Survey of the In Vitro Antifungal Activity of Paeonia broteri Extracts
Ana Margarida Madureira1 and Generosa Teixeira2,3*
1iMed, Faculdade de Farmácia, Universidade de Lisboa, Av. Prof. Gama Pinto, 1649-003, Lisboa, Portugal
2cE3c - Centre for Ecology, Evolution and Environmental Changes & CHANGE - Global Change and Sustainability Institute, Faculdade de Ciências, Universidade de Lisboa, C2, 1749-016, Lisboa, Portugal
3DCFM, Faculdade de Farmácia, Universidade de Lisboa, Av. Prof. Gama Pinto, 1649-003,
Submission: June 20, 2022; Published: July 06, 2022
*Corresponding author: Generosa Teixeira
How to cite this article:AM Madureira, G Teixeira. Survey of the In Vitro Antifungal Activity of Paeonia broteri Extracts. Glob J Pharmaceu Sci. 2022; 9(5): 555775. DOI: 10.19080/GJPPS.2022.09.555775.
Abstract
Objective: Some species of Paeonia are widely studied as they are used in traditional Chinese medicine as an anti-inflammatory, antioxidant, antitumor, immune-system-modulation and antiseptic. There is still a lack of knowledge about other temperate Eurasia species. Such is the case of P. broteri, an Iberian endemism whose preliminary studies have the purpose to evaluate antifungal properties of their roots, leaves and fruits extracts.
Methods: The microdilution method was used to evaluate the minimum inhibitory concentration (MIC) of P. broteri extracts against 13 selected fungi and yeasts: Aspergillus niger, Candida albicans, C. dubliniensis, C. glabrata, C. guilliermondii, C. krusei, C. parapsilosis, C. tropicalis, Cryptococcus neoformans, Geotrichum sp., Rhodotorula rubra, Saccharomyces cerevisiae and Trichosporon cutaneum. P. broteri extracts where obtained by sequential extraction of increasing polarity (n-hexane, CH2Cl2, EtOAc, MeOH and H2O) and their phytochemical profile was screened.
Results: P. broteri roots and leaves extracts were the most active, in particular against Geotrichum sp, T. cutaneum and A. niger. The chemical profile of these extracts, flavonoids, phenols and terpenoids, may be responsible for their antifungal activity.
Conclusion: Studies with P. broteri extracts indicate that they may have antifungal potential.
Keywords: Paeonia broteri; Antifungal activity; Phytochemical screening; Microdilution method
Introduction
Chemotaxonomic studies placed the genus Paeonia L. in the monogeneric family Paeoniaceae Raf. [1-4]. Their 35 species are distributed in 3 sections: Moutan, Oneapia, and Paeonia, all living in the temperate areas of North hemisphere [5]. Some Paeonia species have a long tradition of use in Chinese medicine, among them Paeonia lactiflora Pallas (Syn: P. albiflora Pallas, P. edulis Salisb, P. officinalis Thunb) [6-9]. Phytochemical studies on these and on a few other Paeonia taxa, have revealed more than 262 compounds, belonging to different chemical groups: monoterpenoid glucosides, triterpenoids, steroids, stilbenoids, flavonoids, tannins and phenols [10,7]. Several biological and pharmacological activities, such as an antiseptic, anti-inflammatory, antioxidant, antitumor, immune-system-modulation, cardiovascular-system and central-nervous-system, have been assigned to those chemicals [7,11-13]. In Portugal P. broteri Boiss. & Reut. (Syn: P. broteri, P. lusitanica) is endemic and can be found in understory of oak populations [14]. To our knowledge few local western species have been studied so far. Our objective was to evaluate the antifungal properties of P. broteri extracts, obtained from roots, leaves and fruits, against selected pathogenic fungi.
Materials and Methods
Plant material
Samples of tuberous roots, leaves and fruits of P. broteri were collected from native populations, in a mountainous area of Southwestern Portugal (38º 8’N-8º 33’W). Plant material was identified, and voucher specimens were deposited in the Lisbon Botanical Garden Herbarium (LISU 221345).
Plant extracts
P. broteri samples were dried in the dark, at room temperature and ground into powder separately. About 100g of each powder was extracted sequentially, with increasing polarity solvents, [n-hexane (n-hex), dichloromethane (CH2Cl2), ethyl acetate (AcOEt), methanol (MeOH) and water (H2O)], for 24h, at room temperature with occasional shaking. The obtained extracts were filtered, evaporated under reduced pressure and stored at 4ºC, until use, with the exception of the aqueous extract which has been freeze-dried.
Phytochemical screening
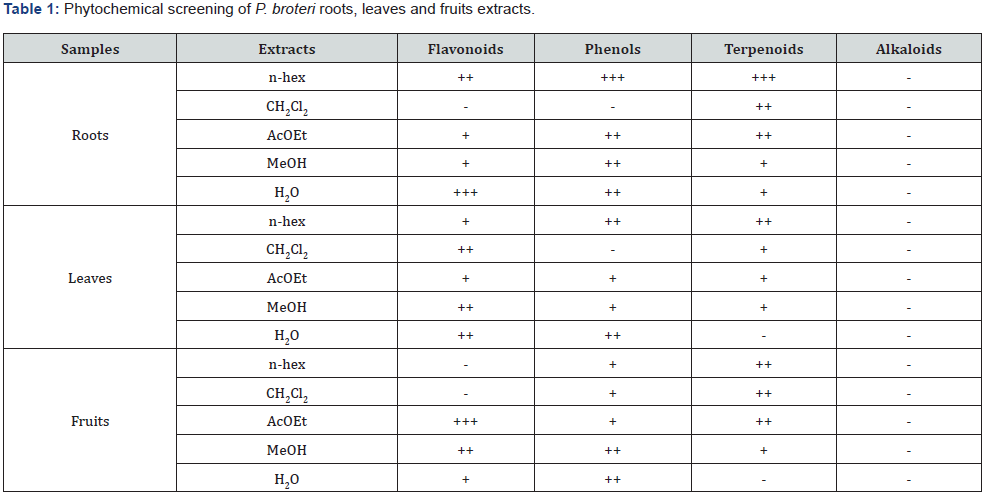
Each extract was dissolved in proper solvents, applied on silica gel thin layer chromatography plates and de¬veloped with appropriate mixtures of eluents. The plates, containing an application of each extract, were revealed with spray specific reagents for the presence of each main class of metabolites NEU for flavonoids, FBS for phenols, anisaldehyde / sulfuric acid for terpenoids and saponins, and Dragendorff for alkaloids [15]. Results were displayed semi-quantitative in a range between absent (-), slight (+), moderate (++) and strongly present (+++).
Fungi strains
The selected fungi included 13 filamentous fungi and yeasts: Aspergillus niger (ATCC 16888), Candida albicans (ATCC 90028), C. dubliniensis (FFUL 21), C. glabrata (FFUL 12B), C. guilliermondii (FFUL 1403), C. krusei (ATCC 6258), C. parapsilosis (ATCC 90018), C. tropicalis (ATCC 750), Cryptococcus neoformans (FFUL 948), Geotrichum sp. (ATCC 96884), Rhodotorula rubra (FFUL 190), Saccharomyces cerevisiae (FFUL 1997) and Trichosporon cutaneum (FFUL 18H).
Minimal inhibitory concentration
The microdilution method was used to evaluate the minimum inhibitory concentration (MIC) of each of the P. broteri extracts against the selected fungi. The MIC is the lowest concentration that inhibits the development of a microorganism. The different extracts were tested in a concentration range between 500 - 7.5μg/ mL and the culture medium used was the Sabouraud liquid. The microplates were incubated at 37°C for 24h, with the exception of A. niger, which required 48h of incubation period. All the assays were repeated at least in three independent experiments. Positive values were considered when MIC<100μg/mL [16] and verified by observing the wells turbidity.
Results
Phytochemical screening
The main classes of metabolites in each extract are shown in Table 1. The samples corresponding to the three plant structures do not have alkaloids, but their phytochemical profile shows some differences regarding the presence of flavonoids, phenols, terpenoids.
Antifungal activity
Table 2 shows the obtained MIC values of the P. broteri extracts against A. niger, C. albicans, C. dubliniensis, Geotrichum sp., R. rubra, and T. cutaneum. No interesting results were obtained against C. glabrata, C. guilliermondii, C. krusei, C. parapsilosis, C. tropicalis, C. neoformans and S. cerevisiae. Considering the root samples, the n-hex extract strongly inhibited (MIC=7.5μg/mL) the growth of A. niger and of G. candidum; the AcOEt extract showed moderate inhibition (MIC=30μg/mL), against C. albicans and C. dubliniensis the MeOH extract moderately inhibited (MIC=30μg/mL) the growth of Geotrichum sp. and slightly inhibited (MIC=62.5μg/ mL) the growth of T. cutaneum the H2O extract strongly inhibited (MIC=7.5μg/mL) the growth of A. niger and of Geotrichum sp. and moderately inhibited (MIC=30μg/mL) the growth of T. cutaneum
The results obtained with the leaves samples indicate that the n-hex extract moderately inhibited (MIC=30μg/mL) the growth of A. niger and strongly inhibited (MIC=7.5μg/mL) the growth of Geotrichum sp, the AcOEt extract showed a slight inhibition (MIC=62.5μg/mL) of growth of Geotrichum sp. and of T. cutaneum the MeOH extract had a slight inhibitory activity (MIC=62.5μg/ mL) on R. rubra the H2O extract inhibited strongly to moderately (MIC=15μg/mL) the growth of A. niger, strongly (MIC=7.5μg/mL) of Geotrichum sp. and moderately (MIC=30μg/ml) of R. rubra and of T. cutaneum. The activity of P. broteri fruit samples show that the n-hex extract moderately inhibited (MIC=30μg/mL) the growth of T. cutaneum the H2O extract had a slight inhibition (MIC=62.5μg/ mL) over A. niger, a moderate inhibition (MIC=30μg/mL) over T. cutaneum and a strong inhibition over Geotrichum sp. (MIC=7,5 μg/mL). Table 2 also shows that CH2Cl2 extracts had no inhibition at all against the tested fungi

Discussion
The phytochemical screening performed on P. broteri roots, leaves and fruits extracts showed some semi-quantitative differences regarding the presence of flavonoids, phenols, terpenoids and verified as well the absence of alkaloids. The MIC evaluations carried out with those extracts revealed antifungal activity against some of the tested fungi. In descending order, the most sensitive fungi were Geotrichum sp., T. cutaneum, A. niger, R. rubra, C. albicans, C. dubliniensis (Table 2). No antifungal activity was noticed against C. tropicalis.
Lai et al. [17] performed a phytochemical screening on crude organic extracts of leaves and fruits of P. broteri and evaluated their antimicrobial activity against standard and resistant strains of Gram-positive, Gram-negative bacteria, Mycobacterium smegmatis and C. albican. The same authors found that the most polar extracts strongly inhibited Staphylococcus aureus standard, VRSA and MRSA strains but no inhibition against C. albicans was noticed [17]. Picerno et al. [18] working with root polar extracts of P. rockii found a moderate inhibition (MIC=30μg/mL) against C. albicans. These same authors attributed the antifungal activity to the chemical composition of the mentioned extract, rich in phenolic compounds, gallic acid derivatives and paeoniflorina, a monoterpene glycoside [18]. The last compound seems to be ubiquitous in all the species examined of the Paeoniaceae family and it is considered as a chemotaxonomic marker [1,19]. Papandreou et al. [20] isolated volatile constituents from roots of 3 Paeonia species and detected their antimicrobial activity against pathogenic bacteria and against C. albicans, C. tropicalis and C. glabrata. P. broteri root n-hex extract revealed activity against A. niger and Geotrichum sp. and only the AcOEt root extract showed activity against C. albicans and C. dubliniensis (Table 2).
Conclusion
The phytochemistry of P. broteri was beyond the scope of our study but considering our results with those of the other authors,it is suggested that the biological activity of P. broteri extracts can be attributed to the synergistic effect of terpenoids, flavonoids and phenolics. The compounds involved and their mechanism of action are unknown and need to be elucidated. The activity of P. broteri extracts indicates that they may have potential as antifungal and as a source of new models of bioactive molecules, but further investigations are needed.
Acknowledgement
This work was funded by national funds through Fundação para a Ciência e a Tecnologia (www.fct.pt/), under the project UIDB/00329/2020.
Conflict of interests
The authors have no conflicts of interest to declare. The authors are entirely responsible for the paper’s content and writing.
References
- Sang T, Crawford D, Stuessy T (2007) Chloroplast DNA phylogeny, reticulate evolution, and biogeography of Paeonia (Paeoniaceae). Am J Bot 84(8): 1120.
- Tamura M (2007) Paeoniaceae In: Klaus Kubitski (Ed.), The Families and Genera of Vascular Plants, Volume IX. Springer-Verlag, Berlin Heidelberg, Germany, pp. 265-269.
- Jian S, Soltis P, Gitzendanner M, Moore M, Li RQ, et al. (2008) Resolving an Ancient, Rapid Radiation in Saxifragales. Syst Biol 57(1): 38-57.
- Cheng Y, Chang Ho K, Dong Il S, Seong Min K, Han Mo K, et al. (2011) Development of simple sequence repeat (SSR) markers to study diversity in the herbaceous peony (Paeonia lactiflora). J Med Plants Res 5: 6744-6751.
- APG III (2009) An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants. Bot J Linnean Soc 161: 105-121.
- (1999) WHO Monographs on Selected Medicinal Plants, Radix Paeoniae.
- He CN, Peng Y, Zhang YC, Xu LJ, Gu J, et al. (2010) Phytochemical and Biological Studies of Paeoniaceae. Chem Biodivers 7(4): 805-834.
- He DY, Dai SM (2011) Anti-inflammatory and immunomodulatory effects of Paeonia lactiflora Pall., a traditional Chinese herbal medicine. Front Pharmacol 2: 10.
- Tan YQ, Chen HW, Li J, Wu QJ (2020) Efficacy, chemical constituents and pharmacological actions of Radix Paeoniae Rubra and Radix Paeoniae Alba. Front Pharmacol 11: 1054.
- Wu SH, Wu DG, Chen YW (2010) Chemical Constituents and Bioactivities of Plants from the Genus Paeonia. Chem Biodivers 7(1): 90-104.
- Blumenthal M (1998) The Complete German Commission E Monographs: Therapeutic Guide to Herbal Medicines (1998) Integrative Medicine Communications, Newton, Germany, p. 364.
- Lin HC, Ding HY, Ko FN, Teng CM, Wu YC (1999) Aggregation inhibitory activity of minor acetophenones from Paeonia species. Planta Med 65: 595-599.
- Halda J, Waddick J (2004) The genus Paeonia. Timber Press: Oregon, USA.
- Garmendia FM, Navarro C, Castroviejo (1993) Flora Iberica-Plantas Vasculares de la Península Ibérica e Islas Baleares 3: 144-150.
- Wagner H, Blader S (1996) Plant drug analysis: A Thin Layer Chromatography Atlas. (2nd edn), Springer-Verlag, Berlin, Heidelberg, Germany, p. 384.
- Cos P, Vlietinck AJ, Berghe DV, Maes L (2006) Anti-infective potential of natural products: How to develop a stronger in vitro “proof-of-concept. J Ethnoph 106: 290-302.
- Lai B, Teixeira G, Moreira I, Correia AI, Duarte A (2012) Evaluation of the antimicrobial activity in species of a Portuguese "Montado" ecosystem against multidrug resistant pathogens. J Med Plants Res 6: 2381-2387.
- Picerno P, Mencherini T, Sansone F, del Gaudio P, Granata I (2011) Screening of a polar extract of Paeonia rockii: Composition and antioxidant and antifungal activities. J Ethnopharmacol 138: 705-712.
- Duan WJ, Yang JY, Chen LX, Zhang LJ, Jiang ZH (2009) Monoterpenes from Paeonia albiflora and their inhibitory activity on nitric oxide production by lipopolysaccharide-activated microglia. J Nat Prod 72(9): 1579-1584.
- Papandreou V, Magiatis P, Chinou I, Kalpoutzakis E, Skaltsounis AL, et al. (2009) Volatiles with antimicrobial activity from the roots of Greek Paeonia taxa. J Ethnopharmacol 81(1): 101-104.






























