- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Exosomes as Endogenous Nanovesicular Delivery System in Combating Various Diseases
Ardhendu Kumar Mandal*
Central Instrumentation Division, CSIR-Indian Institute of Chemical Biology, India
Submission: June 30, 2023; Published: July 10, 2023
*Corresponding author: Ardhendu Kumar Mandal, Central Instrumentation Division, CSIR-Indian Institute of Chemical Biology, 4, Raja S.C. Mullick Road, Jadavpur, Kolkata – 700032, India, Email: ardhendu_mandal@yahoo.co.in
How to cite this article: Ardhendu Kumar M. Exosomes as Endogenous Nanovesicular Delivery System in Combating Various Diseases. Glob J Pharmaceu Sci. 2023; 10(5): 555796. DOI: 10.19080/GJPPS.2023.10.555796.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Abstract
Exogenous nanoparticular systems as drug delivery vehicles have few disadvantages over biomolecular toxicity, immunogenicity, and metabolic stability in comparison to endogenous nanovesicles. Exosomes, the cell-derived extracellular endogenous nanovesicles bearing numerous small biological molecules including lipids, nucleic acids and proteins, may play a significant role in nutrient transport and intercellular signaling communication through their interacted adhesion molecules or ligands with the membranous natural lipid bilayers in cellular homeostasis or in diverse pathophysiological conditions such as infection, neurodegeneration and cancer diseases. Owing to their unique characteristics, such as naturally occurring structure, higher biocompatibility, more innate stability, stable circulating capability, cargo-carriage properties, low immunogenicity, excellent cell / tissue penetrating capacity, favorable safety profile, and the ability to pass through biological barriers, exosomes can hold advanced platform to regulate cellular communication by targeted variable cargos delivery to recipient cells and tissues without rejection. However, their workable applications may be limited for the insufficient targeting capability and low efficiency in a few cases. To survive these barriers, exosomes may be isolated and loaded with exogenous cargos and surface-functionalized with ligands for delivery of therapeutics to specific site/s of interest with enhanced local concentration and minimal side effects for utilization as endogenous efficient drug delivery system against various diseases. This review reveals the recent advances mainly regarding their isolation, cargo-loading, functionalization, and biomedical applications as a suitable targeted nano-exosomal delivery system in combating different diseases.
Keywords: Exosomes; Isolation; Functionalization; Diseases; Nanoexosomal targeted delivery system; Biomedical applications
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Introduction
Intercellular communications occur for the cellular development, immunity, and maintenance of homeostasis under both physiological and pathological conditions through the usages of chemical messengers [1]. Local communications involve direct connection between cells via communication systems such as a gap junction to connect the cytoplasm of adjacent cells allowing signaling substances to transfer between the cells, whereas distant intercellular communications involve molecules like hormones to pass signals via the circulatory system to other parts of the body. However, several communication systems such as artificial exogenous nanocarriers have few biological barriers such as toxicity, insolubility and biostability. To overcome these barriers, recently exosomes, the naturally derived nanocarriers, have garnered considerable research interest owing to their suitable characteristics of encapsulating and attachments various molecules with the enclosed bilayer-phospholipid membrane to mediate intercellular messengers and molecules to specific site/s of interest through surface receptor interactions, receptor-mediated endocytosis, membrane fusion, micro-pinocytosis and / or phagocytosis in patho-physiological processes [2-4]. Exosomes, secreted from direct budding of different cells’ membranes into the biological fluids and the extracellular sites, carry and protect biological compounds such as specific mRNAs, regulatory miRNAs, other small non-coding RNAs, genomic DNAs, proteins and lipids between disease specific neighboring cells, and even to distant organs for viral infections, neurodegenerative diseases and cancer (Figure 1) [5-11]. Exosomes have great potential for targeted drug delivery as they can penetrate anatomical barriers, diffuse into the blood and even cross the blood-brain barrier keeping stability and maintaining sufficient binding effects [12,13]. Exosome-mediated drug delivery can bypass the P-glycoprotein drug efflux system by reducing drug resistance [14]. Exosomes, the non-immunogenic nanocarriers, can protect their cargos from the degradation by serum proteases and immune cells enhancing drug solubility, stability and bioavailability in a controlled release manner [15,16]. Exosome-nanotechnology has provided stable, high compatible and prolonged circulating natural lipid vesicular nanovehicles for overcoming the nanotechnological limitations in drug delivery systems such as their quick eliminating clearances by the reticuloendothelial system (RES), the changes of pharmacokinetic behaviors owing to protein absorptions on their surfaces and low cellular internalizations [17]. As exosomes contain membraneanchored and transmembrane -proteins suitable for enhancing endocytosis, they can promote easy payload delivery to targeted site/s [18]. Moreover, owing to their bio-originated membrane, inert to protein corona formation, they are flexible for exosomal surface modifications with ligands for specific targeted delivery [19-24]. Therefore, to get higher biological efficiencies, exogenous cargos should be loaded into specific isolated and purified exosomes with / without ligand attachments for their delivery to the targeted diseased zone/s. This review demonstrates chiefly the isolation, purification, cargo-loading and characterization of exosomes and their biomedical applications against several diseases to consider them as suitable nanomedicinal carriers.
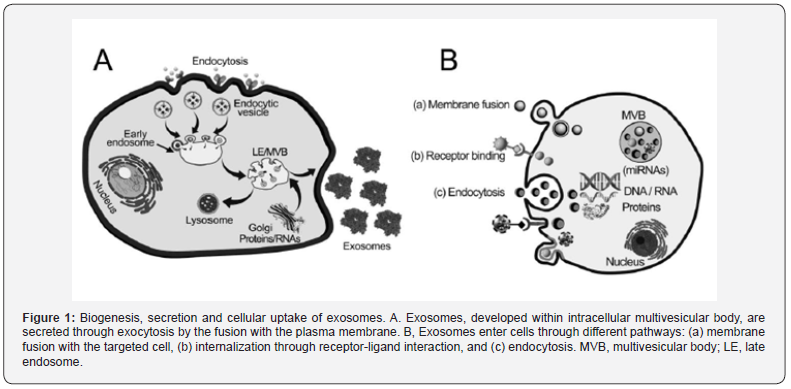
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Isolation of Exosomes
Exosomes are isolated from biological fluids and cell culture supernatants utilizing various methods such as differential (gradient) centrifugation, filtration, sucrose gradient centrifugation, size-exclusion chromatography, polymer precipitation and immune-affinity chromatography [25-29,22]. For gradient centrifugation, at first, cell culture supernatants are cleared of large vesicles and cell debris by sequential centrifugation at 300, 1000 and 10000xg for 10, 20 and 30min respectively accompanied with filtration utilizing 0.2μm syringe filter. The cleansed sample is spun at 100,000xg for 1h to pellet down the exosomes. The acquired exosomes are cleansed twice with phosphate buffer solution (PBS). For avoiding contamination by the FBS-deduced exosomes, FBS is centrifuged at 100,000xg for 2h to detach exosomes before the experiment. Exosome-purity may be ameliorated by spinning the samples utilizing floatation density gradient spin with optiprep or sucrose. The purified enriched exosomes may be determined by measuring the protein concentration through Braford assay and nanoparticle tracking analysis. The derived exosomal fraction is re-suspended in PBS (500μL, 1mg/mL total protein) for further usages. Size-exclusion chromatography, known as molecular sieve chromatography, is used for isolation of exosomes, where different compounds are separated according to their sizes by the penetration of the pores in the stationary phase through the aqueous solvents-used gel filtration or organic solvents-used gel permeation chromatography (polymer analysis). Polymer precipitation based on the formation of a mesh-like net for embedding exosomes is utilized to isolate and purify polymers, while Exo-Quick-TC commercial kit may be used for exosome extraction. In the antibody-dependent method, the antibodies may be immobilized in various media conditions, combined with chromatographic matrix, magnetic beads, microfluidic devices and plates for separations of exosomes, where antibodies are covalently attached to exosomal surface markers or related antigens like tetraspanins, TSG101, or differentiation molecules (CD9, CD63, CD81, CD82, CD9), Rasrelated protein (Rab5), and epithelial cell adhesion molecule (EpCAM), and collected by washing the stationary phase with a low pH buffer. Moreover, exosomes may also be isolated in a highly purified manner on the basis of sizes by using high performance liquid chromatography.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
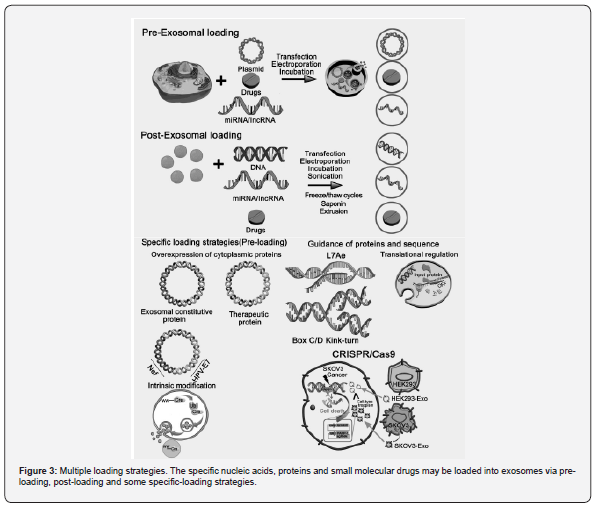
Loading of Exosomes
Loading of cargos in / on exosomes enables them as targeted delivery systems. Multiple approaches such as physical, chemical and biological mechanistic methods and / or in their combinations are utilized for the loading of various active molecules into / on the exosomes (Figure 2 & 3). For physical loading methods such as sonication, electroporation, freeze-thaw and extrusion, physical energy in the forms of electric, mechanical and optical interactions are usually employed for loading therapeutic cargos into the exosomes, while physical destruction may affect the integrity and function of the exosomal phospholipid membranes. For chemical loading processes of exosomes, transfection and saponin-assisted permeation are generally applied for facilitating encapsulations of molecules via electrostatic interactions and membrane permeabilization respectively following purification of exosomes from saponin-contamination and reagents. For biological loadings of exosomes, active molecules are usually loaded via simple incubations or complicated viral transductions on the basis of biological characteristics of the membranes and vectors. As the exosomal structures and functionalities may be affected by cargos-loadings, the key parameters such as the power of physical interactions, operation time, concentration of reagents, cell types and exosomes should be optimized to achieve high loading efficiencies for targeted delivery.
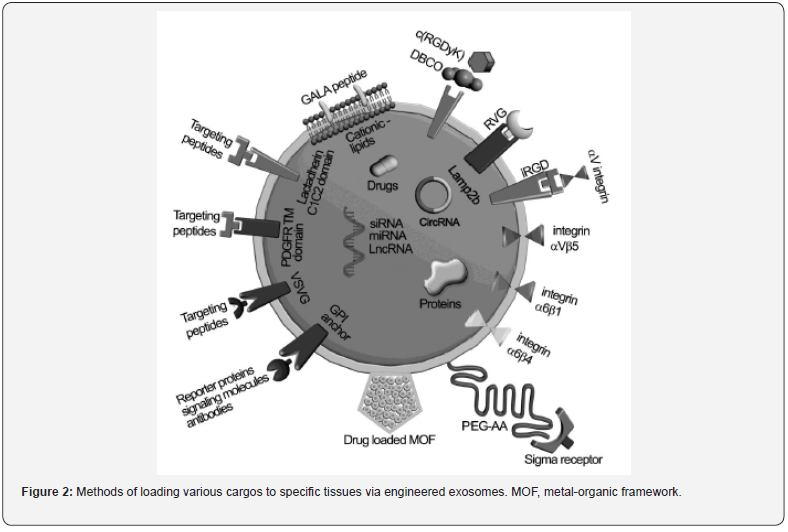
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Sonication
In general, exosomes are mixed with cargos and sonicated by utilizing a probe type homogenizer, while the mechanical shear-force of ultrasound treatment damages the integrity of the exosomal membrane allowing the entry of cargos into the exosome with simple diffusion through the deformed transient membraneous pores, followed by the restoration of the exosomal membrane-integrity by incubation after sonication. In this context, the loading molecules may also be anchored to the outer exosomal membrane-layers. In this process, temperature control, pulse, power output and pH should be optimized to get high drugloading efficiencies. Usually, during sonication, the mixture of drug and exosome is treated with ultrasound at 20% amplitude for 6 cycles following incubation at room temperature (RT, 37°C) for 1h [30-32]. Additionally, for enzyme loading such as catalase, the mixture of enzyme and exosome is sonicated (500v, 2kHz, 20% power, 6 cycles by 4 sec pulse / 2 sec pause), chilled on ice for 2 min and sonicated once more [33]. The cargo-loading process may be accompanied with saponin treatment, freeze-thaw cycling or extrusion, and purification for achieving their highest loading efficiencies.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Electroporation
Under the action of external electric pulse in conductive buffer, the larger cargos such as siRNA, miRNA, DNA, SPIONs, bioactive molecules and chemotherapeutic molecules enter into the exosomes through the electrical field-created recoverable pores of the exosomal phospholipid bilayer membranes by diffusion or membrane permeabilization, followed by the transfer to chilled electroporation cuvettes with applied voltages varying from 0.1-1000 kV, dependent on the exosomal origins and concentrations [30,31]. As the complex formation between electrodes-produced metal ions and the conductive buffergenerated hydroxide ions may develop exosomal aggregations, the optimized buffers such as disaccharide trehalose, citric acid and EDTA are utilized to inhibit the aggregations of exosomal cargos for aiding and maintaining the exosomal structural integrities and their encapsulation-efficiencies [31,30]. Electroporation of antisense miRNA oligonucleotides against miR-21 into exosomes has been performed to treat glioblastoma, while the mixture of nucleotides and exosomes has been electroporated at 400V in a 4mm cuvette, followed by the removal of unloaded nucleotides through super-centrifugation at 100,000xg for 1h [32]. Another group of investigators have reported the loading of microRNA-21 inhibitor and chemotherapeutic drugs into exosomes through electroporation at 1000 V for 10 min to get highest loading efficiency, determined by HPLC and quantitative real time PCR [32]. Other researchers have also performed the exosomal loadings of nucleotides through electroporation at 70V/150mF or 400V/125μF in 4 mm cuvettes to get higher loading efficiencies [32].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Freeze and Thaw Cycles
The freeze-thaw method is generally used to load proteins and miRNA, involving repeated fusion of lipid bilayers, while incubated (30min at RT) cargos with exosomes are rapidly frozen at -80°C or in liquid nitrogen, followed by their thawing at RT for 3~10 cycles to ensure high encapsulation efficiencies of exosomal cargos [31,30,33].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Extrusion
For exosomal drug loading, the mixture of exosomes and drugs are pushed repeatedly (x10 times) for extrusion through a lipid extruder (syringe-based) equipped with porous polycarbonate / whatman membranes (aperture: 100-400nm) and heating block under a controlled temperature (42°C) to get high drug loading efficiency with uniform size of exosomes [30- 33]. During the extrusion, the exosomal membranes are disrupted accompanied with vigorous mixing of drugs, and their loading into them by diffusion, while the excessive harsh mechanical force of extrusion (intensive extrusion and excessive shear stress) may alter the membranous zeta potentials and the structures of surface proteins. The extruded cargo-loaded exosomes may also be refined from free-cargos by gel-filtration chromatography accompanied with Sepharose 6 BCL.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Incubation with or without Surfactant-Assisted Permeation
The cargos-loading with incubation may be achieved by mixing the proposed small denaturation and exosomes or parent cells, while therapeutic molecules can pass passively for concentration gradient or by interactive attachments through / with the hydrophobic lipid bilayers / cholesterol / glycoprotein to enter into the exosomes or donor cells, dependent on the effective optimization of concentration of cargos, incubation temperature, volume and time, as appropriate or inappropriate rise in temperature may increase cargos-loading through the increment of the lipid membrane -fluidity or cause protein denaturation, and correlate the volumetric imbalance between the space and the electrostatic repulsion [30-32,34]. In this concern, donor cells may be treated with cargos for 24h, followed by cleansing and culturing with fresh medium. After 48h of culture, the generated cargo-carrying exosomes are isolated from cell conditioned medium. In general, for an exosomal drug loading, macrophagederived 250μL exosomes (diluted in PBS to a 0.15mg/mL of total protein) and small drug molecules (diluted in PBS to a 0.5mg/mL) are mixed to the final concentration of 0.1mg/mL total protein, followed by incubation at RT (37°C) for 1~18h. The surfactants like saponin or triton, used to increase membrane permeabilized cargos-loading into exosomes through the interactions with the membrane-bound cholesterol to create membranous pores, have been utilized in incubation treatment, while 0.01~0.2% saponin is used for the incubation of exosomes and drugs under shaking (~700 rpm) for 10~30min at 37°C with repetition of 2-7 times.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Transfection with Different Reagents
Cargos may be transfected into exosomes with various transfection agents for getting their higher efficiencies and molecular stabilities. The chemical transfection reagents, such as liposomes, diethylaminoethyl (DEAE)-dextran, polyethylenimine (PEI) and calcium phosphate, are capable to facilitate the nucleic acid loadings into / on exosomes, while calcium phosphates may form co-precipitates with nucleic acids, and others may form electrostatic complexations among positively charged nano / micro carriers and the negatively charged nucleic acids [30]. The commercial reagents, based on cell-penetrating peptides for the direct loadings of miRNAs and siRNAs into exosomes, require additional washings and purifications, as the reagents’ residuals may hamper vesicular reconstitutions as well as functionalities [30]. For reagents-based general transfections, nucleic acids are firstly mixed with appropriate reagents for forming complexes, followed by incubations with parent cells to initiate transfections. The specified nucleic acids are then manifestated as desired peptides or proteins to be packaged into the exosomes afterward through biogenesis [30]. In addition, siRNA / plasmid DNA may be integrated into exosomes through transfection using lipofectamine 2000 by incubation at RT for 30 min, followed by their transfections to recipient cells after being in culture for 24h [34,32].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Viral Transduction
Genetic therapeutic cargos are loaded to exosomes-based delivery systems by viral transduction to get their higher transfection abilities and stabilities [30]. After the infections of lentiviruses / adenoviruses to donor cells, the transduced and over-expressed or transcription-regulated specific genes are loaded into the exosomes or the exosomal membranes during their secretions [30].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Exosomal Surface Functionalizations
In order to utilize exosomes for specific targeting to cells, their membranes-surfaces are modified directly with chemical (click chemistry) methods, indirectly with genetic engineering of exosome-secreting cells, or by fusions of membranes (Figure 4) [30,35,31,34,22]. Direct chemical modifications of exosomes are carried out chiefly via conjugation reactions, hydrophobic insertion, click chemistry and receptor-ligand binding. Conjugation reactions can covalently and stably bind molecules to the exosomal surfaces. Lipophilic, amphipathic or hydrophilic molecules can be inserted or encapsulated into the exosomal lipid bilayers allowing their hydrophilic parts on the exterior or within hydrophilic core. Bioconjugations of macro or small molecules to the exosomal surfaces may be performed by copper-catalyzed azide alkyne cycloaddition named click chemistry, while the alkyne chemical groups react with azide chemical groups for forming triazole linkages. Exosomes cross-linked with alkyne groups may be conjugated to azides utilizing carbodiimide chemistry. Receptor-ligand-mediated cargo-delivery at specific target sites may be utilized by modifications of exosomes with polyethylene glycol-aminoethyl anisamide (PEG-AA), folate, aptamer and other components against various diseases. Modifications of exosomes with targeting molecules are attained by genetic engineering of exosome-producing cells via transfection with plasmid vectors. First, gene sequences of guiding proteins, peptides or ligands are fused with the selected exosomal surface- expressed genetically encodable targeting motifs of the transmembrane proteins (nonspecific or specific receptor membrane proteins). Subsequently, plasmids encoding fusion proteins -transfected donor cells secrete engineered exosomes bearing targeting surface-ligands. In addition, the exosomal lipid bilayer membranes can be fused with other types of membranous structures such as liposomes, virus and virus-stimulating vesicles to form hybrid exosomes through electrostatic interactions of cationic lipids and exosomes for strong cationic bindings to recipient cells and their cellular uptake. Moreover, incubated donor cells with synthetic azidebearing liposomes can secrete azide group bearing exosomal lipidmembranes and their bioconjugation with targeting peptides through click reactions can also attach a targeting moiety to the exosomal surface membranes.
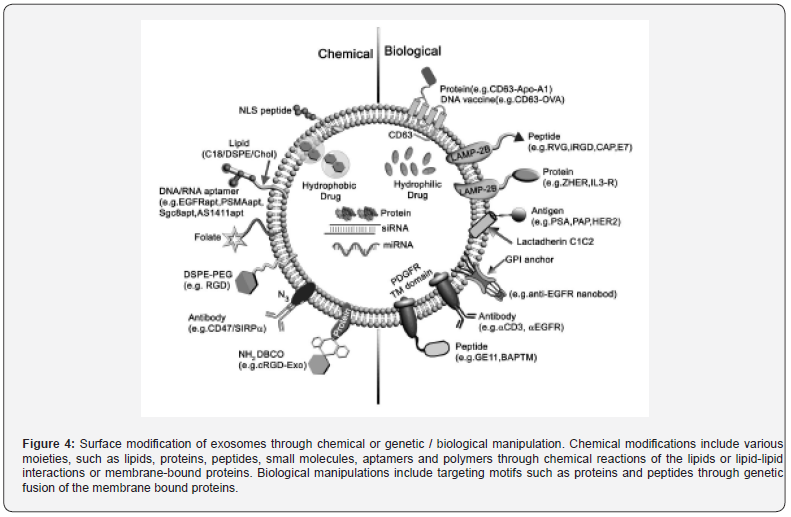
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Characterizations of Empty / Conjugated Exosomes
Empty / conjugated exosomes may be characterized on the basis of their protein and lipid contents, sizes and cargosconjugations. The shape, size (effective hydrodynamic diameter), number and size-distributed morphology of the exosomes (empty or conjugated) may be measured by using dynamic light scattering and nano tracking analysis, quantity by using Braford and BCA assay, topography by utilizing transmission electron microscopy, scanning electron microscopy, atomic force microscopy and Raman microspectroscopy, whereas their biological compositions may be examined by using western blot, fluorescence-activated cell sorting, resistive pulse sensing, flow cytometry, microfluidics, enzyme-linked immunosorbent assay, and electrochemical biosensors [36,37,13]. Various cargosloaded exosomal formulations may also be characterized by hyperspectral microscopy equipped with CytoVivo advanced dark field illumination system as well as diffraction grating hyperspectral imaging system, while the mean spectrum of each sample is determined utilizing the CytoVivo customized ENV1 hyperspectral image analysis software [33].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Therapeutic Applications of Exosomes
Generally, cargos are loaded into endogenous exosomes through their direct loading, or by indirect loading into donor cells and subsequent cargos-loaded exosomes-secretion, utilized for specific therapeutic applications against various diseases.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Delivery of Small Drug Molecules
Different studies have indicated that exosomes are efficient vectors for therapeutic small molecules [30,34,31,38]. The exosomes, produced from lymphoma cell line, encapsulated with curcumin have shown their suppressive affectivity against inflammatory diseases by the reduction of inflammatory markers [30,31,38]. Macrophage-derived exosomes loaded with paclitaxel have exhibited their higher cytotoxicity to multi-drug resistant cancer cells through bypassing the P-glycoprotein (P-gp) drug efflux system as well as anti-neoplastic activity with pulmonary anti-metastases [30,31]. Aminoethylanisamide-polyethylene glycol -incorporated paclitaxel-loaded exosomes have shown their improved anti-carcinogenic therapeutic indices against lung cancer cells through targeting the cancerous cells-expressed sigma receptors [30]. iRGD exosomes and reticulocyte (RTC)- derived exosomes loaded with doxorubicin have been treated against tumor tissue for getting higher anti-carcinogenic efficiency [34]. In addition, exosomes, isolated from colorectal cancer cell line, designed with engineered loading of doxorubicin, iron oxide and A33 antibody, have demonstrated their excellent targeting efficacy against colon cancer accompanied with decreased heart cytotoxicity [30].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Delivery of Large Peptides and Proteins Biomolecules
Various studies have investigated the exosomes-mediated peptides and proteins –deliveries [30,31,39,34,35,22]. The exosomal deliveries of catalase and dopamine have shown their anti-inflammatory, anti-microgliosis, anti-astrocytosis and antineurodegenerative efficiencies against Parkinson’s and other neuro diseases through antioxidant and transferin receptormediated targeting activities [30,31,34,35,39]. Membranous signal regulatory protein α (SIRPα) -loaded exosomes have been used for the treatment of tumor by blocking the tumoric CD47 receptor, and by phagocytosis and growth inhibition of tumor cells [30]. In addition, PH20 hyaluronidase and doxorubicin -loaded exosomes have exhibited their effective enhancement of tumor inhibition through hyaluronan-degraded tumor penetration [30]. Immature dendritic cells (imDCs)-derived exosomes containing over-expressed fusion protein of iRGD and lysosome-associated membrane protein-2b (Lamp 2b) loaded with doxorubicin have been utilized as anticancer treatment against breast cancer cells [34]. Mesenchymal stromal cells (MSCs) -derived curcumin loaded exosomes, conjugated with rabies virus glycoprotein and cyclo (Arg-Gly-Asp-DTyr-Lys) peptide, have been applied to treat cerebral ischemia through targeting integrin αvβ3 in reactive cerebral ischemic vascular endothelial cells [34]. Curcuminloaded exosomes conjugated with c(RGDyK) peptide have been utilized to treat cerebral ischemia through the enhancement of tissue-specific targeting capability [34]. GALA, the repeating sequences of Glu-Ala-Leu-Ala, the negatively charged amphiphilic pH-sensitive peptide, have been conjugated with cationic lipid and exosomes to achieve the higher efficiency of the cytosolic delivery of proteins and peptides by interacting cell membranes to mediate viral gene escape from acidic endosomes [34]. The fused membranes of liposomes and exosomes utilizing freezethaw method for optimizing exosomal surfaces to reduce immunogenicity and enhance colloidal stability and half-life in blood have been applied to transport exogenous hydrophobic and hydrophilic cargos to recipient cells by membrane fusions against diseases [34]. Exosome-coated inorganic metallic cations / clusters and multi-dentate organic ligands nanoparticles have been applied to get higher drug loading capacity and controlled drug release to targeted site/s in combating various diseases [34].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Delivery of Genetic Materials
Exosomes are utilized as vehicles for nucleic acids to deliver them to the targeted site/s of interest and to protect them from degradation in the systemic circulation, while siRNAs are used for disrupting genes of interest, and miRNAs, the short forms of non-coding RNA with non-protein nucleotides, are used to control post-transcriptional gene expressions by their binding to complementary sequences on targeted mRNAs [30,38,31,35]. One study has shown that exosomes have the capability to deliver siRNA against RAD51 gene protein for repressing the growth of proliferative cancerous cells [38]. Another investigation has indicated that exosomes loaded with albumin and siS100A4 have shown their capability to deliver siRNA for the significant inhibition of breast cancer metastasis [30]. Other investigation has exhibited that Lamp2b protein-targeted dendritic exosomes are capable to deliver GADPH siRNA and BACE1siRNA across the animal blood-brain barrier for suppressing mRNA expressions and β-amyloid against neurodegenerative diseases [31]. A few investigators have demonstrated that interleukin-3 (IL-3) exosomes fused with the N-terminal of Lamp2b and loaded with imatinib and BCR-ABL siRNA are able to kill chronic myeloid leukemia cells [35]. Other group of researchers has indicated that exosomes manifesting HER2-binding ankyrin repeat protein (DARPin) have delivered siRNA to HER2-positive tumor cells for their killing [35]. B cells / ovarian cancerous omental fibroblasts -derived exosomes loaded with miRNAs-155 inhibitors / miR- 199a-3p have been applied to inhibit lipopolysaccharide-induced TNFα production in macrophages or suppress c-Met production and inhibit cancerous cells- proliferation and invasion [30]. miR- 140-loaded exosomes functionalized with chondrocyte-affinity peptide have been utilized to inhibit osteoarthritis progression [35]. Exosomes functionalized with epidermal growth factors (EGFs) and epidermal growth factor receptor (EFGR)-specific peptides (GE11) carrying miRNA lethal-7 genes (let-7a) have been used to deliver let-7a to EGFR-expressed cancer cells to suppress tumor and inhibit the progress of cancer through the reduction of RAS and high-mobility group AT-hook protein (HMGA2) expressions [31,35,38]. Exosomes loaded with CRISPRdCas9- VP64 or CRISPR/Cas9 plasmids against poly (ADP-ribose) polymerase-1 (PAPR-1) have been utilized to attenuate hepatic fibrosis through activation of hepato stellate NHF42 expression or to inhibit cancer cells propagation [30].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Exosomes Removal as Disease Therapy
Diseased cells -derived exosomes transport disease-spreading proteins as well as immune suppressive molecules for propagation and progression of diseases, while removal of the exosomes from the systemic circulation to inhibit exosomes-production may be accomplished by targeting microtubules assembly, stability and endosomal sorting pathway and by utilizing proton pump inhibitors [28].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Pharmacokinetics, Biodistribution and Elimination of Exosomes
Exosomal tissue distribution is dependent on the cell sourcing, particle size, composition of lipid bilayer, surface charge, dosages and surface-modification, while most of unmodified exosomes is sequestered by liver, lung and spleen after intravenous administration [40,41]. Exosomes carrying slightly negative charged phosphatidylserine are cleared primarily by mononuclear phagocyte syetem (MPS) [42-44]. Exosomes containing surface-expressed glycosylphosphatidylinositol (GPI)- anchored CD59 and CD55 have been exhibited for protecting exosomes from complement-mediated lysis [45]. It has been investigated that the enhanced dosage of exosomes (400μg) may lead to massive lung accumulation, while a lower dosage (60μg) to the liver and spleen predominantly [46]. Exoxomes ranging from 100-200nm have been exihibited to accumulate into tumors by the enhanced permeability and retention (EPR) effect [47]. In addition, glioblastoma cells-derived exosomes have been utilized to target CCR8-positive glioblastoma cells through triple interactions with exosomes-expressed glycosaminoglycans and CCR8 ligand CCL18 as the bridging molecules [48]. Exosomal positively charged sphingomyelin / ganglioside GM3s have shown their boosted targeting uptakes into cancer cells in acidic tumor microenvironment through the better exosomes-cells fusions [49]. It is investigated that B16BL6 cells-derived luciferasepresenting exosomes have been departed from blood circulation with a half-life of 2 min after intravenous animal administration, followed by bioaccumulation to the liver, lung and spleen [30]. Other investigations have reported that exosomal surface deposition of complement protein C3, phosphatidylserine and CD169 ligand may induct macrophagic capture and systemic uptake [30]. A few investigators have shown the inhibition of macrophagic phagocytosis by exosomal protein CD47 through the interactions with the ligand signal regulatory protein α (SIRPα) followed by exosomal prolonged circulation time and alteration of biodistribution [30]. A few other researchers have exhibited that the biocompatible monomeric / polymeric / cholesterol-DNA tethered surface coating or incorporation onto / into exosomes may prolong circulation time from 10-60min or 4-fold longer circulating retention followed by selective biodistribution [30]. In general, exosomes, after systemic administration, are cleared up by systemic circulatory or residential macrophages, while the biodegradation of exosomal phospholipids takes place in blood circulation by lipases, and the endocytosed phospholipids are hydrolysed by lysozymes to fatty acids which are recycled in the body system, and cholesterols are metabolized to bile salts [42- 44,50,51].
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Conclusions and Future Perspectives
Targeted delivery with well-designed vectors has been utilized against various diseases to get reduced toxicity and high efficacy. Owing to the advancement on biocompatibility, multi cargo loading capability, site-specific surface modification tolerability and deep tissue penetration, exosomes, especially derived from mesenchymal stem cells and immune cells, have attracted interest as natural endogenous carriers against infections, inflammations, neurodegenerative diseases and cancer. However, optimization of exosomal nano formulation should be needed for the largescale production of purified, drug-loaded and quality controllable exosomes. In this context, more extensive and systematic in vivo investigations regarding pharmacokinetics biodistribution, toxicity and biological efficiencies are required to consider exosomes as effective nano medicinal delivery system in favor of clinical translation.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
Acknowledgement
This study was supported by the Council of Scientific and Industrial Research (CSIR), Government of India.
- Review Article
- Abstract
- Introduction
- Isolation of Exosomes
- Loading of Exosomes
- Sonication
- Electroporation
- Freeze and Thaw Cycles
- Extrusion
- Incubation with or without Surfactant-Assisted Permeation
- Transfection with Different Reagents
- Viral Transduction
- Exosomal Surface Functionalizations
- Characterizations of Empty / Conjugated Exosomes
- Therapeutic Applications of Exosomes
- Delivery of Small Drug Molecules
- Delivery of Large Peptides and Proteins Biomolecules
- Delivery of Genetic Materials
- Exosomes Removal as Disease Therapy
- Pharmacokinetics, Biodistribution and Elimination of Exosomes
- Conclusions and Future Perspectives
- Acknowledgement
- References
References
- Zhou QF, Cai YZ, Lin XJ (2020) The dual character of exosomes in osteoarthritis: Antagonists and therapeutic agents. Acta Biomater 105: 15-25.
- Nicholson C, Shah N, Ishii M, Annamalai B, Brandon C, et al. (2020) Mechanisms of extracellular vesicle uptake in stressed retinal pigment epithelial cell monolayers. Biochim Biophys Acta Mol Basis Dis 1866(3): 165608.
- Loggozzi M, Spugnini E, Mizzoni D, Di RR, Fais S (2019) Extracellular acidity and increased exosome release as key phenotypes of malignant tumors. Cancer Metastas Rev 38(1-2): 93-101.
- Riquelme JA, Takov K, Santiago FC, Rossello X, Lavandero S, et al. (2020) Increased production of functional small extracellular vesicles in senescent endothelial cells. J Cell Mol Med 24(8): 4871-4876.
- Palanisamy V, Sharma S, Deshpande A, Zhou H, Gimzewski J, et al. (2010) Nanostructural and transcriptomic analyses of human saliva derived exosomes. PLoS one 5(1): e8577.
- Hood JL (2016) Post-isolation modification of exosomes for nanomedicine applications. Nanomedicine (Lond) 11(13): 1745-1756.
- Vader P, Mol EA, Pasterkamp G, Schiffelers RM (2016) Extracellular vesicles for drug delivery. Adv Drug Deliv Rev 106: 148-156.
- Yuan D, Zhao Y, Banks WA, Bullock KM, Haney M, et al. (2017) Macrophage exosomes as natural nanocarriers for protein delivery to inflamed brain. Biomaterials 142: 1-12.
- Alenquer M, Amorim MJ (2015) Exosome biogenesis, regulation, and function in viral infection. Viruses 7(9): 5066-5083.
- Zhou J, Meng LC, Ye WR, Wang QL, Geng SZ, et al. (2018) A sensitive detection assay based on signal amplification technology for Alzheimer’s disease’s early biomarker in exosome. Anal Chim Acta 1022: 124-130.
- Jalalian SH, Ramezani M, Jalalian SA, Abnous K, Taghdisi SM (2019) Exosomes, new biomarkers in early cancer detection. Anal Biochem 571: 1-13.
- Zheng M, Huang M, Ma X, Chen H, Gao X (2019) Harnessing exosomes for the development of brain drug delivery systems. Bioconjug chem 30(4): 994-1005.
- Meng W, He C, Hao Y, Wang L, Li L, et al. (2020) Prospects and challenges of extracellular vesicle-based drug delivery system: Considering cell source. Drug Deliv 27(1): 585-598.
- Kim MS, Haney MJ, Zhao Y, Mahajan V, Deygen I, et al. (2016) Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine 12(3): 655-664.
- Schey KI, Luther JM, Rose KI (2015) Proteomics characterization of exosome cargo. Methods 87: 75-82.
- Lakhal S, Wood MJ (2011) Exosome nanotechnology: An emerging paradigm shift in drug delivery: Exploitation of exosome nanovesicles for systemic in vivo delivery of RNAi heralds new horizons for drug delivery across biological barriers. Bioessays 33(10): 737-741.
- Zhang Y, Chopp M, Meng Y, Katakowski M, Xin H, et al. (2015) Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurgery 122(4): 856-867.
- Lu M, Zhao X, Xing H, Xun Z, Zhu S, et al. (2018) Comparison of exosome-mimicking liposomes with conventional liposomes for intracellular delivery of siRNA. Int J Pharm 550(1-2): 100-113.
- Liao W, Du Y, Zhang C, Pan F, Yao Y, et al. (2019) Exosomes: The next generation of endogenous nanomaterials for advanced drug delivery and therapy. Acta Biomater 86: 1-14.
- Kamerkar S, LeBleu VS, Sugimoto H, Yang S, Ruivo CF, et al. (2017) Exosomes facilitate therapeutic targeting of oncogenic kRAS in pancreatic cancer. Nature 546(7659): 498-503.
- Johnsen KB, Gudbergsson JM, Skov MN, Pilgaard L, Moos T, et al. (2014) A comprehensive overview of exosomes as drug delivery vehicles-endogenous nanocarriers for targeted cancer therapy. Biochim Biophys Acta 1846(1): 75-87.
- Bunggulawa EJ, Wang W, Yin T, Wang N, Durkan C, et al. (2018) Recent advancements in the use of exosomes as drug delivery systems. J Nanobiotechnol 16: p 81.
- Walker S, Brusatto S, Pham A, Ming T, SuhA, et al. (2019) Extracellular vesicles-based drug delivery system for cancer treatment. Theranostics 9(26): 8001-8017.
- Salunkhe S, Basak M, Chitkara D, Mittal A, Dheeraj (2020) Surface functionalization of exosomes for target-specific delivery and in vivo imaging and tracking: Strategies and significance. J Control Release 326: 599-614.
- Thery C, Amigorena S, Raposo G, Clayton A (2006) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol 3: unit 3.22.
- Lai RC, Yeo RWY, Tan KH, Lim SK (2013) Exosomes for drug delivery - a novel application for the mesenchymal stem cell. Biotechnol Adv 31(5): 543-551.
- Vlassov AV, Magdaleno S, Setterquist R, Conrad R (2012) Exosomes: Current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim Biophys Acta 1820(7): 940-948.
- Oves M, Qari HA, Felamban NM, Khan AAP, Rehan M, et al. (2018) Exosomes: A paradigm in drug development against cancer and infectious diseases. J Nanomater 2018: 6895464.
- Qin J, Xu Q (2014) Functions and applications of exosomes. Acta Pol Pharm 71(4): 537-543.
- Chen H, Wang L, Zeng X, Schwarz H, Nanda HS, et al. (2021) Exosomes, a new star for targeted delivery. Front Cell Develop Biol 9: 751079.
- Luan X, Sansanaphongpricha K, Myers I, Chen H, Yuan H, et al. (2017) Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacolog Sinica 38(6): 754-763.
- Xi XM, Meng C, Xia SJ, Lu R (2021) Drug loading techniques for exosome-based drug delivery systems. Pharmazie 76(2): 61-67.
- Haney MJ, Klyachko NL, Zhao Y, Gupta R, Plotnikova EG, et al. (2015) Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J Control Rel 207: 18-30.
- Xu M, Yang Q, Sun X, Wang Y (2020) Recent advancements in the loading and modification of therapeutic exosomes. Front Bioengineer Biotechnol 8: 586130.
- Liang Y, Duan L, Lu J, Xia J (2021) Engineering exosomes for targeted drug delivery. Theranostics 11(7): 3183-3195.
- Zhang M, Jin K, Gao L, Zhang Z, Li F, et al. (2018) Methods and technologies for exosome isolation and characterization. Small Methods 2(9): 1800021.
- Tatischeff I, Larquet E, Falcon-Perez JM, Turpin PY, Kruglik SG (2012) Fast characterization of cell-derived extracellular vesicles by nanoparticles tracking analysis, cryo-electron microscopy and Raman tweezers microspectroscopy. J Extracell Vesicles 1: 1.
- Ha D, Yang N, Nadithe V (2016) Exosomes as therapeutic drug carriers and delivery vehicles across biological membranes: Current perspectives and future challenges. Acta Pharmaceutica Sinica B 6(4): 287-296.
- Qu M, Lin Q, Huang I, Fu Y, Wang I, et al. (2018) Dopamine-loaded blood exosomes targeted to brain for better treatment of Parkinson’s disease. J Control Rel 287:156-166.
- Armstrong JP, Stevens MM (2018) Strategic design of extracellular vesicle drug delivery systems. Adv Drug Deliv Rev 130: 12-16.
- Morishita M, Takahashi Y, Nishikawa M, Sano K, Kato K, et al. (2015) Quantitative analysis of tissue distribution of the B16BL6-derived exosomes using a streptavidin-lactadherin fusion protein and iodine-125-labeled biotin derivative after intravenous injection in mice. J Pharm Sci 104(2): 705-713.
- Wan Z, Zhao L, Lu F, Gao X, Dong Y, et al. (2020) Mononuclear phagocyte system blockade improves therapeutic exosome delivery to the myocardium. Theranostics 10(1): 218-230.
- Elsharkasy OM, Nordin JZ, Hagey DW, Jong OGD, Schiffelers RM, et al. (2020) Extracellular vesicles as drug delivery systems: Why and how? Adv Drug Deliv Rev 159: 332-343.
- Matsumoto A, Takahashi Y, Nishikawa M, Sano K, Morishita M, et al. (2017) Role of phosphatidylserine-derived negative surface charges in the recognition and uptake of intravenously injected B16BL6-derived exosomes by macrophages. J Pharm sci 106(1): 168-175.
- Clayton A, Harris CL, Court J, Mason MD, Morgan BP (2003) Antigen-presenting cell-exosomes are protected from complement-mediated lysis by expression of CD55 and CD59. Eur J Immunol 33(2): 522-531.
- Smyth T, Kullberg M, Malik N, Smith-Jones P, Graner MW, et al. (2015) Biodistribution and delivery efficiency of unmodified tumor-derived exosomes. J Control Rel 199: 145-155.
- Blanco E, Shen H, Ferrari M (2015) Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat Biotechnol 33(9): 941-951.
- Berenguer J, Lagerweij T, Zhao XW, Dusoswa S, Stoop PVD, et al. (2018) Glycosylated extracellular vesicles released by glioblastoma cells are decorated by CCL18 allowing for cellular uptake via chemokine receptor CCR8. J Extracell Vesicles 7(1): 1446660.
- Parolini I, Federici C, Raggi C, Lugini L, Palleschi S, et al. (2009) Microenvironmental pH is a key factor for exosome traffic in tumor cells. J Biol Chem 284(49): 34211-34222.
- Mandal AK (2018) Vesicular drug delivery against leishmaniasis in animals: A critical evaluation. Int J Curr Res 10(1): 64103-64113.
- Mandal AK (2022) Mitochondrial targeting of potent nanoparticulated drugs in combating diseases. J Biomater Appl 37(4): 614-633.






























