Detection and Characterization of an Unknown Impurity in Levothyroxine Sodium Tablets by UHPLC, LC/MS, MS/MS and MS/MS/MS
Taliha Beril Ünlü, Zeynep Halamoğlu*, Burçin Kıvanç and Mine Uz Gökalp
Department of Analytical Development, Abdi İbrahim R&D Center, Turkey
Submission: December 13, 2021; Published: December 21, 2021
*Corresponding author: Zeynep Halamoğlu, Department of Analytical Development, Abdi İbrahim R&D Center, Turkey
How to cite this article:Taliha Beril Ünlü, Zeynep Halamoğlu, Burçin Kıvanç and Mine Uz Gökalp. Detection and Characterization of an Unknown Impurity in Levothyroxine Sodium Tablets by UHPLC, LC/MS, MS/MS and MS/MS/MS. Glob J Pharmaceu Sci. 2021; 9(3): 555763. DOI: 10.19080/GJPPS.2021.09.555763.
Abstract
Impurities in the active ingredient of a drug are very important for the quality and reliability of the finished product. Therefore, the identification of these impurities constitutes reliability for patients using the drug. Levothyroxine sodium has known but also unknown impurities. Liothyronine sodium is known as the most common impurity of Levothyroxine sodium. Thought to be more active than levothyroxine and with a rapid (few hours) onset and short duration of action, liothyronine sodium is used in the treatment of hypothyroidism, particularly in the cases of hypothyroid coma. In our study, besides known impurities of Levothyroxine sodium, some unknown impurities were observed in samples that were exposed to heat. In these unknown impurities, the most important one that increases the fastest with the temperature that will affect the quality of the product is the impurity at RRT 0.6, according to our impurity method which was developed in-house. The impurity that increases with temperature has been defined by various identification studies, including MS/MS analysis, LC-UV detection etc. The degradation product, which was initially characterized by its retention time as RRT 0.6, was subsequently identified to be a lactose adduct of Levothyroxine API, which is formed by a Maillard reaction between Levothyroxine and lactose.
Keywords: Levothyroxine sodium; Unknown impurity; Lactose adduct; Degradation impurity; LC/DAD; UHPLC/DAD/HRMS; UHPLC/DAD/HRMS/MS; MS/MS/MS
Introduction
Levothyroxine is the sodium salt of the L-isomer of sodium trioxine. Levothyroxine sodium preparations are used to treat or cure deficiency when thyroid hormone is not secreted adequately. It is also used to prevent or treat some thyroid tumors. [1] In patients with hypothyroidism, levothyroxine is used as replacement therapy. For most patients, therapy can be initiated with a full replacement dosage the average full replacement dose of Levothyroxine sodium is approximately 1.7 mcg/kg/day (e.g., 100-125 mcg/day for a 70 kg adult). Older patients may require less than 1 mcg/kg/day [2]. Levothyroxine sodium doses greater than 200 mcg/day are seldom required. An inadequate response to daily doses ≥300 mcg/day is rare and may indicate poor compliance, malabsorption, and/or drug interactions [3,4]. Levothyroxine was first isolated in crystalline form in 1915 its structural formula was discovered in 1926, and it was first synthesized in 1927 [5]. It is on the World Health Organization’s List of Essential Medicines [6]. Levothyroxine is available as a generic medication [7,8] which was the third most prescribed medication in the United States in 2016, with more than 110 million prescriptions [9]. Levothyroxine chemically known as (S)-2-amino-3-[4-(4-hydroxy-3,5diiodophenoxy)-3,5-diiodophenyl] propanoic acid can be synthesized by various methods and as shown in Figure 1 [10]. Related substances or impurities in the form of by products or process impurities are possible during the manufacturing process or storage of the drug substance [10]. Illumination of the nature of impurities in a product plays a significant role in the quality of the product. Levothyroxine sodium molecule has known impurities as Liothyronine Sodium (impurity A), impurity F and impurity G [11]. The preceding known impurity of Levothyroxine sodium according to pharmacopoeias is Liothyronine sodium. Apart from known impurities, an impurity that increases significantly with temperature was detected on our product. Levothyroxine sodium tablets contains many excipients such as lactose monohydrate, starch maize, acacia gum, sodium citrate dihydrate and magnesium stearate. It was predicted that levothyroxine sodium has a high probability of reacting with lactose due to the NH group in its structure and RRT 0.6 impurity was predicted to be formed in this way.
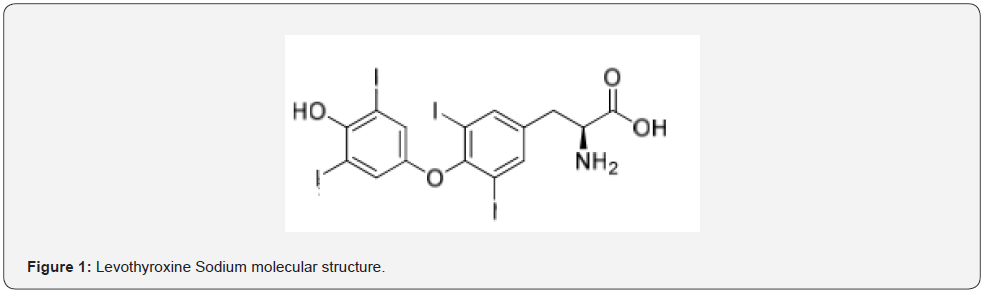
Lactose is widely used as a filler or filler-binder in the manufacture of pharmaceutical tablets. The general properties of lactose that contribute to its popularity as an excipient are its bland taste, low hygroscopicity, compatibility with active ingredients and other excipients, excellent physical and chemical stability and water solubility. Various lactose grades are commercially available that have different physical properties, such as particle size distribution and flow characteristics [12]. Lactose as a reducing sugar, bearing an equilibrium concentration of a free aldehyde (CHO) group, freely undergoes the Maillard reaction with amines yielding the corresponding Schiff’s bases, which further convert into thermodynamically favored six-membered 1-aminopyranose derivatives [13]. The degradation product is formed by the Maillard reaction of lactose and Levothyroxine sodium within the tablet matrix. Levothyroxine sodium as a primary amine reacts with the aldehyde group of the open-ring form of lactose, yielding unstable levothyroxine-lactose-hemiaminal. This hemiaminal undergoes water elimination under the influence of the basic reaction conditions, giving levothyroxine-lactose Schiff’s base. The Schiff’s base further undergoes an intramolecular cyclization reaction by nucleophilic attack of the 5-hydroxy (OH)-group from the open-chain glucose moiety to the electrophilic Schiff’s base imine carbon (C=N), yielding the final degradation product RRT 0.6. The reaction is presumably catalyzed by the equilibrium concentration of hydroxide anions (OH) which are generated from the hydrolysis reaction of levothyroxine anions [13]. The proposed structure of the levothyroxine-lactose adduct at RRT 0.6 was shown in Figure 2
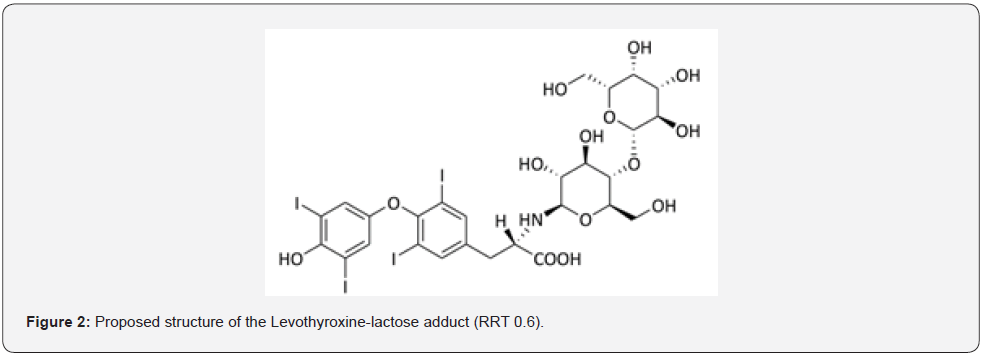
In stability samples of Levothyroxine Tablets of different strengths (all containing lactose as excipient/filler), a degradation product was detected that exceeded the identification and qualification threshold before reaching the intended shelf life. The degradation product, initially characterized by a retention time of RRT 0.6, was thought to be a lactose adduct of API Levothyroxine formed by a Maillard reaction between Levothyroxine and lactose. Since “Lactose adduct” purchased from two different suppliers (Puritan and Simsons) was matched with the impurity in our product with a retention time of RRT 0.6, the studies were started in this direction by MS systems [14]. We suppose that, in the view of previous information, this paper is the first research related with identification of unknown impurity of Levothyroxine sodium tablets, since there is no article published on this subject before in the literature review.
Materıals, Methods and Experımentals
Materials
Phosphoric acid (H3PO4), formic acid (HCOOH), acetonitrile (ACN), ethanol (CH2OH) were purchased from ISOLAB, Merck and JT. Baker. Deionized water was used from PURELAB ® Ultra Elga DV35 (High Wycombe, United Kingdom). Levothyroxine Sodium standard was obtained from Peptido. Levotiron 100μg Tablet provided by Abdi Ibrahim Research and Development Center Department. (Istanbul, Turkey).
Methods
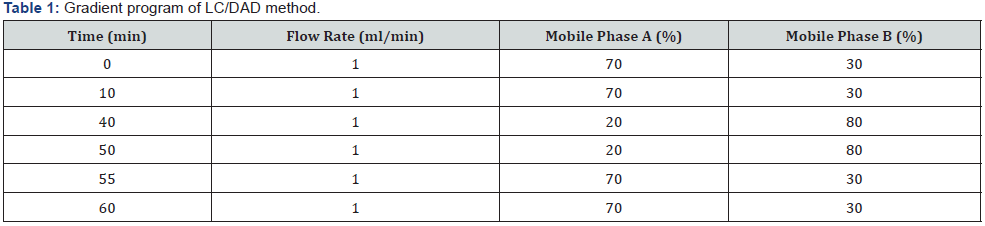
UHPLC/DAD (USP method modification): All required solutions including the mobile phase were prepared according to the modified method based on USP (2010) monograph. Agilent 1290 Infinity UHPLC instrument was used. All analytical chromatographic separations were carried out with Intersil ODS 3V 4.6mm×150mm, 5μm column. The UV detection wavelength was 225 nm. 0.06% phosphoric acid in water and 0.06% phosphoric acid in acetonitrile were used as mobile phase A and mobile phase B respectively. Modified gradient program used for optimal resolution is shown below, injection volume was 20μl and the column temperature was 25 °C. Diluent was prepared by combining mobile phase A and ethanol (1:2) respectively (Table 1).
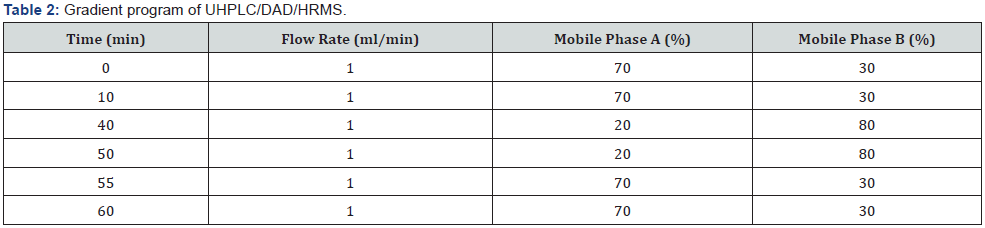
UHPLC/DAD/HRMS equipment for detection of impurity: Agilent 6540 QTOF instrument equipped with Agilent 1290 Infinity UHPLC was used. All analytical chromatographic separations were carried out with Inertsil ODS 3V 4.6 mm×150 mm, 5μm column. The UV detection wavelength was 225 nm. Formic acid was used instead of phosphoric acid. 0.06% formic acid in water and 0.06% formic acid in acetonitrile were used as mobile phase A and mobile phase B respectively. Modified gradient program used for optimal resolution was shown below, injection volume was 20μl and the column temperature was 25 °C. Diluent was prepared by combining mobile phase A and ethanol (1:2) respectively (Table 2 & 3).
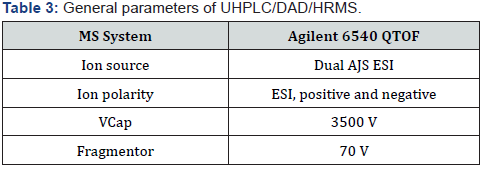
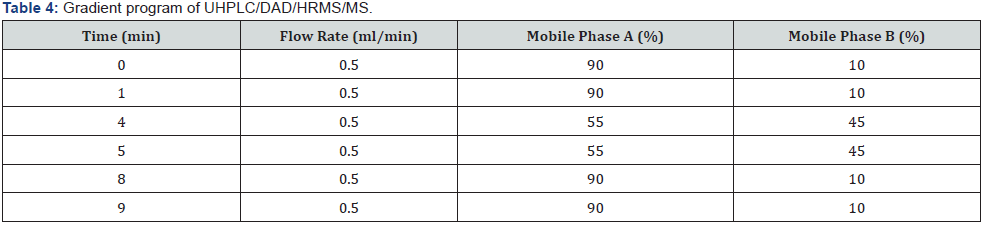
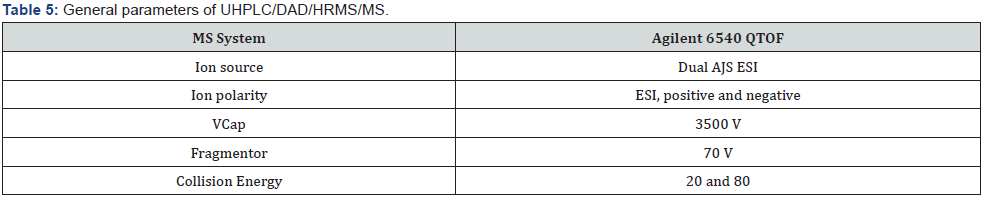
UHPLC/DAD/HRMS/MS equipment for detection of impurity: Agilent 6540 QTOF instrument equipped with an Agilent 1290 Infinity UHPLC was used. All analytical chromatographic separations were carried out with Acquity BEH C18 2.1 mm ID x 50 mm, 1.7 μm column. The UV detection wavelength was 225 nm. Formic acid was used instead of phosphoric acid. 0.06% formic acid in water and 0.06% formic acid in acetonitrile were used as mobile phase A and mobile phase B respectively. Modified gradient program used for optimal resolution is shown below, the injection volume was 20μl and the column temperature was 25 °C. Diluent was prepared by combining mobile phase A and ethanol (1:2) respectively (Table 4 & 5).
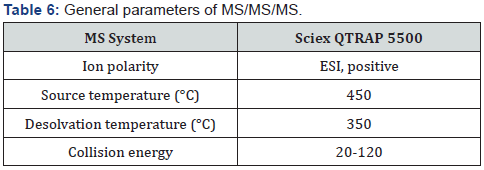
MS/MS/MS equipment for detection of impurity: Sciex QTRAP 6500 and QTRAP 5500 instruments were used and chromatographic conditions were shown in Table 6.
Experimentals
Impurity was detected and defined with different methods and equipment while using the same standard and sample preparations.
Preparation of standard solution of levothyroxine sodium: Levothyroxine Sodium Stock Standard Solution was prepared at a concentration of 0.0125 mg/ml with diluent.
Preparation of levothyroxine sodium l00μg and 25μg tablet: About 500.0 mg of tablet powder was weighed and transferred to a centrifuge tube containing several pieces of glass beads. Diluted to volume with diluent and dissolved with vortex, then it was cenrifuged. Diluted to a concentration of 0.05 mg/ml and filtered through a 0.45μm PVDF filter.
Preparation of placebo for levothyroxine sodium l00μg tablet levothyroxine sodium+ maizer starch, maizer starch (initial and stress): About 500.0 mg of tablet powder was weighed and transferred to a centrifuge tube containing several pieces of glass beads. Diluted to volume with diluent and dissolved with vortex, then it was centrifuged. Filtered through 0.45μm PVDF filter.
Result and Discussion
UHPLC/DAD analysis
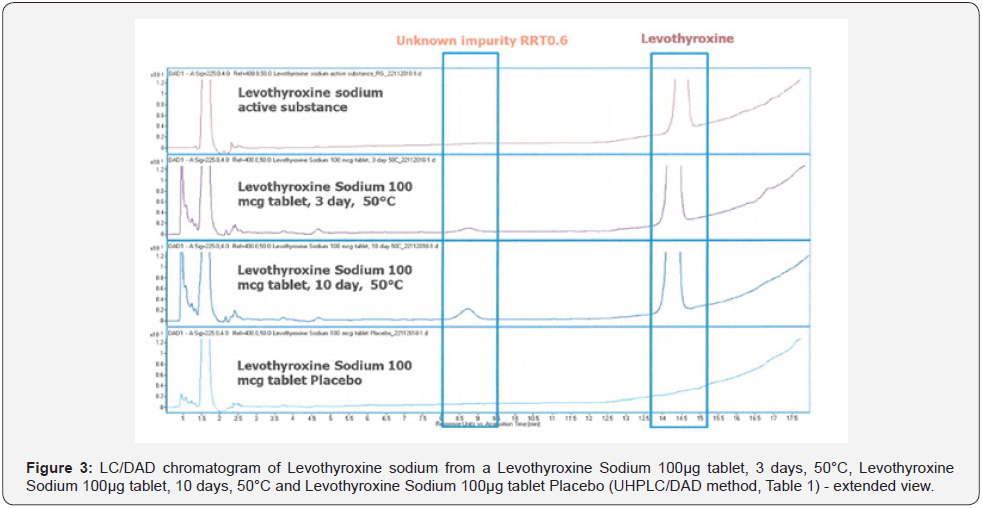
The LC-UV method was used to identify the impurity detected in Levothyroxine Sodium 100μg Tablets. Levothyroxine Sodium API, Levothyroxine Sodium 100μg tablet placebo, Levothyroxine Sodium 100μg tablet exposed to 50 °C for 3 days and Levothyroxine Sodium 100μg tablet exposed to 50°C for 10 days. Although there was an impurity which is called RRT 0.6 impurity in Levothyroxine Sodium 100μg tablet, 10 days-50 °C and Levothyroxine Sodium 100μg tablet, 3 days-50 °C, it was not detected in Levothyroxine API and Levothyroxine Sodium 100μg tablet placebo (Figure 3).
UHPLC/DAD/HRMS analyses
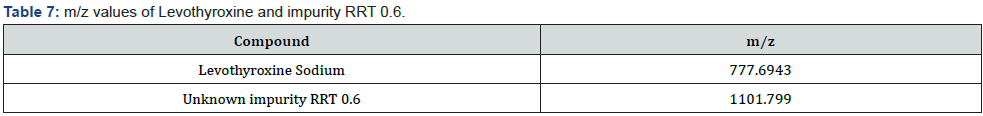
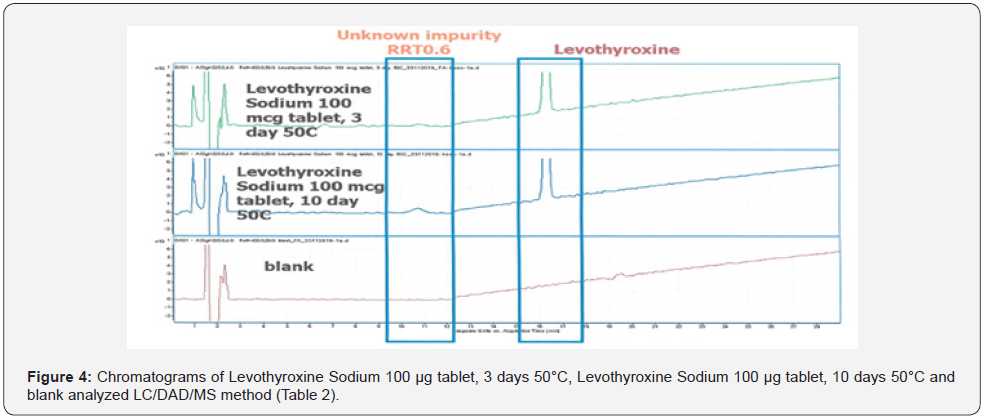
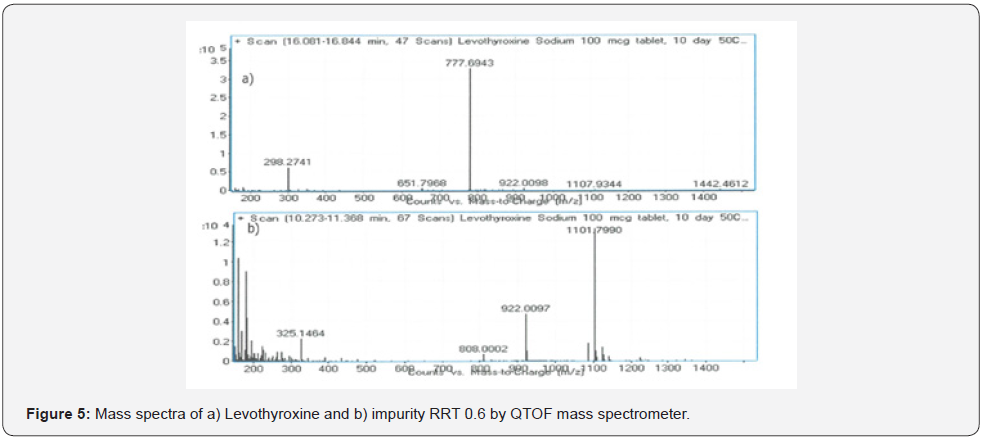
After applying the LC/DAD method and confirming the impurity peak at RRT 0.6, the analytical method was modified for LC/MS and LC/MS/MS analysis by replacing phosphoric acid with formic acid. In this way, it was aimed to detect the molecule in the LC/MS system (Figure 4). The m/z value of molecular peak for Levothyroxine Sodium and RRT 0.6 impurity are 777.6943 Da and 1101.7990 Da respectively in positive ESI mode (Table 7). Mass spectra of Levothyroxine and RRT 0.6 impurity are presented Figure 3 & 5. Potential molecular formulas were obtained from HRMS results which contain C, H, N, O, I (Table 8).
UHPLC/MS/MS analysis
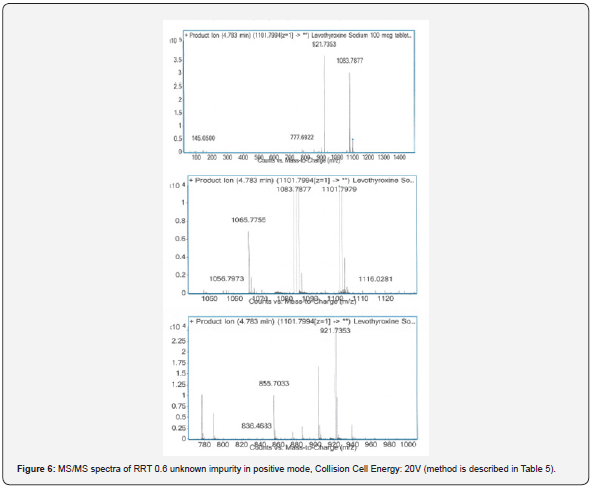
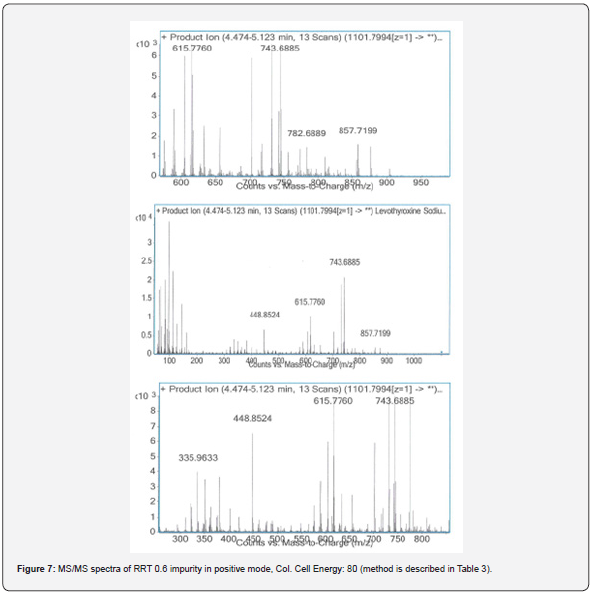
To increase the sensitivity of the method and make a more accurate identification for impurity, the molecular mass for RRT 0.6 impurity was fragmented with collision energy. Therefore, the mass transitions were monitored as a result of different collision energy voltage which is presented in Figure 6 & 7. While the collision energy was applied 20V, the highest abundance fragments were 921.7353 and 1083.7877; the collision energy was applied 80V, they were 743.6885 and 615.7760.

MS/MS/MS analysis
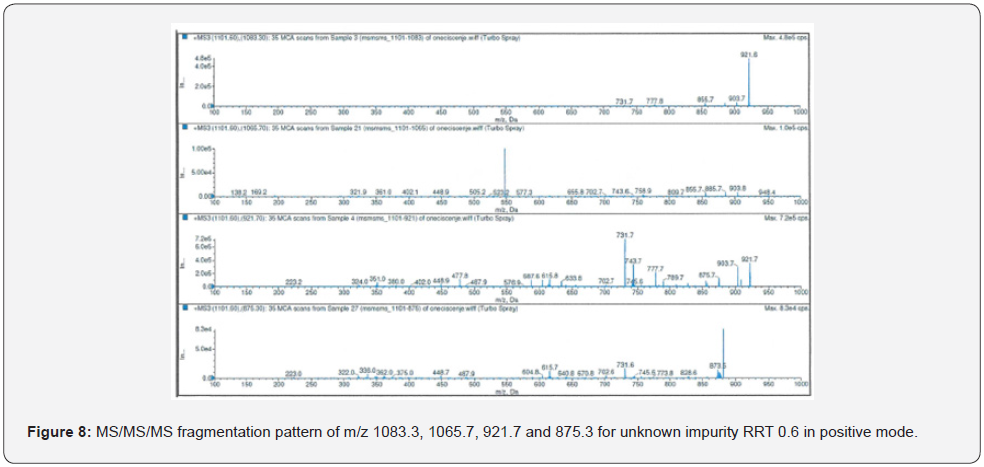
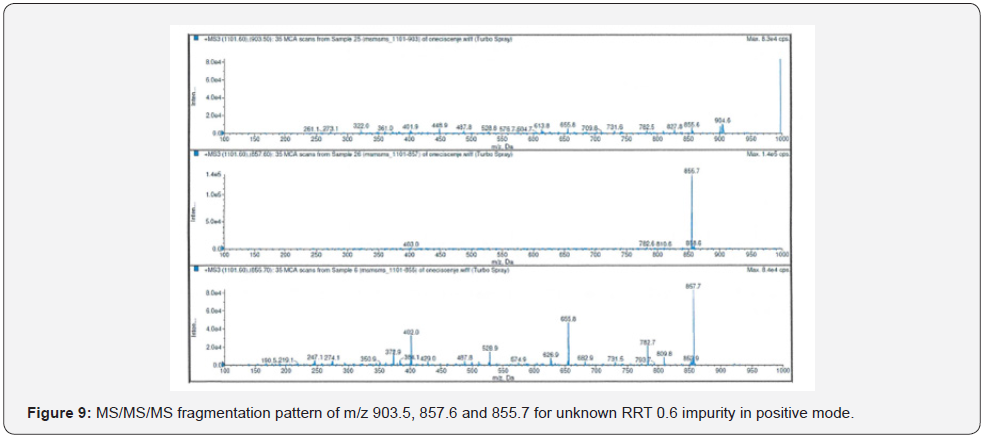
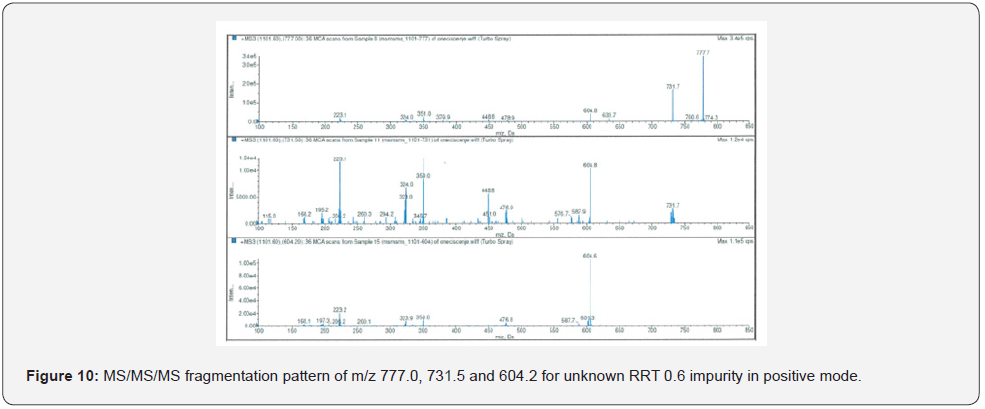
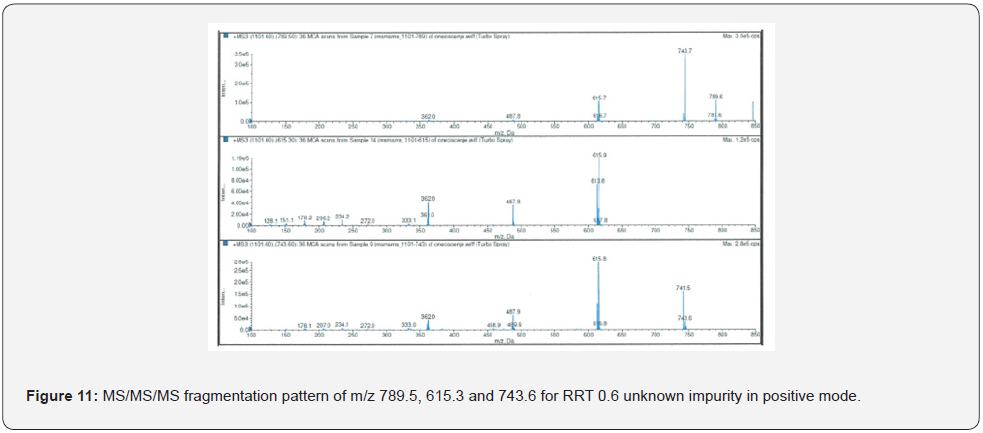
The highest abundance fragments obtained from MS/ MS analysis were fragmented one more time for accurate identification. The unknown RRT 0.6 impurity was analyzed by MS/MS/MS which method described in Table 4. The MS/MS/MS spectra of the impurity were presented in Figure 8-11.
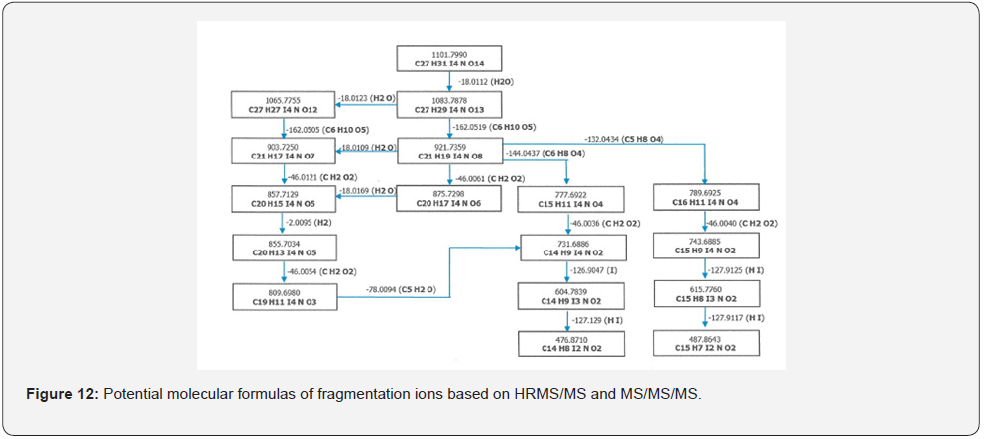
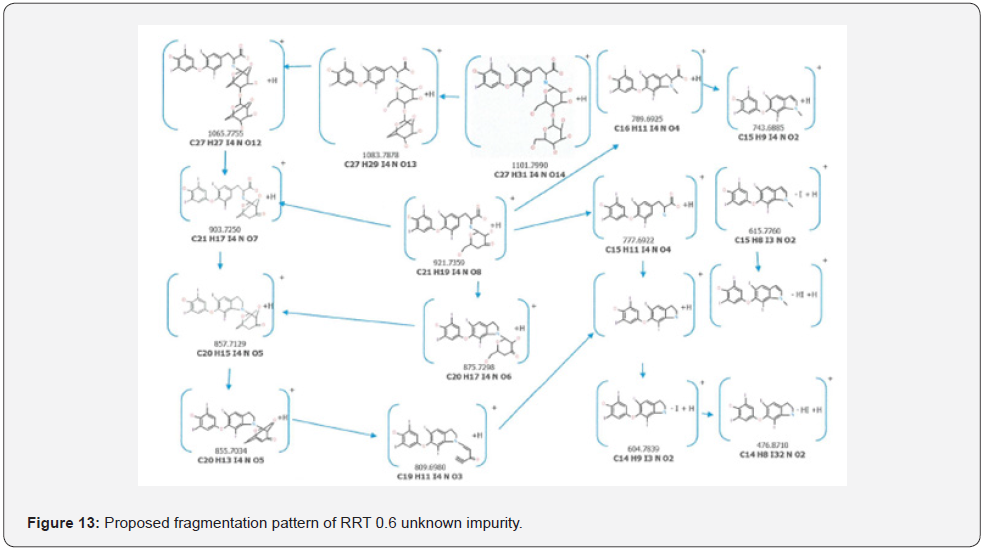
As a result of HRMS/MS and MS/MS/MS analysis for RRT 0.6 unknown impurity, the potential molecular formulas of fragmentation ions were presented in detail in Figure 12. A summary showing the molecular structures and transitions of fragments that may occur in HRMS/MS and MS/MS/MS analysis were presented in Figure 13.
Conclusion
When all the studies were summarized, an impurity was detected in the LC/DAD system. After being detected again in the LC/MS system, the substance had a fragmentation in the LC/ MS/MS system and the m/z ratio of the impurity was obtained. At the end of these studies, MS/MS/MS system was used to make a more precise cutting and to obtain system precision. Molecular mass for impurity was obtained as 1101.7923 m/z with the help of the MS systems. Because of the formation of the lactose adduct via Maillard reaction appears to be a general observation for all levothyroxine formulations containing lactose as an excipient, the unknown impurity was predicted to be a structure formed by Levothyroxine sodium with lactose. According to impurity results,the degradation (Figure 14) product RRT 0.6 was detected in all tested drug products (Levothyroxine Sodium 100μg Tablet, 10 day 50°C, Levothyroxine Sodium 100μg tablet, 3 day 50°C) containing lactose as inactive ingredient (filler). This indirectly supports the proposed structure of the levothyroxine lactose adduct. Also, RRT 0.6 impurity was defined as Lactose adduct because it chromatographically overlaps the Lactose adduct from different suppliers and the MS structure also matches the Lactose adduct (Puritan and Simsons). Considering all the studies conducted, the chromatographic results obtained from stress studies, the structure definitions and predictions obtained from MS studies prove that the impurity RRT 0.6 in our Levothyroxine Sodium 100μg tablet product was lactose-based.
Acknowledgements
The authors would like to thank Fidelta Galapagos Company for their excellent support regarding analytical analysis of Levothyroxine Sodium tablets.

References
- Boulton DW, Fawcett JP, David JW (1996) Stability of an extemporaneously compounded levothyroxine sodium oral liquid. American journal of health-system pharmacy 53(10): 1157-1161.
- SYNTHROID® (Levothyroxine sodium tablets, USP).
- Mandel SJ, Gregory AB, Larsen PR (1993) Levothyroxine therapy in patients with thyroid disease. Annals of internal medicine 119(6): 492-502.
- Ruggenthaler M, Grass J, Schuh W, Huber CG, Reischl RJ (2017) Levothyroxine sodium revisited: A wholistic structural elucidation approach of new impurities via HPLC-HRMS/MS, on-line H/D exchange, NMR spectroscopy and chemical synthesis. Journal of pharmaceutical and biomedical analysis 135: 140-152.
- Tekoa LK, Brucker MC (2010) Pharmacology for Women's Health 19: 544.
- (2019) World Health Organization. World Health Organization model list of essential medicines: 21st list 2019. No. WHO/MVP/EMP/IAU/2019.06. World Health Organization.
- Richheimer SL, Tahani MA (1983) Stability-indicating assay, dissolution, and content uniformity of sodium levothyroxine in tablets. Journal of pharmaceutical sciences 72(11): 1349-1351.
- Brower JF, Duckhee YT, John CR (1984) Determination of sodium levothyroxine in bulk, tablet, and injection formulations by high-performance liquid chromatography. Journal of pharmaceutical sciences 73(9): 1315-1317.
- Cooper DS, Leonidas HD (2019) Thyroid hormone therapy: past, present, and future. Endocrine 66(1): 1-2.
- Roy J (2002) Pharmaceutical impurities - a mini-review. AAPs Pharm Sci Tech 3(2):1-8.
- European Pharmacopoeia, Levothyroxine Sodium (2021) Implementation 05, 3: 3094-3095.
- Drug Development and Delivery (2004) Lactose in Pharmaceutical Applications 4(5).
- Ellis GP (1959) Advances in Carbohydrate Chemistry. The Maillard Reaction 14: 63-134
- (2005) McMaster MC, LC/MS A Practical User’s Guide, John Wiley& Sons, New Jersey, USA.






























