Toxicological and Therapeutic Evaluation of a Poly-Herbal Syrup as Anti-Ulcer Drug
Anjan Adhikari1*, Sankhadip Bose2 and Sabyasachi Banerjee3
1Department of Pharmacology, Coochbehar Government Medical College, India
2Department of Pharmacy, Sanaka Educational Trust’s Group of Institutions, India
3Department of Phytochemistry, Gupta College of Technological Sciences, India
Submission: January 19, 2022; Published: January 31, 2022
*Corresponding author: Anjan Adhikari, Department of Pharmacology, Coochbehar Government Medical College, Coochbehar, West Bengal, India
How to cite this article:Anjan A, Sankhadip B, Sabyasachi B. Toxicological and Therapeutic Evaluation of a Poly-Herbal Syrup as Anti-Ulcer Drug. Glob J Pharmaceu Sci. 2022; 9(3): 555765. DOI: 10.19080/GJPPS.2022.09.555765.
Abstract
Objective: This experiment has been intended to demonstrate the antiulcer activities of a polyherbal syrup prepared by Diastase, Papain, Nux-vomica Tincture, Compound Cardamom Tincture and Casein hydrolysed. The herbs Papain, Nux-Vomica, Cardamom and fungal Diastase are described in Ayurveda as herbal medicine which have astringent, anti-phlogistic and acid neutralization properties. These properties are anticipated for the treatment of gastric ulcer.
Materials and Methods: To assess the formulation’s toxicity, an acute as well as sub-acute toxicity study were carried out. In anti-ulcer activity, ethanol induced ulcers have been identified in the rat’s stomach and the volume of gastric juice, free acidity, pH, gastric wall mucus content, total acidity, total protein, pepsin activity, peristaltic movement, lipase activity, amylase activity and gastric emptying activity were performed.
Results: By performing all the parameters it can be concluded that the formulation has the anti-ulcer property and also has the property to treat gastric diseases.
Conclusion: It can normalize the total digestion system for the gastric irritant and gastric malfunction patients.
Keywords: Antiulcer; Free acidity; Gastric juice; Gastric wall mucus content; Total acidity; Total Protein; Pepsin activity; Peristaltic movement; Lipase activity; Amylase activity; Gastric emptying activity
Introduction
Peptic ulcer, a global health problem, is the utmost collective gastrointestinal disorders which affect a major group of people across the world [1]. In the tropical countries, gastric ulcer marks around sixty percent of the adults and eighty percent of the child populace. The most prevalent anomalies of this disorder are gastric and duodenal ulcers, as well as gastro-oesophageal reflux. In the pathogenesis of gastric ulcer, it has been observed that the major reason of peptic ulcer and other gastrointestinal diseases are oxidative stress only [2]. The acute gastric lesions may be created in the human body through nonsteroidal anti-inflammatory drugs, ethanol or by infection of Helicobacter pylori [3,4]. According to the review of literature it has been concluded that oxygen-derived free radicals and lipid per-oxidation are responsible for the development of those gastric lesions [3,4]. Cyto-protective drugs may be the one option to minimize tissue injury due to gastric problems [5-7]. The remedy for such ailments primarily focused on rectification of an ostensible inequity among the pepsin activity, acid, and mucosal resistances. The achievement of the rehabilitation is restrained in the status of ulcer recovery, symptom management, and disease recurrence rate. Due to the difficulties concomitant with ulcer recurrence and side effects of prevailing medications, it’s urgently required for unconventional therapies which have antiulcer properties [8]. Treatment with natural drugs is frequently observed in developing countries to fight with different diseases. Now a days, this practice has become an alternative to pharmacotherapy against many diseases [9]. Due to safety, availability and low cost, herbal drugs are used in the treatment of a wide range of gastrointestinal disorders. The herbs Nux-Vomica and Cardamom are stated in Ayurveda as anti-phlogistic, astringent and acid neutralizing which are required for treating gastric ulcer. In Ayurveda, those have also been indicated to elevate the mucosal secretions, inhibiting those enzymes responsible for degrading the gastro cytoprotective prostaglandin E and prostaglandin Fα, thereby increasing their concentrations. These herbs exhibit the ability to form a complex with proteins. It has been demonstrated that this complex is resistant to proteolytic enzymes. Such attributes of those medications, as well as the methodologies proposed for the treatment of gastric ulcers, prompted the development of a polyherbal formulation including the aforementioned plants [1-9].
Materials and Methods
Preparation of formulation
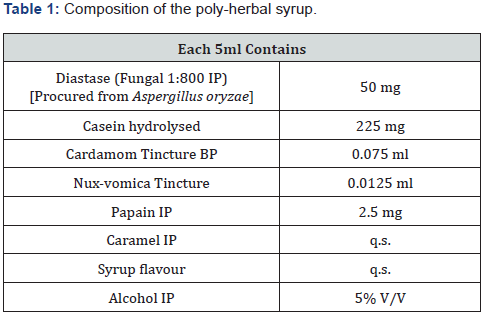
A syrup of Diastase, Papain, Nux-vomica Tincture, Compound Cardamom Tincture and Casein hydrolysed was prepared by using following composition (Table 1). The prepared syrup was evaluated biologically as anti-ulcer drug.
Animals
Healthy 8–12-week-old Wistar rats (female) were utilized. The animals were procured from the Animal House of Bengal School of Technology, Chuchura, Hooghly. Evaluation of physical and behavioral attributes of the animals was done before initiation of the experiment. The weights of the animals were within plus/ minus twenty percent of the mean weight of the animals. The protocols and the experiments were previously approved by IAEC (Registration Number -1726/PO/Re/S/14/CPCSEA). All the selected rodents have been placed under laboratory conditions for seven days for acclimatization. The female rats were kept in standard polypropylene cages. Five rats were kept in each cage. Paddy husk was used for the bedding of the cages. The bedding was changed frequently, to maintain hygiene of the animals. Twelvehour light/dark cycle was used for the animals as per prescribed guidelines. A temperature of 22±3oC was maintained by use of air conditioners. Relative humidity of 30-70 percent was used. The rats have been provided regular rat pellets. Food and drink were offered ad libitum. Apart from that, the animals were fasted over night before drug administration. Stainless steel sippers were used to feed water ad libitum throughout study period.
Study for acute toxicity
The analysis has been designed in accordance with Organization for Economic Cooperation and Development (OECD) Guideline - 420 (Acute Oral Toxicity - Fixed Dose Procedure).
The experiment employs healthy female rats weigh around 150 to 200g. All through the study, the rats were given unrestricted amounts of food and drink. Rats were administered at different doses with a polypropylene syringe. Sixteen-gauge oral feeding needle was used to administer the test sample. All the animals were fasted overnight prior to administration of sample. Only water was provided. This was to ensure that the food in GIT did not influence drug absorption or interacted with test sample. Food was given only after three to four hours of administration of the sample [10-13].
Study for sub-acute toxicity
The analysis was designed in accordance with Organization for Economic Cooperation and Development (OECD) Guideline - 407 (Repeated Dose 28-Day Oral Toxicity Study in Rodents). Healthy rats (M/F), weighing between 120-150g were used. The rats had unrestricted accessibility to food and drink. Rats were administered at different doses with a polypropylene syringe connected to a stainless steel 16 gauge oral intubation needle. All the animals were fasted overnight before drug administration. Food was given only after three to four hours of formulation administration. 10 rats (5 male and 5 females) were taken in each group. The animals were randomly assigned to each group.
Additional ten animals were taken for reversibility studies. Delayed occurrence of adverse/toxic effects was also noted. The animals were dosed daily for 28days [14-18].
Group I: Fed only vehicle (5%Tween 80) as a control (1ml/100gm b.w.) P.O.
Group II: Formulation (1.8ml/kg b.w.) P.O.
Group III: Formulation (2 ml/kg b.w.) P.O.
Ethanol-induced ulcer
The stomach ulcer will be induced in the rat according to previously explained method [12-23]. First the rats were kept in fasting for 24 hours. Then they had been arbitrarily distributed among five group of 6 rats each.
Group-I (Control group): was given distilled water and one ml 100% ethanol per kg b.w. orally.
Group-II and -III (Test groups): was given prepared formulation 1.8 and 2 ml/kg respectively.
Group-IV (Standard group): was given Famotidine at a dose of 20 mg per kg.
Group-V (Normal group): was given only distilled water.
All animals were given one ml per kg of 100% ethanol orally after 45 minutes of formulation and famotidine administration. To prevent coprophagia, those were housed in specifically designed cages. The rodents had been anaesthetized with anaesthetic ether 1 hour after ethanol injection. Their stomachs were immediately removed and preserved with 10% buffered formalin. The ulcer index was calculated by dividing the total area of stomach lesions by the size of the glandular section of the stomach [19-30].
Measurement of gastric lesions
Gastric ulcers are measured microscopically utilizing a light after they have been induced. The below ratings/scores, as stated by Takagi and Okabe [19-30], could be utilized to determine the ulcer index including the severeness of gastric lesions:
0 = no lesions, 1 =mucosal oedema and petechiae, 2 =1-5 smaller lesions (1 to 2 mm), 3 =>5 smaller or else one intermediate lesion (3 to 4 mm), 4 =≥ 2 intermediate lesions or 1 gross lesion (more than 4 millimetre), 5 =perforated ulcers.
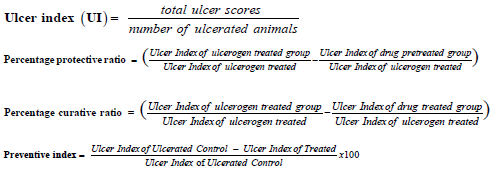
Ulcer index, percent protective ratio, and percent curative ratio are calculated utilizing the corresponding equations:
Biochemical estimations
The gastric fluid of ulcerated rats was obtained.
Gastric juice collection: One hour after ulcer onset, animals were anaesthetized for 2-5 minutes in a tiny chamber with a 1.9 percent diethyl ether-saturated cotton ball, and then sacrificed by cervical dislodgement. The stomachs of the animals had been quickly taken off and dissected across the larger curving, wherein its contents have been procured for volume as well as pH assessment. The gastro discharge was preserved to estimate pepsin activities. Following that, gastro tissue specimens were carefully cleaned utilizing phosphate buffer saline (PBS) to eliminate unwanted thrombus before being evaluated morphologically to determine the gastric ulcer index. Second, individual stomach was dichotomizing carefully, with 1 portion submerged in formaldehyde (10%) for histopathological analysis and another one at a 1:10 (w/v) ratio, homogenised in potassium phosphate buffer pH 7.4 (0.1 M). The homogenates have been taken for centrifugation (3000 rpm for 10 minutes at 4°C) in a 3-18KS cooling centrifuge from Sigma, Germany. Supernatants were kept at -80°C for future biochemical studies [19-30]. The collected gastric fluid was centrifuged, and the pH and the volume of the same were recorded. Then the gastric fluid was exposed to the following biochemical estimations
Determining total and free acidity [31]: Gastric fluid (1 millilitre) had been pipetted out and [31] taken in a conical flask (100 millilitre). To it two or three drops of Topfer’s reagent had been mixed, and titration was done adding sodium hydroxide (0.01 N) till entire residues of red disappeared, leaving behind yellowy-orange color. The amount of base used had been recorded. That quantity is equivalent to free acidity. After mixing two or three drops of phenolphthalein solution, the titration was resumed until a clear red tint appeared. The total amount of alkali used was recorded. Total acidity is represented by the quantity. The following formula was utilised to determine acidity
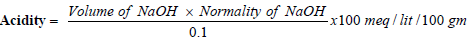
The student’s t-test and ANOVA were used to evaluate statistical significance.
Estimating total proteins : The dispersed proteins within gastric fluid [32] were measured utilising an alcoholic precipitate produced by mixing alcohol (90%) and gastric juice at a ratio 9:1. The alcoholic precipitation of gastric secretion was then dispersed in one millilitre of sodium hydroxide (0.01 N) and 0.05 ml of that had been placed into other test tube. 4 mL of basic mixture was transferred to this and left for 10 minutes. After some time, few drops of phenol reagent (0.4 ml) have been mixed, and the colour began to emerge again after 10 minutes. A Hitachi 15-20 spectrophotometer had been used to take reading at 610 nm using distilled water as blank. The protein content was then estimated using a standard curve generated with bovine albumin and reported in microgram per millilitre of gastric secretion. The statistical evaluation employed the student’s t-test [19-30].
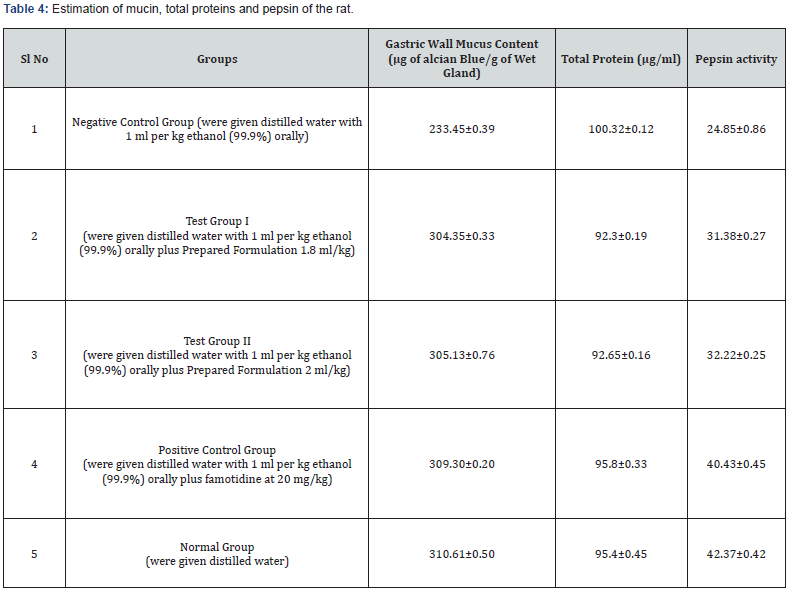
Estimating mucin: Following the collecting of stomachic secretion, the glanduliferous [33] region incisions which exposed the less curvature have been unveiled. The anterior end of the stomach had been immersed for two hours in alcian blue 8GX (0.1 percent) diluted in sucrose 0.16 (M) buffered with 0.05 M sodium acetate and pH adjusted with hydrochloric acid. Un-complexed dye was eliminated with 2 washing at fifteen and forty-five minutes in sucrose (0.25 M) solution. For 2 hours, the dye mixture including mucus had been diluted in ten millilitre magnesium chloride (0.5 M). The resultant blue liquefaction had been gently stirred including an equivalent amount of diethyl ether before measuring the optical density of the aqueous phase with Hitachi 15-20 spectrophotometer at 605 nm. The sample’s mucin content was calculated using the mucin standard curve, which was expressed in micrograms per gramme of moistened glandular tissue.
Estimating pepsin: At 37°C, 20 μl of stomach juice have been incubated for [34] ten minutes utilizing 500μl of albumin solution (5 milligram per millilitre of HCl (0.06 N)). The action was interrupted using 200μl of ten percent trichloroacetic acid, and the contents had been centrifuged for twenty minutes at the rpm 1500. The procured supernatant was alkalinised using 0.55 M sodium carbonate (2.5ml), and 1.0 N Folin’s reagent (400μl) had been put in the tubes and incubated for half an hour at normal temperatures. A spectrophotometer set to 660 nm was used to measure the absorbance of the sample. A standard curve is used to calculate the concentration of pepsin.
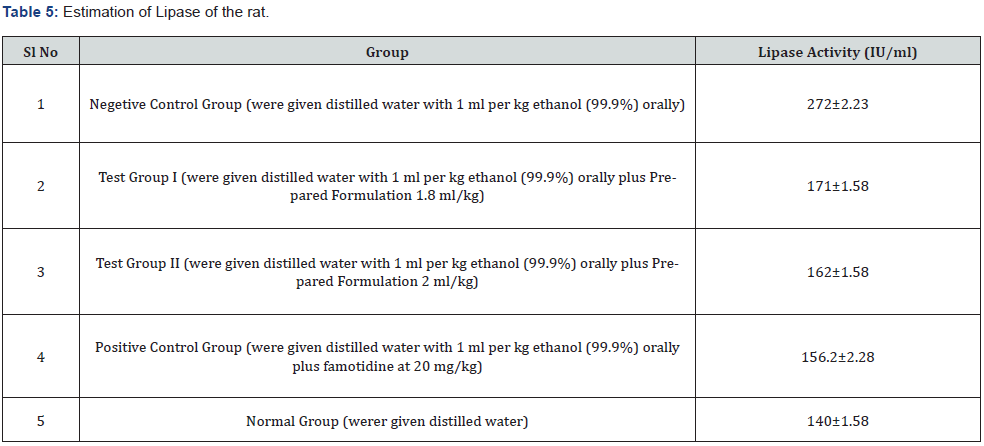
Estimating Lipase: Lipases, like pancreas lipase, are essential for lipid metabolism [35]. Lipases are also important in functions including cell signalling and inflammation. Lipase action assays are frequently utilized to test for pancreas damage or syndrome, as well as to evaluate disorders like Crohn’s disease, cystic fibrosis, and celiac disease. The Lipase Activity Assay kit was used to make this assessment.
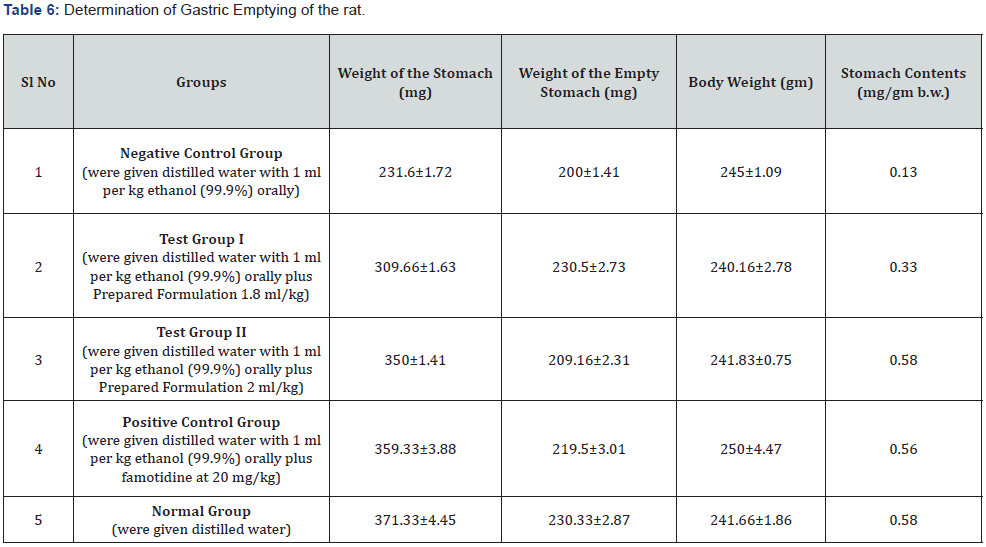
Gastric emptying test: The mass of foodstuff residual remaining in the stomach of rats fed a regular solid meal has been measured in animals. In rats fed a regular feeding diet, the quantity of residual in stomach should be inversely proportional to time after meal ingestion and the pace of gastric emptying. Prior to the beginning of the investigation, the previously grouped rats consuming regular feeding pellets had been relocated for cleaning polypropylene cages with grate flooring for one hour habituation session. After that, the animals have been slaughtered, laparotomies, and the stomach was taken. An analytical balance was used to weigh the full stomach. Every stomach was cut and opened and washed with water. The excessive wetness has been eliminated by sponging gently using tissue paper, and the emptied stomach was weighed. To evaluate gastric emptying, 3 weights had been required: (a) full stomach weight, (b) emptied stomach weight and (c) body weight. The weight of foods left within stomach has been represented per gramme body weight to account for body size variation, which has been demonstrated in rats to impact the transit of a feed across the gastrointestinal tract [36-39]. The stomach contents were estimated in the following:
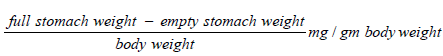
Estimating amylase: This assay methodology seems suitable for the colorimetric measurement [40] of amylase activities in tissue and cell culture supernatants, plasma, urine, serum, and other biologic fluids using the Amylase Activity Assay Kit (MAK009). Amylase function has been measured utilizing a coupled enzymatic assay, which yields a colorimetric outcome at 405 nm, proportionate towards the quantity of substrates cleaving by the amylase, ethylidene-pNP-G7. At 25°C, 1 unit seems to be the quantity of amylase which results in cleaving ethylidene-pNP-G7 to produce 1μmole of p-nitrophenol in every minute.
Assessment of gastrointestinal motility: For ten days prior to the study, 20 mature female Swiss Albino mice weight in between thirty to forty grams had been chosen and maintained in polypropylene cages (30 x 20 x 13 centimetre) in normal circumstances (21±1oC, relative humidity fifty to sixty percent with a twelve-hour reversing dark-light cycle). The rodents received unlimited accessibility for drinking as well as standard commercial lab feeding. The mice were separated among 5 groups of 6 each on the test day. Mice were weighed and given free access to water while being denied of food. The mice in Group-I served as the control group 180 minutes post food deprivation, receiving distilled water with 1 ml per kg ethanol (100%) orally. Groups II and III were given 1.8 and 2 ml per kg of the specified mixture, accordingly. Famotidine (20 milligram per kilogram b.w.) was orally given to Group-IV as the positive control medicine, whereas Group-V was given simply distilled water. After 1.5 hour, each mice received 0.3 ml slurry of 5% charcoal in 10% water orally by gavage (time 1.5 hour). Those rodents received unrestricted accessibility to foods after 1 hours (time 2.5 hours). Those mice had been monitored in every five minutes till faeces containing charcoal were removed (maximal observing time was 7.5 hours). Charcoal had been noticed within the faeces utilizing whether natural light or a microscope to facilitate the identifying of the black dots. The time it took for the charcoal to be eradicated was used to calculate the findings [41-42].
Result and Discussion
Acute toxicity
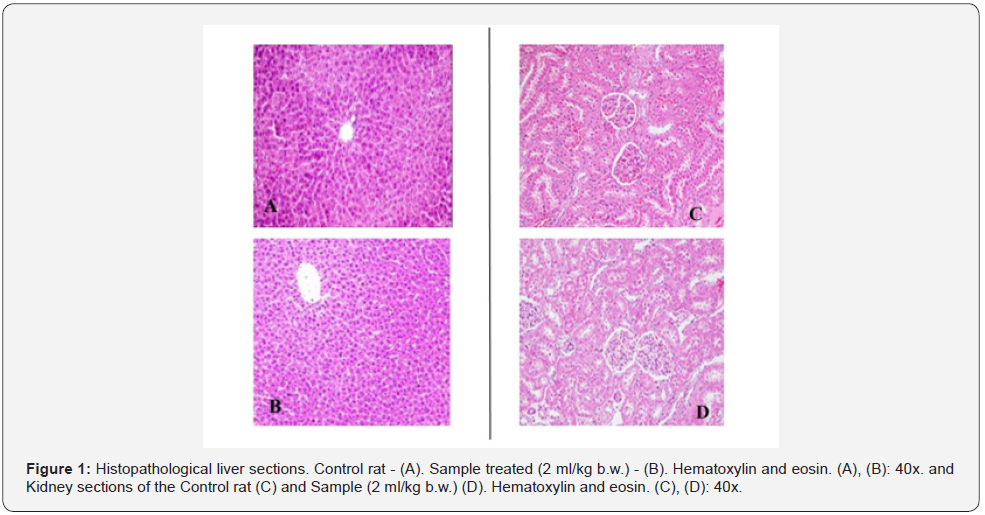
During the acute toxicity study all the animals showed their normal activities. Gross necropsy of the test rodents didn’t exhibit any gross pathological changes. There is ≥1 rats with evident toxicity and/or <1 death at an oral dosage of 2 ml/kg body weight. Thus, sample may be classified as GHS (Globally Harmonized Classification System for Chemical Substances and Mixtures) Category - 5. Category 5 is generally given to compounds which are relatively safe, and they have low values of acute toxicity. Histopathological studies of liver and kidney were conducted and seems to be normal Figure 1.
Sub-acute toxicity study
Variation in average body weight, variation in food intake behavior, blood parameters and biochemical analysis of the blood of male and female rats were normal. Lipid metabolism was found to be normal in female as well as male rats. With regards to the kidney function tests, the urea levels had found to be considerably elevated (p<0.05) in the male and female rats receiving highest dose of test sample (2ml /kg b.w. P.O.). The creatinine levels and urea levels did not show much variation with regards to the control group. No major alterations had been noticed in the relative organ weights of treated rats with provided test sample after 28-day period of repeated oral administration at varying doses of sample. Histopathological evaluation of kidney and liver portions of Wistar rats receiving Control treatment and provided test sample (1.8 and 2ml/kg b.w.) have been shown in Figure 2. The sections of animals receiving lower and mid dosage of test sample show normal cellular architecture when compared with control group.

Ethanol induced ulcer
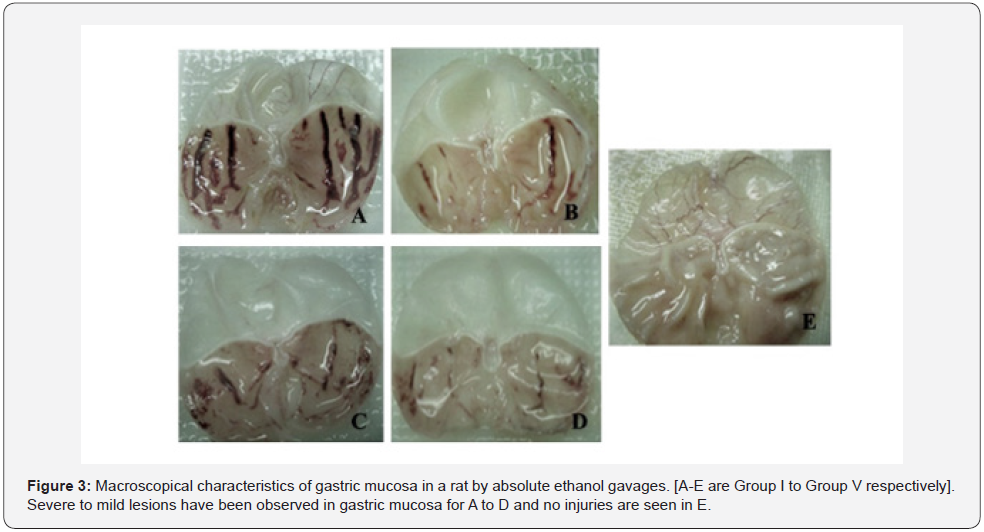
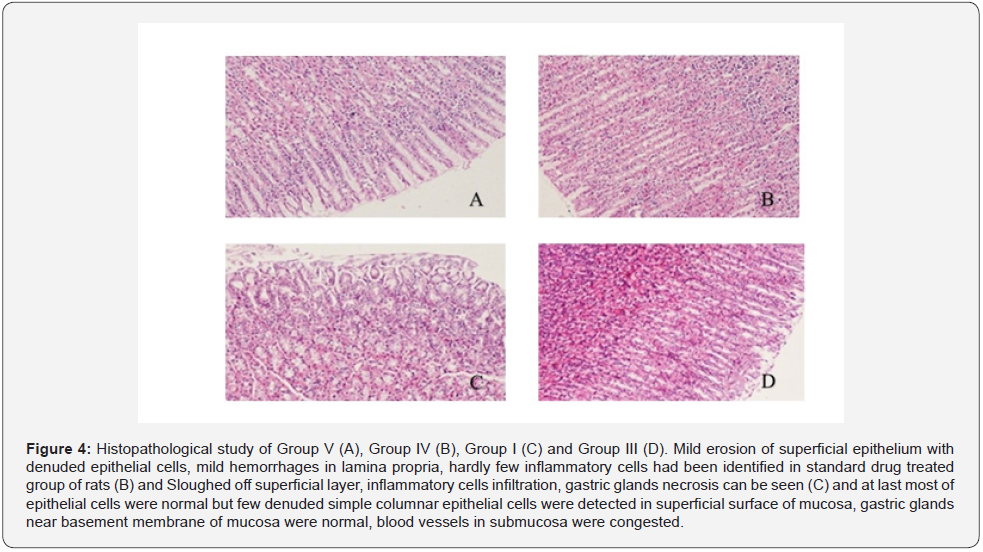
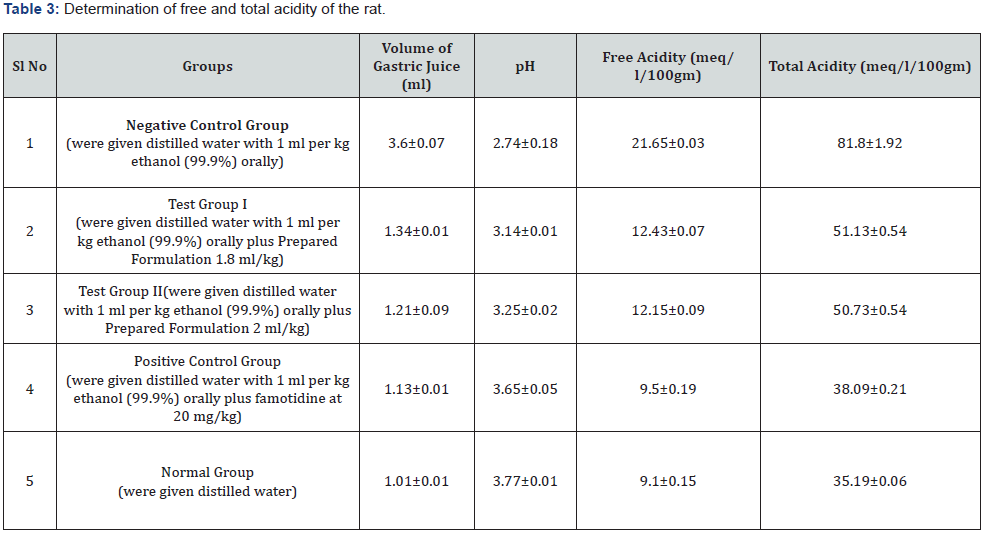
The present study was considered as gastro-protective efficacy of a prepared poly herbal preparation over ethanolincited gastric ulcer with famotidine as standard drug, commonly accepted for gastric ulcer treatment. In this work, an acute ulcer had been noticed in rats induced by ethanol (99.99%) (Table 2). It was evidently established by organoleptic and histological conclusions that showed significant haemorrhage, seemed to be acute congestions in the lamina propria submucosa and inter-villus extravasation of RBCs within the mucosal villi of the gastric tissues. Such results might be attributed to ethanolic toxicological impacts that leads reduce in coagulopathy function, resulting in the continuation of haemorrhage [32]. Besides, in the ethanol treated rats, significant coagulate necrosis had been noted within few portions of gastric mucosa. When comparing with the ulcerated group, pre-treatment of rats with prepared formulation substantially decreased the ulcer index at both doses (1.8ml/kg b.w. and 2ml/kg b.w.). Furthermore, ulcerated animals treated with formulation had a lower ulcer index than those treated with the standard drug famotidine, suggesting that formulation could be useful in treating gastric ulcers (Table 3) (Figure 3 & 4).
The stomach pH level had been observed to be significantly decreased in ethanol-treated rats as compared to the normal control group. In comparison to the ethanol group, formulation pre-treatment dramatically increased gastric pH levels while simultaneously decreasing gastric secretion. Furthermore, pretreatment with this formulation found to have the same effect on stomach pH as the standard drug famotidine. That seems to have a strong capability to reduce gastric acid productions and neutralise the acidic condition in the stomach (Table 3). Additionally, when compared to the control group, ethanol ulcerated rats exhibit a substantial drop-in pepsin functionality. Pre-treatment of the formulation, on the other hand, enhanced pepsin functionality in gastric secretion in a dose-dependent way. It may demonstrating that the formulation can modulate the ethanol influence on peptic activity (Table 4).
Estimation of lipase
Gut perforation and renal failure are two diseases that can lead to hyperamylasemia or hyperlipasemia. In between them, increased lipase and amylase were most likely caused by acute gastritis as well as chronic alcoholism. In this study, the amount of lypase was increased in the negative control group due the formation of gastric ulcer flowed by pancreatitis but both the doses (1.8ml/kg b.w.& 2ml/kg b.w.) of prepared formulation decreased the level of lypase near to normal similar to standard drug (Table 5).
Gastric emptying test
In the current investigation, it’s been observed that the formation of gastric ulcer increases the tendency of gastric emptying of the rat stomach. But the both the dose of prepared formulation (1.8ml/kg b.w.& 2ml/kg b.w.) have made the gastric emptying near to normalize like standard drug Famotidine (Table 6).
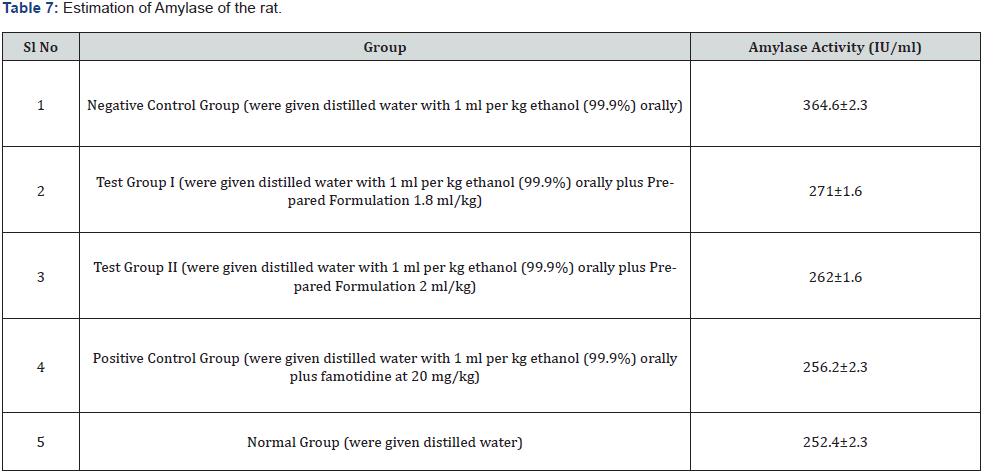
Amylase estimation
Normal blood amylase levels in healthy rats on a typical diet are around 250 IU per ml, and 65 IU per ml after twelve hours of fasting, according to earlier research. The activity of pancreatic enzymes in serum changes as a result of food and pancreatic function. Acute pancreatitis is thought to be caused by a penetrating ulcer in the pancreatic parenchyma caused by a posterior gastric wall penetrating ulcer. Though, various additional disorders, including renal failure and intestinal perforation, can cause hyperamylasemia or hyperlipasemia. The increased lipase and amylase levels in this case were most likely caused by acute gastritis and chronic alcoholism. In this study, the amount of amylase was increased in the negative control group due the formation of gastric ulcer flowed by pancreatitis but both the doses (1.8ml/kg b.w.& 2ml/kg b.w.) of prepared formulation decreased the level of amylase near to normal similar to standard drug (Table 7).
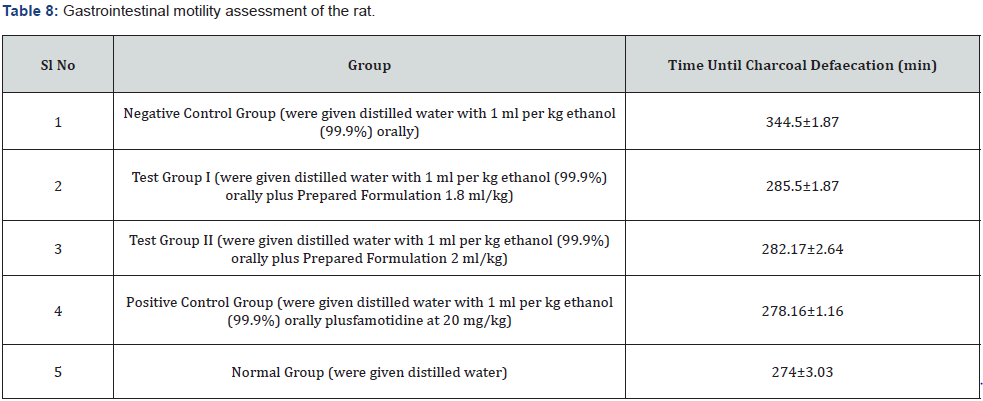
Gastrointestinal motility assessment
Disturbances in gastric motility can cause dyspeptic symptoms like nausea, vomiting and pain, which can be resistant to gastric acid suppression. Numerous investigations on individuals with active gastric ulcer have documented motility abnormalities. Here in this study also same phenomena have been observed. Due to the gastric ulcer formation the time of charcoal defaecation increased in negative control group which has been normalised by both the doses (1.8ml/kg b.w.& 2ml/kg b.w.) of test formulations like standard drug (Table 8).
Conclusion
By performing all the parameters, it can be concluded that the prepared formulation has the anti-ulcer property and also has the property to treat gastric diseases. It can normalize the total digestion system for the gastric irritant and gastric malfunction patients. The herbal industry can manufacture this poly-herbal syrup containing Diastase, Papain, Nux-vomica Tincture, Compound Cardamom Tincture and Casein hydrolysed for treating gastric ulcers, gastric irritant as well as gastric malfunction of the patients. This poly-herbal formulation can fight for a numerous people of the World against gastric disorders.
Conflict of interest
The authors declared no conflicts of interest. The authors are entirely responsible for the paper’s content and writing.
References
- Chan FK, Leung WK (2002) Peptic ulcer disease. Lancet 360(9337): 933-941.
- Das D, Banerjee RK (1993) Effect of stress on the antioxidant enzymes and gastric ulceration. Mol Cell Biochem 125(2): 115-125.
- Niki E, Yoshida Y, Saito Y, Noguchi N (2005) Lipid peroxidation: mechanisms, inhibition, and biological effects. Biochem Biophys Res Commun 338(1): 668-676.
- Hollander D, Tarnawski A, Krause WJ, Gergely H (1985) Protective effect of sucralfate against alcohol induced gastric mucosal injury in the rat. Macroscopic, histologic, ultra structural and functional time sequence analysis. Gastroenterology 88(1-2): 366-374.
- Halliwell B (1991) Drug antioxidant effects: a basis for drug selection? Drugs 42(4): 569-605.
- Hoogerwerf WA, Pasricha PJ (2001) Agent used for control of gastric acidity and treatment of peptic ulcers and gastro esophageal reflux disease. In: Hardman JG, Limbird LE (eds.) Goodman & Gilman’s the pharmacological basis of therapeutics, (10th), McGraw-Hill, New York, USA, pp. 1005-1020.
- Valle DL (2005) Peptic ulcer diseases and related disorders. In: Braunwald E, Fauci AS, Kasper DL, Hauser SL, Longo DL, Jameson JL (Eds.), Harrison’s principles of internal medicine, (16th), McGraw-Hill, New York, USA, pp. 1746.
- Rang HP, Dale MM, Ritter M, Moore PK (2003) Pharmacology (5th ), Churchill Livingstones, Edinburgh, UK.
- Dahanukar SA, Kulkarni RA, Nrege N (2000) Pharmacology of medicinal plants and natural products. Indian J Pharmacol 32(4): S81-S118.
- Pandy V (2020) A Simple Method for Animal Dose Calculation in Preclinical Research. EC Pharmacol Toxicol 3: 01-02.
- Anroop BN, Shery J (2016) A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm 7(2): 27-31.
- (1992) OECD, Guidelines for the Testing of Chemicals: Test No. 420: Acute Oral Toxicity-Fixed Dose Procedure Repeated Dose 90-Day Oral Toxicity Study in Rodents.
- Ghosh MN (2005) Toxicity Studies. In: Fundamental of Experimental Pharmacology. Hilton and Company, Kolkata, India 3: 190-197.
- Kulkarni SK (1999) Handbook of Experimental Pharmacology. Vallabh Prakashan, New Delhi 3: 148-149.
- Turner RA (1965) Acute toxicity. In: Screening Methods in Pharmacology. Academic Press, New York 1: 302-304.
- Hilaly JE, Israili ZH, Lyouss B (2004) Acute and chronic toxicological studies of Ajuva iva in experimental animals. J Ethnopharmacol 91(1): 43-50.
- Dixit VP, Joshi S (1982) Effect of chronic administration of garlic (Allium salivum Linn.) on testicular function. Indian J Exp Biol 20(7): 534-536.
- (1998) OECD, Guidelines for the Testing of Chemicals: Test No. 407: Repeated Dose 28-Day Oral Toxicity Study in Rodents.
- (2001) OECD, Guidelines for the Testing of Chemicals: Test No. 414: Prenatal Developmental Toxicity Study.
- (2001) OECD, Guidelines for the Testing of Chemicals: Test No. 416: Two-Generation Reproduction Toxicity Study.
- Alkofahi A, Atta AH (1999) Pharmacological screening of the anti-ulcerogenic effects of some Jordanian medicinal plants in rats. J Ethnopharmacol 67(3): 341-345.
- Schmeda HG, Yesilada E (2005) Traditional medicine and gastroprotective crude drugs. J Ethnopharmacol 100(1-2): 61-66.
- Megraud F, Lehours P (2007) Helicobacter pylori detection and antimicrobial susceptibility testing. Clin Microbiol Rev 20(2): 280-322.
- Senay EC, Levine RJ (1967) Synergism between cold and restraint for rapid production of stress ulcers in rats. Proceedings Society Exp Biol Med 124(4): 1221-1223.
- Levine RJ (1971) A method for rapid production of stress ulcers in rats, in Peptic Ulcer. In: Pfeiffer CJ (Ed.), Munksgaard, Copenhagen, Denmark, pp. 92-97.
- Vincent G, Glavin G, Rutkowski J, Pare W (1977) Body orientation, food deprivation; and potentiation of restraint induced gastric lesions. Gastroenterologie Clinique et Biologique 1(6-7): 539-543.
- Davenport HW (1967) Salicylate damage to the gastric mucosal barrier. New England J Med 276(23): 1307-1312.
- Djahanguiri B (1969) The production of acute gastric ulceration by indomethacin in the rat. Scandinavian J Gastroenterol 4(3): 265-267.
- Bhargava KP, Gupta MB, Tangri KK (1973) Mechanism of ulcerogenic activity of indomethacin and oxyphenbutazone. Eur J Pharmacol 22(2): 191-195.
- Cho CH, Ogle CW (1979) The pharmacological differences and similarities between stress- and ethanol-induced gastric mucosal damage. Life Sci 51(24): 1833-1842.
- Oates PJ, Hakkinen JP (1988) Studies on the mechanism of ethanol-induced gastric damage in rats. Gastroenterol 94(1): 10-21.
- Takagi K, Okabe S, Saziki R (1970) A new method for the production of chronic gastric ulcer in rats and the effect of several drugs on its healing. Jap J Pharmacol 19(3): 418-421.
- Sener G, Paskaloglu K, Ayanoglu DG (2004) Protective effect of increasing doses of famotidine, omeprazole, lansoprazole, and melatonin against ethanol-induced gastric damage in rats. Ind J Pharmacol 36(3): 171-174.
- Lowry CH, Randall RJ, Rosebrough NJ, Farr AL (1951) Protein measurement with Folin phenol reagent. J Biol Chem 193(1): 265-275.
- Lüllmann H, Mohr K, Ziegler A, Bieger D (2000) Color atlas of pharmacology. New York: Thieme, Stuttgart.
- Puurunen J (1982) Effect of ethanol on peptic activity in the rat stomach. Digestion 23(2): 97-103.
- Droppleman DA, Gregory RL, Alphin RS (1980) A simplified method for assessing drug effects on gastric emptying in rats. J Pharmacol Methods 4(3): 227-230.
- Figueroa WG, Jordan T, Bassett SH (1968) Use of barium sulfate as an unabsorbable fecal marker. American Journal of Clinical Nutrition 21(11): 1239-1245.
- Franklin RA (1977) The influence of gastric emptying on plasma doses of the analgesic, meptazinol. Br J Pharmacol 59(4): 565-569.
- Gelencser F, Gati T, Hideg J (1973) Role of adrenergic receptors in the regulation of gastric motility in the rat. Acta Physiological Hungarian Academy of Sciences 44: 327.
- Brunton LL (1996) Agents affecting gastrointestinal water efflux and motility; emesis and antiemetics; bile acids and pancreatic enzymes. In: Goodman AG (9th), The Pharmacological Basis of Therapeutics. McGrawHill, New York, USA, pp. 917-936.
- Das AK, Mandal SC, Banerjee SK, Sinha S, Das J, et al. (1999) Studies on antidiarrhoeal activity of Punica granatum seed extract in rats. J Ethnopharmacol 68(1-3): 205-208.






























