Effect of γ -Cyclodextrin on the Dissolution of Ginsenosides Rg5 and Rk1 from Red Ginseng Chewable Tablets
Ginosyan Aghavni1*, Prazyan Anna1, Davinyan Andranik1 and Hovhannisyan Areg2
1Scientific Center of Drug and Medical Technologies Expertise of the Ministry of Health of the Republic of Armenia Yerevan, Armenia
2Institute of Fine Organic Chemistry of the National Academy of Science of the Republic of Armenia, Yerevan, Armenia
Submission: February 4, 2022; Published: February 17, 2022
*Corresponding author: Ginosyan Aghavni, Scientific Center of Drug and Medical Technologies Expertise of the Ministry of Health of the Republic of Armenia Yerevan, Armenia
How to cite this article:Ginosyan A, Prazyan A, Davinyan A, Hovhannisyan A. Effect of γ -Cyclodextrin on the Dissolution of Ginsenosides Rg5 and Rk1 from Red Ginseng Chewable Tablets. Glob J Pharmaceu Sci. 2022; 9(4): 555766. DOI: 10.19080/GJPPS.2022.09.555766.
Abstract
This study aimed to assess the water solubility of Ginsenosides Rg5 and Rk1 released from chewable tablets containing Red Ginseng preparation HRG80 incorporated γ-cyclodextrin (GCD) complex compared with γ-cyclodextrin free drug preparation. The dissolution rate of Ginsenosides Rg5 and Rk1 was increased three times after γ-cyclodextrin inclusion. The relative solubility of Ginsenosides Rg5 and Rk1 calculated from the ratio of Area under the curve AUC 0 – 90 min was correspondingly 221% and 227%. This study for the first reports that Red Ginseng preparation HRG80 inclusion into GCD significantly improves (increases) the water solubility of active constituents Ginsenosides Rg5 and Rk1.
Keywords: Ginsenosides Rg5 and Rk1; γ-cyclodextrin; Red ginseng; HRG80; Dissolution profile
Introduction
Ginsenosides Rg5 and Rk1 are major active constituents of Red Ginseng, comprising correspondingly about 1.9% and 1.0% of herbal preparation HRG80 of Panax ginseng C.A. Mey [1-4]. Both ginsenosides exhibit pleiotropic pharmacological activity [1-13], but their content in the blood circulation system is low due to poor solubility in water [14-16]. A growing body of evidence suggests that gamma-cyclodextrin (GCD) can increase the clinical efficacy of water-insoluble biologically active compounds, which have low bioavailability [17]. GCD is the most bio adaptable and applicable to increase the absorption of many drugs [18], including ginsenosides of Panax ginseng [19,20], by forming inclusion complexes or the form of GCD/drug conjugates [19,21,22]. Since GCD inclusion method is known to improve the dissolution of poorly soluble compounds including ginsenoside Re and compound K [19,20], we used GCD as an inclusion host for the complex with red ginseng preparation of HRG80. The aim of this study was to compare the water solubility of Ginsenosides Rg5 and Rk1 from the γ-cyclodextrin complex of red ginseng containing chewable tablets with the solubility of the same, but cyclodextrin free, Red Ginseng commercial preparation HRG80 capsules.
Materials and Methods
Chemicals
Test Samples of Red Ginseng Energy Chewable Tablets (Lot 2241-0201, Exp. date Jan 2023), and Red Ginseng Energy Capsules (Lot 201049, Exp. date Jan 2023), containing correspondingly 100 mg and 200 mg of red ginseng preparation HRG80, were obtained from Euro pharma USA Inc. Green Bay, USA. The red ginseng HRG80TM preparation was standardized for the content of total ginsenosides Rg1, Re, Rf, Rb1, Rg2, Rc, Rh1, Rb2, F1, Rd, Rg6, F2, Rh4, Rg3-(S-R), PPT (20-R), Rk1, C(k), Rg5, Rh2, Rh3, 20S-PPT, and PPD (7.6%, including 1.9% Rg5 and 1.0 % Rk1) [2]. The content of ginsenoside Rg5 is 1.124 mg/CD-tablet and 2.628 mg/capsule, the range of ginsenoside Rk1 is 0.89 mg/CD-tablet and 2.19 mg/capsule. The content of GCD (CAVAMAX W8 Food, MW 1297, Wacker Chemie AG, Germany) in c Red Ginseng Energy Chewable Tablets was 400 mg per tablet. Reference Standards of Ginsenosides Rg5 (CAS#186763-78-0, (purity > 98%)) and Ginsenosides Rk1, (CAS Number: 494753-69-4: (purity > 98%)) were purchased from Shanghai Standard Technology Co., Ltd., Reagents and HPLC Solvents: Methanol (purity > 99,9%, Gold Ultragradient, Carlo-Erba), acetonitrile (purity > 99,9%, Carlo Erba), ammonium acetate (purity > 99%, ChemLab, ACS).
Dissolution test
The dissolution test was conducted by the dissolution tester (Erweka, DT800, Germany), containing 500 ml or 900ml of water correspondingly for tablets and capsules at 37.0℃±0.5℃ according to USPNF [23]. The speed was set at 75 rpm. The test samples of 5 ml were collected in 0,15,30,45,60,90 min followed by the adjusting of the equilibration media volume. The collected samples were filtered using Millex-HV 045 syringe filters before HPLC analysis.
Analytical method
The analysis was performed using a Shimadzu LC/MS instrument (LC-20AD/T, Shimadzu Corporation, Kyoto, Japan) consisting of autosampler (LC-20AD/SIL-20A), HPLC column (Symmetry Shield, RP18, 5μm, 250x4.6mm), UV-diode array detector (SPD-M20A IVDD), and Data acquisition and analysis software (LabSolutions, Version 3.40.299. Shimadzu). The column and autosampler temperatures were maintained at 30℃ and 10℃, respectively. The injection volume was 20μl. The mobile phase consisted of 1mM ammonium acetate in water (solvent A) and ACN (solvent B). The binary gradient elution conditions were optimized as follows: linear gradient from 35 to 60%B (0-4 min), 60% B (4-18 min), 60-80% B (18-20 min), 80% B (20-22 min), 80-35% B (22-25 min). The flow rate was set at 0.5 mL/min Quantification was carried out at 203nm. Calibration curves were prepared using the reference standards of Rg5 and Rk1 in four concentrations: 1.0,2.0,3.0,4.0μg/ml in methanol. The linearity coefficients were R2=0.9928 and R2=0.9942, correspondingly for Ginsenoside Rg5 and Ginsenoside Rk1, the method was validated for selectivity, accuracy, and precision (RSD < 5%).
Statistical analysis
The column statistics, the area under the curve, and ANOVA analysis were conducted using GraphPad Prism Software 3.0 (GraphPad Software Inc., San Diego, CA, USA). The statistical significance of differences between the dissolution profiles of ginsenosides Rg5 and Rk1 released from HRG80 capsules and GCD complex containing tablets over time were assessed using two-way between/within ANOVA wherein an interaction effect indicates a different response over time between the two dosage forms.
Results
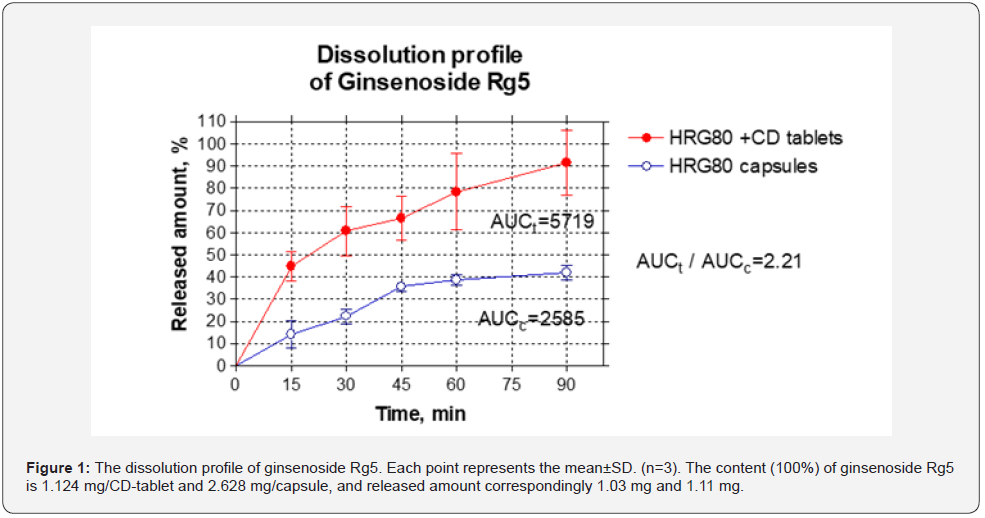
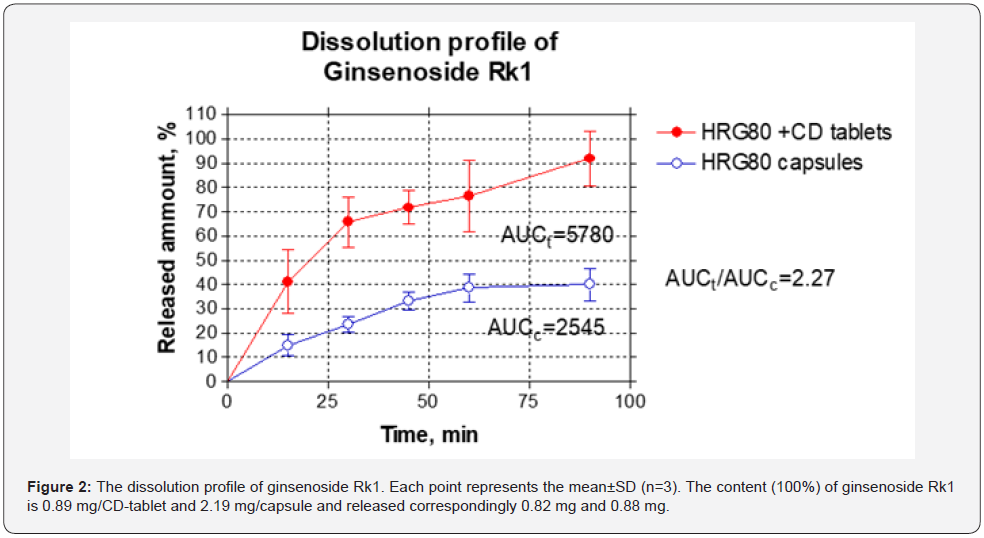
The dissolution profiles of ginsenosides Rg5 and Rk1 released from both preparations are shown in Figure 1 & 2. The dissolution rate of both Ginsenosides Rg5 and Rk1 began to reach equilibrium at 60 min. At that time, the dissolution rate of ginsenosides Rg5 release from HRG80 capsules and GCD complex containing tablets were 40%, and 80%, respectively. Compared with the GCD free capsules, the dissolution rate of the GCD complex increased 2-fold, indicating that GCD enhanced the solubility on Rg5.
Discussion
This study shows that the dissolution rate of both ginsenosides Rg5 and Rk1 from the GCD inclusion complex was significantly enhanced presumably due to both the surface-active volatilization of γ -CD in the aqueous solution [19] and intermolecular interactions by hydrogen bonds. The Ginsenosides Rg5 and Rk1 amounts released from one GCD tablet were the same as from GCD free capsule containing 2.2-2.3-times greater dose of HRG80 (Figure 1 & 2). The ratios of Areas Under the Curves (AUC) of ginsenosides Rg5 and Rk1 dissolved from capsules and tablets correspondingly are 2.21 (Figure 1) and 2.27 (Figure 2), suggesting that incorporation of cyclodextrin in the chewing tablets increases the solubility of ginsenosides Rg5 and Rk1 for 221% and 227% correspondingly.
Conclusions
In this study, we, for the first time, have demonstrated that the water dissolution rate of Ginsenosides Rg5 and Rk1 released from chewable tablets containing Red Ginseng incorporated cyclodextrin complex is about 2.2 - 2.3-fold higher of cyclodextrin free drug preparation.
Acknowledgments
The authors gratefully acknowledge valuable comments and recommendations by Professor Alexander Panossian.
References
- Panossian A, Abdelfatah S, Efferth T (2021) Network Pharmacology of Red Ginseng (Part I): Effects of Ginsenoside Rg5 at Physiological and Sub-Physiological Concentrations. Pharmaceuticals (Basel) 14(10): 999.
- Panossian A, Abdelfatah S, Efferth T (2021) Network Pharmacology of Ginseng (Part II): The Differential Effects of Red Ginseng and Ginsenoside Rg5 in Cancer and Heart Diseases as Determined by Transcriptomics. Pharmaceuticals (Basel) 14(10): 1010.
- Mariage PA, Hovhannisyan A, Panossian AG (2020) Efficacy of Panax ginseng Meyer herbal preparation HRG80 in preventing and mitigating stress-induced failure of cognitive functions in healthy subjects: A pilot, randomized, double-blind, placebo-controlled crossover trial. Pharmaceuticals (Basel) 13(4): 57.
- Dimpfel W, Mariage PA, Panossian A (2021) Effects of Red and white ginseng Preparations on Electrical Activity of the Brain in Elderly Subjects: A Randomized, Double-Blind, Placebo-Controlled, Three-Armed Cross-Over Study. Pharmaceuticals (Basel) 14(3): 182.
- Choi SY, Kim KJ, Song JH, Lee BY (2018) Ginsenoside Rg5 prevents apoptosis by modulating heme-oxygenase-1/nuclear factor E2-related factor 2 signaling and alters the expression of cognitive impairment-associated genes in thermal stress-exposed HT22 cells. J Ginseng Res. 42(2):225-228.
- Zhu Y, Zhu C, Yang H, Deng J, Fan D (2020) Protective effect of ginsenoside Rg5 against kidney injury via inhibition of NLRP3 inflammasome activation and the MAPK signaling pathway in high-fat diet/streptozotocin-induced diabetic mice. Pharmacol Res. 155: 104746.
- Elshafay A, Tinh NX, Salman S, Shaheen YS, Othman EB, et al. (2017) Ginsenoside Rk1 bioactivity: a systematic review. PeerJ 17(5): e3993.
- Hong Y, Fan D (2019) Ginsenoside Rk1 induces cell cycle arrest and apoptosis in MDA-MB-231 triple negative breast cancer cells. Toxicology 15(418): 22-31.
- Hong Y, Fan D (2019) Ginsenoside Rk1 induces cell death through ROS-mediated PTEN/PI3K/Akt/mTOR signaling pathway in MCF-7 cells. Journal of Functional Foods 57: 255-265.
- Song L, Yang F, Wang Z, Yang L, Zhou Y (2021) Ginsenoside Rg5 inhibits cancer cell migration by inhibiting the nuclear factor-κB and erythropoietin-producing hepatocellular receptor A2 signaling pathways. Oncol Lett 21(6): 452.
- Xu D, Wang C, Zhao W, Gao S, Cui Z (2017) Antidepressant-like effects of ginsenoside Rg5 in mice: Involving of hippocampus BDNF signaling pathway. Neurosci Lett 645: 97-105.
- Liu Y, Fan D (2018) Ginsenoside Rg5 induces apoptosis and autophagy via the inhibition of the PI3K/Akt pathway against breast cancer in a mouse model. Food Funct 9(11): 5513-5527.
- Liu Y, Fan D (2019) Ginsenoside Rg5 induces G2/M phase arrest, apoptosis and autophagy via regulating ROS-mediated MAPK pathways against human gastric cancer. Biochem Pharmacol 168: 285-304.
- Yoo S, Park BI, Kim DH, Sooyoung L, Seung HL, et al. (2021) Ginsenoside Absorption Rate and Extent Enhancement of Black Ginseng (CJ EnerG) over Red Ginseng in Healthy Adults. Pharmaceutics 13(4): 487.
- Ma C, Lin Q, Xue Y, Ju Z, Deng G, et al. (2021) Pharmacokinetic studies of ginsenosides Rk1 and Rg5 in rats by UFLC-MS/MS. Biomed Chromatogr 35(8): e5108.
- Pan W, Xue B, Yang C, Miao L, Zhou L, et al. (2018) Biopharmaceutical characters and bioavailability improving strategies of ginsenosides. Fitoterapia 129: 272-282.
- Uekama K (2004) Design and evaluation of cyclodextrin-based drug formulation. Chem Pharm Bull (Tokyo) 52(8): 900-915.
- Loftsson T, Moya-Ortega MD, Alvarez-Lorenzo C, Concheiro A (2016) Pharmacokinetics of cyclodextrins and drugs after oral and parenteral administration of drug/cyclodextrin complexes. J Pharm Pharmacol 68(5): 544-555.
- Li H, Zhang G, Wang W, Chen C, Jiao L, et al. (2021) Preparation, Characterization, and Bioavailability of Host-Guest Inclusion Complex of Ginsenoside Re with Gamma-Cyclodextrin. Molecules 26(23): 7227.
- Sharma A, Lee HJ (2020) Ginsenoside Compound K: Insights into Recent Studies on Pharmacokinetics and Health-Promoting Activities. Biomolecules 10(7): 1007-1028.
- Tannous M, Caldera F, Hoti G, Dianzani U, Cavalli R, et al. (2021) Drug-Encapsulated Cyclodextrin Nanosponges. Methods Mol Biol 2207: 247-283.
- Rivero-Barbarroja G, Benito JM, Ortiz Mellet C, García Fernández JM (2020) Cyclodextrin-Based Functional Glyconanomaterials. Nanomaterials (Basel) 10(12): 2517.
- (2021) General chapter: 711 Dissolution requirements. USPNF 3.






























