]. But none of the formulations are capable of achieving qualities for uniform and controlled delivery of the actives. Transdermal drug delivery systems (TDDS) is a well-established route of administration for therapeutically beneficial actives due to their potential qualities such as uniform controlled release, patient compliance and reduction of the dose of potent actives [5]. The conventional transdermal patches such as Daytrana©, Catapres-TTS© and Emsam© have gained significant commercial success but posing the limitations with therapeutic efficacy of drugs.
Nanofiber (NF) represents one of the newest, promising and advanced technology in transdermal and oromucosal drug delivery due to its unique advantages such as high porosity, surface area, least fiber to fiber distance, high surface to volume ratio, design flexibility, good mechanical strength and diversity in the surface functionalities [6,7]. Electrospinning (E-spin) is one of the most reliable techniques for nanofiber formation. Among all the techniques, electrospinning has gained more importance because of its ability to fabricate thin uniform sized NF with the high surface area and its fibrous porosity [8]. E-spin technology is mostly applied in tissue and material engineering, wound dressing, prosthesis and drug delivery [9,10].
K-SR is a blend of polyvinyl acetate and povidone (K-30) in the ratio of 8:2 which is used in pharmaceutical industry as a matrix forming agent, dry binder, film-former, stabilizer, dispersant for pigments, enzyme stabilizer and for improvement of bioavailability [11]. The polymer contains 80 % polyvinyl acetate and 19 % povidone in physical mixture with 0.8 % sodium lauryl sulphate (SLS) and about 0.2 % of silica as stabilizer. It is generally used as a rate-retarding agent in matrix tablet formulations and other dosage forms such as microcapsules and beads. It is used in the TDDS as a matrix retarding agent [12]. However, K-SR is not explored to develop the NF mat by the electrospray technology. Paying attention to this area, the present work deals with investigating the feasibility of K-SR in the fabrication of NF using Eprosartan (EPR) as a model drug. The EPR has broad spectrum antihypertensive activity having poor aqueous solubility of 0.00866mg/mL with the molecular weight of 424.515g/mol. The pKa and log P value of the drug is 3.63 and 3.9 respectively emphasizing its lipophilic character. The poor aqueous solubility and lack of absorption at the site of application makes EPR a challenging candidate in the fabrication of electros punned mat.
The objective of present study was to explore the feasibility of K-SR to fabricate NF as TDDS by using electrospinning process. The EPR was entrapped in polymeric NF for its delivery to the systemic circulation through skin in the controlled release manner with high pharmacological action. The NF mat was electro spanned by dissolving EPR and K-SR in dichloromethane (DCM) along with sodium iodide (SI) and compared with conventional film of EPR prepared by film casting method. The fabricated mat was evaluated for mechanical properties (thickness, folding endurance) and physicochemical properties including weight variation test, drug content, scanning electron microscopy (SEM), infrared spectroscopy (IR), X-ray diffraction studies (PXRD) and differential scanning calorimetry (DSC). Further, fabricated NF was evaluated for in-vitro and ex-vivo diffusion study, skin irritation study and antihypertensive activity.
Methods
Eprosartan (EPR) was obtained as gift sample from Glenmark Pharmaceuticals Ltd, Navi Mumbai, India. Kollidon® SR (Polyvinyl Acetate and Povidone) polymer was kindly given as a gift sample by BASF India Ltd, Navi Mumbai, India. Sodium iodide (SI), Dichloromethane (DCM) and all other solvents used in the study were purchased from Merck Ltd, Mumbai, India.
Development of nanofiber mat
Preparation of electrospinning solution: An accurately weighed 1gm of K-SR powder was dissolved in 10mL of DCM to obtain 10% w/v of solution. The 2mg of SI was added in above solution. 100 mg of EPR was added to the above polymer solution with continuous stirring to obtain a homogenous solution [13,14].
Fabrication of nanofiber mat: The prepared solution was withdrawn into 5mL syringe fitted with a metallic needle of 0.6mm inner diameter. The whole syringe setup is fixed horizontally on the syringe pump. The feeding rate of 2mL/h was controlled by the syringe pump. A high voltage supply of 25kV was applied to the metallic needle. A piece of aluminum foil covered on collector drum was adjusted at distance of 10cm from the needle tip. The electrospinning was carried out at 750RPM. The formed nanofibers are collected on aluminum foil which was carefully removed from the collector drum and placed in the desiccator till further use. This process was carried out under ambient condition using an instrument E-Spin Nano (PECO-Chennai, India) enclosed in a Plexiglass box [15].
Casting of polymeric backing film: Backing film of the NF mat was cast by dissolving hydroxyl propyl methyl cellulose (HPMC) in a 2:1 mixture of deionized water and propan-2-ol. Glycerol was added to the above solution as a plasticizer at concentration of 70% of the polymer mass. This solution was poured on the aluminum foil in rectangular mould and was placed in desiccator for 2 days for complete evaporation of solvent [16].
Preparation of mat by solvent casting method: The conventional mat of K-SR was prepared by solvent casting method. The 100 mg of EPR and 1 gm of K-SR was taken and mixed in the mortar pestle with continuous trituration. The 10 mL of DCM was poured in the above mixture and vortexed to get the homogenous dispersion. This dispersion was poured in the petri plate which was previously lubricated using paraffin wax for the easy removal of the film from the petri plate. The petri plate was kept in the clean room for 24 h for solvent evaporation and film formation. The developed film was stored in desiccator for further use [17].
Characterization of nanofiber mat
Entrapment efficiency: UV spectroscopic method was used to quantify the amount of the EPR loaded in the NF. The NF mat was cut into an area of 16cm2 and dissolved in 10mL of methanol. From the above solution 1mL was taken and diluted to 10mL with the buffer solution (pH 7.4) and again 1mL was taken and diluted to 10mL using the same buffer solution. Absorbance of resulted solution was taken at 234 nm using UV spectrophotometer (Shimadzu UV-1601, Kyoto, Japan) in order to obtain exact EPR content [18] . The % EE was determined by using following equation
% EE = Weight of EPR in NF mat / Total weight of EPR feeded.
Folding endurance: The folding endurance was determined by repeatedly folding the strip containing 9 cm2 area of film at one specific point till it was broken. The number of times the film was folded at same place which without breaking gave the value of folding endurance [19] .
Thickness: Thickness is one of the most important physical evaluations of a transdermal mat as it reveals the uniformity of the mat. This property is measured at three different places on the same mat using screw gauge and the mean value was calculated.
Uniformity of weight: To test the weight variation, 6 different mats were weighed individually from each batch. The average weight was calculated and difference between average weight and individual weight was determined to know the variation of weight from the mean weight.
Fourier transforms infrared spectroscopy: The chemical structure of fiber was examined using FTIR (4100, JASCO International Co., Ltd., Tokyo, Japan). The EPR, K-SR, EPR loaded NF mat were analyzed. The samples were mixed with dry potassium bromide (1mg sample in 100mg KBr) and placed in the mold. The IR spectra for the same were recorded in the region from 3500 to 500cm-1 [20].
Scanning electron microscopy: The SEM (SEM Jeol, Japan) was used to characterize morphology of prepared EPR loaded NFs. The mat was mounted on the aluminium stud and sputtered with a thin layer of platinum using auto fine coater (VG-Microtech, UK) [21,22].
Powder x-ray diffraction: This detects crystal lattice and is a powerful tool for studying polymorphism, salts and crystalline phases. The PXRD was recorded on sample exposed to nickel filtrate radiation (40KV, 30mA) and were scanned from 10-40ºC with 2θ at a size of 0.045 degree and step time of 0.5s [23].
Differential scanning calorimetry: A thermal property of EPR, K-SR and NF mat was studied using DSC in a calorimeter equipped with an intra-cooler (Mettler-Toledo, Greifensee, Switzerland). Indium standards were used to calibrate the temperature and enthalpy scale. Approximately 5mg of sample was hermetically sealed in an aluminum pan with a hole and heated at a constant rate of 10ºC /min over a temperature range of 50-350ºC. The inert atmosphere was maintained by purging nitrogen gas at a flow rate of 50mL/min [2].
In-vitro diffusion study: The in vitro diffusion study of the EPR from the NF and film casted mat was carried out in phosphate buffer saline (PBS) of pH 7.4 using Franz diffusion cell (Dolphin Instruments, Mumbai, India) through dialysis bag membrane. Both mats were cut into 6cm2 sizes which adhered onto the membrane. The reception compartment in the cell was filled with the 20mL of the PBS upon which the dialysis membrane was placed. The whole setup was placed on magnetic stirrer and diffusion was carried out till 24h and the amount of EPR diffused through membrane was measured by withdrawing the samples at the time interval of 0.5 ,1 ,2 ,4 ,6 ,8 ,10 ,12 and 24 h. The sink condition was maintained by replacing equal amount of PBS. The samples collected were filtered through what man filter paper and diluted as needed with the PBS and absorbance of each diluted solution was measured at fixed wavelength of 234nm using UV spectroscopy [13].
Ex-vivo permeation study: Ex vivo skin permeation study was performed for both NF and film casted mat in PBS of pH 7.4 using Franz diffusion cell (Dolphin Instruments, Mumbai, India) through excised rat skin. Hair on the waster rat skin in the abdominal area was removed after its scarification by ether inhalation method. Subcutaneous tissue was surgically removed, and isopropyl alcohol was used to wipe the dermis side in order to remove all the fat on its surface. The distilled water was used to wash the skin followed by treating the skin with the 2M sodium bromide solution for 7h. The epidermis cleaned with water was separated with the cotton swab. A research proposal was prepared according to the guidelines of the Committee for the Purpose of Control and Supervision of the Experiment of Animals (CPCSEA). The experiment protocol was approved by the Institutional Animal Ethics Committee (IAEC) (CPCSEA/12/2020).
Skin irritation test: Skin irritation test was performed on male wistar rats weighing between 220-250gm. Adhesive tape USP was used as a control mat. The mat of 6cm2 was used as test samples. The study was conducted on abdominal skin of rats. The control mat was placed on left dorsal surface of the rat, whereas test formulations were placed on identical side of the right dorsal surface of the rat. The mats were removed after 24h with the help of an alcohol swab and skin was examined for any erythema or edema on skin where patch was adhered.
Antihypertensive activity: Methyl prednisolone acetate (20mg/kg/week) for 3 weeks was administered via the subcutaneous injection to 24 wistar rats for the induction of hypertension and the systolic blood pressure (SBP) of rats was measured by tail-cuff method. Animals were divided into three groups (n=8). Hypertension was successfully induced in all rats with minimum SBP of 150mm Hg and was selected for further study. Oral suspension of the brand product Teveten™ was administered to group 1 using an oral-feeding needle. Group 2 was applied to the EPR-NF formulation and group 3 was applied with the film prepared by casting method. The SBP of each rat was measured at predetermined time intervals of 1, 2, 3, 4, 6, 8, 12 and 24 h using the tail-cuff system [1].
Statistical Method: All the experiments were performed in triplicates. Data were recorded as mean ± standard deviation. Statistical analysis was carried out using online Quick Calcs Graph Pad software. The data analyzed in following manner; *p≤0.1(not statistically significant), **p ≤ 0.01 (statistically significant), ***p ≤ 0.001 (very statistically significant) and **** p≤0.0001 (extremely statistically significant).
Result
Preparation of EPR loaded NF mats
The polymer concentration was optimized at the standard processing conditions of flow rate, distance of the syringe tip from the collector surface and applied voltage to get the NF mat. Initially, the flow rate was adjusted from 0.5-3mL/h. At 0.5mL/h, the available solution for process was very less resulting formation of the droplets on collector. The uniform and optimum fibers were formed at 2mL/h but upon further increase in flow rate (3mL/h), the formation of higher diameter fiber and bead was observed. Further, the distance between the syringe tip and collector was set at optimum (10cm), so that the flight time was maximum in the applied electrical field which was further sufficient for evaporation of solvent. The applied voltage of 25kV was functional to form the Taylor cone of K-SR solution. Moreover, the syringe (5mL) was used to form the small droplets which will be supportive to form thin, uniform fibers without formation of any beads. The K-SR was explored from 1% to 15% to fabricate the NF mat. The batch having 10% K-SR has depicted the uniform, ultrathin, nanosized polymeric mat as depicted in (Figure 1A &1B). The further increase in concentration of K-SR up to 15% resulted in the thick fiber comprising uneven beads. These beads were observed due to the higher concentration of polymer.

Characterization of nanofiber Mat and solvent casting film
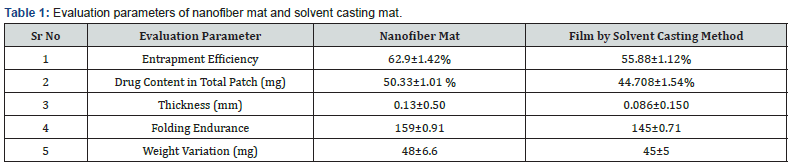
Mechanical characterization: NF mat was evaluated for the entrapment efficiency, folding endurance, thickness, weight variation. It was observed that the entrapment efficiency was found to be 62.9% with the drug content 50.33mg as depicted in table 1. The folding endurance of NF mat was depicted 159. Further, The Thickness and mean weight of developed NF mat was found to be 0.13mm and 48±6.6mg.
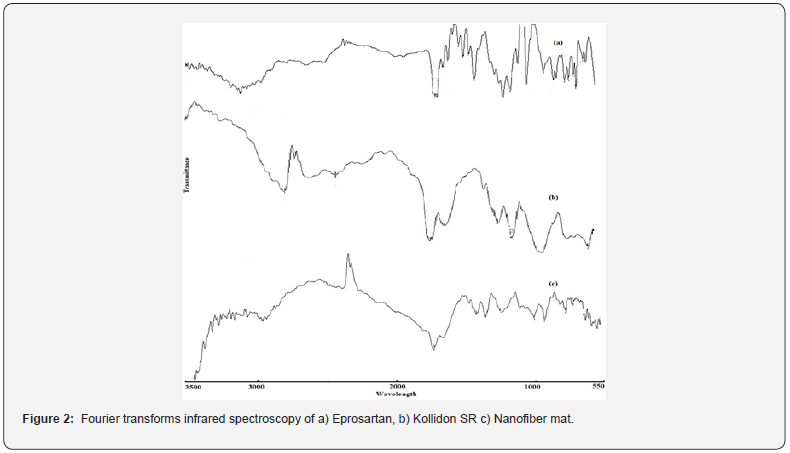
Fourier transforms infrared spectroscopy
FTIR spectra were recorded for EPR, K-SR and EPR-NF mat. EPR alone showed characteristic bands at 1710 cm-1 (C=O), 3174 cm-1 (COOH), 2850-2970 cm-1 (aliphatic C-H), 3000-3100 cm-1 (aromatic C-H) as shown in figure 2.

Scanning electron microscopy
The SEM of fabricated electros punned mat was observed to confirm the formation of NFs. The SEM images depicted in figure 3 represent the uniform NFs within the range below 500 nm.
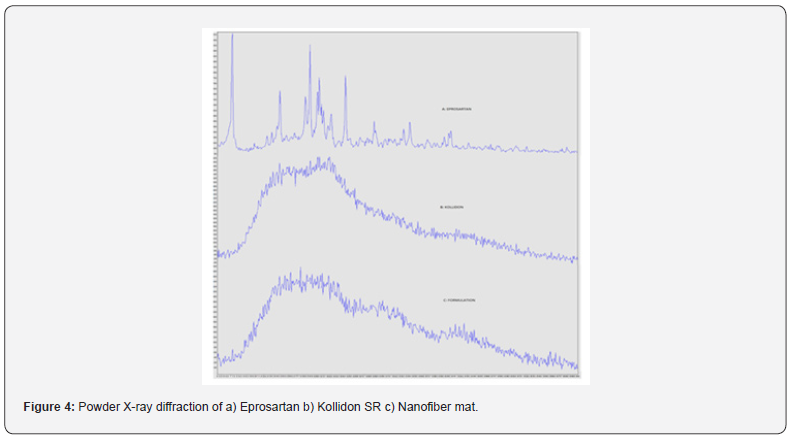
Powder X-ray diffraction
The PXRD study was commonly performed to elucidate the drug and excipient interaction, physical properties and also structural characterization of the material. The PXRD analysis of the prepared NFs, K-SR and EPR was performed in order to analyze polymorphic transitions that take place in EPR when they were formulated in the electrospun NFs. The XRD pattern can also reveal the degree of crystallinity of sample using relative integrated intensity of reflection peaks in the given range of reflecting angle 2θ. The XRD pattern of EPR, K-SR, and EPR loaded NFs were shown in figure 4. The EPR alone showed characteristic diffraction lines at 2θ values such as 7.2, 7.3, 19.1 and NF formulation has shown many different lines along with the broad peaks which confirmed its amorphous nature.
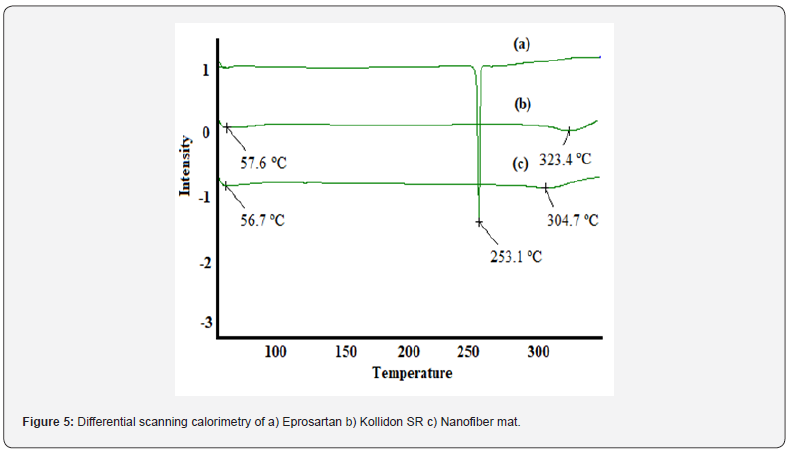
Differential scanning calorimetry
The thermal properties of EPR, K-SR and NF mat were investigated by DSC. The DSC thermo gram of EPR showed a sharp endothermic peak at around 253 ºC as shown in figure 5 which corresponds to the melting point of EPR. For NF mat, the DSC thermogram did not illustrate any melting peak for EPR suggesting that EPR was completely dispersed in the fiber matrix without any crystalline aggregation [9]. Both NF mat and K-SR showed a broad endothermic peak between 50-60 ºC due to loss of water and the melting of polymer.
In- vitro diffusion study
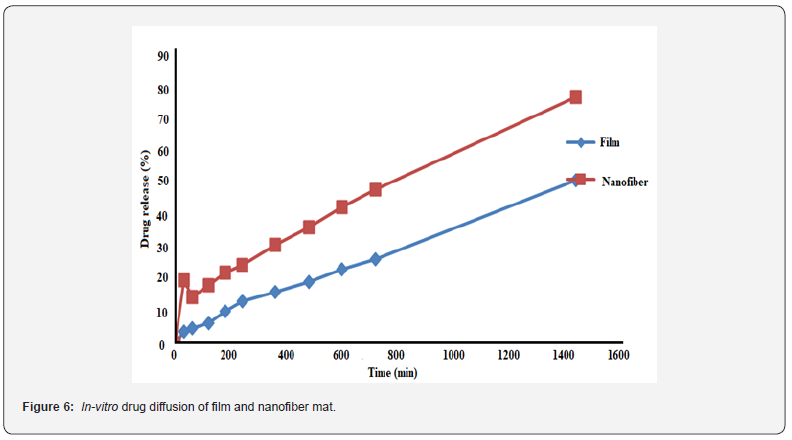
The release pattern of NF mat and cast film was studied using Franz diffusion cell in phosphate buffer of pH 7.4 through dialysis membrane. The fabricated NF initially depicted the burst release and then gradual release of EPR from the mat was observed (Figure 6). The initial burst release of EPR was observed due the diffusion of solubilized drug through the formed pores due to the uptake of the moisture and consequent leaching of water-soluble povidone from the matrix. Further, the cast film has shown the gradual diffusion without any burst release. The EPR release from the NF mat was more efficient as compared to that of the film fabricated by casting method. The EPR release from the NF was found to be 80% as compared to the 50% of cast film in 24h. This difference could be because of the amorphous nature of the NF mat [12]. Further, drug diffusion from the NF mat and film was confirmed by plotting the graphs and subjecting to the model fitting such as zero order, first order, higuchi, Hixson-crowell and korsmeyer-peppas. Both the EPR release curves of the mat and the cast film showed the zero-order kinetics with R2 value as 0.9898 and 0.9958 respectively. It revealed the controlled diffusion of EPR from both the NF mat and cast film.
Ex-vivo permeation study
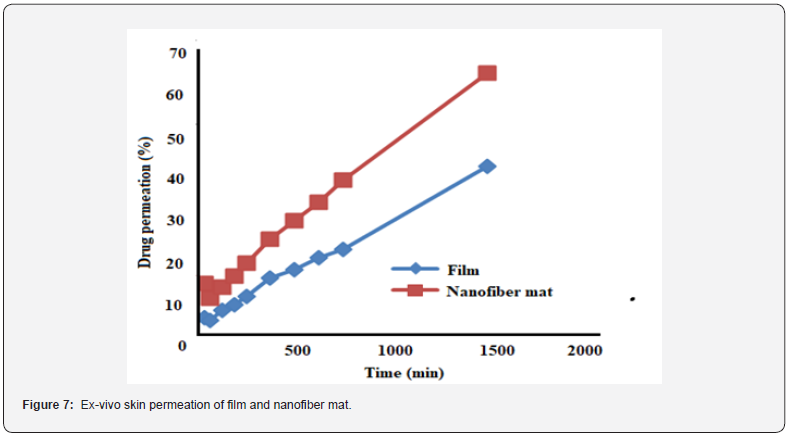
Ex-vivo skin permeation study was carried out by the diffusion method using Franz diffusion cell through the exercised rat skin in the pH 7.4 phosphate buffer. This study also referred to the superiority of the NF mat as the drug diffusion from the mat was 62% in 24h whereas drug diffusion from the mat was 40%. This difference of drug diffusion from the mat and film may be due to fast swelling of the NF mat due to small size and also increased surface area resulting in the high solubility of the drug (Figure 7). To confirm the drug diffusion, graph was plotted for the drug release and subjected to the various model [15]. The graph showed control release of the EPR from the mat and also from the cast film as both the graphs followed zero order with the R2 value as 0.9923 and 0.9959 for the NF mat and film respectively [24].
Skin irritation test
The skin irritation study revealed that there was no erythema or edema on the abdominal area where patch was adhered. Further, there was not any staining of the skin also was seen. This study also revealed that this formulation can be given through skin without any irritation or redness or any swelling.
Antihypertensive activity
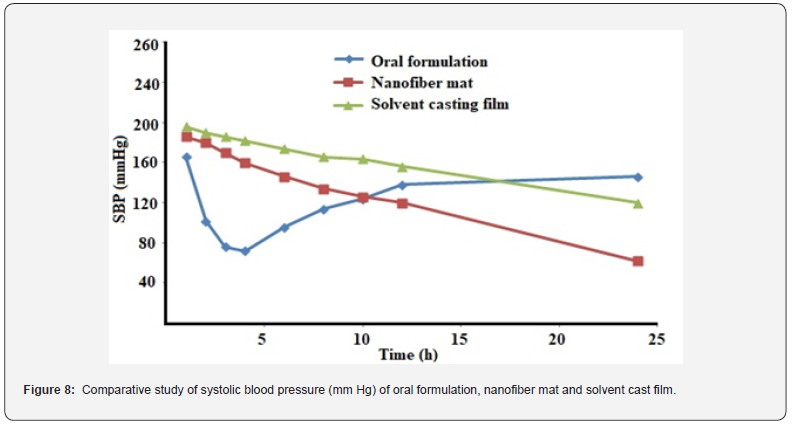
During antihypertensive study, hypertension was induced in all rats in a successful manner after subcutaneous injection of methyl prednisolone acetate (20mg/kg/week for 3 weeks). There is a significant difference in SBP values in pre- and post-treatment with methyl prednisolone acetate in Wistar rats. An early drug action was observed in Group 1 after oral administration of EM; hypertension was significantly reduced to normal value with the maximum reduction in SBP (120±2mm Hg) observed at 2h [16]. After 6 h of oral administration, SBP progressively started to rise and reached up to 142±2mm Hg at 8 h and reached 155±2 mm Hg at 24 h after oral administration. On the other hand, Eprosartan loaded nanofiber formulation steadily decreases the SBP and maximum action of drug observed at 3h after transdermal application of mat (SPB 110±3mm Hg) in hypertensive rats. The percentage reductions in mean SBP of Wistar rates by mat formulation and oral suspension were 25% and 15%, respectively, at 24h time point. SBP lowering effect of mat formulation was sustained and maintained up to the complete duration of study i.e. 24 h. At the 24 h time point, SBP values of Group 3 rats were much less when compared to oral and mat (SBP was 135±3mmHg at 2h). It is concluded that developed Eprosartan nanofiber mat formulation released the drug gradually and efficaciously controlled the SBP in rats up to 24 h.
Discussion
EPR was chosen as a model drug in the present work. The key intent of this research work was to develop EPR loaded K-SR nanofiber mat by electrospinning method to get the better release profile and therapeutic efficacy of the model drug by overcoming its existing limitations. NF mat representing nano porous structure instantly absorbs moisture through nano voids leading to strong adhesion. Further, the delivery of EPR by using NF mat helps to retard the EPR for a longer period of time with direct systemic delivery by skipping the rate limiting step such as solubility and absorption. It also bypasses the first pass effect leading to higher bioavailability and pharmacological action leading to the direct discharge of the actives at the site of action with prolonged pharmacological action.
The K-SR and DMSO combination should be carefully conceded out to obtain high-quality EPR loaded NF mat. In the present work K-SR was selected due to its film forming property and compatibility with various drugs. The EPR and K-SR were both soluble in DMSO. SI salt was used due to its conductivity nature. It helps to facilitate nanometer size fibers due to increased conductivity of the polymer solution. Moreover, it helps to reduce the bead formation in nanofibers. Folding is defined as the number of folds at specific place to break the film or to develop any crack on the film. This test plays important role in checking the ability of the film to withstand folding. It is also an indication of brittleness of the mat. The higher range of folding endurance indicates less brittleness of the prepared mat.
The spectra of EPR loaded fibers showed retention of all characteristics band of EPR and the K-SR which states that there was absence of interaction between drug and polymer. There was no presence of drug crystals or an aggregate which was clear confirmation that the EPR was distributed uniformly in the NFs. The images also revealed that the formed NFs have smooth surface without any pores. The diffusion from mat was high enough due to the increased surface area of NF. This high surface area resulted in an elevated dissolution rate leading to high diffusion of the EPR from the NFs (Figure 8). Whereas the slower swelling of polymer in cast film has resulted in the minimum diffusion of the EPR. The presence of aggregates on the surface of the cast film due to retention of crystalline nature of EPR might be the factors for the limited release which further reflected in low solubility and less diffusion from film.
Conclusion
In this study, NFs were successfully prepared using K-SR by electrospinning method. The polymer also showed good compatibility with the EPR. Further, the reduction in the crystalline nature of the EPR to amorphous state resulted in the high solubility of the EPR. The skin irritation study revealed that there was no redness or any irritation on the skin of the rat. The in-vitro drug diffusion and ex-vivo skin permeation study revealed the initial burst release of drug from NF which further supported by gradual diffusion for a longer period of time. The higher drug diffusion was observed from the NF mat as compared to the cast film. Further, the promising results were observed with the fabricated NF in the management of the hypertension due to controlled release of the EPR. This study concludes that the electro spanned biocompatible polyvinyl acetate/povidone-based nanofiber mat can be explored for the successful delivery of therapeutic actives.
References
- Ahad A (2017) Pharmacodynamic study of eprosartan mesylate-loaded transferosomes Carbopol® gel under Derma roller® on rats with methyl prednisolone acetate-induced hypertension. Biomed Pharmacother 89: 177-184.
- Bhandaru JS, Malothu N, Akkinepally RR (2015) Characterization and solubility studies of pharmaceutical cocrystals of eprosartan mesylate. Crystal Growth and Design 15(3).
- Bhatta R, Hossain MS (2011) Evaluation of kallidin SR based ketorolac tromethamine loaded transdermal film. Journal of Applied Pharmaceutical Science 1(8).
- Chong EJ (2007) Evaluation of electro spun PCL/gelatin nanofibrous scaffold for wound healing and layered dermal reconstitution. Acta Biomater 3(3): 321-330.
- Cuevas AG, Williams DR, Albert MA (2017) Psychosocial Factors and Hypertension: A Review of the Literature. Cardiol Clin 35(2): 223-230.
- Delacroix S, Chokka RG (2014) Hypertension: Pathophysiology and Treatment. Journal of Neurology & Neurophysiology 05(06).
- Dott C (2013) A mucoadhesive electro spun nanofibrous matrix for rapid or mucosal drug delivery. Journal of Nanomaterials p: 3.
- Fetih G, Ibrahim MA, Amin MA (2011) Design and characterization of transdermal films containing ketorolac tromethamine. International Journal of PharmTech Research 3(1).
- Haider A, Haider S, Kang IK (2018) A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arabian Journal of Chemistry 11(8): 1165-1188.
- Huang ZM (2003) A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Composites Science and Technology 63(15): 2223-2253.
- Jannesari M (2011) Composite poly(vinyl alcohol)/poly(vinyl acetate) electro spun nanofibrous mats as a novel wound dressing matrix for controlled release of drugs. Int J Nanomedicine 6: 993-1003.
- Kamble RN (2016) Fabrication of electro spun nanofibers of BCS II drug for enhanced dissolution and permeation across skin. J Adv Res 7(3): 483-489.
- Mahajan NM (2018) Formulation development and evaluation of transdermal patch of piroxicam for treating dysmenorrhea. Journal of Applied Pharmaceutical Science 8(11).
- Maher MM (2020) Electro spun Fibrous Mat of Cellulose Acetate: Influence of Solvent System (Acetic Acid/Acetone) on Fibers Morphology. International Journal of Pharmaceutical Investigation 10(1).
- Potrac T, Sasa B, Robert R, Odon P, Zoran L (2015) Electro spun polycaprolactone nanofibers as a potential or mucosal delivery system for poorly water-soluble drugs. Eur J Pharm Sci 75: 101-113.
- Prisant LM (1992) Novel drug-delivery systems for hypertension. Am J Med 93(2A): 45S-55S.
- Sarwar MS, Hossain MS (2012) Development and evaluation of sustained release losartan potassium matrix tablet using kallidin SR as release retardant. Braz J Pharm Sci 48(4).
- Satyavati K (2014) Formulation and evaluation of immediate release tablets of eprosartan mesylate. International Journal of Pharmaceutical Sciences Review and Research 25(1).
- Siu AL (2015) Screening for high blood pressure in adults: U.S. preventive services task force recommendation statement. Ann Intern Med 163(10): 778-786.
- Stanek ZA (2019) Recent approaches in transdermal drug delivery system. Research Journal of Pharmacy and Technology.
- Subbiah T (2005) Electrospinning of nanofibers. Journal of Applied Polymer Science 96(2): 557-569.
- Tyagi C (2014) Electro spun Nanofiber Matrix with a Mucoadhesive Backing Film for Or mucosal Drug Delivery. International Journal of Materials, Mechanics and Manufacturing.
- Venkatesh KK (2009) Preparation and in vitro characterization of valsartan solid dispersions using skimmed milk powder as carrier. International Journal of PharmTech Research 1(3).






























