Prevalence of Endocrinological Medications among Pregnant Women in the Military and Khartoum Teaching Hospitals, Khartoum, Sudan, 2016
Haj Elamin AE1*, Mohammed RR2, Haj Elamin OE3 and Iman AM4
1Department of Clinical Pharmacy and Pharmacy Practice, Alfajr College and Khartoum University, Sudan
2Omdurman Islamic University, Sudan
3Sudan Medical Specialization Board, Khartoum Teaching Hospital, Sudan
4Omdurman Islamic University, Sudan
Submission: February 14, 2023; Published: March 29, 2023
*Corresponding author: Azhari Elnour Haj Elamin, Department of Clinical Pharmacy and Pharmacy Practice, Alfajr College and Khartoum University, Sudan
How to cite this article: Haj Elamin AE, Mohammed RR, Haj Elamin OE and Iman AM. Prevalence of Endocrinological Medications among Pregnant Women in the Military and Khartoum Teaching Hospitals, Khartoum, Sudan, 2016. Glob J Pharmaceu Sci. 2023; 10(4): 555791. DOI: 10.19080/GJPPS.2023.10.555791.
Abstract
Background: Normal women have intricate modifications to their maternal endocrine systems as a result of the heightened metabolic demands of pregnancy. Endocrinologic system disorders may have harmful repercussions on the mother, the embryo, or the fetus. Increases in infertility, spontaneous abortions, congenital deformities, abnormalities in maternal/ fetal metabolism, and maternal and fetal deaths are some of these impacts. Certain endocrine disorders, including gestational diabetes mellitus is emerge spontaneously during pregnancy. While pre-existing endocrine conditions (such thyroid issues) may aggravate, improve, or remain stable throughout gestation.
Objectives: Our goal is to enlighten readers on the prevalence and rate /frequency with which pregnant women use endocrinological drugs in outpatient settings of Hospitals in Khartoum, Sudan. Moreover, to determine the correlation and association between the characteristics of pregnant women and the endocrinological medications.
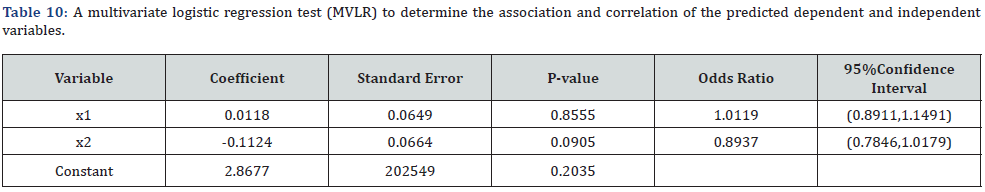
Materials and Methods: The Military Hospitals and Khartoum Teaching Hospital served as the sites for this descriptive cross-sectional hospital study, Sudan (between Jan. 2015 and Jan. 2016). The information was taken from the files of 650 pregnant subjects using a data collection form. Age, trimester, parity, medical history, maternal problems, and endocrinological medications were among the details that were noted. The sample size was a total coverage (N = 134) to all records of subjects attended the outpatient clinics of the two hospitals in the study period. On the Microsoft Windows 10 Professional operating system, data were analyzed using the SPSS16 program, and the Chi square test, which considers p 0.05 statistically significant, was used. The anticipated different variables (dependent and independent) were tested for the correlation using a multivariate logistic regression test (MVLR).
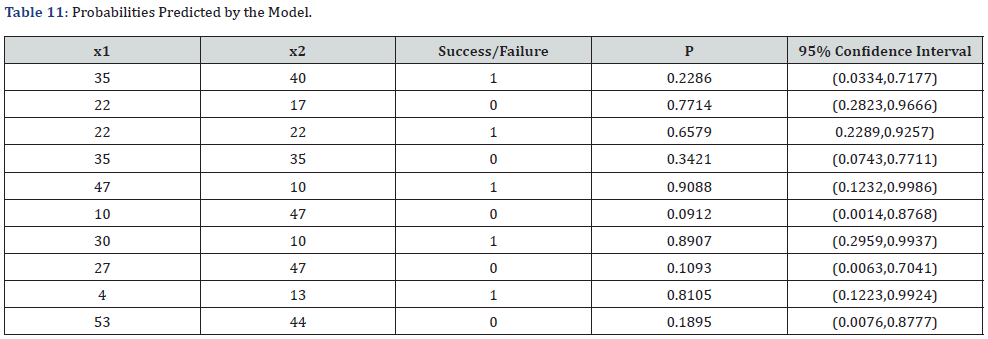
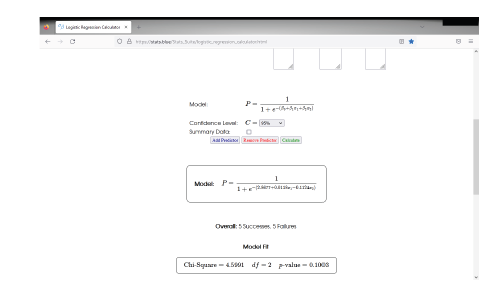
Results: The 650 pregnant women at Khartoum Teaching Hospital and Military Hospitals in Sudan met the criteria for the study between January 2015 and January 2016; about 20.6% (N = 134) received endocrinological drugs. Among the study subjects (650), insulin and levothyroxine were the two most frequently prescribed endocrinological drugs (No=82; 12.6%) and (No=52; 8 %). Of 134 (20.6%) medications, 22 (16.4%) used levothyroxine in the second trimester (4-6 months) and 30 (22.4%) in the third trimester. About 35 (26.1%) of the pregnant women used insulin in the second trimester (4-6 months) and 47 (35.1%) in the third trimester (7-9 months). While in medical history, 8 (6.0%) with hyperthyroidism got levothyroxine and 40 (29.9%) with diabetes got Insulin. Of 650 subjects we found that the age of 256(39.4%) was between (15-25 years), 310 (47.7%) between (26-35 years) and 84 (13%) between (36-45 years). In trimester, bar-wise, 9 (1.4%) were in the first trimester (1-3 months), 78 (12%) in the second trimester (4-6 months) and 563 (86.6%) in third trimester (7-9 months). In parity, primigravida were 210 (32%) while multigravida were 440 (67.7%). About 154 (23.7%) of the pregnant women had maternal disorders, however, most of them 15(2.3%) had hypertension. The MVLR test determined the correlation and association between predictor’s different variables in term of (correlation coefficient, standard error, P-value, df, odds ratio “OR” and OR (95% CI)). I.e., the characteristics of pregnant women and the frequency and rates of endocrinological drugs. (Chi- square 4.5091, df 2, P-value=0.1003).
Conclusion and Recommendations: According to the study, the uses of endocrinological medication were evaluated rigorously and mainly it was focus in the treatment of pregnant diabetics and hypothyroidism patients; included their different conditions, pregnancy stages, maternal disorders, and medical histories’. However, the sample size was a total coverage (N = 134) of subjects had adequate information/ data, the study results / findings of prevalence of endocrinological drugs among pregnant women in Sudan may consider reasonable in light of literature review (mainly in effectiveness and toxicity). Pregnant diabetics should know that maintaining strict glucose control, particularly in the first trimester, will increase the likelihood of having a healthy baby. Contrarily, a healthy pregnancy causes a variety of significant hormonal and physiological changes that affect thyroid functions. Due to these modifications, thyroid function tests must be evaluated carefully during pregnancy. Before getting pregnant, parents ought to learn about the good habits they may start practicing right now to increase the likelihood of a healthy pregnancy. Usage of endocrinological medications among pregnant women should be prescribed carefully by physicians and verified by pharmacists for appropriateness of prescribing to optimize the treatment and to avoid drug related problems (DRPs).
Keywords: Pregnant Women; Trimester; Endocrinological Medications; Military Hospitals; Khartoum Teaching Hospital; Insulin; Levothyroxine
Introduction
Due to the heightened metabolic needs of pregnancy, normal women experience complex alterations in their maternal endocrine system. Endocrinologic system disorders may have negative effects on the mother, the embryo, or the fetus. Increased infertility, spontaneous miscarriage, fetal deformities, abnormalities in the mother’s / fetus’ metabolism, maternal development, and moreover fetus’ deaths are a result of these consequences [1,2,3]. While some endocrine conditions, like gestational diabetes mellitus, develop on their own during pregnancy; others, like thyroid issues, may worsen, get better, or stay stable during gestation [1,4,5].
Diabetes mellitus in pregnancy
Diabetes mellitus is a major endocrine disorder. A partial or complete shortage of insulin results in diabetes mellitus, a chronic illness; there is 0.2-0.3% of all pregnancies is complicated by it 3,4,5]]. Clinical signs and symptoms might range from an asymptomatic hyperglycemic condition to severe diabetic ketoacidosis, unconsciousness, and even death. In about 2 to 3 percent of pregnancies, there is a condition known as gestational diabetes mellitus that is defined by glucose intolerance that appears in the second to third trimesters [3,6,7].
Diabetic embryopathy
Compared to the general population, children of women who had diabetes mellitus prior to becoming pregnant had a two- to four-fold higher rate of congenital abnormalities. Prior to the 8th week of gestation, when organ development takes place, is the crucial window of time when the teratogenic effect of overt maternal diabetes manifests [3]. Cardiovascular, skeletal, and central nervous system birth abnormalities are frequently observed in offspring of diabetic mothers. Babies born to diabetic mothers today have chances of being healthy as those born to babies to non-diabetic mothers, that is due to the development of tools and strategies to maintain normoglycemia before, during, and between all pregnancies It is crucial to remember, however, that because gestational diabetes mellitus exposure occurs outside of the crucial phase of organogenesis, newborns of women who develop the condition do not have an elevated risk for such problems [ 3,6,7].
Fetal complications
Significant neonatal morbidity is a danger for babies born to women who had diabetes previous to becoming pregnant as well as those who developed gestational diabetes mellitus. The objective of insulin therapy is to achieve and maintain normoglycemia before, during, and after all pregnancies complicated by diabetes because research conclusively identifies glucose as the teratogen. The risk of respiratory distress syndrome (RDS), macrosomia, hypoglycemia, hyperbilirubinemia, and hypocalcemia in these newborns is elevated. The risk of fetal death is also two to three times higher than it is in the general population [3,6,7]. Higher amounts of glycosylated hemoglobin (HbAlc) are directly correlated with more abnormalities occurring. Compared to women with HbAlc values that are closer to the normal range during this time, those with higher HbA1c values have a much higher risk of newborns with abnormalities. In 20% to 25% of pregnancies complicated by diabetes, macrosomia—defined as a birth weight >4 kg—occurs. During the neonatal period, infants of diabetes mothers (IDM) are also more likely to experience prolonged hypoglycemia after birth, respiratory distress syndrome (RDS), hypocalcemia, polycythemia, and hyperbilirubinemia [1,2, 3, 5,7]. Although there is still some debate, it is generally agreed that these issues can be less frequent in pregnant women with diabetes. [3,6,7]. Strict maternal metabolic management, enhanced prenatal surveillance, and newborn intensive care have all significantly decreased the perinatal mortality rate for children of diabetic mothers.
Management of pregnancy diabetic patient
Several features distinguish the management of diabetes during pregnancy from the general therapy of diabetes. These include the following:
(1) Oral hypoglycemic agents are contraindicated. It is not advised to use oral hypoglycemic medication.
(2) Weight reduction is not advised since fatal nutrition can be adversely affected.
(3) To increase the likelihood of having healthy, normal kids, intensive insulin therapy with frequent self-monitoring of blood glucose is generally recommended. Every effort should be made, utilizing multiple injections of insulin or a continuous infusion of insulin by pump, to maintain near normalization of fasting and preprandial blood glucose values while avoiding hypoglycemia. Glycohemoglobin should be maintained in the normal range. Since many diabetic pregnancies persist beyond the expected term or because the infants are usually large and hyramnios may be present it has been suggested that the baby should be delivered early (37-38 weeks), especially if glycemic control during pregnancy have been inadequate (e.g., Glycohemoglobin >10%). There is a current tendency toward inducement of labor rather than elective cesarean sections Pregnancy and the diabetic patient [1, 2, 3, 5, 7].
Hypothyroidism in pregnancy
In a healthy pregnancy, thyroid hormone production is raised to keep up with the expanding fetal placental unit’s increased physiologic demands. Pregnancy-related changes in thyroid function result from a number of processes. Notably, the formation of thyroxine binding globulin (TBG) in the liver is up-regulated by two or threefold during the first half of gestation, with serum estrogen levels rising to 500–1000 pg/mL [8,9,10]. Increased TBG levels change the equilibrium between bound and free thyroxine (FT4), resulting in a brief drop in FT4 levels. This temporary drop in FT4 levels then triggers increased thyroid gland stimulation by the hormone thyrotropin (TSH), leading to the physiologic restoration of FT4 at the expense of higher serum total T4 (TT4) levels. The increased placental synthesis of human chorionic gonadotropin (HCG), which peaked at about 50,000–75,000 IU/L at 8–11 weeks, is a second contributor. This is important because HCG directly stimulates thyrocytes, and this impact is mediated by binding to the TSH receptor. Thirdly, there is the increased requirement for iodine during pregnancy, which is necessary to support the rise in thyroid hormone production and made worse by the iodine loss as a result of enhanced renal clearance [8,9,11]. Therefore, between 250 and 500 ug/d is the suggested range for daily iodine intake during pregnancy [12,13]. The existence of placental iodothyronine deiodinase type III, which modifies T4 availability, distribution, and metabolism for both mother and fetus, is a last contributing factor [11]. It is believed that 0.3-0.5% of pregnant women have overt hypothyroidism (OH) and 2-3% have subclinical hypothyroidism (SH). In women of childbearing age, thyroid autoantibodies are present in 5–18% of cases, and chronic autoimmune thyroiditis (AITD) [8,14,15].
Fetal complications
Hypothyroidism, if left untreated, can reduce fertility, and increase the risk of congenital abnormalities, spontaneous abortions, and still births.
Constipation, dry skin, coarse hair, irritability, and intolerance to the cold are other symptoms. Due to the fact that many of these symptoms are typical of healthy pregnancy, clinical diagnosis may be challenging. It is unclear how maternal hypothyroidism impacts the developing fetus.
According to some research, there is little cause for alarm, while other reports indicate that the children of hypothyroid women have a high prevalence of congenital deformities and poor mental and physical development [8,9,11].
Management of hypothyroidism
Thyroxine is the drug of choice. In most young healthy patients with disease of short duration, therapy can be started with 100-150μg daily. In the elderly or those with cardiac disease the initial dose should be lower (12.5 – 25μg daily). This dose may be increased by 25-50μg (12.5 - 25μg in elderly or cardiac disease) at intervals of at least 4 weeks. The usual maintenance dose to relieve hypothyroidism is 100-200μg daily. In children a daily dose of 10μg/kg up to a maximum of 50μg daily should be given.
Adverse effects of thyroxine include anginal pain, tachycardia, cramps in skeletal muscle, headaches, restlessness, excitability, flushing, sweating, diarrhoea, excessive weight loss and arrhythmias.
Triiodothyronine (Liothyronine sodium, T3) may also be used as replacement therapy; it has a short half-life and the resultant fluctuations in serum T3 concentrations above and below normal can predispose the typical elderly patient to supraventricular tachyarrhythmias. The initial dose in adults is 10-20μg daily in two or three divided doses, gradually increasing to 20μg three times daily [12,13,14,15].
Special circumstances in the management of hypothyroidism
• Ischemic heart disease – If angina is present, thyroid hormone replacement may increase the risk of myocardial infarction; in this case treatment of hypothyroidism should start with a low dose of thyroxine, that is, 25μg daily. The patient should be closely observed.
• Congenital (neonatal) hypothyroidism – Infants with evidence of hypothyroidism should be treated with 8-10μg/kg/ day of thyroxine, if possible, by the 14th day postnatal. Treatment can then be reduced to 6μg/kg/day from the age of six and 3μg/kg/day from the age of 12.
• Pregnancy – Patients with established hypothyroidism may require their thyroxine dose to be increased dependent on TSH levels. After delivery, the dose should be reduced to previous levels.
• Temporary post-partum hypothyroidism – Thyroiditis may occur 6-12 months post-partum, resulting in transient hypothyroidism. In most cases replacement therapy is not required [12,13,14,15].
Patient monitoring in hypothyroidism
In patients with hypothyroidism the pharmacist should monitor the following:
• T4 and TSH level (TSH levels once replacement therapy has commenced).
• Patient symptoms, that is, weakness, fatigue weight gain, cold intolerance.
• Cardiovascular monitoring, to check for heart failure, angina, or bradycardia.
• Signs of drug toxicity, that is, tachycardia, arrhythmias, due to thyroxine therapy.
• Drug therapy that may precipitate hypothyroidism [13, 14,15].
Objectives
Our aim is to enlighten readers on use and rate /frequency with which pregnant women administered endocrinological drugs in outpatient settings of the Military and Khartoum Teaching Hospitals in Khartoum, Sudan. Furthermore, to determine the correlation and association between the characteristics of pregnant women and the endocrinological drugs.
Materials and Methods
From January 2015 to January 2016, Khartoum Teaching Hospital (Sudan) and Military Hospitals conducted a descriptive cross-sectional hospital study. The information was taken from the files of 650 pregnant patients using a data collection form. The sample size was a total coverage (N = 134) to all records of the pregnant women attended the outpatient clinics of the two hospitals in the study period. Age, trimester, medical history, medical investigations, and endocrinological medications were all listed in the information records. The information extracted included characteristics of subjects (age, trimester, parity), medical history, maternal disorders (independent variables) and endocrinological medications (dependent variable) to determine the correlation and association between the characteristics of the pregnant women and prescription of endocrinological drugs. On the Microsoft Windows 10 Professional operating system, data were analyzed using the SPSS16 program, and the Chi square test was used, which considers p 0.05 to be statistically significant. A multivariate logistic regression test (MVLR) was used to determine the association and correlation of the predicted dependent and independent variables.
Results
Sample:
The study sample consisted of 650 patients of pregnant women.
Age:
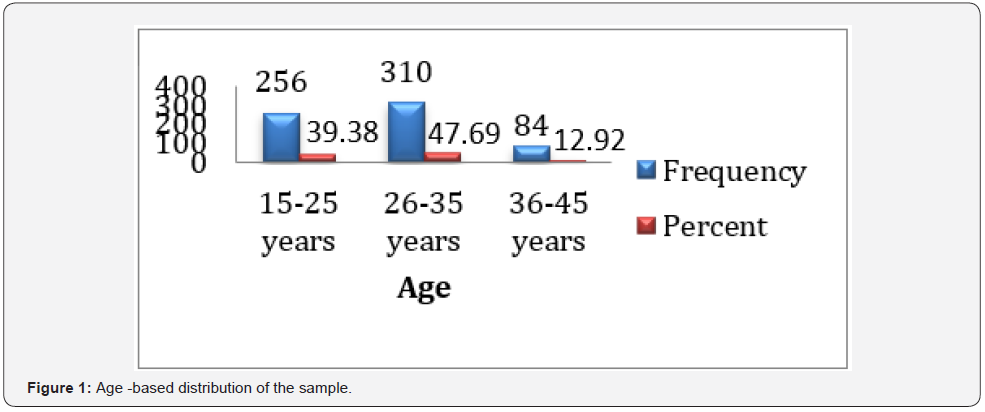
Of a total of 650 pregnant women included in this study we found that the age of 256(39.38%) was between (15-25 years), 310 (47.7%) between (26-35 years) and 84 (13%) between (36- 45 years), as shown by (Table 1) and (Figure 1).
Trimester:
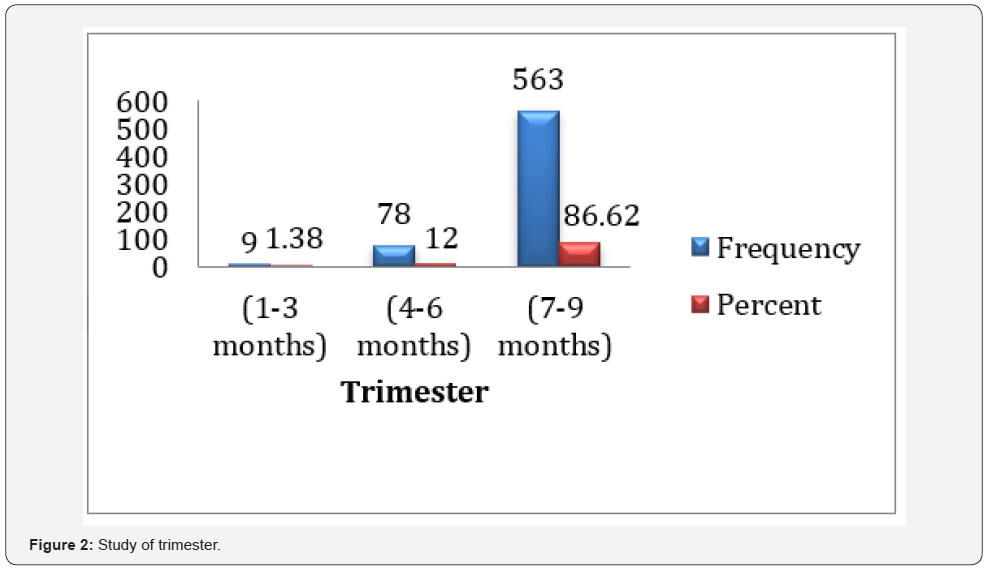
Trimester bar-wise, 9 (1.38%) were in the first trimester (1-3 months), 78 (12%) were in the second trimester (4-6 months) and 563 (86.62%) were in third trimester (7-9 months), as shown by (Table 2) and (Figure 2).
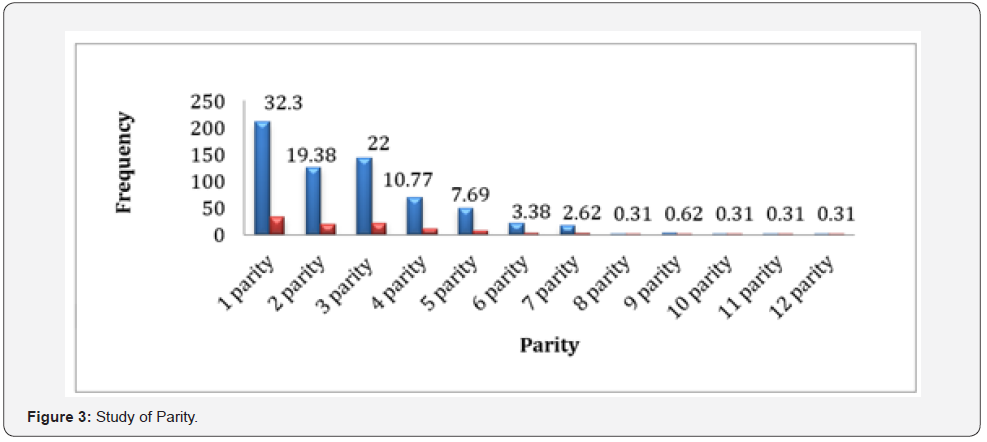
Parity:
Primigravida were 210 (32%) while multigravida were 440 (67.7%), as shown by (Table 3) and (Figure 3).
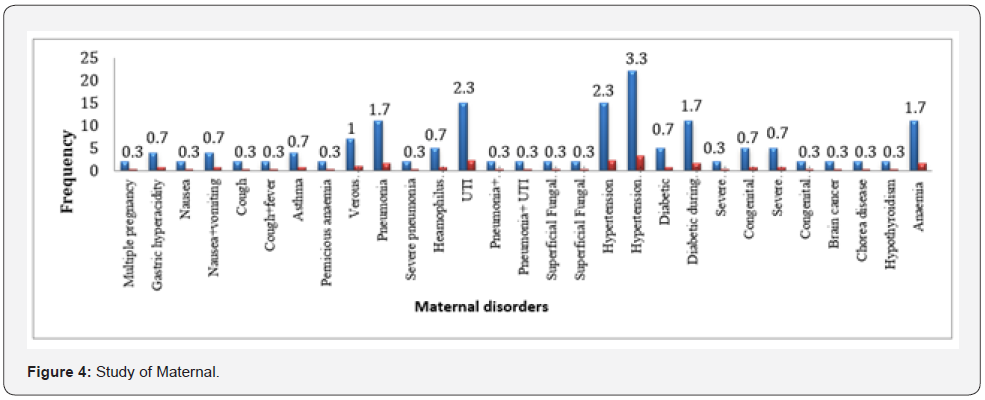
Maternal Disorders:
One hundred and fifty four 154 (23.7%) of the pregnant women had maternal disorders, however, most of them 15(2.3%) had hypertension during pregnancy; this is shown in (Table 4) and (Figure 4).
Endocrinoogical Drugs:
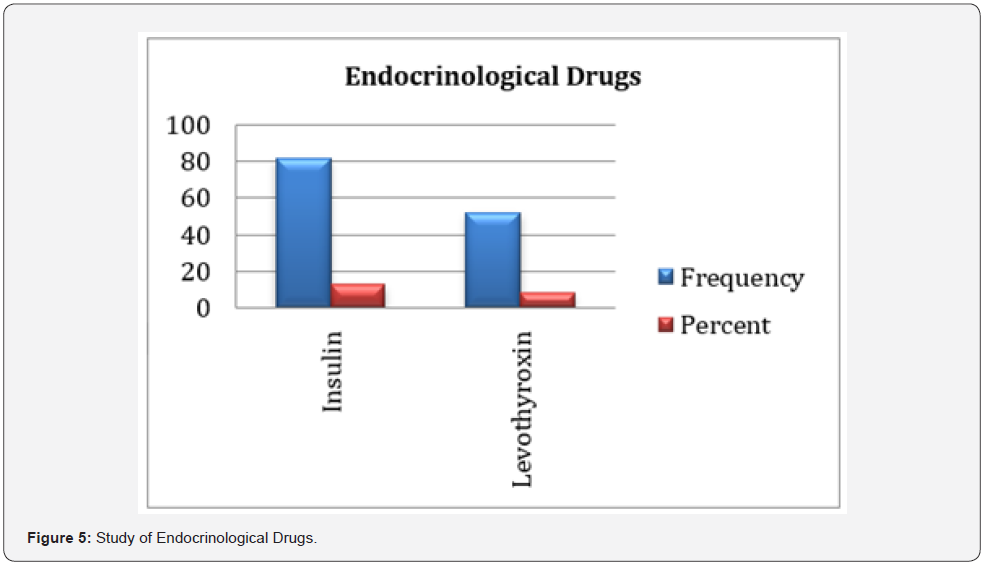
One hundred and thirty four (20.62%) were on endocrinological drugs. 82 (12.62%) on insulin therapy and 52 (8%) on levothyroxine therapy. This is shown in (Table 5) and (Figure 5).
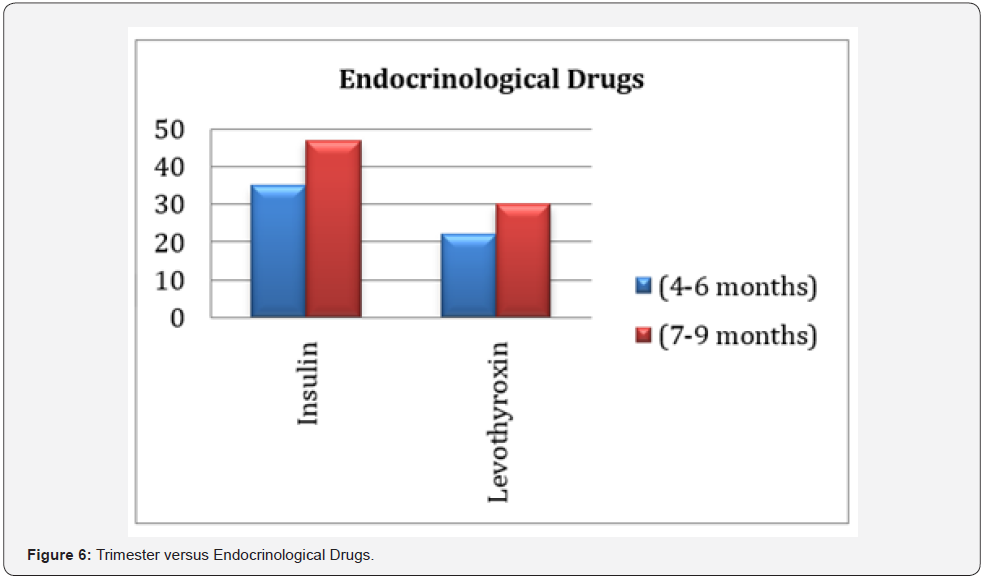
Trimester versus Endocrinological Drugs:
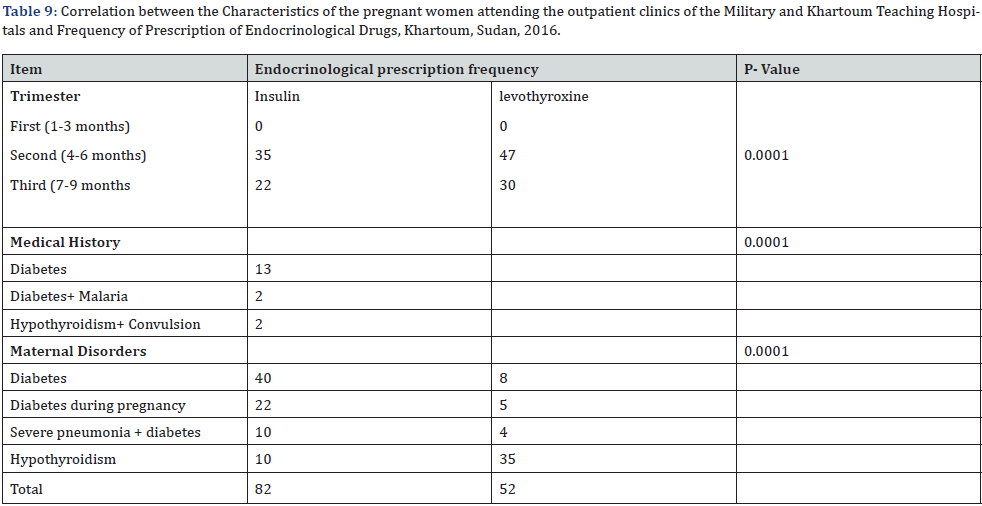
About 35 (26.1%) of the pregnant women used insulin in the second trimester (4-6 months), 47 (35.1%) used in the third trimester (7-9 months), while 22 (16.4%) used levothyroxine in the second trimester (4-6 months) and 30 (22.4%) used in the third trimester (7-9 months). This is given in (Table 6) and (Figure 6). The difference of use of insulin and levothyroxine in second and third trimesters was very significant with p<0.0001 at 95% confidence (0.000-0.01).
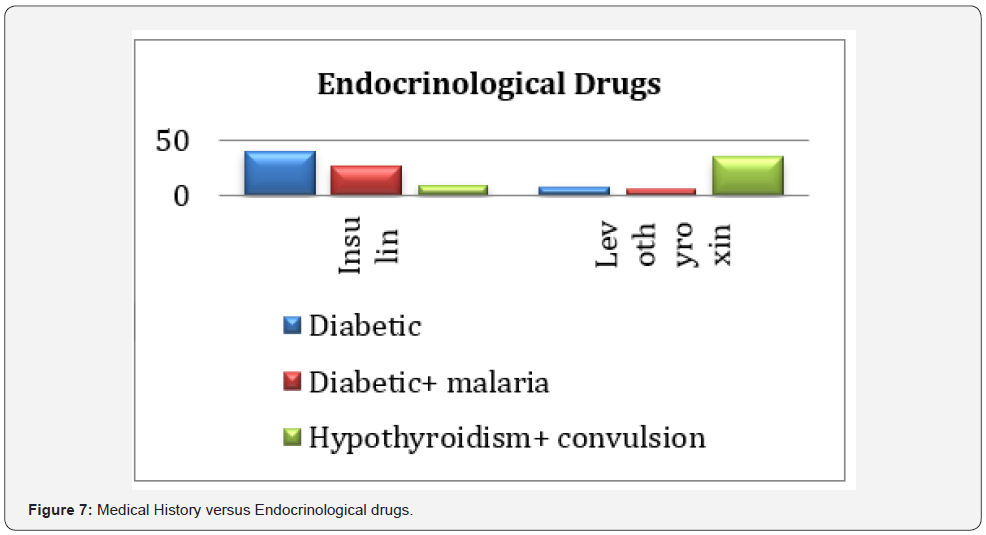
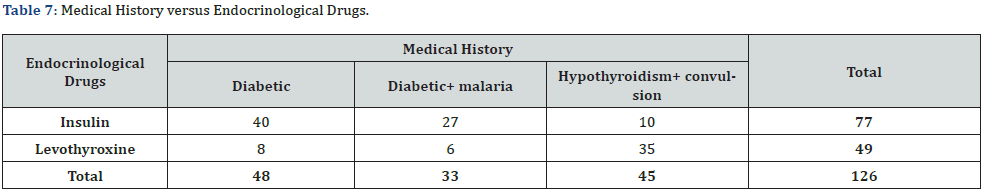
Medical History versus Endocrinological Drugs
There were 40 (29.85%) of diabetic pregnant women had received insulin and 8 (6%) with hyperthyroidism had received Levothyroxine, this is given in (Table 7) and (Figure 7). The difference of use of insulin and levothyroxine in medical history was very significant with p<0.0001 at 95% confidence (0.0001-0.01).
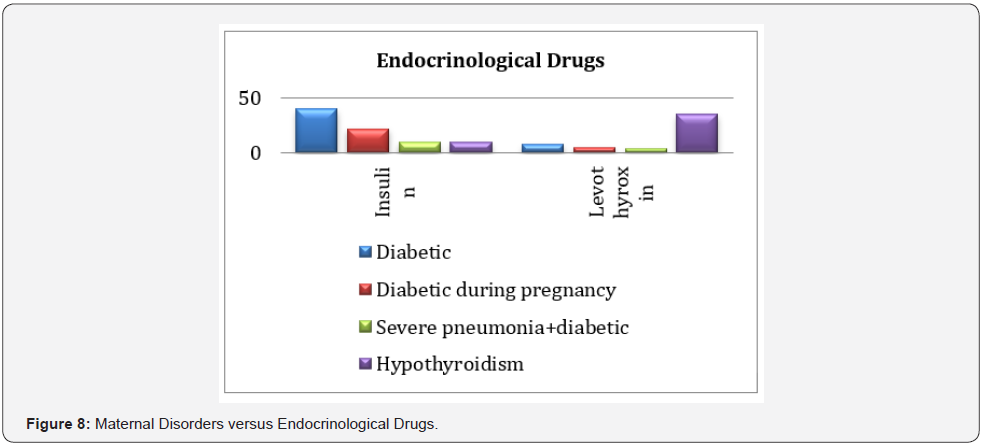
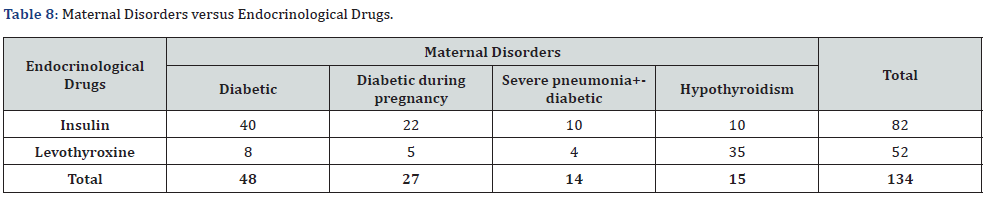
Maternal Disorders versus Endocrinological Drugs:
40 (6.2%) of the pregnant women were diabetic while 22 (3.4%) were diabetic during pregnancy, this is given in (Table 8) and (Figure 8). The difference of use of insulin and levothyroxine in maternal disorders was very significant with p<0.0001 at 95% confidence (0.0001-0.01) (Table 9-11).
Discussion
The study described the uses of endocrinological medication were evaluated rigorously and mainly in the treatment of pregnant diabetics and hypothyroidism patients; included their different conditions, pregnancy stages, maternal disorders, and medical histories’. However, the sample size was a total coverage (N = 134) of subjects had adequate information and data.
In normal situations, the beta cells of the pancreas create the hormone insulin, which controls the metabolism of glucose and other substances. The placenta does not allow for physiologically relevant levels of human insulin to pass. Insulin is often given by subcutaneous injection, although it can also be given intravenously in an emergency or under pressure when tight control is required (such as during delivery), lowering blood sugar levels quickly. Due to insulin significantly adverse drug reactions (ADRs) and potential drug interactions PDIs care should be taken. For its lower antigenicity (e.g., ADR), human insulin (semisynthetic or biosynthetic) is recommended over animal insulin. Because maternal insulin antibodies have the potential to change the pharmacokinetics of insulin and cross the placenta. They are crucial because they can cause fetal hypoglycemia, beta-cell hyperplasia, and hyperinsulinemia [3,4,5]. Therefore, the majority of endocrinologists concur that pregnant women shouldn’t use immunogenic (animal) insulin. Early research revealed that both free insulin and insulin antibody complexes were impermeable to the human placenta. However, it now appears that significant amounts of animal insulin that has been antibody-bound can pass through the placenta. Exenatide is a non-oral medication that can be used to treat diabetes; however, it has not been researched during pregnancy [3,4,5].
It has been observed pregnancies complicated by diabetes were decreased in Developed Country.as a result of strict maternal metabolic control/follow-up, improved fetal surveillance, and neonatal intensive care. Although, due to lack of facilities the perinatal complication and mortality for infants of diabetic mothers were observed in many health care systems of Developing Countries. (2. 5) Infants of diabetic mothers (IDM) are at increased risk for prolonged hypoglycemia after delivery, respiratory distress syndrome (RDS), hypocalcemia, polycythemia, and hyperbilirubinemia during the neonatal period were also observed in pregnant diabetic women.
Preconception Management
To reduce the risk of significant congenital malformations, good glycemic control (HbA1c) levels up to >1% above normal) should be attained months before conception. A direct correlation exists between higher glycosylated hemoglobin levels and increased frequency of anomalies. Women with higher HbA1c values during this time have a significantly higher incidence of infants with anomalies compared with women with HbAlc closer to the normal range. Pregnant diabetics should know that maintaining strict glucose control, particularly in the first trimester, will increase the likelihood of having a healthy baby. Before getting pregnant, they ought to learn about the good habits they may start practicing right now to increase the likelihood of a healthy pregnancy [1,2,3,5,7].
Pregnant diabetics should be started on prenatal vitamins containing at least 400 mcg of folic acid and re-educated about diet and glycemic control of her diabetes. Her insulin therapy should be titrated to reduce her preprandial (capillary whole blood) glucose to 70 to 100 mg/dL and her 2-hour postprandial glucose to below 140 mg/dL. Good glycemic control (HbA1c levels up to >1% above normal) should be achieved months before conception to minimize risks of major congenital anomalies [1,2,3,5,7].
High hypertension of pregnant diabetics should be decreased to a diastolic BP of about 80 mm Hg to minimize risks for pre-eclampsia or exacerbation of her disorder. Many women with pregestational diabetes are likely on an angiotensin converting enzyme inhibitor (ACEI) or angiotensin receptor blocker for hypertension treatment. We should consider other antihypertensives (e.g., methyldopa or calcium channel blockers) because recent studies have observed increased rates of congenital cardiac malformation associated with the use of ACEI in the first trimester [1, 2, 3, 5, 7].
Care should be taken in the management of pregnant diabetic and hypertensive women in Developing Countries (e.g., Sudan); however, in general, aspects for consideration in the management of diabetes include (Glycemic control optimization. Blood pressure management Dietary modification. Exercise. Weight loss/ treatment of obesity. Smoking cessation and stress reduction. Treatment of hyperlipidemia. Antiplatelet agents. Treatment of infection).
The thyroid gland typically produces the hormone L-thyroxine (T4) It is used to treat hypothyroidism and is safe to take while pregnant. Among537 pregnancies exposed to exogenous thyroxine or thyroid hormone during the first trimester and 1605 pregnancies exposed at any point during pregnancy did not result in an increased frequency of congenital abnormalities. The results in humans were consistent with experimental investigations. It should be understood that using thyroxine while pregnant is safe.
In the study, of a total of 650 pregnant women it was found that the age of 256(39.38%) was between (15-25 years), 310 (47.7%) between (26-35 years) and 84 (13%) between (36-45 years); Trimester bar-wise, 9 (1.38%) were in the first trimester (1-3 months), 78 (12%) were in the second trimester (4-6 months) and 563 (86.62%) were in third trimester (7-9 months), primigravida were 210 (32%) while multigravida were 440 (67.7%), 154 (23.7%) of the pregnant women had maternal disorders, however, most of them 15(2.3%) had hypertension during pregnancy; .Roughly 40 (6.2%) of the pregnant women had diabetes, whereas 22 (3.4%) developed it during pregnancy. About 134 (20.62%) were on endocrinological drugs; 82 (12.62%) on insulin therapy and 52 (8%) on levothyroxine therapy. Approximately 35 (26.12%) of the pregnant women used insulin in the second trimester (4-6 months), and 47 (35.07%) in the third trimester (7-9 months). About 30 (22.38%) in the second trimester (4-6 months) and 22 (16.4%) of the participants utilized levothyroxine during the third trimester (7-9 months).
The rate of ADRs and PDIs of insulin therapy and levothyroxine were few. The difference of use of insulin and levothyroxine in second and third trimesters was very significant with p<0.0001 at 95% confidence (0.000-0.01). The difference of use of insulin and levothyroxine in medical history was very significant with p<0.0001 at 95% confidence (0.0001-0.01). The difference of use of insulin and levothyroxine in maternal disorders was very significant with p<0.0001 at 95% confidence (0.0001-0.01). Furthermore, a MVLR test determined the correlation and association between predictor’s different variables in term of (correlation coefficient, standard error, P-value, df, odds ratio “OR” and OR (95% CI)). I.e., the characteristics of pregnant women and the frequency and rates of prescription endocrinological medications. (Chi- square 4.5091, df 2, P-value=0.1003) The percent of use of insulin (29.9%) and levothyroxine (6.0%) indicate rate/frequency of prescribing in pregnant women in Sudan may be considered fair. However, the sample size was a total coverage (N = 134) of subjects had adequate information and data. The study results / findings of prevalence of endocrinological drugs among pregnant women in Sudan may consider reasonable in light of literature review (mainly in effectiveness and toxicity).
Conclusion and Recommendations
According to the study, the uses of endocrinological medication were evaluated rigorously and mainly in the treatment of pregnant diabetics and hypothyroidism patients; included their different conditions, pregnancy stages, maternal disorders, and medical histories’.
The difference of use of insulin and levothyroxine in second and third trimesters was very significant with p<0.0001 at 95% confidence (0.000-0.01). The difference of use of insulin and levothyroxine in medical history was very significant with p<0.0001 at 95% confidence (0.0001-0.01). The difference of use of insulin and levothyroxine in maternal disorders was very significant with p<0.0001 at 95% confidence (0.0001-0.01). Furthermore, MVLR test determined the correlation and association between predictor’s different variables in term of (correlation coefficient, standard error, P-value, df, odds ratio “OR” and OR (95% CI)). I.e., the characteristics of pregnant women and the frequency and rates of prescription of endocrinological drugs. (Chi- square 4.5091, df 2, P-value=0.1003).
The percent of use of insulin (29.9%) and levothyroxine (6.0%) indicate rate/frequency of prescribing in pregnant women in Sudan may be considered fair. However, the sample size was a total coverage (N = 134) of subjects had adequate information and data. The study results/findings of prevalence of endocrinological drugs among pregnant women in Sudan may consider reasonable in light of literature review (mainly in effectiveness and toxicity).
Pregnant diabetics should know that maintaining strict glucose control, particularly in the first trimester, will increase the likelihood of having a healthy baby. Contrarily, a healthy pregnancy causes a variety of significant hormonal and physiological changes that affect thyroid functions. Due to these modifications, thyroid function tests must be evaluated carefully during pregnancy. Human chorionic gonadotropin (HCG) and estrogen are the two main hormones that have an impact on thyroid functions testing during pregnancy. Before getting pregnant, parents ought to learn about the good habits they may start practicing right now to increase the likelihood of a healthy pregnancy.
Women and their doctors should however be reassured that there are safe options to treat endocrinological disorders, during pregnancy and breastfeeding. Usage of endocrinological medications among pregnant women should be prescribed carefully by physicians and verified by pharmacists for appropriateness of prescribing to optimize the treatment and to avoid drug related problems (DRPs).
References
- Kimble MK, Young LY, Kradjan WA, Applied Therapeutics. The Clinical Use of Drugs. 9th edition. Lippincott Williams & Wilkins Press, US.
- Patrick M Malone, Karen L Kier, John E Stanovich (2016) Drug Information: A Guide for Pharmacists, 4th
- Little B Bertis (2006) Drugs and pregnancy handbook, Endocrine disorders, contraception, and hormone therapy during pregnancy, Oxford University, New York, chapter 4: 77-83.
- Briggs GG, Freeman RK, Yaffe SJ In: Mitchell CW, ed. (2002) Drugs in pregnancy and lactation: a reference guide to fetal and neonatal risk. 6th Lippincott Williams and Wilkins, Philadelphia, United States.
- Joseph T, DiPiro R, Robert L, Talbert G, Yee C, et al. (2008) Pharmacotherapy- A Pathophysiologic Approach (Pharmacotherapy), 7th edition, McGraw-Hill Medical Posey Press, New York.
- Andrade SE, Gurwitz JH, Davis RL, Chan KA, Finkelstein JA, et al. (2004) Prescription drug use in pregnancy. Am J Obstet Gynecol 191(2): 398-407.
- Brunton L, Chabner BA, Knollman B (2011) Goodman and Gilman's (The Pharmacological Basis of Therapeutics). 12Edition McGraw-Hill Medical Press -New York.
- Glinoer D (1997) The regulation of thyroid function in pregnancy: pathways of endocrine adaptation from physiology to pathology. Endocr Rev 18(3): 404-433.
- Glinoer D (2004) The regulation of thyroid function during normal pregnancy: importance of the iodine nutrition status. Best Pract Res Clin Endocrinol Metab 18(2): 133-152.
- Krassas GE, Poppe K, Glinoer D (2010) Thyroid function and human reproductive health. Endocr Rev 31(5): 702-755.
- Smallridge RC, Glinoer D, Hollowell JG, Brent G (2005) Thyroid function inside and outside of pregnancy: what do we know and what don’t we know? Thyroid 15(1): 54-59.
- Allan WC, Haddow JE, Palomaki GE, Williams JR, Mitchell ML, et al. (2000) Maternal thyroid deficiency and pregnancy complications: implications for population screening. J Med Screen 7(3): 127-130.
- Abalovich M, Amino N, Barbour LA, Cobin RH, De Groot LJ, et al. (2007) Clinical practice guideline: management of thyroid dysfunction during pregnancy and postpartum: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 92(8 Suppl): S1-S47.
- Klein RZ, HaddowJE, Faix JD, Bown RS, Hermos RJ, et al. (2007) Prevalence of thyroid deficiency in pregnant women. Clin Endocrinol (Oxf) 35(1): 41-46.
- WHO (2013): Model List of Essential Medicines (2011).






























